Abstract
Cerebral microinfarcts (CMIs) are small ischemic lesions invisible to the naked eye at brain autopsy, while the larger ones (0.5–4 mm in diameter) have been visualized in-vivo on magnetic resonance imaging (MRI). CMIs can be detected on diffusion-weighted imaging (DWI) as incidental small DWI-positive lesions (ISDPLs) and on structural MRI for those confined to the cortex and in the chronic phase. ISDPLs may evolve into old cortical-CMIs, white matter hyperintensities or disappear depending on their location and size. Novel techniques in neuropathology and neuroimaging facilitate the detection of CMIs, which promotes understanding of these lesions. CMIs have heterogeneous causes, involving both cerebral small- and large-vessel disease as well as heart diseases such as atrial fibrillation and congestive heart failure. The underlying mechanisms incorporate vascular remodeling, inflammation, blood–brain barrier leakage, penetrating venule congestion, cerebral hypoperfusion, and microembolism. CMIs lead to clinical outcomes, including cognitive decline, a higher risk of stroke and mortality, and accelerated neurobehavioral disturbances. It has been suggested that CMIs can impair brain function and connectivity beyond the microinfarct core and are also associated with perilesional and global cortical atrophy. This review aims to summarize recent progress in studies involving both cortical-CMIs and ISDPLs since 2017, including their detection, etiology, risk factors, MRI correlates, and clinical consequences.
Introduction
Cerebral microinfarcts (CMIs) are considered the most widespread form of brain infarction invisible to naked eyes on gross pathological examination. 1 They are commonly found in aging brains with a prevalence of 24% in older adults without dementia, compared to 43% in patients with Alzheimer’s disease and 62% in those with vascular dementia. 2
Although most CMIs remain invisible on magnetic resonance imaging (MRI), larger CMIs (0.5–4 mm) can be detected in the cortex using structural MRI (i.e. cortical-CMIs) and as hyperintense lesions on diffusion-weighted imaging (DWI) throughout the brain (i.e. incidental small DWI-positive lesions (ISDPLs)). 3 “ISDPLs” is a very recent terminology which has been recommended by Standards for Reporting Vascular Changes on Neuroimaging 2 (STRIVE-2) to describe acute small infarction on DWI, comprising both small subcortical and tiny cortical lesions. 3 These lesions may evolve into old cortical-CMIs, white matter hyperintensities (WMHs) or disappear on subsequent MRI scans. 3 ISDPLs in the cortex are expected to share the same etiology, risk factors, and clinical outcomes with old cortical-CMIs and are considered a different phase of the disease.
Since the last major review of CMIs in 2017, 4 significant progress has been made in this research field. The visualization of cortical-CMIs with lower-resolution (i.e. 1.5 T) MRI has greatly facilitated their study in diverse populations. 5 Moreover, the utility of 3D double-inversion recovery (DIR) imaging 6 and iron-sensitive MRI sequences 7 has been shown to improve the accuracy and detection rate of cortical-CMIs. Recent studies have shown that cortical-CMIs are associated with cognitive decline, 8 increased risks of death and stroke, 5 and accelerated deterioration of neurobehavioral disturbances. 9
This review mainly focuses on cortical-CMIs by summarizing recent advances in their detection, etiology, risk factors, MRI correlates, and clinical consequences.
Detection of CMIs
Neuropathological assessment is considered the reference standard of identifying CMIs, due to its high specificity in detecting both acute and chronic lesions, encompassing cortical and subcortical regions. Although CMIs have been visualized in-vivo, the relatively low sensitivity of detecting CMIs on MRI remains a concern. However, recent advances in neuropathological and neuroimaging techniques can provide more reliable strategies for CMI detection.
