Abstract
Introduction:
Aneurysmal subarachnoid hemorrhage (aSAH) and intracerebral hemorrhage (ICH) are main forms of hemorrhagic stroke. Data regarding cerebral small vessel disease (SVD) burden and incidental small lesions on diffusion-weighted imaging (DWI) following aSAH are sparse.
Patients and methods:
We retrospectively analyzed a prospective cohort of aSAH and ICH patients with brain MRI within 30 days after onset from March 2015 to January 2023. White matter hyperintensity (WMH), lacune, perivascular space, cerebral microbleed (CMB), total SVD score, and incidental DWI lesions were assessed and compared between aSAH and ICH. Clinical and radiological characteristics associated with small DWI lesions in aSAH were investigated.
Results:
We included 180 patients with aSAH (median age [IQR] 53 [47–61] years) and 299 with ICH (63 [53–73] years). DWI lesions were more common in aSAH than ICH (47.8% vs 14.4%, p < 0.001). Higher total SVD score was associated with ICH versus aSAH irrespective of hematoma location, whereas DWI lesions and strictly lobar CMBs were correlated with aSAH. Multivariable analysis showed that shorter time from onset to MRI, anterior circulation aneurysm rupture, CMB ⩾ 5, and total SVD score were associated with DWI lesions in aSAH.
Discussion and conclusion:
Incidental DWI lesions and strictly lobar CMBs were more frequent in aSAH versus ICH whereas ICH had higher SVD burden. Incidental DWI lesions in aSAH were associated with multiple clinical and imaging factors. Longitudinal studies to investigate the dynamic change and prognostic value of the covert hemorrhagic and ischemic lesions in aSAH seem justified.

Introduction
Hemorrhagic stroke constitutes approximately 30%–40% of annual stroke incidence worldwide. 1 Spontaneous intracerebral hemorrhage (ICH) and aneurysmal subarachnoid hemorrhage (aSAH) are the two forms of hemorrhagic stroke that share similarly high rates of disability and mortality.2,3
Cerebral small vessel disease (SVD) encompasses a variety of cerebral microvascular pathological changes and is closely associated with ICH. 4 Magnetic resonance imaging (MRI) enables in vivo detection of multiple SVD markers including white matter hyperintensity (WMH), perivascular space (PVS), lacune, and cerebral microbleed (CMB), while diverse mechanisms exist for pathogenesis of small vessel abnormalities. 5 Despite that microvascular dysfunction after aSAH has garnered increasing attention, 3 there was limited evidence illustrating the prevalence of small-vessel damage using multimodal MRI among aSAH patients. Although ICH and aSAH may share similar risk factors such as advanced age, hypertension, smoking, and heavy alcohol consumption, as well as overlapping imaging appearance such as subarachnoid extension after ICH and vice versa, and intraventricular extension in both severe conditions, there is a paucity of studies comparing clinical and neuroimaging features between ICH and aSAH. 6 Interestingly, a small MRI-based study of SAH patients 7 found that CMBs were not uncommon after SAH, indicating that microangiopathic injury are readily detected on MRI in the acute phase of SAH.
In addition, there is emerging evidence that incidental punctate hyperintense lesions on diffusion-weighted imaging (DWI) are prevalent in both ICH 8 and SAH. 9 A growing body of evidence suggested that incidental DWI-positive lesions may occur in 11%–40% of ICH patients and are related to SVD. 8 For patients with aSAH, DWI hyperintensities may represent a more complex scenario due to diffuse subarachnoid blood and early endovascular treatment of intracranial aneurysm.10,11 Additionally, microbleeds were reported to be associated with DWI lesions in SAH patients. 7
Given the aforementioned evidence, the primary objective of our study was to explore the prevalence of SVD and incidental DWI lesions after aSAH and compare the clinical and neuroimaging profile between ICH and aSAH. Additionally, we aim to investigate potential factors associated with small DWI lesions after aSAH.
Patients and methods
Study population
This is a retrospective analysis of prospectively collected hemorrhagic stroke cohort admitted to a tertiary teaching hospital from March 2015 to January 2023. Adult patients with ICH or SAH and brain MRI including blood sensitive susceptibility-weighted imaging (SWI) were included in our study. ICH was diagnosed by acute neurological deficit and presence of intraparenchymal hematoma on brain computed tomography (CT) and/or MRI. Non-traumatic SAH was diagnosed based on brain CT and/or lumbar puncture. Exclusion criteria were: (1) ICH secondary to arteriovenous malformation, cavernous angioma, trauma, brain neoplasm, hemorrhagic transformation of ischemic infarct, and anticoagulant-associated ICH, (2) primary intraventricular hemorrhage, (3) non-aneurysmal SAH, (4) hematoma evacuation performed before MRI scan, (5) MRI scan performed >30 days after symptom onset, and (6) poor image quality for analysis. Intraparenchymal hemorrhage concurrent with adjacent or remote subarachnoid extension is considered as ICH after excluding any potential secondary causes, and patients with hematoma secondary to intracranial aneurysm rupture were included into SAH group. The study was reported according to the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines.
Data collection and image analysis
The baseline clinical characteristics including age, sex, vascular risk factors, and admission blood pressure were collected by reviewing the medical records and interviewing with the patients or their relatives. For patients with aSAH, ruptured intracranial aneurysm and their sizes were confirmed by CT angiography and/or catheter angiography. Anterior circulation aneurysms were defined as those in internal carotid artery, anterior communicating artery, middle cerebral artery, and posterior communicating artery. Posterior circulation aneurysms were defined as those located in vertebrobasilar system. The amount of subarachnoid blood was evaluated by the modified Fisher scale, and the clinical severity of aSAH were assessed by Hunt-Hess scale. Presence of vasospasm was identified using transcranial Doppler, CT angiography, or catheter angiography.
