Abstract
Academic research from years past has increasingly come under scrutiny for a variety of reasons. While it is imperative that flawed studies based on obvious research misconduct receive notices of retraction or expressions of concern, it is unclear to what extent these corrective actions apply to published research that is historical, based on clearly unethical human experimentation, and already de facto retracted based on unanimous views within the academic community. The controversy over the use of unethically obtained specimens and data derived from them for research and education is not new to bioethics, but the US Public Health Service project, the Tuskegee Study of Untreated Syphilis (TS), has not been subjected to such scrutiny. We identified 19 articles published under 13 different journal titles between 1936 and 1973 that detailed the results of TS. We examined journal records and found that none of the 19 selected publications derived from TS ever received a retraction notice or expression of concern. Only one of these publications has an accompanying commentary for its online publication context. We discussed various options for potentially amending journal archives containing TS publications, reviewed guidelines from CIOMS, COPE, and ICMJE. We suggest that academic journals establish a clear policy and guidelines on publications based on unethical research, for issuing a retraction for unethical research, as clear as the policy for research misconduct, and that a grace period be introduced into academic publishing to cover unethical research. To promote the publication ethics of correcting the scientific record, we urge the original or successor journals that published TS to consider adding notices to the articles as they deem appropriate.
Introduction
Academic journals have renewed their interest in amending the scientific record by focusing on their journal archives. Increasingly, academic research from years past has come under scrutiny for a variety of reasons. High-profile journals have also begun to purge their archives of articles based on flawed scientific research. Most recently, Nature has been investigating its role throughout history in publishing harmful contributions based on discrimination and intolerance (Nature, 2022). However, some journals have grown more conscious of purging their archives of papers that propagate violations of the protection of human subjects or other ethically indefensible features (Marcus and Oransky, 2020).