Neuropathological examination
On histological sections using standard hematoxylin and eosin (H&E) staining, CMIs are characterized by focal lesions with neuronal cell loss and other features such as macrophages or gliosis depending on the timepoint of injury (Figure 1). 4 The appearance of hemorrhagic components (e.g. hemosiderin-laden macrophages) in the location may indicate a hemorrhagic transformation. 10 Several studies have reported a varied prevalence of CMIs in different regions of the brain which may be due to the sampling strategy used in neuropathological analysis.11,12
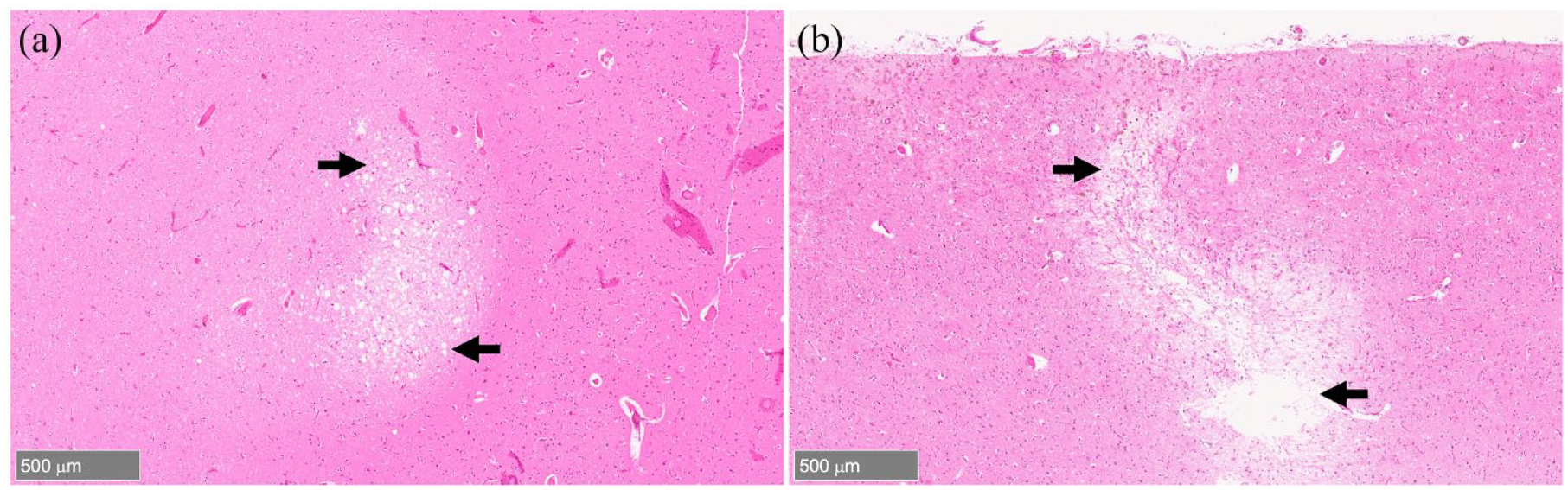
Cerebral microinfarcts on neuropathological examination. (A) An acute cerebral microinfarct (black arrows) with neuronal cell injury and no cell loss on histological sections using hematoxylin and eosin staining. (B) A chronic cerebral microinfarct (black arrows) characterized by tissue cavitation and puckering. (Image courtesy: Dr Susanne J van Veluw, Massachusetts General Hospital).
Neuropathological examination is limited in its ability to detect the true burden of CMIs in the brain due to the small proportion of brain tissue examined and the limitations of traditional H&E staining in detecting all types of CMI lesions.11,13 Recent developments in histological methods such as the use of Nissl stain and Collagen 4-immunohistochemistry have improved the ability to identify CMIs which are undetectable with H&E staining. 14 Similarly, the development of two histological protocols for thick sections has elevated the detection rate of CMIs by detecting lipofuscin granules and macrophages in CMI location, as well as better visualization of abnormalities within the capillary network in relation to CMIs. 15
High resolution and conventional structural MRI
Currently, structural MRI can reliably detect cortical-CMIs. In rating criteria from previous literatures,16,17 cortical-CMIs are hypointense lesions on T1-weighted images, hyperintense or isointense (with the surrounding tissue) on fluid-attenuated inversion recovery (FLAIR) sequences, hyperintense on T2-weighted images, <4 mm in diameter, restricted to the cortex, and perpendicular to the cortical surface (Figure 2).
