Abstract
Purpose:
Cerebral small vessel disease (cSVD) is a highly prevalent disorder leading to physical, cognitive and functional decline. We report key barriers in the management of individuals with cSVD, the potential benefit of cSVD-dedicated health services, and evidence from existing models of care for adults with cSVD.
Methods:
We examined information from a scientific seminar developed between seven experts in cSVD during the eighth European Stroke Organisation Conference that discussed the optimal health care for adults with cSVD and what health services dedicated to cSVD should include.
Findings:
Barriers in cSVD care include unrecognised symptoms and modifiable risk factors, heterogeneity of clinical presentations, inefficient inter/intra-clinical services communication/organisation, and uncertainties regarding what assessments/treatments should be routinely done, when and by whom. However, existing health services and research studies suggest models of care in cSVD. Multi-step approaches can be used for identification and aetiological workup in individuals with cSVD, including basic and selected advanced evaluations, for example, monogenic cSVD testing. Although current guidelines for management of cSVD illlustrate limitations, there are recommendations on risk factors and lifestyle considerations, as well as pharmacological and non-pharmacological interventions for people with cSVD.
Discussion and conclusion:
Existing healthcare models do not provide optimal care for individuals with cSVD. Lack of awareness of heterogeneous clinical presentations and uncertainty in threshold of cSVD ‘burden’ for referral to specialist multidisciplinary services, are key challenges for health services to overcome. Creating cSVD-dedicated services may prevent underdiagnosing and achieve standardised holistic management to improve outcomes in people with cSVD. However, adequate prevention and early management should be offered at all levels of care.
Keywords
Introduction
Cerebral small vessel disease (cSVD) is a common cause of ischaemic and haemorrhagic stroke, dementia, neurobehavioural symptoms and death. 1 cSVD is a disorder of the brain’s microvasculature that results in several characteristic features seen on brain imaging including white matter hyperintensities (WMH), lacunes, perivascular spaces, cerebral microbleeds (CMB) and covert brain infarcts (CBI) where these findings vary in prevalence and severity between individual subjects.2,3 cSVD encompasses sporadic and monogenic forms, 2 and may be divided clinically into covert ‘asymptomatic/subclinical/incidental’ (found on scanning) and overt ‘symptomatic’ (stroke and/or cognitive syndromes amongst other features). 4 cSVD is clinically important, doubling the risk of stroke and dementia. 5 Improved clinical management might help to reduce this risk.