The controversy over the use of unethically obtained specimens and the data derived from them for scientific publications and medical teaching is not new to bioethics but has been extensively discussed in the context of Nazi experiments (Czech et al., 2021). In particular, Eduard Pernkopf’s Anatomical Atlas forced German and Austrian anatomical institutes to confront their past, especially their widespread procurement of bodies from victims of National Socialism (Angetter, 2000). Surprisingly, however, the United States Public Health Service (USPHS) project, the Tuskegee Study of Untreated Syphilis (also known as TSUS; TS), has not been subjected to such scrutiny (White, 2005).
While it is imperative that flawed studies based on obvious research misconduct receive retractions or expressions of concern, it is unclear to what extent these corrective actions apply to published research that (1) is more than 50 years old, (2) is based on clearly unethical human subject experimentation, and (3) has already been de facto retracted based on the academic community’s unanimous view.
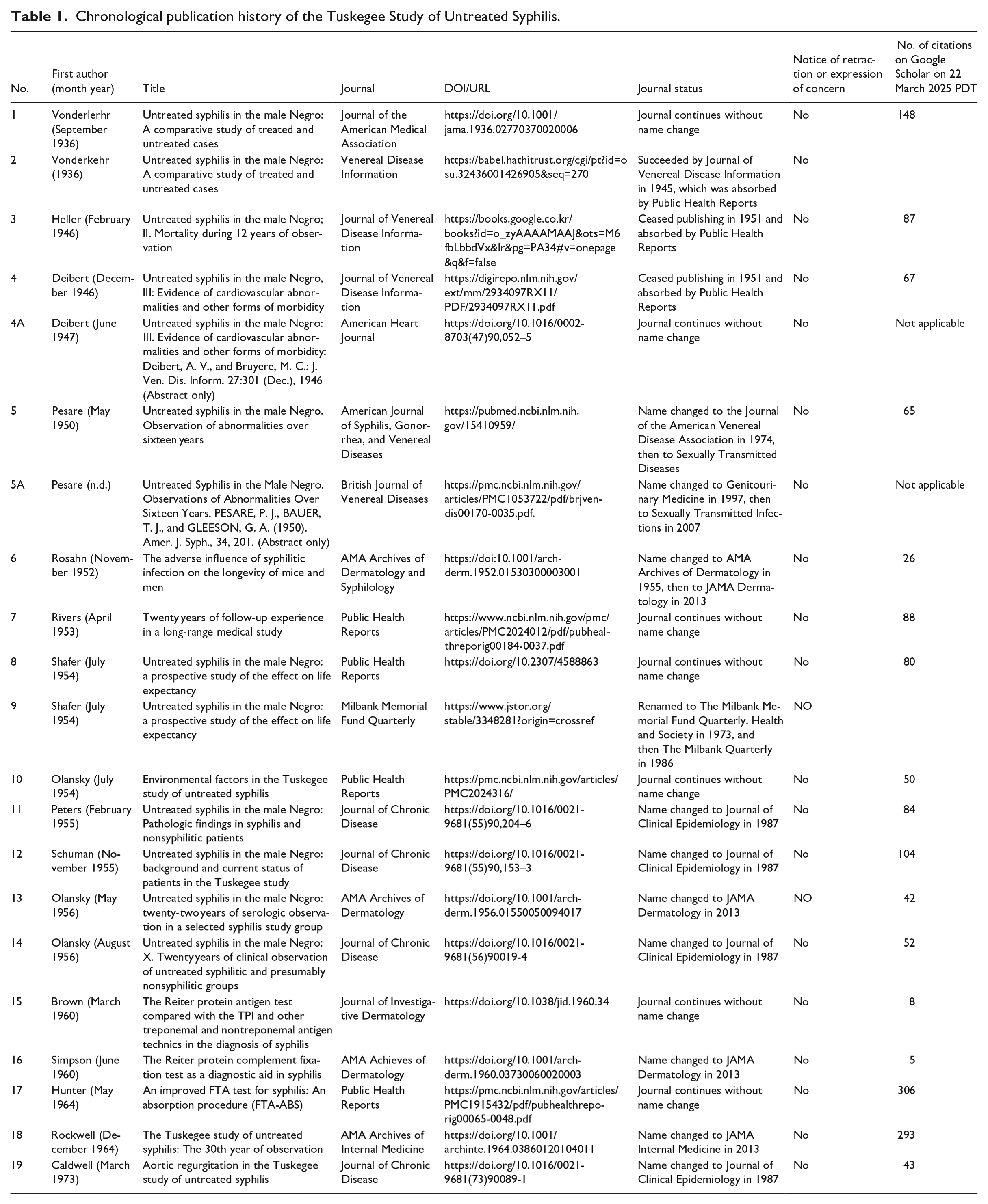
This study presents 19 articles reporting the results of TS. These articles were published under 13 different journal titles between 1936 and 1973 and can still be searched for. The study raises the question of whether the journals that published these articles based on unethical research should, 52–89 years later, consider adding notices to them as they deem appropriate. The study reports on the publication history, post-publication notices and citations of these articles, and discusses potential challenges alongside guidance from others and our own recommendations.
Methodology
Given these factors, we assess the role of these publication practices based on TS. Academic publications derived from TS remain part of the scientific record in their current form, despite the notoriety of the study and scholarly agreement that the study was conducted unethically (U.S. Department of Health, Education, and Welfare Public Health Service, 1973, Final report of the Tuskegee syphilis study ad hoc advisory panel, April 28).
We have identified publications from 1936 to 1973 that detail results derived from TS (White, 2000: 589) provided an exhaustive summary of the publications, counting “14 known articles (two articles were published twice) about TSUS published under nine different journal titles.” Additionally, we found that two of White’s were published in abstract form only under two other journal titles. We also found that among 143 publications citing the first article reported by Vonderlehr et al. (1936), there were five other publications based on the results of TS. Based on this list of publications, we examined journal records to determine whether any retractions or expressions of concern had been published, and we searched Google Scholar for the number of citations to each of the 19 publications. We also looked at other journal publication practices related to the studies.
