Abstract
Under Karstedt’s catalyst, an organic–inorganic hybrid fluorescent nano-molecular dendrimer has been synthesized by grafting silafluorene units onto the polyhedral oligomeric silsesquioxane core through hydrosilylation reaction. The new hybrid molecule exhibits high solid fluorescence quantum efficiency because of the nano-size and large steric hindrance of the polyhedral oligomeric silsesquioxane core. Thermogravimetric analysis shows that the thermal stability of the target compound is effectively improved by the presence of polyhedral oligomeric silsesquioxane.
Keywords
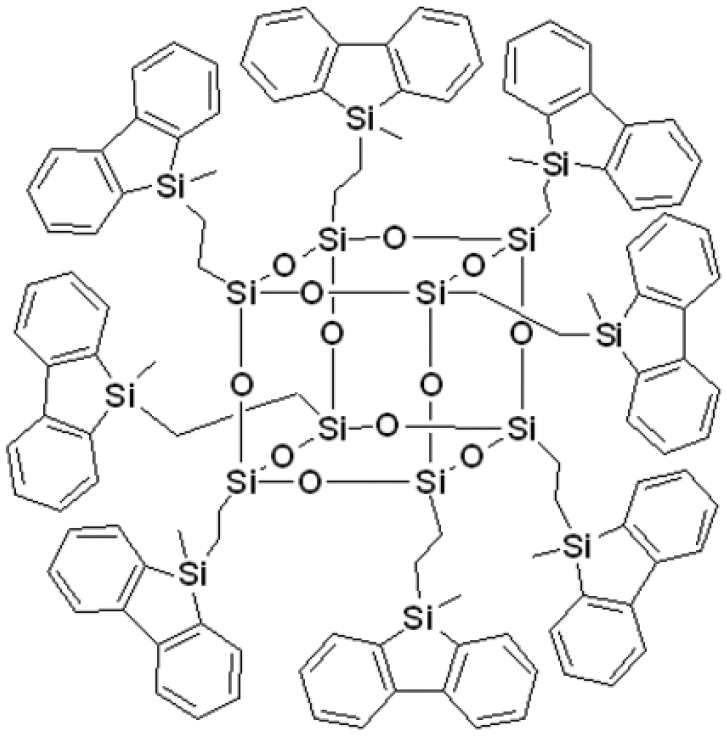
The organic–inorganic hybrid nano-molecular dendrimer based on polyhedral oligomeric silsesquioxane core exhibits high solid fluorescence quantum efficiency and the improved thermal stability
Introduction
Silicon-containing π-conjugated compounds are attractive and interesting molecules due to their excellent photo-electrical properties and thermal stability.1–5 As the most typical representatives, silacyclopentadienes (siloles) have gained much attention as a new class of optoelectro-active materials with aggregation-induced emission (AIE) and/or good electron transport properties in organo-electronics.6–15 Siloles have a relatively low-lying lowest unoccupied molecular orbital (LUMO) level due to the π*–σ* conjugation between the σ* orbital of the exocyclic Si–C bond and the π*orbital of the butadiene fragment, resulting in a high electron affinity. Moreover, silafluorene-containing compounds,16–20 which are the analogue of fluorene, integrate the silacyclopentadiene character into the structure of fluorene; the introduction of a silicon atom in the 9-position of fluorene could retain the fluorescent properties of fluorene, such as the strong blue emission. The combination of the electrical effect of the silicon element with a silicon-bridge structure could tune the opto-electrical properties, and even improve the quantum yield of fluorescence and the thermal stability. Silafluorene derivatives are widely applied in the field of OLED, PSC, OFET and so on,21–23 by using their unique electronic structure and photoelectric properties. However, the tendency for crystallization, the solid fluorescent yield and thermal properties are also important issues in developing and exploiting silafluorene-containing compounds.
Polyhedral oligomeric silsesquioxane (POSS) possesses a cage-like nanometer-sized structure and demonstrates high thermal stability and facile chemical modification.24–27 In particular, the highly symmetrical and topologically ideal cube octasilsesquioxae (T8-POSS) with a cage size of approximately 0.5–0.7 nm has drawn intensive attention as a constructive unit for fabrication of various functional organic–inorganic hybrid materials. Design and preparation of new hybrid optoelectronic materials through chromophore units covalently bounded to POSS has been an active research topic. Many POSS-based fluorescent materials have been developed by combination of POSS and various chromophores in different modes.28–33 For example, attached mono-functionalized POSS as pendant groups onto conjugated polymers 28 incorporated multi-functionalized POSS as a core in conjugated dendrimers 29 and hyperbranched polymers. 30 We have also developed the POSS-based organic–inorganic hybrid AIEgens. 34 , 35 They exhibit outstanding thermal stability, improved quantum efficiencies and excellent fluorescent detection of explosives and metal ions compared with their parent molecules without the POSS unit.
There are reports about silafluorene and tetraphenylsilole groups constructed in the oligo-siloxane chains, which show interesting photophysical properties. 36 , 37 In this paper, under Karstedt’s catalyst, an organic–inorganic hybrid fluorescent nano-molecular dendrimer has been synthesized by grafting silafluorene units onto the octavinylsilsesquioxane (octavinyl-POSS) core through hydrosilylation reaction. The new hybrid molecule exhibits high solid fluorescence quantum efficiency because of the nano-size and large steric hindrance of the POSS core. Thermogravimetric analysis (TGA) shows that the thermal stability of the target compound can be effectively improved by the existence of POSS. The incorporation of POSS and fluorescent units may provide a new method or route to improving the fluorescent quantum efficiency and thermal stability of the organic fluorescent molecules.
Results and discussion
Synthesis and characterization
As shown in Scheme 1, octavinylsilsesquioxane (