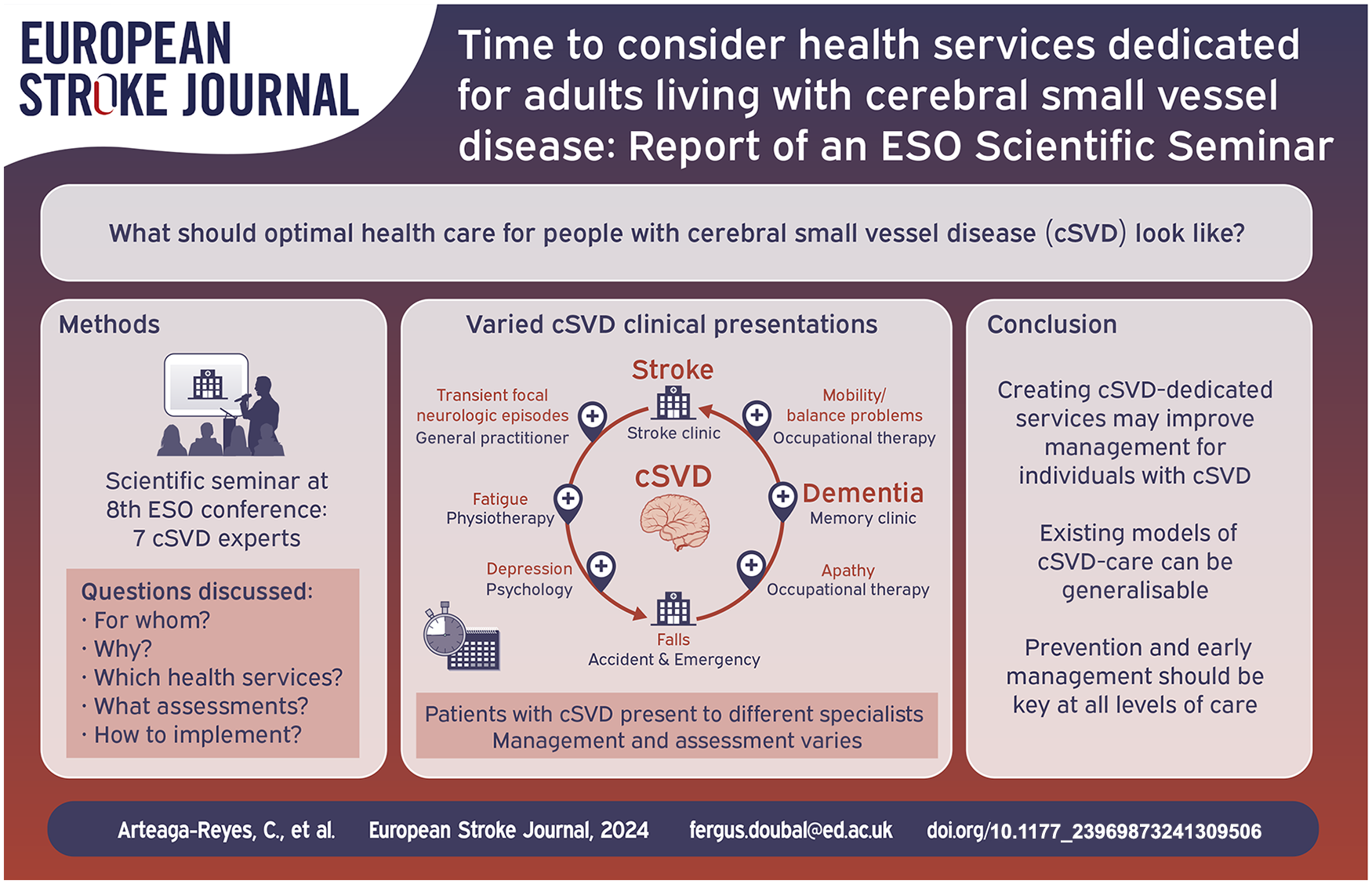
Many individuals with cSVD are diagnosed after presentation with stroke/cognitive impairment. Others may present with multiple overlapping clinical syndromes, such as apathy, delirium, transient focal neurologic episodes, depression, fatigue, Parkinsonism, falls and/or sphincter control disturbances. 4
The wide range of clinical syndromes in patients with cSVD leads to presentation to multiple clinical services with different levels of expertise, fragmented evaluation and management, and the severity of the condition being overlooked, losing an opportunity to prevent stroke and dementia.
This report examines gaps in cSVD care based on a scientific seminar held during the eighth European Stroke Organisation Conference (ESOC). It highlights unanswered questions in the management of patients with cSVD and the potential benefit of dedicated-cSVD services.
Procedure
Five clinical researchers with expertise in cSVD delivered talks reflecting on the design of optimal clinical services for patients with cSVD based on their experience. Presentations addressed the five questions described below and were followed by audience discussion coordinated by two additional experts. The seven experts represented individuals who have already started an cSVD clinic (3/7), are actively involved in cSVD clinical research, or have prepared guidelines for the diagnosis and management of cSVD and related topics (7/7).
Findings
A dedicated clinic for cSVD – For whom and why?
The natural history of cSVD represents a cumulative burden of disability that results in functional decline, institutionalisation, and death. Despite current ‘adequate’ secondary preventive treatment after an ischaemic stroke (e.g. ‘lacunar’ infarct), some individuals with cSVD continue to decline with recurrent strokes (ischaemic/haemorrhagic) or cognitive impairment, indicating that current management may be suboptimal.6–10
Gaps in cSVD care arise from several factors, including: (a) Existence of several underlying pathophysiology hypotheses; (b) Lack of awareness and under-recognition of cSVD symptoms by individuals and clinicians; (c) Frequent multi-morbidity in people with cSVD especially at late-stages; (d) Multiple clinical presentations being managed in isolation by several healthcare professionals resulting in unstructured management; (e) A need for neuroimaging confirmation of cSVD features with standardised radiological reporting; (f) Uncertainty over where to put the threshold for assessing people with cSVD in dedicated services and (g) An attitude that because there is no current specific treatment for cSVD, management should reside mainly in primary care and research settings (sBox1).