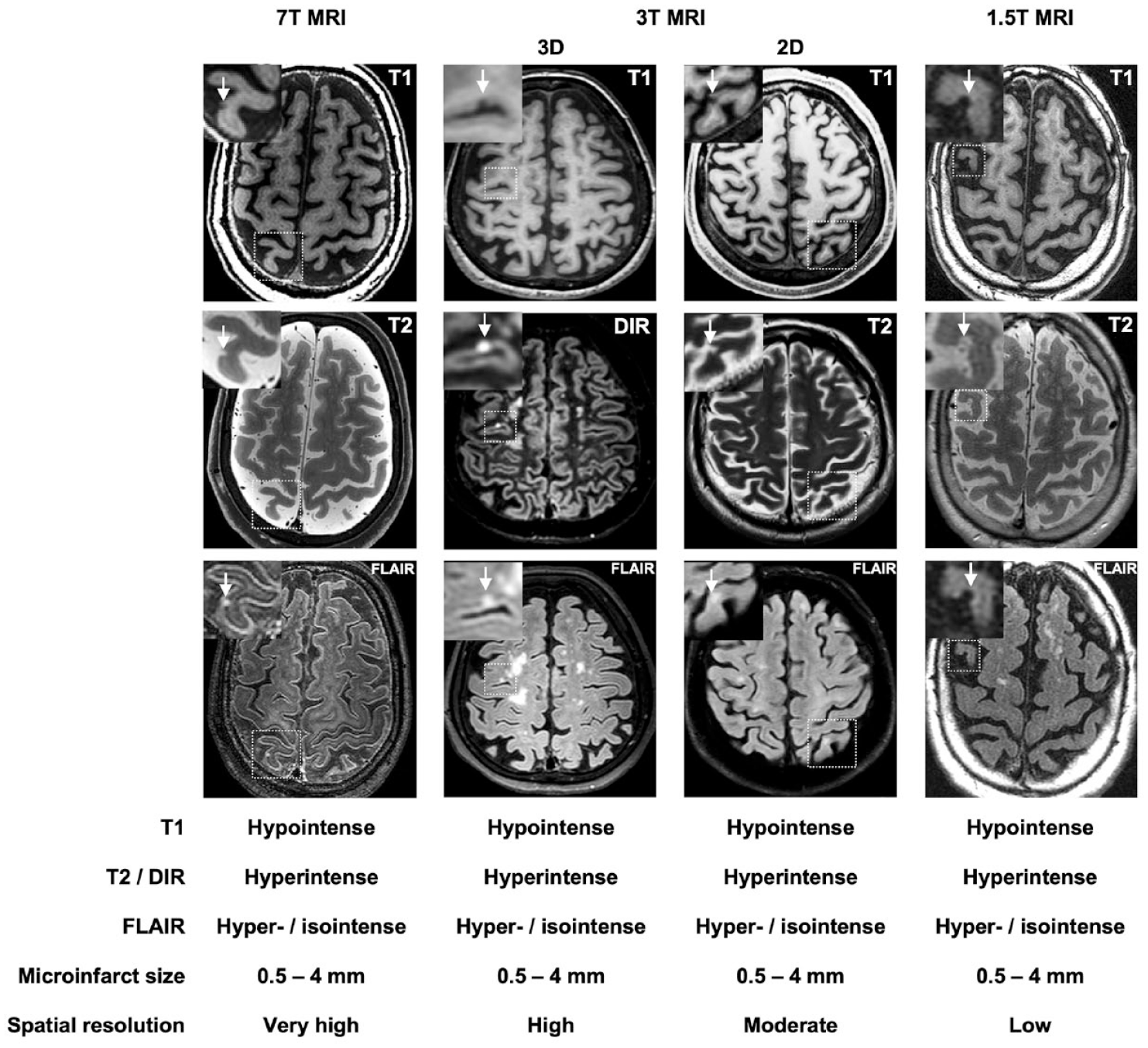
Cortical cerebral microinfarcts on structural MRI. T1 = T1-weighted MRI; T2 = T2-weighted MRI; DIR = Double inversion recovery; FLAIR = Fluid-attenuated inversion recovery. (Image courtesy for 7T MRI images: Dr Alberto de Luca, UMC Utrecht).
Cortical-CMIs were first detected in-vivo using 3D-DIR and 3D-FLAIR images on 3 T MRI, 18 and subsequently reported on high-resolution 7 T MRI with histopathologic verification as microinfarction. 19 In recent years, cortical-CMIs have been visualized on lower-resolution 1.5 T MRI platforms with more widespread availability, which facilitates their study in diverse populations. 5 Although few studies published head-to-head comparison of cortical-CMI detection between high and low field strength MRI in the same subjects, overall, lower field strength studies reported lower prevalence rates of cortical-CMIs, due to the comparatively lower sensitivity. 20 Studies performed on 3 T MRI showed a prevalence of cortical-CMIs at 6.3–11.5% in population-based cohorts, 20% in patients with vascular cognitive impairment, and 57.1% in patients with cerebral amyloid angiopathy (CAA), 21 while a study using 1.5 T MRI found a 10.5% prevalence of cortical-CMIs in a population-based setting. 5 However, even 7 T MRI cannot fully detect the whole-brain burden of cortical-CMIs as it is estimated that only less than 1% of total lesions can be detected with MRI. 7 Hence, the detection of a single cortical-CMI may indicate the presence of hundreds to thousands more in the rest of the brain.22,23
Cortical-CMIs are reported to have a predilection for the parietal lobe (59%), followed by frontal lobe (48%), and are least observed in occipital (11.4%) and temporal lobes (5.2%). 5 However, some studies have reported the predilection of cortical-CMIs in watershed areas,24–26 while others reported no regional predilection throughout the cortex.21,27 These controversial findings may be due to differences in study populations, sample size, and rating criteria.