The synthetic route to the target compound
The hydrosilylation reaction results were confirmed by the nuclear magnetic resonance (NMR) analyses. Figure 1 shows the 1H spectra of

1
H NMR spectra of (a) POSS

13
C NMR spectra of (a) POSS
Thermal properties
The thermal stability of

TGA curves of compound
Photophysical properties
The UV-vis absorption spectra of

UV-vis spectra of compound
The photoluminescence (PL) spectra of
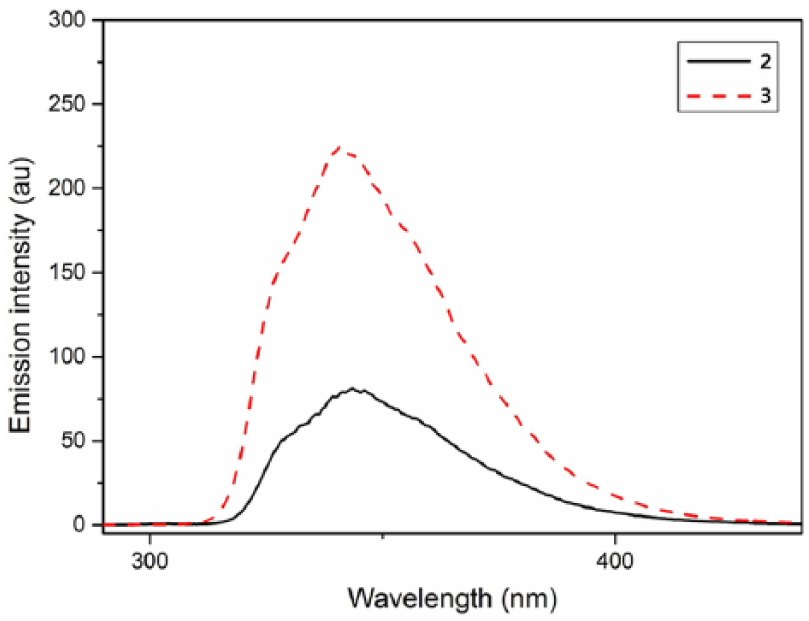
PL spectra of compound
To have a quantitative comparison, we measured the fluorescence quantum yields of the molecule and hybrid dendrimer in solution and solid state. The nano-size POSS core can improve the fluorescence quantum yield (ΦF) of chromophore both in solution and in the solid state. Although the ΦF values of POSS in THF are low, the ΦF value of the
Conclusion
The organic–inorganic hybrid fluorescent nano-molecular dendrimer (
Experimental section
Materials and instruments
Unless otherwise mentioned, materials were obtained from commercial suppliers and used without further purification. The key starting compounds
1 H and 13C NMR data were recorded on a Bruker Avance III 400 HD spectrophotometer using CDCl3 as solvent. 29Si NMR was recorded on a Bruker DMX 300 spectrophotometer. Chemical shifts are referenced to the solvent peak (for CDCl3, 1H NMR: 7.26 ppm, 13C NMR: 77.0 ppm) and tetramethylsilane (TMS) (29Si). The UV-vis and fluorescence spectra were recorded with a Shimadzu UV-2600 spectrophotometer and a Hitachi F-4500 spectrofluorophotometer, respectively. TGA was carried on a Hitachi STA 7300 instrument under N2 or air at a heating rate of 10 °C min−1. Differential scanning calorimetry (DSC) was carried out on a Seiko EXSTAR DSC6220 under N2 at a heating rate of 10 °C min−1. Absolute fluorescence quantum yields were tested on a Hamamatsu C11347 Quantaurus-QY instrument with a calibrated integrating sphere. High-resolution mass spectra (HRMS) were recorded with a Bruker SolariX 9.4T spectrometer operating in MALDI-FT mode.
Preparation of target compound 3
Under N2, to a THF (10 mL), solution of
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