These gaps contribute to the challenge in cSVD to define referral criteria and clinical pathways providing meaningful impact on person-centred care (Box 1). Creating cSVD clinics can offer a formal diagnosis and accurate information sharing, standardised structured aetiological workup, clinical classification and phenotyping, treatment as per evidence base, monitoring of outcomes, and improved risk factor management. In addition, cSVD centres can help translation from research into clinics and vice versa to better understand the pathophysiology of cSVD and develop targeted therapeutics beyond conventional cardiovascular risk reduction.
Challenges in defining eligibility criteria for referral to cSVD services.
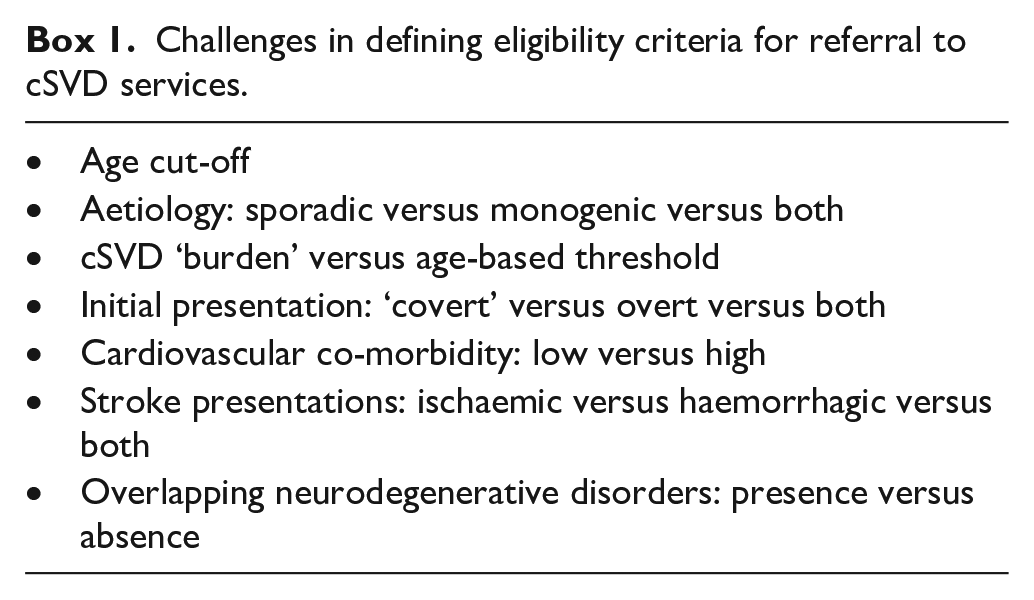
Referral of patients into a cSVD-dedicated service leans on pre-existing services (e.g. geriatric medicine, general practice). These services could refer patients presenting with cSVD-related stroke, cognitive decline, Parkinsonism, suspicion of monogenic cSVD or cerebral amyloid angiopathy (CAA). However, covert cSVD requires developing neuroradiology screening services to identify patients. Since the number of potential cSVD patients requiring assessment could overwhelm any clinical service, cSVD services should work strategically with pre-existing services to avoid duplication and empower existing services to provide guideline based care.
Traditionally, clinical outcomes (stroke/function/cognition) have been measured in patients with cSVD. It is also important to measure process and patient-reported outcome measures and satisfaction as a marker of high quality clinical services.
The Manchester Centre for Clinical Neurosciences cSVD model (United Kingdom)
In the established Manchester cSVD-dedicated clinical service, the clinical pathway starts with discussion of referred patients at a clinical multidisciplinary team meeting with an aim to confirm neuroimaging cSVD features with exclusion of other differential diagnosis, offer a provisional aetiological classification, and grade the severity based on WMH-burden. Subsequent aetiological workup involves four-steps: (a) Vascular risk factor screening; (b) Decision on neurogenetic testing (sBox2); (c) Standardised clinical assessment of cSVD-associated syndromes and (d) Decision on treatment and follow-up based on clinical syndromes identified (sTable1). The current Manchester model attempts to link research to the clinic through projects on neurogenetics, and novel magnetic resonance imaging (MRI) techniques.