Novel neuroimaging strategies to detect cortical-CMIs
Conventional structural MRI has limitations in detecting small cortical-CMIs, as traditional MRI techniques can only detect lesions greater than 1 mm in size. 10 Therefore, many studies have applied new neuroimaging strategies such as DIR and 3D-MRI to improve the detection rate of cortical-CMIs in-vivo. DIR images have been reported to have better intracortical localization than conventional MRI, 28 and 3D-MRI is superior to 2D-MRI in terms of spatial resolution. 29 Therefore, more and more studies have used 3D-DIR combined with 3D-FLAIR images in detecting cortical-CMIs instead of conventional 2D-MRI.6,10,30–32 However, the longer scan time required for 3D-MRI scan has restricted its use in clinical practice.
Iron accumulation in cortical-CMI lesions can increase their detectability on MRI, especially with iron-sensitive MRI sequences such as susceptibility weighted imaging (SWI) or T2*-weighted images. 14 Some cortical-CMIs that are undetectable on T1- and T2-weighted images may be visible on these sequences due to iron accumulation in the microinfarct core. 7 Therefore, SWI or T2*-weighed images can provide complementary information for detecting cortical-CMIs beyond T1- and T2-weighed images and improve the detection rate. 7
These findings demonstrate the potential of new neuroimaging strategies to detect more cortical-CMIs in-vivo and emphasize the need to develop techniques with higher sensitivity and specificity. Moreover, visual ratings of cortical-CMIs on MRI are time-intensive and operator-dependent, making automated methods for detection necessary to support large-scale studies.
Identification and evolution of ISDPLs
ISDPLs are small hyperintense lesions on DWI, with corresponding hypointense or isointense signals on apparent diffusion coefficient maps. 3 ISDPLs overlap with recent small subcortical infarcts and acute cortical microinfarction (<5 mm in diameter) 3 and can be mostly observed in subcortical white matter, followed by cortical gray matter and then the deep gray matter. 33
Although DWI has high sensitivity in detecting ISDPLs of 1–2 mm in diameter, 34 it is unable to capture the entire burden of such lesions in the brain. However, submillimeter DWI has shown promise in detecting smaller lesions that are invisible on conventional DWI. 35 Moreover, detecting a single ISDPL on DWI may suggest an annual incidence of several hundred new lesions in the brain. 36
ISDPLs typically disappear on DWI within 2 weeks and may evolve into other chronic lesions or disappear on follow-up FLAIR or T2-weighted images. 37 A previous study has reported that 58.8% of ISDPLs appeared as WMHs on follow-up MRI, 17.6% developed cavitation, 5.9% became old cortical-CMIs, and 17.6% disappeared after a mean follow-up of 4.7 years. 33 A possible reason for disappearance of these lesions on follow-up imaging might be due to salvageable tissue which can undergo tissue repair and lead to their disappearance. 38 Besides, it has been reported that the lesions visible on follow-up imaging tend to be larger initially and the smaller lesions tend to disappear or become undetectable due to limited image resolution. 39 Moreover, another study has shown that some lesions invisible on follow-up T2-weighted images appeared as hypointense signals on SWI sequences, indicating a probable hemorrhagic transformation. 7
ISDPLs are more commonly observed in patients with acute intracerebral hemorrhage (ICH) compared to the general population 40 and were indeed shown to be acute microinfarction on histology in CAA-related ICH. 41 ISDPLs following acute ICH are associated with microangiopathy such as WMH and cerebral microbleeds (CMBs) as well as ICH characteristics such as hematoma volumes.42,43 Based on current findings, it is likely that ISDPLs after ICH are primarily a consequence of the progression of cerebral small vessel disease (CSVD) and are influenced by secondary risk factors associated with ICH, such as intensive blood pressure reduction, cerebral blood flow (CBF) dysregulation, inflammation, and blood–brain barrier leakage.42,43 Further investigations are necessary to elucidate the intricate interactions among these mechanisms in the development of ISDPLs.