The Retraction Watch Database contained 64,772 retracted publications as of 27 May 2025 (GitLab, 2025). An initial search was conducted using the Retraction Watch online database of retracted publications. Title searches for keywords including “untreated syphilis,” as well as searches of each publication individually, yielded no results for these 19 articles. These results were then cross-referenced with the original and current journal pages. Keyword searches were also conducted in PubMed and Google Scholar to identify potential duplicate publications of the Tuskegee articles. The keywords used were “Tuskegee,” “Macon County,” and “untreated syphilis.”
Results
We trace the origins of TS through its published reports over several decades in the academic literature, revealing its legacy in the archives of major journals today. We also report on five other publications, including two in the journal of the American Medical Association (AMA). These are new additions to other scholarly investigations of the publications generated by TS. We do not intend this list of 19 publications to be exhaustive.
These publications derived from TS did not contain any retractions or expressions of concern. Viewed in isolation, only one of these publications has an accompanying editorial comment for its online publication context (Buncher, 2006: 234). Here the author of the Commentary, C. Ralph Buncher, stated that all medical researchers who should have read the study and did not react to the word “untreated” should be ashamed of themselves.
Chronological Publication History
A series of reports on a study of acquired syphilis in the male negro in Macon County, Alabama were published over the years from 1936 to 1973. Table 1 in the Appendix gives a chronological list of the 19 reports on TS published in 13 different medical journals. Two of the 19 articles were published twice (Nos. 1 and 2; and Nos. 8 and 9). These duplicate publications occurred before policies against duplicate publication, such as the Ingelfinger rule, were formalized (New England Journal of Medicine, 1969). The abstracts of a further two articles were published under different journal titles to the original publication in 1947 and in about 1950 (Nos. 4A and 5A).
All the 13 journals still exist, although nine have changed their names. Three of these are very well-known journals, such as the Journal of the American Medical Association (JAMA), JAMA Internal Medicine and JAMA Dermatology.
Publication in September 1936
A comparative study of treated and untreated cases from TS was read to the Section on Dermatology and Syphilology at the 87th Annual Session of the AMA conference in Kansas City, Missouri, on 15 May 1936. It was discussed in academic circles but did not attract public attention. This research was reported in both JAMA and Venereal Disease Information (Nos. 1 and 2). The Venereal Disease Information was succeeded in 1945 by the Journal of Venereal Disease Information, which continued until 1951 when it was absorbed by Public Health Reports (Annals of Internal Medicine, 1934).
Publication in February 1946
Mortality during the 12-year observation period of TS was reported in the Journal of Venereal Disease Information, February 1946 (No. 3). As mentioned above the Journal of Venereal Disease Information continued until 1951 and was absorbed by Public Health Reports.
Publication in December 1946 and June 1947
The evidence of cardiovascular abnormalities and other forms of morbidity from TS was reported in the Journal of Venereal Disease Information in December 1946 (No. 4). The abstract of this report was also published in the American Heart Journal in June 1947 (No. 4A).
Publication in May 1950
The observation of abnormalities over 16 years of TS was reported in the American Journal of Syphilis, Gonorrhea, and Venereal Diseases in May 1950 (No. 5). The abstract of this report was also published in the British Journal of Venereal Diseases (No. 5A). The American Journal of Syphilis, Gonorrhea, and Venereal Diseases was renamed the Journal of the American Venereal Disease Association in 1974, and later Sexually Transmitted Diseases in 1977 (Welch Medical Library, n.d.a, n.d.b). The British Journal of Venereal Diseases was also renamed the Genitourinary Medicine in 1985, and then Sexually Transmitted Infections in 1998 (The Online Books Page, n.d).
Publication in November 1952
Mortality among syphilitic subjects and with the influence of syphilitic infection on longevity were reported and included four earlier reports from TS (Nos. 2, 3, 4, 5) under “Evidence from clinical findings and vital statistics” (No. 6). The AMA Archives of Dermatology and Syphilology was renamed the AMA Archives of Dermatology in 1955, and then JAMA Dermatology in 2013 (AMA Archives of Dermatology Syphilology, 1955).
Publication in April 1953
As the 20 years of follow-up of TS, the experience in a long-range medical study was reported in Public Health Reports in April 1953 (No. 7).