In future, the Manchester model aims to develop cSVD services in the district general hospitals with expertise to provide standardised aetiological workup, assessments and treatment (steps a–d); and referral for individuals with monogenic cSVD, uncertain or complex diagnosis to the Manchester Neurosciences centre.
Which clinical services could be used as a model for cSVD dedicated services?
The increasingly high prevalence of detected cSVD, and the clinical and socio-economic consequences of the disease, illustrate the need for dedicated services for patients with cSVD. To efficiently address the associations between cSVD and cognitive outcomes (e.g. higher WMH burden leading to higher risk of cognitive impairment and all-cause dementia) 11 ; it might be necessary to assess people with cSVD at earlier stages.
Due to the heterogenous presentations of cSVD, the few cSVD centres that do exist overlap with other services (sTable2); and patients with different cSVD presentations, that is, acute/chronic clinical presentations, suspicion/known monogenic cSVD, do not receive standardised multidisciplinary assessments. Since cognitive impairment might be absent or unrecognised in people with cSVD at the initial stages, 4 assessment of these individuals should not be limited to highly specialised services, such as memory services. Assessments should start from primary care with management tackling the basics (modifiable risk factors).
Well-known examples of clinical services that could be used as models for a cSVD service are memory services, and some translatable research-based models, such as the LADIS study (Leukoaraiosis and Disability study) 12 (sTable2). Although the models from non-cSVD services offer guidance to develop a cSVD-dedicated service, several items should be explicitly addressed for cSVD as outlined in Box 1, as well as referral pathways within existing services, and haemorrhagic risk in people with cSVD (including decisions over blood-thinner medications).
In addition, barriers for the management of people with cSVD needing to be addressed when developing a cSVD clinic include: strategies for identification of cSVD; risk stratification; effective guidance for assessments, imaging and functional screening, management and systematic follow-up; focus on person-centred outcomes; and available resources at primary care and specialised services.
Which routine assessments should be performed in cSVD patients?
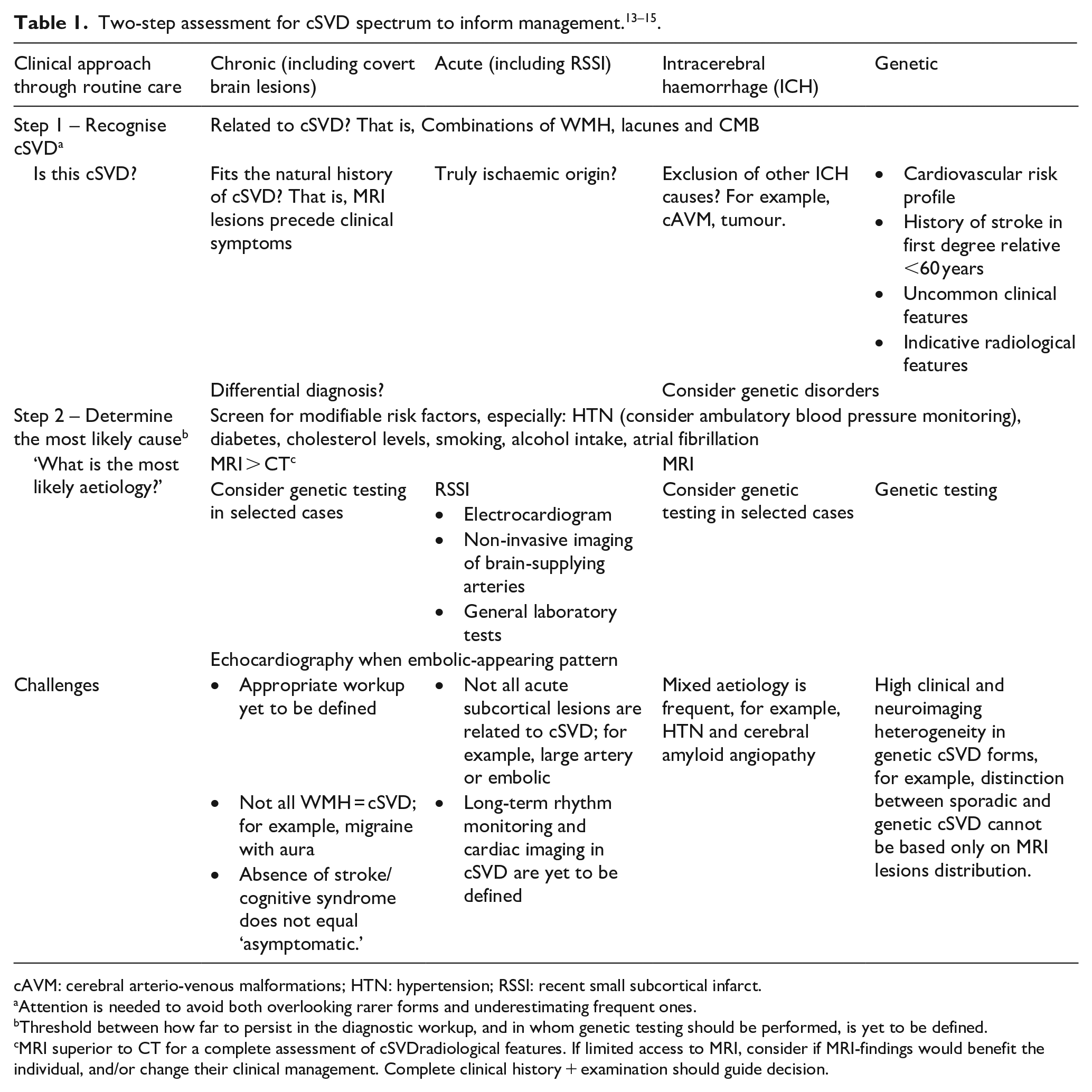