Risk factors of cortical-CMIs
The association of cortical-CMIs with traditional cardiovascular risk factors (e.g. hypertension, hyperlipidemia, and diabetes) has been widely studied in recent years, but the results have been inconsistent. 20 Many studies have demonstrated that the presence of cortical-CMIs was associated with hypertension44,45 or hyperlipidemia5,46 or both.25,47,48 By contrast, some studies showed no relationship between cortical-CMIs and cardiovascular risk factors.20,26,49 For example, one study investigating the risk factors of cortical-CMIs on 7 T MRI reported that the risk of cortical-CMIs did not change with the presence of hypertension, hyperlipidemia, or diabetes. 20 Moreover, only a few animal experiments have shown the temporal relationship between cardiovascular risk factors and incident cortical-CMIs, 50 which, however, has not been verified in human studies yet.
MRI correlates of cortical-CMIs
Many studies have shown that the presence of cortical-CMIs was associated with MRI markers of CSVD such as lacunes,5,6,9,51 WMH,9,25,26,52,53 CMBs,9,25,31,48,53 and brain atrophy,5,6,25,27,44,53 indicating a shared etiology such as CAA and arteriolosclerosis. It has been reported that CAA is highly related to the occurrence of CSVD including lobar lacunes,54,55 WMH, 56 CMBs, 10 and cortical-CMIs, 57 while arteriolosclerosis also contributes to the pathogenesis of CSVD albeit in a different manner. 58 Moreover, a study found that the presence of cortical-CMIs was significantly associated with total MRI burden of CSVD in CAA indicating more severe changes in CAA-related vascular pathology. 32 In comparison to cortical-CMIs due to microemboli, cortical-CMIs due to CAA on MRI were shown localized within the cortex, smaller in size, lower in lesion counts, and predominantly in the occipital lobe. 30
In addition, an association between cortical-CMIs and cerebral macroinfarcts on MRI has been confirmed based on different study populations, including patients with heart failure (HF), 44 carotid artery stenosis, 6 and memory clinic patients.25,26,51 Previous studies have also found that the presence of cerebral large-vessel disease such as internal carotid artery (ICA) occlusion, 48 carotid artery stenosis (>30%), 6 and middle cerebral artery (MCA) stenosis (>50%) 46 was associated with cortical-CMIs.
In addition to the relationship between cortical-CMIs and other ischemic cerebrovascular disease (e.g. lacunar and non-lacunar infarcts, WMH) which suggests an ischemic origin, the association between cortical-CMIs and CMBs in patients with CAA may indicate a shared etiology with different mechanisms. A previous study combined neuropathological examination with postmortem MRI to detect both cortical-CMIs and CMBs in 12 CAA cases and the different features of cortical-CMIs and CMBs by location, pathological changes, and timepoint of occurrence suggested that there may be different mechanisms involved in their development and impact on the brain, 59 which needs further investigation.
Clinical relevance of cortical-CMIs
Cortical-CMIs and cognitive dysfunction
Previous studies have shown an association between cortical-CMIs and cognitive impairment using cross-sectional data.16,17 Limited data have also emerged using longitudinal follow-up and demonstrated that the increased number of cortical-CMIs predicted cognitive decline in memory clinic patients. 8 It was further shown that patients with increased cortical-CMIs declined on average by 2 points on Montreal Cognitive Assessment (MoCA) and 0.5 on global cognition scores per year, corresponding to around 110% and 185% greater decline compared to patients without cortical-CMIs. 8 This finding is corroborated by another study in a post-stroke population. 45
A recent study has suggested that the combined presence of cortical-CMIs and retinal pathology (reflected by increased retinal venular tortuosity or reduced ganglion cell-inner plexiform layer thickness) may have a synergistic effect on cognitive impairment highlighting the importance of considering both cerebral and retinal microvascular health in the assessment of cognitive dysfunction. 60
Cortical-CMIs and other adverse clinical outcomes
Apart from cognitive impairment, cortical-CMIs are also associated with other adverse clinical consequences. A recent study involving 2156 participants from a population-based study showed that participants with cortical-CMIs had 18% increased risk of stroke and 9% increased risk of mortality compared to those without cortical-CMIs, independent of demographics, vascular risk factors, and other MRI markers. 5 Similarly, another study of 414 patients with acute ischemic stroke showed that patients with cortical-CMIs were about 1.5 times more likely to suffer from functional dependence, stroke recurrence, or death at 90 days, compared to those without cortical-CMIs. 49
Moreover, a study involving 496 memory clinic patients found that the presence of cortical-CMIs was associated with a higher incidence of neurobehavioral disturbances at baseline. 9 Patients with multiple cortical-CMIs (⩾2) also showed accelerated deterioration of neurobehavioral disturbances, reflected by total neuropsychiatric scale and hyperactivity subsyndrome scores. 9
Pathogenesis and remote effects of cortical-CMIs
The heterogeneous causes and risk factors of cortical-CMIs lead to a series of changes in vascular pathology and hemodynamics, ultimately resulting in microinfarct lesions via mechanisms such as cerebral hypoperfusion and microembolism. Cortical-CMIs can cause clinical manifestations through both local and remote brain tissue damage (Figure 3).