The brain MRI scan was performed with our institutional stroke imaging protocol including T1-weighted, T2-weighted, fluid attenuated inversion recovery (FLAIR), DWI, and SWI on 1.5 Tesla or 3.0 Tesla MRI scanners. Details of MRI parameters were provided in Supplemental Table S1. Two neurologists who were blinded to clinical information independently reviewed all MRI scans. ICH location was dichotomized into lobar (supratentorial cortical and/or subcortical white matter) and non-lobar (basal ganglia, thalamus, cerebellum, and brainstem) based on the epicenter of hematoma. 12 The SVD markers on MRI were rated according to the STandards for ReportIng Vascular changes on nEuroimaging-2 (STRIVE-2) criteria. 13 The severity of deep and periventricular WMH was assessed separately using the Fazekas et al. 14 scale. Dilated PVS in basal ganglia and centrum semiovale was visually assessed on T2-weighted sequence. A cutoff burden of ⩽20 (mild) and >20 (severe) was used in line with previous studies. 15 Lacune was defined as ovoid to round cavities with or without a light surrounding rim on FLAIR. 13 CMB was identified as homogenous hypointense small round foci on SWI. The number of CMB was stratified into 1–4 and ⩾5 if present. Incidental DWI-positive lesions were defined as punctuate hyperintense foci on DWI with or without corresponding hypointense signal on apparent diffusion coefficient mapping according to previous consensus.13,16 The location of CMBs and DWI-positive lesions was classified into deep (basal ganglia, thalamus, deep periventricular white matter, and brainstem) and lobar (cortical and subcortical white matter) in line with the Microbleed Anatomical Rating Scale. 17 CMB distribution was further classified as strictly lobar, strictly deep, or mixed (i.e. CMBs in both deep and lobar regions). Interrater agreement on a random sample of 50 MRI scans was good to excellent for WMH (interclass correlation = 0.819), lacune (κ = 0.848), CMB (κ = 0.848), CSO-PVS (κ = 0.822), BG-PVS (κ = 1.00), and incidental DWI-positive lesions (κ = 0.896). Any discrepancy was resolved after joint discussion until a consensus was reached. Supplemental Figure S1 displays representative images of small DWI-positive lesions and CMBs in patients with aSAH and ICH. The composite SVD burden was calculated by total SVD score, a well-validated ordinal scale from 0 to 4 points. 18 Each point was given by the presence of moderate to severe WMH (deep WMH Fazekas score 2–3 and/or periventricular WMH Fazekas score 3), PVS in basal ganglia >10, any lacune, or any CMB.
Statistical analysis
Descriptive analysis of patients with ICH and aSAH was reported as number (percentage) for categorical variables and mean (±standard deviation) or median (interquartile range [IQR]) for continuous variables as appropriate. Continuous variables were compared by two-sample t test or Mann-Whitney U test, and categorical variables by the χ2 test or the Fisher exact test. Multivariable binary logistic regression analysis was conducted to assess the consistency of the associations of neuroimaging SVD markers with the event types. Clinical and imaging characteristics associated with