Publications in July 1954
A prospective study of the effect of untreated syphilis on the life expectancy from TS was reported in both Public Health Reports and the Milbank Memorial Fund Quarterly in July 1954 (Nos. 8 and 9). The Milbank Memorial Fund Quarterly was renamed the Milbank Memorial Fund Quarterly Health and Society in 1973, and then The Milbank Quarterly in 1986 (JSTOR, n.d).
In addition, a study of the background of the syphilitic and non-syphilitic participants in TS was reported, such as blood test surveys of Negroes in Macon County, trend of population distribution by race in Macon County, medical facilities, socio-economic data, diet, and age (No. 10).
Publication in February 1955
The pathological findings in syphilitic and non-syphilitic participants in TS were reported in the Journal of Chronic Disease in February 1955 (No. 11).
The Journal of Chronic Diseases published its first issue in January 1955 (Journal of Chronic Diseases, 1955). Its masthead described it as “A Journal Devoted to the Problems of Chronic Illness of All Age Groups.” The co-editors, Joseph Earle Moore of Johns Hopkins University and David Seegal of Columbia University, were established experts in the field of chronic diseases. Moore would later be described as a “noted authority on venereal diseases,” and Seegal as a “leader in chronic diseases research” (New York Times, 1957, 1972). Whatever their respective backgrounds, their journal soon became involved in the USPHS project, the “USPHS Syphilis Study at Tuskegee” (U.S. Centers for Disease Control and Prevention, 2024).
The second issue of the Journal of Chronic Diseases, published in February 1955, included the results of TS (No. 11). This report was one of a series of similar updates that had “appeared regularly in the medical press” from the early years of TS (Brandt, 1978). The Journal of Chronic Diseases changed its name to the Journal of Clinical Epidemiology in 1988 (Feinstein and Spitzer, 1988). At the time, its editors suggested that the name change was superficial and that the journal should continue its research uninterrupted by the change to reflect a broader perspective (Feinstein and Spitzer, 1988).
Publication in November 1955
The background to TS and a description of the current status of the study population were reported in the Journal of Chronic Disease in November 1955 (No. 12).
Publication in May 1956
The 22-year serological follow-up of a selected syphilis study group from TS was reported in the AMA Archives of Dermatology (No. 13). The AMA Archives of Dermatology was renamed JAMA Dermatology in January 2013.
Publication in August 1956
The 20 years of clinical observation of untreated syphilitic and presumably non-syphilitic groups of TS was reported in the Journal of Chronic Disease in August 1956 (No. 14).
Publications on serological tests for syphilis in March 1960, June 1960, and May 1964
The development of serological tests for syphilis using the specimens obtained from TS was reported in three publications as follows. Brown’s 1960 paper was presented at the 20th Annual Meeting of the society for Investigative Dermatology, Atlantic City, N. J., 7 June 1959 (No. 15; Brown et al., 1960). Simpson’s 1960 paper was read to the Section on Dermatology at the 108th Annual Meeting of the American Medical Association in Atlantic City, N. J., 12 June 1959 (No. 16; Simpson et al., 1960). Simpson’s paper also included additional details on cases of TS. Hunter’s 1964 paper used 82 specimens from the Olansky’s previous two 1962 papers (No. 17; Hunter et al., 1964: 410).
Publication in December 1964
The 30th year of observation from TS was reported in the AMA Archives of Internal Medicine in December 1964 (No. 18). JAMA Internal Medicine is the current name of the AMA Archives of Internal Medicine.
Publication in March 1973
Findings of aortic regurgitation in syphilitics and controls with TS were reported in the Journal of Chronic Disease in March 1973 (No. 19).