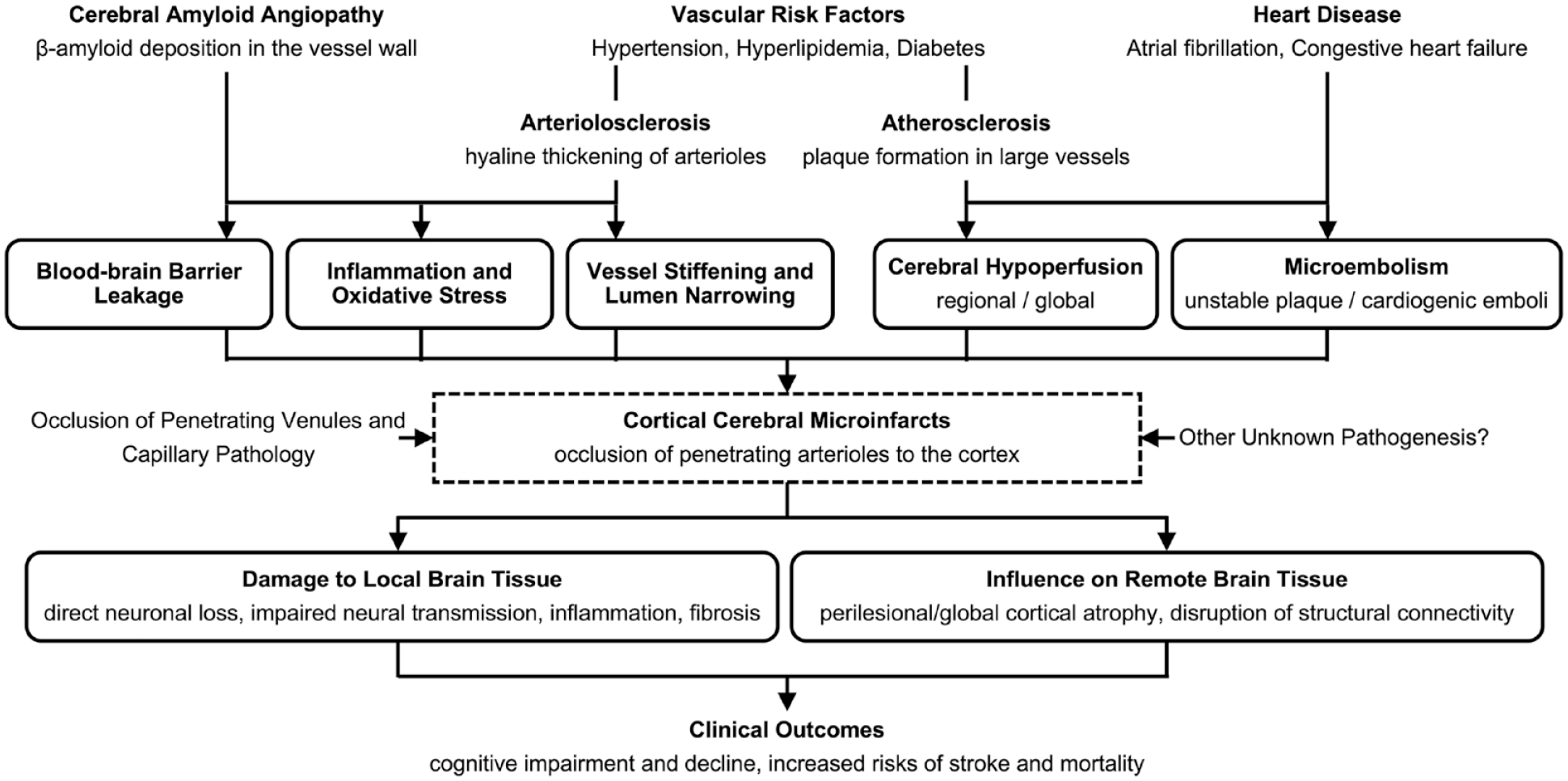
Risk factors, etiology, and clinical relevance of cortical cerebral microinfarcts. Heterogeneous causes and vascular risk factors lead to a series of changes in vascular pathology and hemodynamics resulting in cortical-CMIs via mechanisms such as cerebral hypoperfusion and microembolism. Cortical-CMIs can generate clinical manifestations via influences on both local and remote brain tissue.
