Abstract
The heterogeneous gold(I)-catalyzed highly

Introduction
Over the past few decades, C–H bond functionalization has been developed into an efficient and straightforward synthetic strategy for the construction of complex molecules.1–3 In order to make this strategy generally useful in organic synthesis, the development of highly site-selective functionalization of inert C–H bonds has to be addressed. Substituted benzenes exist widely in nature and materials-related fields,4,5 and they constitute a frequently employed skeleton in small-molecule drugs.
6
Therefore, developing site-selective C–H bond functionalization of a benzene ring is highly attractive to the synthetic community and very useful for post-modification of bioactive compounds. Transition-metal-catalyzed, directing-group-assisted methods have proven to be one of the most efficient and popular solutions to the site-selectivity problem.7–16 A wide range of directing groups (DGs) have been used for the
Transition-metal-catalyzed insertion of a carbene into a C–H bond reaction has proven to be a promising and efficient strategy for undirected C–H functionalization.17–19 However, highly site-selective C(sp)2–H bond functionalization of unactivated arenes with diazo compounds remains a challenge due to the presence of several competing reactions when mixing an alkyl-substituted benzene with diazo esters under the catalysis of transition metals. For instance, the rhodium-catalyzed benzylic C(sp)3–H functionalization of toluene has been reported by the groups of Thu and Davies.20,21 Recently, the application of gold catalysts to carbene transfer reactions has attracted considerable interest since gold carbenes display very unique reactivity and selectivity compared to those of other noble metals such as rhodium, copper, and palladium.22–25 Gold(I)-catalyzed C(sp)2–H bond functionalizations of activated arenes such as phenols,
Recently, mesoporous silica MCM-41 has been widely used as an ideal support for anchoring homogeneous metal complexes, owing to its excellent properties such as ultrahigh surface area, large and defined pore sizes, high pore volume, and high thermal stability.
39
In recent years, functionalized MCM-41-bound Au(I) or Au(III) complexes have been successfully applied to various organic reactions as highly effective and recyclable catalysts.40–44 To further expand our Au(I)-MCM-41 chemistry toolbox,42–44 herein we report the first heterogeneous gold(I)-catalyzed

Heterogeneous gold(I)-catalyzed
Results and discussion
Several MCM-41-anchored diphenylphosphine gold(I) complexes [MCM-41-PPh2-AuX] were easily prepared via a simple procedure, as illustrated in Scheme 2. 44 First, MCM-41-anchored diphenylphosphine ligand (MCM-41-PPh2) was prepared by the condensation of commercially available 2-(diphenylphosphino)ethyltriethoxysilane with mesoporous MCM-41 at 100°C in toluene for 24 h, followed by the reaction with Me3SiCl at room temperature in toluene for 24 h. Next, MCM-41-PPh2 was reacted with Me2SAuCl at room temperature in dichloromethane for 8 h, followed by treatment with various silver salts (AgX = AgOTf, AgNTf2, AgSbF6, and AgBF4) at room temperature for 0.5 h to afford the MCM-41-PPh2-AuX complexes as gray powders.