2006 Commentary on the July 1954 publication
In 2006, Public Health Reports, which inherited four TS-derived articles as a successor journal in 1951 and itself published four more TS-derived articles between 1953 and 1964, took an ameliorative approach to its publications of TS, specifically referencing its three articles published in 1953 and 1954 that contained the word “untreated” in their titles (see Appendix Table 1). To mark the digitization of its archives, Public Health Reports devoted its Volume 121 Issue 1 Supplement for January–February 2006 to a series of more than 30 commentaries, each of which introduced and retroactively responded to historic Public Health Reports publications from the 20th and 19th centuries. One of these was a commentary responding to the 1954 publication, more than 50 years after the publication of the article and the death of all the participants in the study, and criticizing the practices that led to the mistreatment of the participants (Buncher, 2006: 234). Its author, Buncher, highlighted the “misconceptions” about the publications arising from TS. Buncher wrote: “It was not a secret study. It was published in this peer-reviewed and widely read journal [Public Health Reports] in 1954, 18 years before the study was ended. It was published in other medical journals as well.” Buncher’s comment is by no means the longest time ever elapsed between an original article and a commentary in response, with several authors responding to Public Health Reports articles from the late 19th century.
Post-publication notices and citations
Our analysis shows that none of the 19 selected publications derived from TS ever received a retraction or expression of concern. We also find that the 19 papers have been cited in between 5 and 305 subsequent articles (see Appendix Table 1).
Discussion
We now turn to the publication ethics of our findings and the ethical obligations of JAMA and other journals.
For its violations of informed consent and its disregard of the risks to the research participants and their families, TS is universally viewed as falling below contemporary standards for research ethics, particularly those found in the Belmont Report, which was a result of TS. As noted above, none of the 19 selected publications derived from TS has ever received a retraction or expression of concern issued by publishing journals, and all the reports have been cited in varying numbers of subsequent articles. Acknowledgment of that, as part of the publication records, seems important to us in at least two ways. First, it should promote more ethical research in the future. Public retraction or other negative statements about previously published work, now recognized as unethical, increases the costs of unethical research and may deter such misconduct. It also reminds current researchers of the importance of doing ethical work, thus encouraging more compliance with ethical guidance. And second, one might argue that publication of unethical research makes a journal complicit in the wrongdoing as an “appropriator” (Kaveny, 2000). A retraction or expression of concern might be an ethical action by, at least partially and retroactively, meeting a duty of the journal itself—or its editors—to behave ethically.
In our view, journals need to balance the risks of de-historicizing and obscuring these publications with the visibility afforded by the digital transformation of journal publishing. For one thing, although TS was widely circulated prior to its closure following media exposure, there was no publicly available evidence of the unethical nature of TS. While documents in the National Archives detailed misconduct such as patient deception, journal editors and peer reviewers had no such information at the time the publications were received (White, 2000). Therefore, the potential retraction or expression of concern regarding the 19 publications derived from TS does not imply any misconduct or negligence on the part of those previously involved in their publication.
There are several ways to potentially modify the journal archives containing these publications resulting from the study.
Retractions
Undoubtedly, the most serious course of action would be to retract these articles. Even if it is necessary to cite a retracted paper, there are ways in which researchers can acknowledge this practice, such as adding a parenthetical to the retraction notice (APA, 2013; Retraction Watch, 2018). Retractions do not remove the publication entirely from the journal’s history but add more information to the retraction notice about the reasons why it is no longer considered suitable for publication. If these articles were retracted, they would remain part of the publication history, but possibly without the assumption that the science in them is still valid. While the statistics, figures and clinical conclusions may be free from error, the same cannot be said for the study as a whole.
Expressions of concern
Short of a full retraction, expressions of concern allow journals to make an interim statement about potential flaws in the study, pending an investigation. They are a stop-gap measure that can be followed up later with a retraction, but in the short term they inform readers that there may be potential problems with the publication despite a concrete conclusion. This solution is unlikely to be applicable to the 19 publications here, as there is no disagreement about the misconduct that occurred during TS. If an expression of concern were necessary, it would have been timelier in the aftermath of the investigations by Jean Heller of the Associated Press on 25 July 1972, when she broke the story of TS.
Peter Buxtun, who overhead colleagues at the Public Health Service speaking about Tuskegee, requested and received information on the then unpublicized syphilis study in Alabama (Breed, 2022). Those documents, enclosed in a plain manila envelope, were then disclosed to the Associated Press.