CSVD
Pathological changes in cerebral small vessels (e.g. CAA and arteriolosclerosis) have been proved to be associated with the occurrence of cortical-CMIs.22,61 The incidence of cortical-CMIs in individuals with moderate-to-severe CAA or arteriolosclerosis was reported to be about twice the incidence in the healthy elderly. 62 Furthermore, an increased severity of CAA can increase the odds of having cortical-CMIs by 13%, and arteriolosclerosis increases the odds by 20%. 57 Both CAA and arteriolosclerosis can cause direct lumen narrowing or impairment of CBF regulation via impact on normal vessel function, leading to decreased CBF to the cortex and consequently ischemic tissue damage. 21 The perforating end-arteries, frequently involved in cortical-CMIs, are more susceptible to ischemic injury caused by decreased CBF. 59 Arteriolosclerotic vasculopathy or β-amyloid deposition can also activate a cascade of pathological changes including vascular remodeling, blood–brain barrier leakage, inflammation, oxidative stress, and hypercoagulable state, all of which participate in the pathogenesis of cortical-CMIs.22,62–64
Cerebral hypoperfusion and microembolism
The association of cortical-CMIs with cerebral hypoperfusion has been confirmed in studies on patients with cerebral large-vessel disease. In a previous study involving patients with ICA occlusion, patients with unilateral ICA occlusion and cortical-CMIs showed 12% lower global CBF compared to those without cortical-CMIs. 48
Cortical-CMIs may be related to reduced CBF in the whole brain rather than that around the microinfarct core. It was found that although patients with cortical-CMIs had a 12% lower global CBF and 22% higher arterial transit time than those without cortical-CMIs, no difference was observed between CBF in a region of 2 cm around the microinfarct core and CBF in a reference area in the contralateral hemisphere. 25 This finding was also demonstrated by another study in patients with heart disease, which showed that patients with congestive HF had twice the risk of cortical-CMIs compared to those with normal cardiac function. 47
However, cardiac microemboli may also be a likely cause of cortical-CMIs, as thrombotic implications are common in HF patients. 65 One study involving patients with HF further investigated the role of cerebral hypoperfusion in the relationship between impaired cardiac function and cortical-CMIs and concluded that CBF did not mediate this relationship. 44 It was also reported that patients with elevated P-wave terminal force in lead V1 (PTFV1), an indicator of left atrial abnormalities, had higher burden of cortical-CMIs. 66 Abnormal PTFV1 has been verified to be related to embolic stroke. 67 These findings point to microemboli from atrial dysfunction as another possible pathogenesis of cortical-CMIs.
A probable source of microemboli is the heart, as several studies have confirmed the association between cortical-CMIs and atrial fibrillation,12,22,47,49 which is known as an important risk factor for cardioembolism. Another reason may be large-artery atherosclerosis, as it has been reported that the unstable plaques in cerebral arteries are important sources of microemboli to the cortex.6,48,68 Cerebral hypoperfusion can also cause microembolism via washout disturbances in arterioles and/or venules with insufficient perfusion. 48
Venous and capillary pathology
Penetrating venule occlusion is considered a possible cause of cortical-CMIs, as previous studies have suggested that some cortical-CMIs are located near draining venules and are associated with local increase in venule diameter, indicating potential venous congestion or obstruction.69–71 Occlusion of penetrating venules will lead to stagnation of blood flow in corresponding penetrating arterioles, and thus result in oxygen shortage in the affected cortex and finally ischemic tissue injury. 70 Considering the ratio of penetrating arterioles to venules (4-5:1 for human), it is conceivable that occlusion of one penetrating venule will cause a much bigger CMI lesion than that caused by occlusion of one penetrating arteriole. It is important to note that these findings are based on animal experiments, and it is still difficult to detect and confirm the occlusion of penetrating venules as a cause of cortical-CMIs in clinical practice. It has also been suggested that capillary pathology might play a role in the development of cortical-CMIs. 72 Further studies are needed to fully understand the role of penetrating venule occlusion and capillary pathology in the occurrence of cortical-CMIs.