the presence of incidental DWI-positive lesions in aSAH were investigated by univariable analysis and subsequent multivariable analysis including variables with p-values less than 0.1 in the univariable analysis. Variables with variance inflation factors greater than 5 were excluded from multivariable analysis. A two-tailed p < 0.05 was considered statistically significant. Bonferroni correction was used for multiple testing corrections in multivariable analysis between aSAH and ICH with adjusted significance threshold of 0.008 (0.05/6). All analyses were performed using SPSS (version 26.0).
Results
We examined a total of 614 patients admitted to our hospital during the study period. After applying the exclusion criteria, a total of 479 patients including 299 patients with ICH and 180 patients with aSAH were included in the final analysis (Supplemental Figure S2).
Table 1 summarizes the baseline characteristics of the study population. Patients with aSAH were younger (median age [IQR] 53 [47–61] years vs 63 [53–73] years, p < 0.001), less likely to be male (34.4% vs 64.5%, p < 0.001), and had less vascular risk factors. Aneurysms were confirmed by catheter angiography in 178 (98.9%) aSAH patients. Endovascular treatment was performed in 177 (98.3%) aSAH patients, and one patient received clipping whereas two patients refused any surgical intervention (Supplemental Table S2).
Patient characteristics.
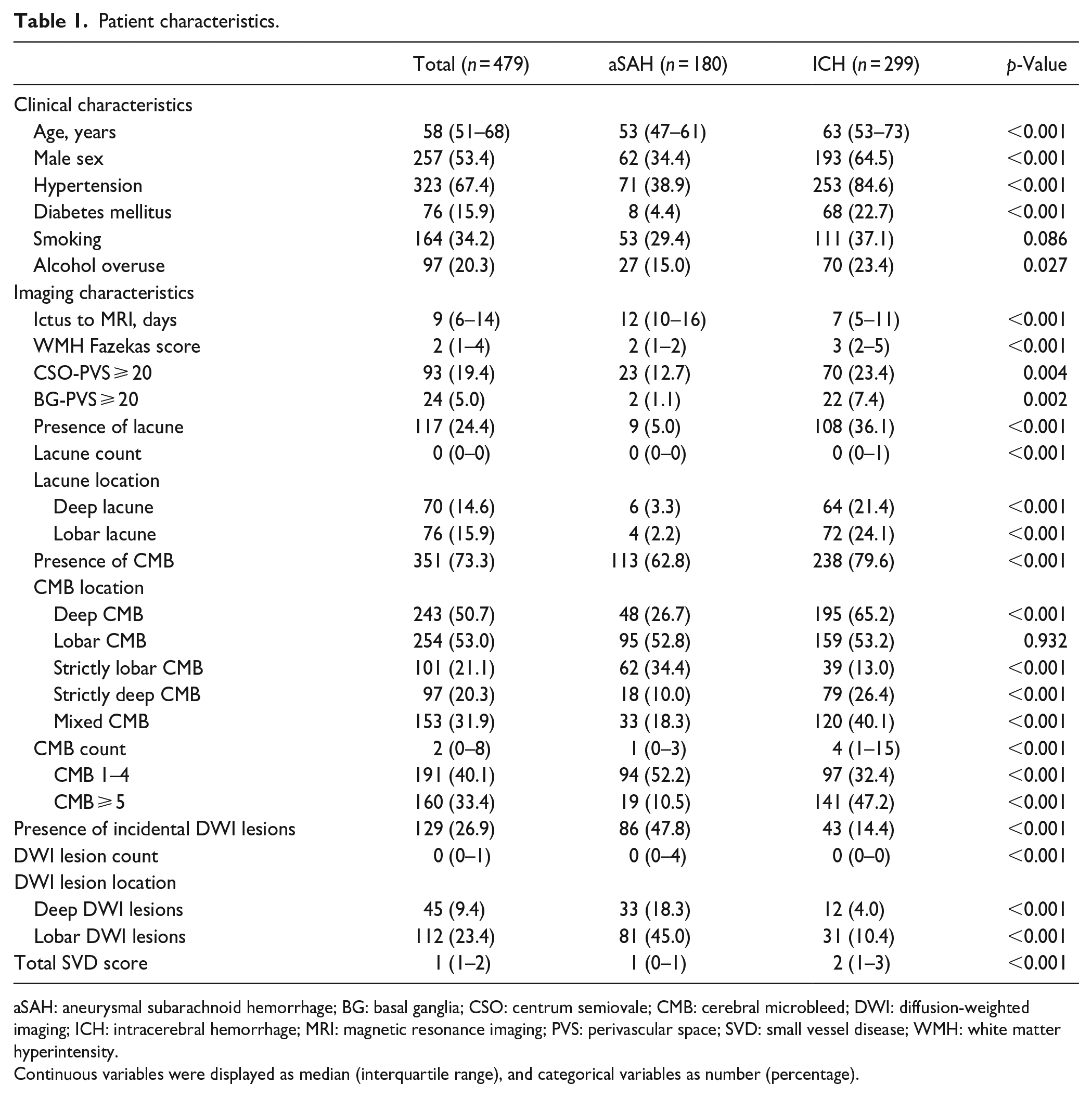
aSAH: aneurysmal subarachnoid hemorrhage; BG: basal ganglia; CSO: centrum semiovale; CMB: cerebral microbleed; DWI: diffusion-weighted imaging; ICH: intracerebral hemorrhage; MRI: magnetic resonance imaging; PVS: perivascular space; SVD: small vessel disease; WMH: white matter hyperintensity.
Continuous variables were displayed as median (interquartile range), and categorical variables as number (percentage).
SVD and incidental DWI-positive lesions between aSAH and ICH
Overall, ICH patients had higher total SVD burden (median [IQR] 2 [1–3] vs 1 [0–1], p < 0.001) than aSAH patients. Figure 1 compares the distribution of total SVD score in ICH and aSAH patients. It is notable that 68.9% of aSAH patients had at least one SVD markers. WMH (median Fazekas score [IQR] 3 [2–5] vs 2 [1–2], p < 0.001), lacune (36.1% vs 5.0%, p < 0.001), CSO-PVS ⩾ 20 (23.4% vs 12.7%, p = 0.001), BG-PVS ⩾ 20 (7.4% vs 1.1%, p = 0.001) were more common in ICH. Although the prevalence of CMBs (79.6%) was higher in ICH group CMBs (62.8%) were not uncommon in SAH group. Strictly lobar CMBs (34.4% vs 13.0%, p < 0.001) were significantly more common in aSAH patients, while strictly deep CMB (26.4% vs 10.0%, p < 0.001) and mixed CMB (40.1% vs 18.3%, p < 0.001) were more prevalent in ICH patients. Incidental DWI-positive lesions were observed in 47.8% patients with aSAH versus 14.4% in those with ICH (p < 0.001). Lobar DWI lesions were more frequent than deep DWI lesions (45.0% vs 18.3%) in aSAH (Table 1).
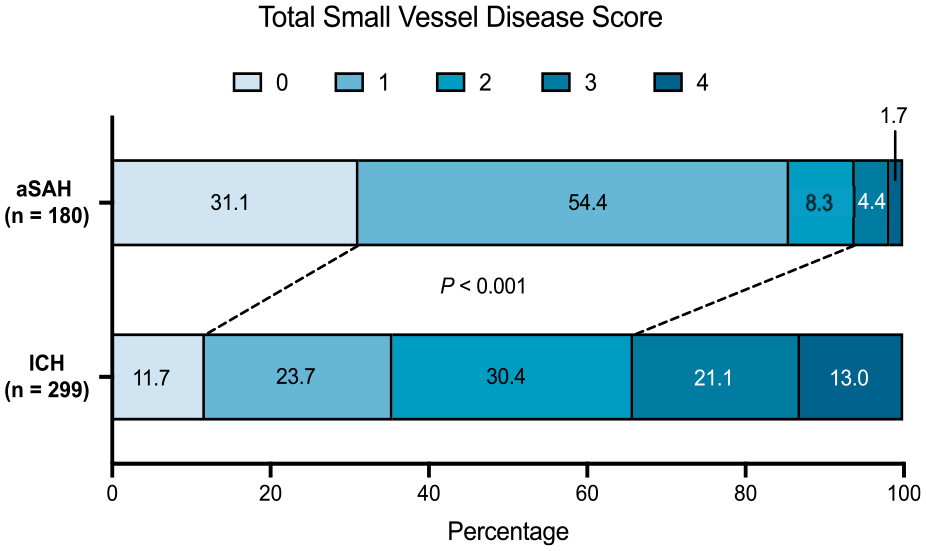
Distribution of total small vessel disease (SVD) score between patients with intracerebral hemorrhage and aneurysmal subarachnoid hemorrhage. Total SVD score was compared by χ2 test.