Before publishing the study, Heller wanted to make sure that there was additional published evidence of research misconduct. Heller placed a high value on her discovery of published results from the study in academic journals, as this convinced her that the study was a real experiment that was actually taking place (Glenn, 2022). She repeatedly cited an “obscure medical journal” that had “tracked statistics from the study” as evidence that justified her decision to go ahead with the story (Glenn, 2022).
This sentiment was echoed by Peter Buxtun in testimony before the US Senate, where he stated that the results were “regularly published” and “usually in an obscure medical journal” (U.S. Senate, Committee on Labor and Public Welfare, Subcommittee on Health, 1973).
However, it is unclear which academic journal article she was referring to, as several articles had been published at the behest of Buxtun by the time of her investigation. Although it had been circulated among medical professionals, TS was not as widely known as it is today, at least in the journalistic community.
Therefore, while an expression of concern for the 19 reports is surely unnecessary today, given the minimal visibility of some of the studies at the time of their investigation by the Associated Press and the subsequent US Senate hearings, it is debatable to what extent an expression of concern would have been an appropriate remedy for TS’s publication ethics even in 1973.
Prefacing
The publication practice of adding a contextual preface to these publications, rather than a retraction, might better support the goals of publication and research ethics. Buncher’s preface to Public Health Reports offers both condemnation and contextualization of the study itself. While not as severe in its effect as a retraction, it is perhaps in some ways more critical. It is clearly attached to publications derived from TS; therefore, any reader of these publications will see the accompanying commentary first.
Another reason why prefaces may be more appropriate is to deal with the risk of citing retracted research. When a paper is retracted, the likelihood of further citations is reduced. However, these papers are not at risk of being cited by other researchers in the field of infectious diseases who have moved on from these topics. Instead, they are being cited by researchers who need to understand the failures of the study and the tragedies of the Tuskegee Project.
Potential challenges
Firstly, before any corrections are made, the question of why it is crucial to correct historical literature on unethical research, despite the passage of time and the potential challenges that this may entail, must be answered. There must also be a consensus among publication ethics stakeholders.
Even if a consensus is reached, there will be many challenges involved in correcting historical cases of unethical research in published literature. One of these is defining unethical research, given that the definitions and criteria for what constitutes it are constantly changing. Another challenge is establishing guidelines for retraction of publications based on unethical research. Other challenges include deciding whether and how to issue notices on the publication of historical unethical research and how to remove knowledge gained from unethical studies from biomedicine.
Moreover, retracting publications on ethical grounds requires journals to conduct more extensive assessments. These assessments must consider both the scientific validity of the publications and the ethics of the research they report. However, such assessments could lead to inefficient retraction processes. This has prompted recent proposals calling for more efficient, expedited processes to verify publication integrity by focusing solely on the validity of the publication (Thorp, 2022). In practice, retractions often address both the invalidity and unethical nature of the research (Xu and Hu, 2022).
In the hypothetical case of a journal that ceased publication in the 1950s or 1960s, there is no reasonable expectation that the defunct journal should issue a retraction, expression of concern, or other clarification in the present. Modern scholars are at low risk of citing such literature for research purposes, and the current scholars familiar with these publications are often bioethicists, research ethicists, and others interested in the context of such publications rather than their scientific content. Practically speaking, if the journal is dead, who would publish such a notice in the first place?
The responsibility of journals today for their previous versions and previous namesakes should not be overstated. Journals change in scope and focus from time to time, and their publishers also influence the choice of content. Organizations that oversee or are affiliated with journals exercise their ability to shape editorial content, even if that content is the result of unethical research.
When the original journals cease publication, it is reasonable to argue that the obligation to correct the literature should lie with the publishers. However, when the original publisher no longer exists, the question arises as to which entity should assume responsibility for issuing literature-correcting notices.