The clinical-radiological spectrum of cSVD includes both frequent sporadic disease and rarer monogenic forms of cSVD. A two-step approach could be applied in people with cSVD.13–15 All clinicians approaching individuals with cSVD could answer two main questions. (1) ‘Is this cSVD or could a differential diagnosis explain brain imaging lesions?’ For instance, in an individual presenting with a lacunar syndrome that corresponds with a recent small subcortical infarct without other cSVD features on brain scan, clinicians may consider other diagnoses, such as large artery atheroembolism or microatheroma in distal arterioles. (2) ‘What is the most likely aetiology?’ To answer this, neuroimaging plays a fundamental role, whether CT or MRI (ideally) according to local clinical availability; in addition to screening for modifiable risk factors; clinical appearance, and genetic testing in selected cases (Table 1).
cAVM: cerebral arterio-venous malformations; HTN: hypertension; RSSI: recent small subcortical infarct.
Attention is needed to avoid both overlooking rarer forms and underestimating frequent ones.
Threshold between how far to persist in the diagnostic workup, and in whom genetic testing should be performed, is yet to be defined.
MRI superior to CT for a complete assessment of cSVDradiological features. If limited access to MRI, consider if MRI-findings would benefit the individual, and/or change their clinical management. Complete clinical history + examination should guide decision.
Although this two-step approach may facilitate clinical practice, there are limitations and uncertainties. For instance, studies have reported almost similar AF detection at 12-months follow-up in participants with stroke attributed to large vessel and small vessel causes (11.7% vs 12.7%, respectively) 16 ; and new subcortical CBI (NS-CBI) on MRI after 2-years in 2.7% (n = 33/1227) of AF-participants, despite that 78.8% (n = 26/33 NS-CBI) of them were anticoagulated at the 2-year scan time. 17 Yet, the clinical relevance of insertable cardiac monitoring in people with cSVD remains unknown, and current clinical guidelines do not recommend routine long-term continuous monitoring for AF detection for individuals with stroke attributed to cSVD.6,7 Furthermore, the diagnostic workup for patients with cSVD proposed by current guidelines is mostly based on non-cSVD-related stroke trials.18–21 This emphasises the need for further research and the development of optimal clinical diagnostic and management strategies for cSVD.
How to handle the not-so-rare genetic causes of cSVD?
As with the sporadic cSVD form, the genetic forms of cSVD represent a diagnostic challenge. Clinical and neuroimaging characteristics could help identify monogenic causes of cSVD (sTable3). However, it is common to find patients with overlapping phenotypes and sometimes with more than one genetic cause of cSVD. Some clinical services dedicated to genetic forms of cSVD have responded by developing their gene panel strategy, for example, the CERVCO 22 (‘Centre de référence pour les maladies vasculaires rares du cerveau et de l’œil’) (sBox3); but no standardised gene panels exist between centres or countries.