In multivariable analysis of individual imaging markers susceptible to aSAH or ICH, WMH severity and lacune were significantly correlated with increased odds of ICH irrespective of ICH location, while small DWI lesions (adjusted odds ratio (aOR) 0.11, 95% confidence interval (CI) 0.06–0.20) after correction for multiple comparisons. Strictly lobar CMBs were associated with aSAH when compared with overall ICH (aOR 0.49, 95% CI 0.27–0.92) and had stronger association with aSAH versus non-lobar ICH (aOR 0.20, 95% CI 0.08–0.48). Strictly deep CMBs were likely to be correlated with overall ICH and non-lobar ICH. Higher total SVD score (aOR 1.96 per point, 95% CI 1.47–2.61) was associated with increased odds of ICH after adjusting for age, sex, vascular risk factors, and time from onset to MRI. These results remained similar when comparing aSAH with both lobar (aOR 1.81 per point, 95% CI 1.27–2.58) and non-lobar ICH (aOR 2.06 per point, 95% CI 1.48–2.87, Table 2).
Odds ratio for intracerebral hemorrhage versus aneurysmal subarachnoid hemorrhage based on neuroimaging findings.
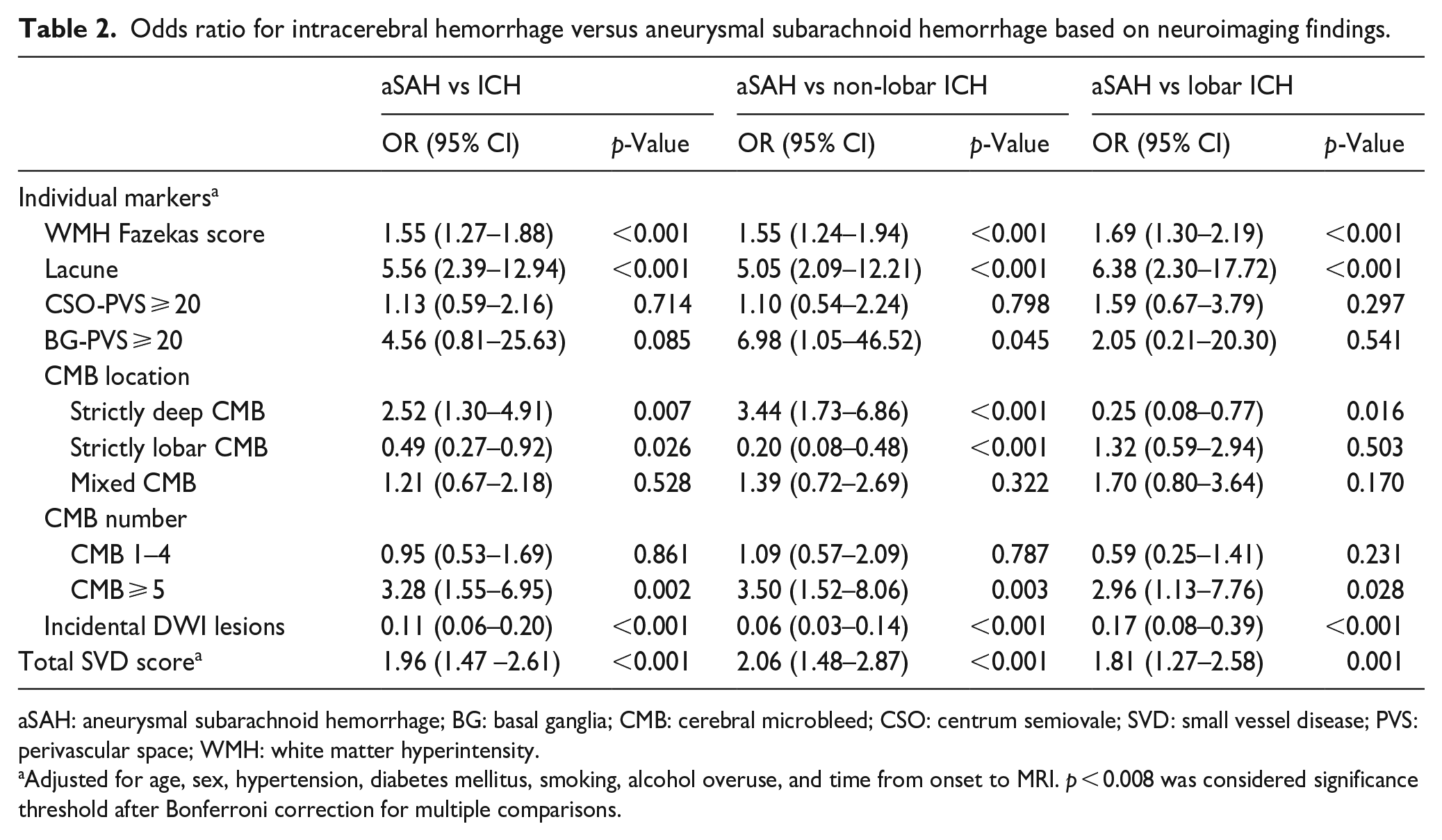
aSAH: aneurysmal subarachnoid hemorrhage; BG: basal ganglia; CMB: cerebral microbleed; CSO: centrum semiovale; SVD: small vessel disease; PVS: perivascular space; WMH: white matter hyperintensity.
Adjusted for age, sex, hypertension, diabetes mellitus, smoking, alcohol overuse, and time from onset to MRI. p < 0.008 was considered significance threshold after Bonferroni correction for multiple comparisons.