Preparation of the MCM-41-PPh2-AuX complexes.
In our initial screening experiments, the reaction of toluene (
Optimization of the reaction conditions. a
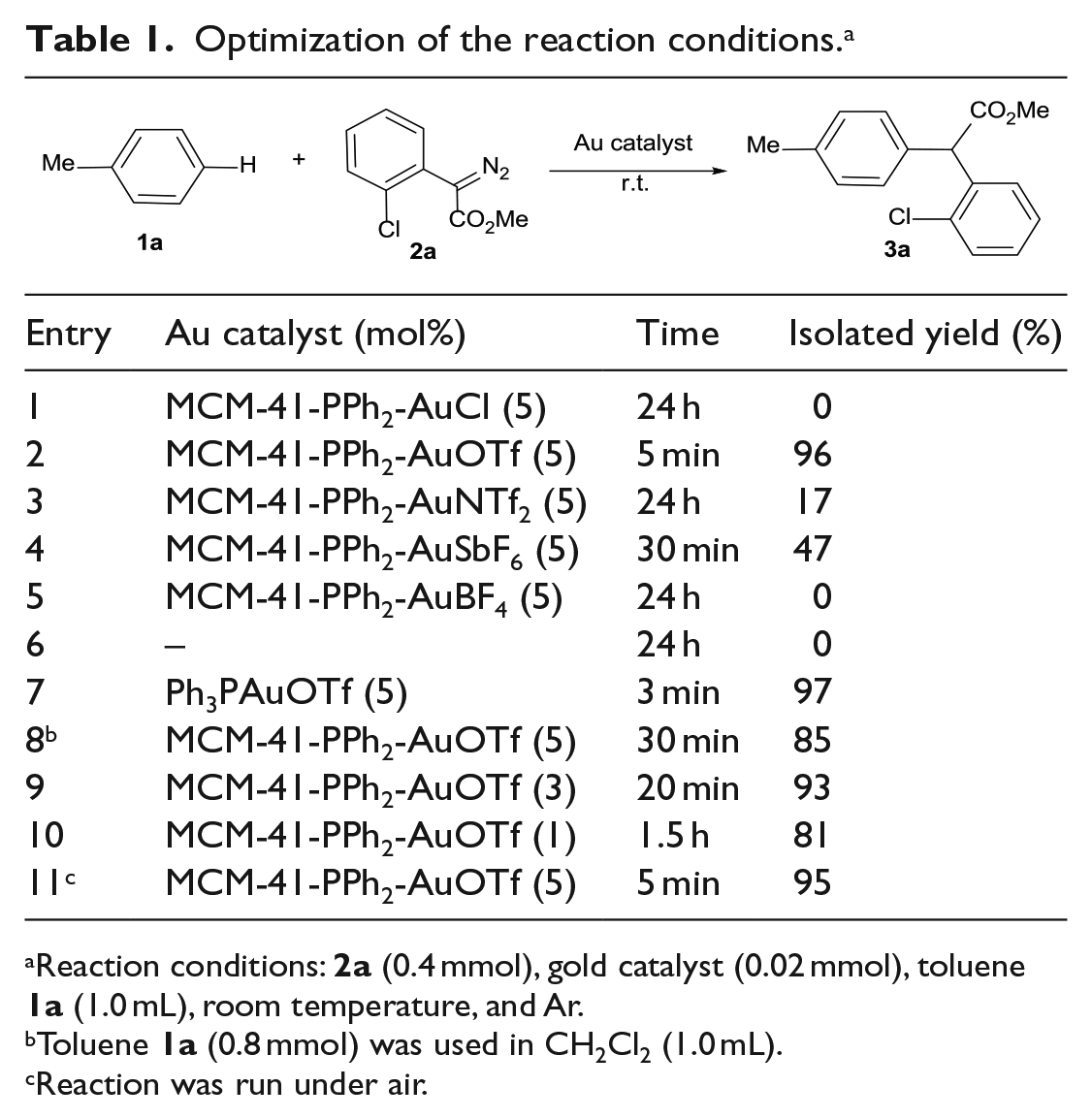
Reaction conditions:
Toluene
Reaction was run under air.
The optimized conditions were then used in the reactions of a wide range of unactivated arenes

Reaction conditions:
Isolated yield.
The gram scale reaction was carried out with
We next investigated the substrate scope of this heterogeneous gold(I)-catalyzed, highly
A plausible mechanism for this heterogeneous gold(I)-catalyzed C(sp)2–H bond functionalization of unactivated arenes is shown in Scheme 3.27,28 First, the cationic gold MCM-41-PPh2-AuOTf undergoes ligand exchange with the diazoester

Proposed catalytic cycle.
For the practical application of a heterogeneous precious metal catalyst, its stability and reusability are important factors to be examined. The MCM-41-PPh2-AuOTf complex can be easily recovered by simple filtration of the reaction solution. We next examined the recycling of the catalyst in the reaction of toluene (
Conclusion
In conclusion, we have developed a novel, efficient, and practical heterogeneous gold(I)-catalyzed C(sp)2–H bond functionalization of benzene derivatives with alkyl α-aryl-α-diazoesters in good to excellent yields with high chemo- and site-selectivities. In this method, the electron-withdrawing groups on the phenyl moiety of the diazoesters effectively promoted the chemo- and site-selective C(sp)2–H bond functionalization. Importantly, this heterogeneous gold(I) catalyst can be easily recovered by simple filtration of the reaction solution and recycled at least seven times with almost consistent activity, thus making this protocol economically and environmentally more acceptable.
Experimental
All reagents were used as received without further purification. The MCM-41-PPh2-AuOTf, MCM-41-PPh2-AuNTf2, MCM-41-PPh2-AuSbF6, and MCM-41-PPh2-AuBF4 complexes were prepared according to our previous procedure. 44 The gold contents were determined to be 0.37, 0.35, 0.36, and 0.38 mmol g−1, respectively, based on inductively coupled plasma atomic emission spectroscopy (ICP-AES) analysis (Atomscan 16; TJA Corporation). All the α-aryl-α-diazoesters were prepared according to the literature method. 32 Dichloromethane was dried over P2O5 and distilled before use. All reactions were carried out under Ar in oven-dried glassware with magnetic stirring. 1H NMR spectra were recorded on a Bruker Avance 400 (400 MHz) spectrometer with tetramethylsilane (TMS) as an internal standard using CDCl3 as the solvent. 13C NMR spectra were recorded on a Bruker Avance 400 (100 MHz) spectrometer using CDCl3 as the solvent. We have identified all the products by comparing their spectroscopic and physical data with that available for the products in the Supplementary Information provided by Ma et al. 32
Heterogeneous gold(I)-catalyzed C(sp2)–H bond alkylation of benzene derivatives with α-aryl-α-diazoesters: general procedure
A 10 mL Schlenk tube was charged with MCM-41-PPh2-AuOTf (54 mg, 5 mol%) and arene
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank the National Natural Science Foundation of China (No. 21462021), the Natural Science Foundation of Jiangxi Province of China (No. 20161BAB203086), and the Key Laboratory of Functional Small Organic Molecules, Ministry of Education (No. KLFS-KF-201704) for financial support.






