Abstract
Background:
Heterozygous HTRA1 mutations are the second most common cause of monogenic dominant cerebral small vessel disease (HTRA1-AD-cSVD or CADASIL2), after cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) due to NOTCH3 cysteine-altering mutations. However, there have been few studies of cohorts of HTRA1-AD-cSVD and whether it can be differentiated clinically and on neuroimaging from CADASIL is unclear.
Aims:
This retrospective study aims to characterize and compare the clinical and neuroimaging features of HTRA1-AD-cSVD with those of CADASIL.
Methods:
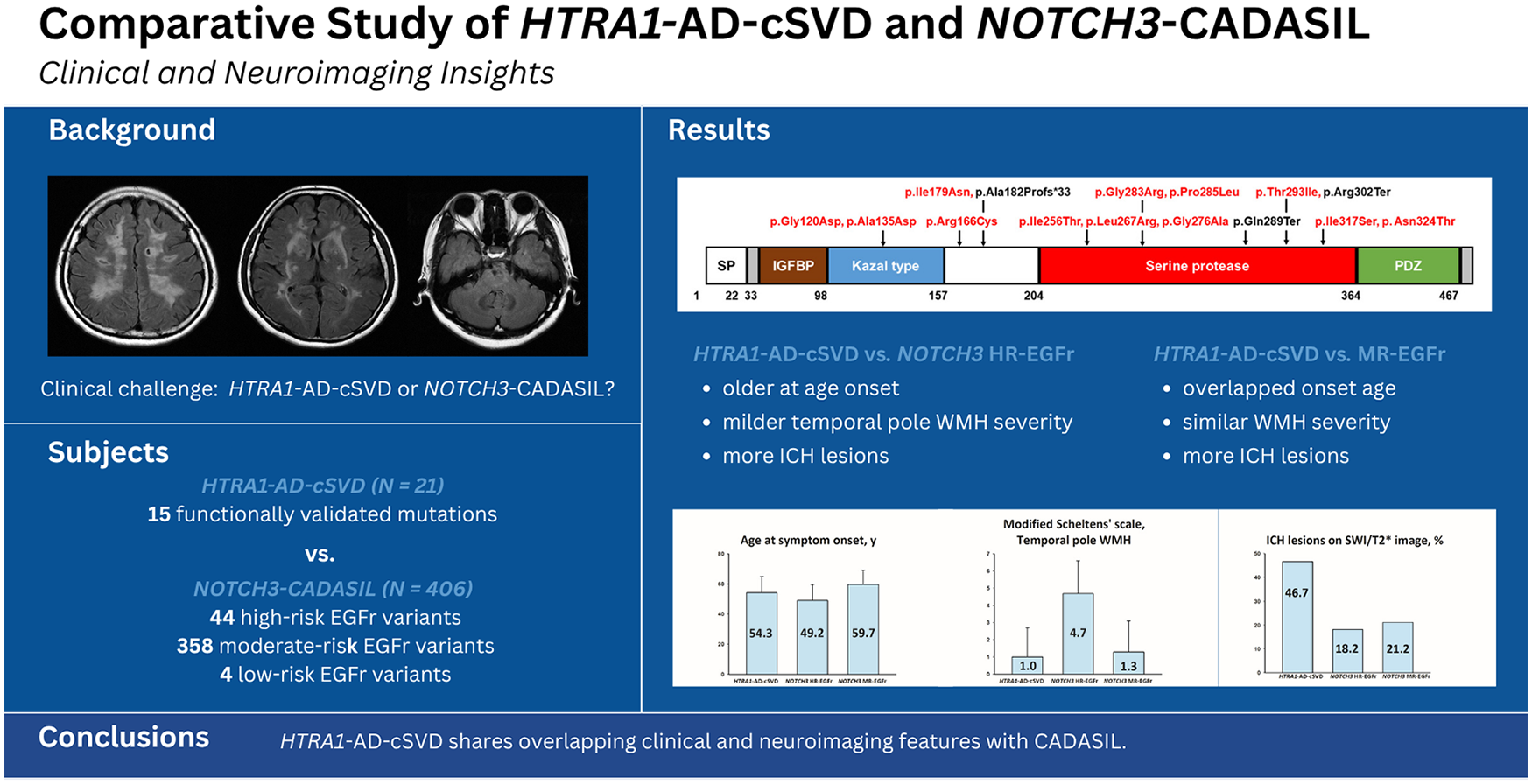
We identified 21 unrelated Taiwanese subjects carrying 15 heterozygous HTRA1 variants, all functionally validated as pathogenic through in vitro protease activity assays. HTRA1-AD-cSVD patients were compared with 406 CADASIL patients, including 44 cases carrying NOTCH3 mutations within the high-risk epidermal growth factor-like repeat domains (EGFr), 358 with moderate-risk EGFr mutations, and 4 with low-risk EGFr mutations. Multivariate regression analyses were conducted with adjustments for age at MRI examination and hypertension.
Results:
Stroke occurred in 81.0% of HTRA1-AD-cSVD patients, and 47.6% exhibited cognitive dysfunction. MRI revealed moderate-to-severe white matter hyperintensity (WMH) in the deep white matter and external capsule (modified Scheltens’ scale: 5.3 ± 1.0 and 4.1 ± 1.7), mild WMH in the temporal pole (1.0 ± 1.7), lacunes in 90.5%, ⩾10 cerebral microbleeds (CMBs) in 66.7%, and intracranial hemorrhage (ICH) lesions in 46.7%, indicating susceptibility to both ischemic and hemorrhagic strokes. Patients with HTRA1 loss-of-function mutations or protease domain missense mutations exhibited a higher prevalence of ⩾10 CMBs on SWI/T2* imaging (100% and 83.3%) compared to those with missense mutations outside this domain (20%). Symptom onset occurred earliest in patients with NOTCH3 high-risk EGFr mutations (49.2 ± 10.5 years), followed by those with heterozygous HTRA1 mutations (54.3 ± 10.7 years), and latest in NOTCH3 moderate-risk EGFr mutations carriers (59.7 ± 9.5 years). Temporal pole involvement was most prevalent in NOTCH3 high-risk EGFr mutations (88.6%), followed by NOTCH3 moderate-risk EGFr mutations (32.4%), and least common in heterozygous HTRA1 mutations (28.6%). Even after adjusting for age and hypertension, HTRA1-AD-cSVD patients exhibited significantly milder temporal pole WMH severity compared to NOTCH3 high-risk EGFr mutation carriers (adjusted p < 0.001). In addition, ICH lesions were more frequently observed in HTRA1-AD-cSVD patients (46.7%) than in patients with NOTCH3 high-risk or moderate-risk EGFr mutations (18.2% and 21.2%), although the difference was not statistically significant.
Conclusion:
HTRA1-AD-cSVD shares overlapping clinical and neuroimaging features with CADASIL. Temporal pole WMH involvement can occur in HTRA1-AD-cSVD but is more common in CADASIL. The high prevalence of ICH in HTRA1-AD-cSVD has been under-recognized.
Data access statement:
Data are available upon reasonable request from third parties.
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