Incidental DWI-positive lesions in aSAH
Among patients with incidental DWI-positive lesions, 41 (47.7%) were ipsilateral to the treated aneurysm, 10 (11.6%) contralateral to the treated aneurysm, and 27 (31.4%) occurred in both hemispheres. In univariable analysis, incidental DWI lesions after aSAH were related to earlier MRI scan, rupture of anterior circulation aneurysm, higher CMB burden, and higher total SVD score, and marginally associated with stent-assisted coiling, mixed CMB pattern and higher WMH burden (Table 3). The incidental DWI lesion burden according to WMH severity, CMB burden, and total SVD score in aSAH patients is shown in Figure 2. In multivariable analysis including individual SVD markers, time from onset to MRI (aOR 0.90, 95% CI 0.84–0.96), anterior circulation aneurysm (aOR 5.03, 95% CI 1.06–23.93), and CMB ⩾ 5 (aOR 14.17, 95% CI 2.35–85.57) were independently correlated with DWI lesions in aSAH (Table 4, Model 1). In the model incorporating cumulative SVD burden, time from onset to MRI (aOR 0.91, 95% CI 0.86–0.97), anterior circulation aneurysm (aOR 5.98, 95% CI 1.27–28.16), and total SVD score (aOR 1.52 per point, 95% CI 1.02–2.27) remained associated with incidental small DWI lesions in aSAH (Table 4, Model 2).
Univariable analysis of characteristics associated with incidental DWI lesions in patients with aneurysmal subarachnoid hemorrhage.
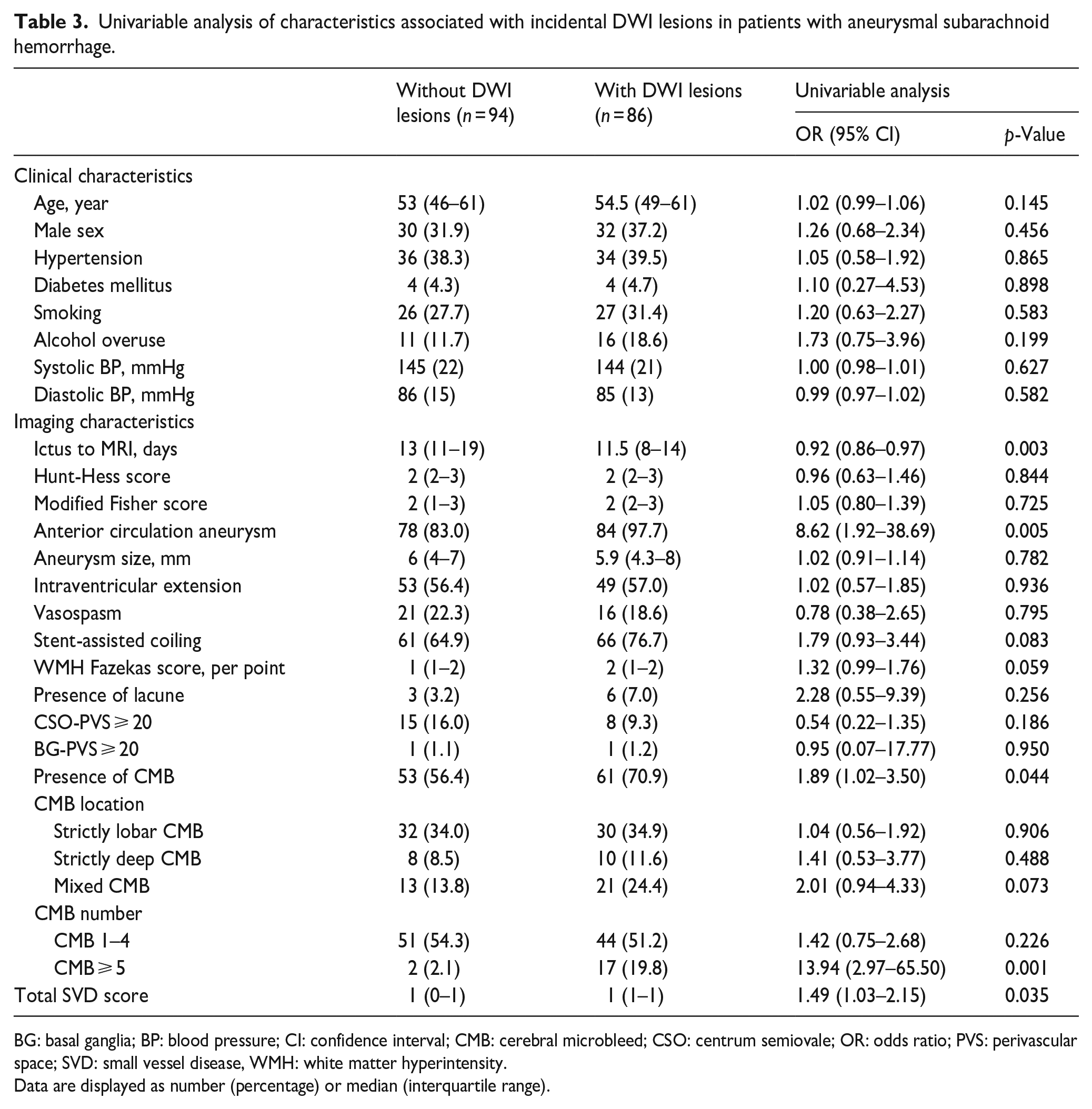
BG: basal ganglia; BP: blood pressure; CI: confidence interval; CMB: cerebral microbleed; CSO: centrum semiovale; OR: odds ratio; PVS: perivascular space; SVD: small vessel disease, WMH: white matter hyperintensity.
Data are displayed as number (percentage) or median (interquartile range).