There are additional considerations for journals that are still publishing but have undergone one or more name changes. For example, the following titles may not be immediately recognizable to many researchers: Venereal Disease Information, Journal of Venereal Disease Information, American Journal of Syphilis, Gonorrhea and Venereal Diseases, Milbank Memorial Fund Quarterly, Journal of Chronic Diseases, AMA Archives of Dermatology and Syphilology, AMA Archives of Dermatology, and AMA Archives of Internal Medicine. However, these journals have all changed their names to more recognizable journals, such as JAMA Dermatology. If it is a successor journal, simply under a different name, then the current version should have the authority (and obligation) to deal with its publications, albeit under a different title. This is probably permissible, provided that other factors about the journal, such as editorial content and staff, remain sufficiently similar over time. For example, the overlap between AMA Archives of Internal Medicine and JAMA Internal Medicine is significant, and readers would not be misled by a retraction notice about content from AMA Archives of Internal Medicine in JAMA Internal Medicine. The fact that they are the same journal is not surprising from the name alone.
For those journals that have used the same name throughout their publication history, there is even more evidence that they should be allowed, or even encouraged, to print retractions from more historical events. Public Health Reports has published under the same name since the publication of TS in 1953 and 1954. Although neither of these publications were retracted or received expressions of concern, the journal took a significant step into these areas of publication ethics by publishing Buncher’s commentary as a preface to the 1954 study (though not the 1953 study). It is interesting that the other publications of TS did not receive similar treatment, and that only Public Health Reports took this step. It is interesting to hypothesize a relationship between the use of journal names over time and greater scrutiny of past publications. In essence, not having published unethical research or the results of unethical research under the same name provides less pressure to revise publication history.
Nevertheless, there is an opportunity to extend current journal practices to one of the most significant bioethical issues of the 20th century, rivaling some of the worst experiments of the Second World War. While the threat of litigation may cause some journals to be cautious about making corrections to scientific publications, the seemingly universal agreement on the unethical nature of the research, as well as the distance between the present and the original publication dates, suggests that this is a remote possibility for TS publications (Oransky, 2022). For papers published several decades ago, there is precedent for offering an expression of concern rather than a retraction for research that fails to meet modern ethical standards (Retraction Watch, 2020).
Guidance from others
The Council for International Organizations of Medical Sciences (CIOMS), in collaboration with the World Health Organization (WHO), have previously emphasized the Tuskegee study as one of the most infamous cases of unethical research in the 20th century in its International Ethical Guidelines for Epidemiological Studies. About sanctions against researchers who violate ethical standards in the conduct of research involving human subjects, the CIOMS stated that “unless there are persuasive reasons to do otherwise, editors should refuse to publish the results of research conducted unethically and retract any articles that are subsequently found to have been based on unethical research.” The CIOMS also noted that “such sanctions, however, may deprive of benefit not only the errant researcher or sponsor but also that segment of society intended to benefit from the research,” and therefore “such possible consequences merit careful consideration.”
The Committee for Publication Ethics (COPE) has also formulated guidelines for publication ethics on the Tuskegee syphilis study, citing it as a case “in the scholarly record of violations of human rights in research where there was no standard voluntary or informed agreement by the subjects of the research as a condition of participation” (COPE Council, 2021). The Retraction Guidelines issued by COPE state that editors should consider retracting a publication “if it reports unethical research” and state that retraction, as relatively severe sanctions, may be used to alert readers to instances of unethical research that would have unduly influenced interpretations of the work or recommendations by peer reviewers (COPE Council, 2019, 2021).
The International Committee of Medical Journal Editors (ICMJE) Recommendations for the Conduct, Reporting, Editing, and Publication of Scholarly Work in Medical Journals, updated in January 2025, state that all investigators should ensure that the design, conduct, and reporting of research involving human subjects comply with the Declaration of Helsinki as revised in 2013. It also states that all authors should seek approval to conduct research from an independent review body, such as an institutional review board, and be prepared to provide documentation when requested by editors.