Furthermore, when considering a person with a confirmed diagnosis of a genetic cSVD cause, healthcare should be extended to family members including pre-symptomatic individuals. Some genetic cSVD services have developed well-established programmes for this.22,23
The CERVCO (Paris, France) experience for pre-symptomatic genetic testing
The pre-symptomatic genetic testing for cSVD is a multidisciplinary and multi-step strategy that includes three initial separate interviews with a neurologist, geneticist and psychologist. Afterwards, the eligible pre-symptomatic individual is given ⩾2-months time before attending for blood sampling conducted in the presence of a neurologist once the person has consented. Results are delivered at least 2-months after blood sampling to provide time to the person to re-consent about the procedure. The individuals can withdraw from the genetic testing at any time.
The pre-symptomatic genetic testing for CADASIL or COL4A1/A2 gly mutation is offered to a relative of a genetically confirmed patient at CERVCO. CADASIL testing aims to help the person anticipate personal and social adjustments to cope with the results. The clinical assessments include motivations to take the test, the individual’s perception of the disease, and the provision of information regarding CADASIL.
The COL4A1/A2 gly mutation testing aims to provide information. Despite there being no treatment of proven efficacy, genetic testing could provide medical benefit in terms of preventive measures to reduce the risk of haemorrhagic complications; including blood pressure control, vigorous or high-impact exercise not recommended, caesarean section to be considered and dedicated follow-up for aneurysm or renal failure.14,15,24
After the confirmation of a genetic cSVD diagnosis, a multidisciplinary team provides psychological support (e.g. risk of being a carrier for relatives), rehabilitation (e.g. cognitive therapy), disease information (e.g. SOS information card, patient’s association), pregnancy and labour information, social worker support (e.g. optimise financial help, maintain social/professional inclusion, optimal return to home); and for their relatives’ disclosure of genetic information and information regarding pre-symptomatic diagnosis. In addition, the patients receive clinical and imaging follow-up performed on a hospitalisation day at least every 2 years. 22
Currently, the care organisation for genetic cSVD in France involves 24 network expertise centres distributed around the country and the CERVCO centre in Paris. Similar national models of care could be implemented in other countries.
What and how to implement optimal current therapeutics for sporadic csvd?
Although sporadic cSVD has been traditionally divided in two clinical settings, covert and overt, both forms of the disease co-exist in the same person, and individuals do not follow a clear lineal evolution. 25 Therefore assessment and management should be repeated and standardised.
In the past years, much attention has been addressed to the ‘classical’ vascular risk factors, as hypertension is considered the main preventable and treatable risk factor for cSVD. Although lifestyle interventions may have limited impact on cognitive decline, 26 there is evidence that cSVD patients have similar vascular risk factors as other stroke subgroups 27 and that effectively treating hypertension can slow down the progression of WMH on MRI, and reduce stroke and dementia risk. 7 Yet, prevention and management of hypertension and other modifiable risk factors (e.g. diabetes, overweight) is suboptimal, in all but especially in low- and middle-income countries. 28
Guidelines for covert cSVD have explicitly been based on WMH and lacunes features of cSVD7,8,13 (sTable4). A recent guideline addressed six questions to assist the clinical decisions for the management of people with covert cSVD. 7 However, the topic suffers from insufficient evidence in several areas and it is important to consider that some of this scarcity of knowledge in the covert cohort derives from a lack of standardised management and poor follow-up.
Considering the symptomatic forms of cSVD, guidelines approach these individuals based on their main clinical presentation, which reflects the fragmented clinical attention that this cohort receives instead of a multidisciplinary approach6,8,18,20,29–32 (sTable5).