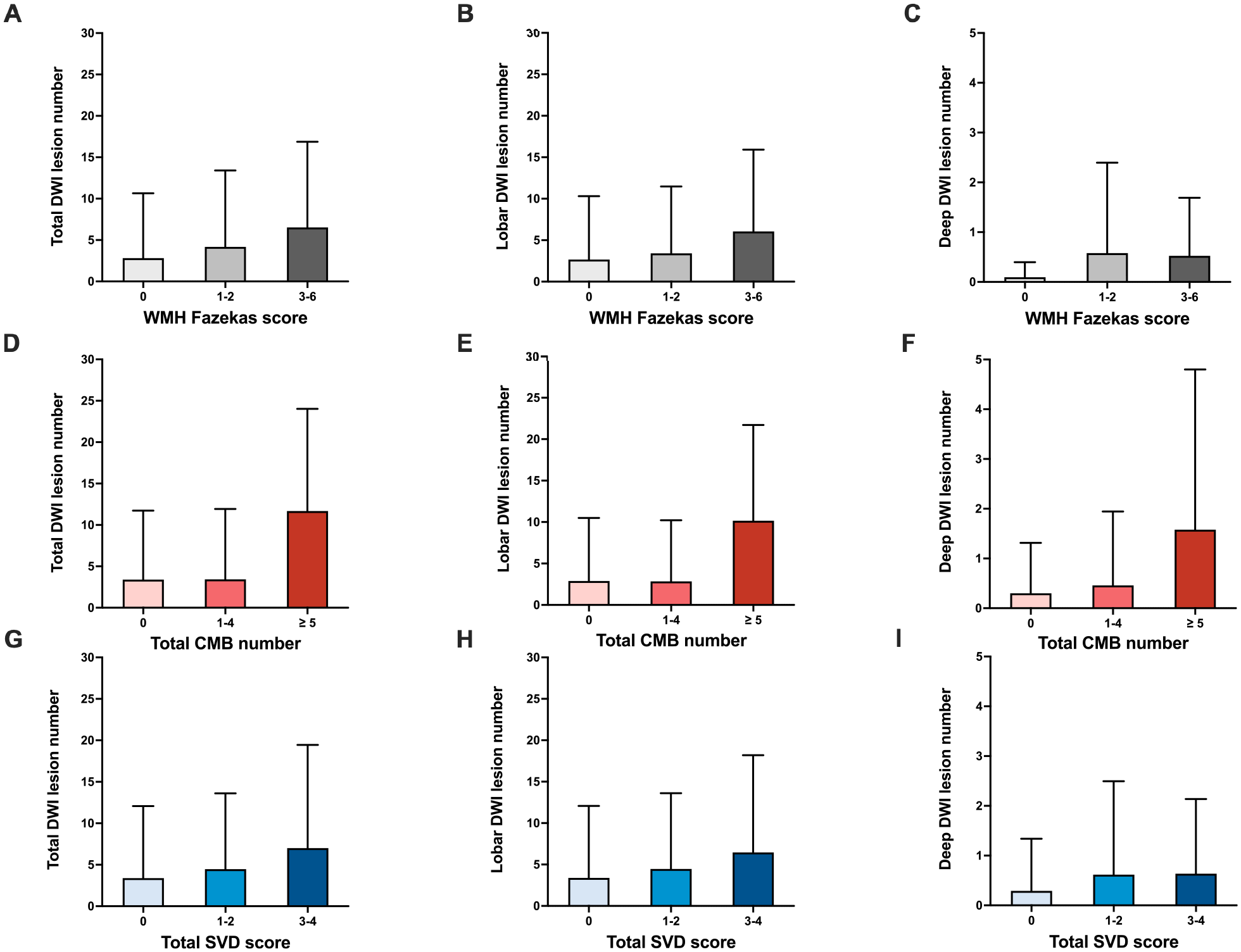
Total, lobar, and deep incidental DWI lesion number stratified by the Fazekas score of white matter hyperintensity (WMH) (a–c), cerebral microbleed (CMB) count (d–f), and total small vessel disease (SVD) score (g–i) in patients with aneurysmal subarachnoid hemorrhage. The number of DWI lesions was presented as mean with standard deviation.
Multivariable logistic regression analysis of characteristics associated with the presence of incidental DWI lesions in patients with aneurysmal subarachnoid hemorrhage.
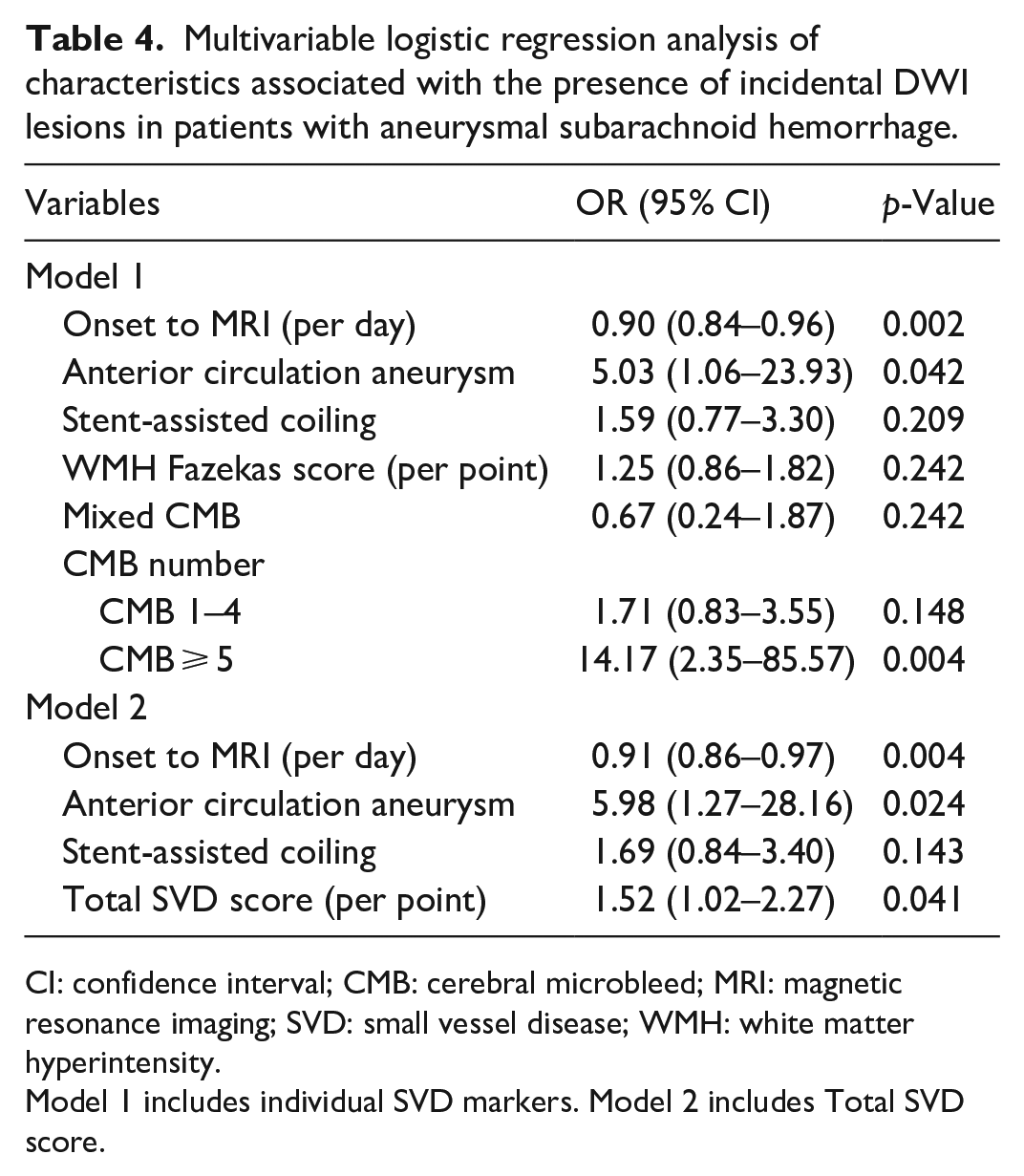
CI: confidence interval; CMB: cerebral microbleed; MRI: magnetic resonance imaging; SVD: small vessel disease; WMH: white matter hyperintensity.