Effects of cortical-CMIs on surrounding and remote brain tissue
As the most widespread form of infarction throughout the brain, 39 the presence of cortical-CMIs can cause both direct neuronal loss within the microinfarct core and disruption of neuronal function and connectivity surrounding and beyond the microinfarct core, leading to clinical manifestations. 27
Although a single cortical-CMI indicates hundreds to thousands of such lesions in the brain, the total volume of all these lesions still does not exceed 1–2 ml. 71 Therefore, it is conceivable that cortical-CMIs impair normal function of the brain via their impact on surrounding and remote tissue5,23 through the following mechanisms: first, the molecular organization of survival axons around the microinfarct core is abnormal, and the functional basis for neural transmission is lost. 73 These microstructural injuries of surrounding tissue will interrupt cortico-cortical and cortico-subcortical circuits.8,45 Second, cortical-CMIs are reported to influence cognition via disrupting brain structural connectivity.74,75 Third, cortical-CMIs have been found to impact a perilesional area up to 150 times greater than the microinfarct core and perilesional cortical atrophy has been observed at least 20 mm away from the microinfarct core. 53 Furthermore, the presence of cortical-CMIs was reported to cause remote cortical atrophy over 2 years of follow-up. 23
Conclusion and future directions
Substantial developments have been made in the CMI research in recent years. New histological techniques have been developed that allow for a more detailed examination of the pathological changes in microinfarcts. The advances in neuroimaging techniques have greatly improved the ability to detect CMIs in the brain non-invasively, providing a more complete understanding of the true burden of these lesions. Visualization of cortical-CMIs on lower-resolution MRI facilitates more in-depth studies on the risk factors and clinical relevance of such lesions in diverse study populations. Heterogeneous causes and risk factors can lead to cortical-CMIs via mechanisms like cerebral hypoperfusion and microembolism. The association of cortical-CMIs with brain structural dysconnectivity and cortical atrophy might be underlying mechanisms of the clinical consequences caused by such lesions.
Further development in the field of CMI research may depend on semi-automated CMI detection. Efforts are underway in developing a semi-automated machine-learning algorithm, for accurate and time-efficient detection of cortical-CMIs on 3 T MRI. Despite the advances in our knowledge about CMIs, there are still some controversies requiring further investigation. First, most previous studies used cross-sectional settings and a causality was difficult to infer. Second, the mechanisms of how the existence of CMIs impairs neuronal function and structure beyond the microinfarct core still need further elucidation. Third, possible differences in pathogenesis, risk factors, and clinical relevance of CMIs with different features (e.g. acute versus chronic, cortical versus subcortical) are complex and warrant clarification. These future advances will enhance our understanding of CMIs and their impact on brain function. This, in turn, can guide the development of preventive and therapeutic strategies for CMIs and related clinical consequences.
Search strategy and selection criteria
To conduct this narrative review, we searched PubMed for articles published in English from 1 January 2017 to 1 February 2023 using terms: microinfarct* AND (brain or cerebral) AND (autopsy or neuropathology or MRI or imaging or diffusion) and identified 220 results. One author (J.H.) screened the titles and abstracts, and the relevant articles (n = 69) were selected after discussion with another author (S.H.). Full-text articles were then retrieved and reviewed. The remaining 151 articles were not relevant for the study. Additional sources of relevant articles were obtained by manually screening the reference lists of reviewed articles (n = 15) and the authors’ own published works (n = 9). The final selection included studies that provided relevant findings and novel insights on the topic to be reviewed.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the National Medical Research Council Singapore, Transition Award (grant no. A-0006310-00-00) and Ministry of Education Singapore, Academic Research Fund Tier 1 (grant no. A-0006106-00-00).