Conclusion
cSVD has a high prevalence and severe clinical and socio-economic impacts worldwide. To date, healthcare systems have not been able to provide a successful standard identification, assessment and management of the whole spectrum of the disease in part due to a lack of general population and healthcare professional awareness of cSVD, and the shortage of dedicated services for cSVD. Genetic cSVD clinics have been developed in some countries, and successful national strategies have been used. However, many sporadic cSVD individuals are left without standardised holistic management. Creating cSVD-dedicated services would (a) Standardise the assessment and management for these individuals; (b) Avoid duplicating consultations; (c) Prevent underdiagnosis; (d) Help to prevent future stroke and dementia and (e) Provide more insight into the pathophysiology of cSVD and opportunities to develop targeted treatments. Accomplishing such change requires efficient and practical healthcare systems, pathways and policies that can address global inequalities in accessibility to healthcare. Additional global challenges, such as shortage of staff, and population and medical education and training, should be addressed to bridge the systemic failure to translate available scientific evidence into standard operational clinical practices covering different levels of care.
Recommendations
Future steps should include identifying other cSVD-dedicated clinical service models and gathering information regarding current experience. Additionally, information from experienced researchers in the field will help translate clinical research models in cSVD to real-world experience. A cSVD clinic should provide care to people with overt and covert cSVD, and avoid duplication with existing clinical services. Multidisciplinary team sessions and developing local, national and international networks are necessary to build standardised care programmes for individuals with cSVD. Specific recommendations for implementing cSVD-dedicated services is beyond the role of this paper. However, the information provided here may be generalisable to existing/future services. Further research studies on cSVD patients should be nested in the clinical services to achieve more impact in the benefit of people with cSVD.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241309506 – Supplemental material for Time to consider health services dedicated for adults living with cerebral small vessel disease: Report of a ESO scientific seminar
Supplemental material, sj-docx-1-eso-10.1177_23969873241309506 for Time to consider health services dedicated for adults living with cerebral small vessel disease: Report of a ESO scientific seminar by Carmen Arteaga-Reyes, Dwaipayan Sen, Salvatore Rudilosso, Eric Jouvent, Dominique Hervé, Arne G Lindgren, Joanna M Wardlaw, Susanna Melkas and Fergus N Doubal in European Stroke Journal
Footnotes
Acknowledgements
We thank all the participants of the discussion held during the eighth ESOC scientific session in cSVD and the ESOC Planning Group for accepting the Scientific Seminar proposal.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors hold academic grants from government and charitable funding agencies as outlined below. A.G.L reports personal fees from Bayer, NovoNordisk, Astra Zeneca, Arega and BMS Pfizer outside this work. S.R., D.H., A.G.L., J.M.W., F.N.D. acknowledged involvement in the ESO guidelines. J.M.W. chaired the ESOC-2022 planning group; the scientific session proposal relevant to this work was assessed independently by other ESOC planning group members. The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the UK Dementia Research Institute which receives its funding from DRI Ltd, funded by the UK Medical Research Council, Alzheimer’s Society and Alzheimer’s Research UK (C.A.R., J.M.W.); The Row Fogo Centre for Research into Ageing and the Brain (ref 486 (AS-CP-18b-001);C.A.R., J.M.W.); The Mexican National Council of Humanities Sciences and Technology (CONAHCYT, 2021-000007-01EXTF-00234) and the Rowling Clinic (C.A.R.); the Stroke Association Garfield Weston Foundation Senior Clinical Lectureship (TSALECT 2015/04) and NHS Research Scotland (F.N.D.). Additional funding reported: the ‘Instituto de Salud Carlos III’ Grant for Health Research (RH041992; JR21/00011; S.R.); Centre de Neurologie Vasculaire Translationnelle, Assistance Publique Hôpitaux de Paris, Hôpital Lariboisière, France (D.H., E.J.); and the Lund University, Skane University Hospital, Swedish Heart and Lung Foundation, Swedish Research Council (A.G.L.)
Ethical Approval – Informed Consent
This work did not require an ethical approval or written informed consent according to the study design (review article).
Guarantor
F.N.D
Contributorship
C.A.R., F.N.D., J.M.W. – Conceptualisation, methodology. J.M.W. – Proposed scientific seminar to ESOC Planning Group. F.N.D., S.M. – Coordinated scientific session. D.S., S.R., E.J., D.H., A.G.L. – Spoke at scientific session, each speaker addressed one main topic from this work. C.A.R. – Investigation, writing original draft. All authors – Writing, review and editing of the manuscript, and approved the final version of the manuscript.
Data availability
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.