Model 1 includes individual SVD markers. Model 2 includes Total SVD score.
Discussion
We demonstrated that ICH and aSAH exhibit divergent neuroimaging profiles. Patients with ICH were older, had greater vascular risk factor burden, and associated with higher overall SVD severity, whereas strictly lobar CMBs and incidental small DWI lesions were more common in patients with aSAH. Approximately half of incidental DWI lesions were observed ipsilateral to the treated aneurysm. The DWI-positive lesions in aSAH were associated with shorter time from onset to MRI, rupture of anterior circulation aneurysm, greater CMB number, and total SVD burden.
There is limited data comparing clinical and radiological factors between ICH and aSAH. Our results delineated the divergent imaging profile between ICH and aSAH. In our study population, we observed significantly greater SVD burden in ICH than aSAH, while strictly lobar CMB and small DWI lesions were more frequent in aSAH. WMH and lacune as hallmarks of chronic small vessel damage were remarkably more frequent in ICH patients, which may be attributed to older age and a higher prevalence of vascular risk factors that promoted more severe preexisting microvascular alterations before ICH onset, whereas the presence of CMBs and incidental DWI-positive lesions in aSAH may be presumably related to different mechanisms from ICH.
Previous literature reported that CMB is observed in 3%–34% of general population, 19 around 30% of patients with ischemic stroke, 20 and greater than 60% of patients with ICH, 21 while the prevalence of CMBs in aSAH patients remains unclear. Our study which represents a relatively large sample size of SAH patients provides important findings that aSAH patients also harbored a particularly high prevalence of CMB and divergent spatial CMB distribution in contrast to ICH. Over 60% of patients with aSAH had concomitant CMBs, which was higher than previously reported. 7 Jeon et al. 7 reported that 46% had microbleeds in a small cohort of 39 SAH patients, while another European-based observational study 22 found that only 10 out of 102 (9.8%) patients with aSAH had microbleeds. These results suggest that there may be ethnic differences in prevalence of CMB in aSAH, which need further validation.
Pathological studies demonstrated that the majority of MRI-visible CMBs correspond to recent or old microhemorrhages. 23 However, there is a lack of discussion regarding the possible mechanisms and histopathological correlates of CMB in SAH.23,24 An autopsy case of aSAH found evidence of putamen CMB on in vivo MRI and corresponding focal hemosiderin deposition, while this may only be the manifestation of arteriolosclerosis. 25 In our study population, CMBs in aSAH were most commonly exhibited a strictly lobar pattern (34.4%), followed by a mixed pattern (18.3%), and strictly deep pattern (10.3%). We proposed that new CMB may occur in SAH patients predominantly as a result of sudden extravasation of blood into subarachnoid space and secondary microvascular disturbance such as endothelial activation and leakage of blood-brain barrier in the acute phase as aSAH patients were younger and had less hypertension, indicating less likelihood of significant amyloid deposition and longstanding hypertensive injury compared with ICH patients. 7 This is supported by pathological observation of erythrocyte infiltration in the perivascular space and petechial hemorrhages in parenchyma in swine model of SAH. 26 Longitudinal studies with serial MRI scans are needed to observe the temporary dynamics of CMBs in aSAH.
Small hyperintense lesions on DWI have emerged as a novel imaging finding after ICH relating to SVD severity. 8 In our study, we reported a significantly higher prevalence of DWI-visible lesions following aSAH than ICH (47.8% vs 14.4%) and investigated potential factors associated with this imaging finding. Considering the presence of diffuse subarachnoid blood and endovascular therapy after SAH occurrence, the development of DWI lesions after SAH may represent a more complex scenario compared with ICH. The DWI lesions were more frequently observed in lobar regions and more common in SAH with anterior circulation aneurysm rupture, indicating that diffuse subarachnoid blood may contribute greatly to formation of DWI lesions. Accumulating evidence suggested that DWI lesions as secondary brain injury were spatially and quantitatively related to convexity SAH and cortical superficial siderosis in cases with progressive cerebral amyloid angiopathy.27 –29 Fresh subarachnoid blood can promote early brain infarction and microthrombosis by the mechanism of the cortical spreading depolarization, coagulation activation and fibrinolytic dysfunction.26,30 Our results also suggest that DWI lesions in aSAH may primarily occur in the acute phase with a shorter interval from onset to MRI scan. However, as subgroup analysis of The REstart or STop Antithrombotics Randomized Trial (RESTART) indicated that DWI lesions could be detected in 30% of ICH survivors even after about 2 months from ICH onset, the dynamic change of DWI lesions after SAH remains to be investigated by serial neuroimaging study. 31 In addition, we observed that nearly half of small DWI lesions occurred ipsilateral to the treated aneurysm, in line with a previous meta-analysis demonstrating that new DWI lesions were not uncommon after endovascular treatment of aneurysm irrespective of procedure methods. 11 Accumulating evidence suggested that incidental DWI-positive lesions may represent intrinsic small-vessel abnormalities. 13 In line with a previous study, we found that incidental DWI lesions following aSAH are correlated with greater CMB burden, which highlighted coexistence of both hemorrhagic and presumably ischemic manifestations of microvascular injury immediately after aSAH. 7 Mixed CMB was numerically more common in aSAH patients with DWI lesions, suggesting possibly more severe underlying arteriolosclerosis, while the association was attenuated in multivariable analysis and should be further validated in future studies. SVD burden may also be associated with elevated risk of small DWI lesions possibly explained by impaired microvascular autoregulation. More work is needed to elucidate the crosstalk between SAH and subsequent microvascular disturbance.
MRI has been increasingly used in the clinical workup of ICH patients to determine the etiology and evaluate the risk of recurrence. 32 However, MRI examination is not routinely used in the diagnostic workup of SAH. To our knowledge, our study may represent one of the largest SAH cohorts with multimodal MRI scan. Our study provides evidence that CMBs and small DWI lesions are also highly prevalent in SAH patients and show divergent distribution pattern compared with ICH, emphasizing an important role of MRI examination for detecting variable brain injuries in aSAH. The high prevalence of microbleeds and DWI lesions highlights marked deleterious effects to microvasculature after aSAH onset and warrants further research on potential therapeutic strategies for restoration of microvascular function. Both CMBs and DWI-positive lesions in ICH have been proved to be associated with unfavorable outcomes 33 and recurrent stroke.31,34,35 Given that severe disability, cognitive decline, and mood disorders were highly present after aSAH, 3 whether the covert punctuate hemorrhagic and ischemic lesions detected immediately after SAH may render short or long-term impact to prognosis requires investigation in future prospective studies.
Our study has several limitations. First, retrospective analysis in a single institution may render selection bias and alleviate the generalizability. Second, the clinical decision of MRI examination was at the discretion of the physicians and MRI would not be performed in critically ill patients and those with contraindications, which may also lead to potential selection bias. Thirdly, the predominance of aSAH patients underwent surgical procedure before MRI scan as per institutional workup because early aneurysm coiling is the priority to attenuate the risk of rebleeding, therefore the influence of invasive angiography to incident DWI lesions could not be avoided. Future studies investigating the MRI appearance of aSAH before surgical procedure seem justified.
Conclusion
Our study found that strictly lobar CMBs and incidental DWI-positive lesions are more prevalent immediately after aSAH compared with ICH whereas ICH patients have higher total SVD burden. Incident DWI lesions in aSAH may be developed through multiple mechanisms. Future prospective studies are needed to confirm their clinical implications in patients with aSAH.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241232327 – Supplemental material for Prevalence of small vessel disease and incidental DWI-positive lesions in patients with aneurysmal subarachnoid hemorrhage versus intracerebral hemorrhage
Supplemental material, sj-docx-1-eso-10.1177_23969873241232327 for Prevalence of small vessel disease and incidental DWI-positive lesions in patients with aneurysmal subarachnoid hemorrhage versus intracerebral hemorrhage by Zi-Jie Wang, Xiao Hu, Yan-Fang Xie, Wen-Jun Yao, Lan Deng, Zuo-Qiao Li, Ming-Jun Pu, Xin-Ni Lv, Zi-Cheng Hu, Jiang-Tao Zhang and Qi Li in European Stroke Journal
Supplemental Material
sj-docx-2-eso-10.1177_23969873241232327 – Supplemental material for Prevalence of small vessel disease and incidental DWI-positive lesions in patients with aneurysmal subarachnoid hemorrhage versus intracerebral hemorrhage
Supplemental material, sj-docx-2-eso-10.1177_23969873241232327 for Prevalence of small vessel disease and incidental DWI-positive lesions in patients with aneurysmal subarachnoid hemorrhage versus intracerebral hemorrhage by Zi-Jie Wang, Xiao Hu, Yan-Fang Xie, Wen-Jun Yao, Lan Deng, Zuo-Qiao Li, Ming-Jun Pu, Xin-Ni Lv, Zi-Cheng Hu, Jiang-Tao Zhang and Qi Li in European Stroke Journal
Footnotes
Acknowledgements
We would like to thank all investigators and health care providers who participated in the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation of China (No. 82071337), the National Key R&D Program of China (No. 2018YFC1312200 and No. 2018YFC1312203), Chongqing Science Fund for Distinguished Young Scholars (No. cstc2021jcyj-jqX0029), Chongqing High-end Young Investigator Project (No. 2019GDRC005), S&T Program of Hebei (No. 20377764D), and Research Fund of Anhui Institute of Translational Medicine (No. 2022zhyx-C38).
Ethical approval
The study was approved by Ethics Committee of Chongqing Medical University (No. 2017-075).
Informed consent
Written informed consent was obtained from all subjects before the study.
Guarantor
Qi Li.
Contributorship
QL and ZJW designed the study; ZJW and XH wrote the manuscript; QL provided critical revisions of the manuscript; QL, ZJW, XH, and YFX analyzed the data; ZJW, XH, YFX, WJY, LD, ZQL, MJP, XNL, ZCH, and JTZ contributed to data collection; QL and JTZ were involved in funding acquisition. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Data availability
Anonymized data and material supporting the research is available by request from qualified investigators.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