Recommendations
We recommend that academic journals establish a clear policy and guidelines for retracting unethical research, as clear as the policy and guidelines for research misconduct such as fabrication, falsification, and plagiarism. Each scientific journal need to develop its own ethical framework or quality standards for research ethics that its editors can accept, that is, when an unethical study is disqualified from publication and what ethical parameters must be considered in deciding qualification for scientific publication. For publications related to a past wrong, such as the Nazi experiment and TS, the journal should be able to recognize potential appropriation problems as described by Kaveny (2000). At the very least, all editors of scientific journals should raise awareness of the ethics of the origins of tissues and data used in human research by establishing a practice of full transparency regarding them.
Retraction of research is a serious matter and very compromising for researchers. It is therefore important to clarify what forms and degrees of unethical behavior are sufficient to warrant retraction. The journal should provide examples of each category of unethical research. All retractions should be recorded in the journal and the reasons for the retraction should be stated in terms of both the type and degree of unethical behavior. For example, in addition to the 2006 editorial expression of concern and retraction (Kennedy, 2006a, 2006b) based on research misconduct and fabrication of data, Science might consider publishing a new notice on each of Hwang Woo Suk’s two papers, published in 2004 (Hwang et al., 2004) and 2005 (Hwang et al., 2005), that the papers violate Science’s current Human Subjects Protection Policy as well as the Korean Bioethics and Safety Act, retroactive to 2025, based on the institutional or court finding of unethical oocyte donation, if any.
We do not propose that this obligation apply to all unethical research, no matter how old. At some past date, a large portion of research would violate current ethical standards, and only a small part of that research would ever be cited or read. Otherwise, journals may argue that the volume of early 20th-century publications that no longer pass the ethical research smell test is too great to retract individually, and that trying to do so would have the effect of diluting their entire archives and render much of their historical research obsolete. Thus, journals might commit to retracting all their publications of unethical research where they became aware of the problem within, say, 50 years of publication. Fifty years should normally cover the span of the career of most authors and thus might be reasonable date, although the policies should allow retractions of older research in exceptional cases, such as when the research continues to be important, scientifically, or ethically, as is the case with TS publications, all of which are more than 50 years old.
TS is different from other cases because of scale and prominence. That could support the view that only the Tuskegee publications should be withdrawn in the interest of justice, partly in response to the exceptional nature of Tuskegee and its role in the Belmont Report. We believe, however, that a more general policy is more ethically appropriate and, if generally limited in time, is unlikely to impose substantial burdens on journals.
Tuskegee has also raised issues of “restorative justice,” including highly publicized acknowledgments and apologies, and even some financial compensation (Mitchell, 1997). However, this article has been concerned with the ethical issues surrounding the correction of the scientific record. We do not consider the extent to which retractions or other measures might be part of such restorative justice.
Therefore, to promote the publication ethic of correcting the scientific record, we urge the original or successor journals that published TS to consider adding notices to the articles as they deem appropriate, announcing that the serious ethical violations of TS, set out in the Final Report of the Tuskegee Syphilis Study Ad Hoc Advisory Panel, violate the current publication policies of journals and the Common Rule.
Footnotes
Appendix
Chronological publication history of the Tuskegee Study of Untreated Syphilis.
| No. | First author (month year) | Title | Journal | DOI/URL | Journal status | Notice of retraction or expression of concern | No. of citations on Google Scholar on 22 March 2025 PDT |
|---|---|---|---|---|---|---|---|
| 1 | Vonderlerhr (September 1936) | Untreated syphilis in the male Negro: A comparative study of treated and untreated cases | Journal of the American Medical Association | https://doi.org/10.1001/jama.1936.02770370020006 | Journal continues without name change | No | 148 |
| 2 | Vonderkehr (1936) | Untreated syphilis in the male Negro: A comparative study of treated and untreated cases | Venereal Disease Information | https://babel.hathitrust.org/cgi/pt?id=osu.32436001426905&seq=270 | Succeeded by Journal of Venereal Disease Information in 1945, which was absorbed by Public Health Reports | No | |
| 3 | Heller (February 1946) | Untreated syphilis in the male Negro; II. Mortality during 12 years of observation | Journal of Venereal Disease Information | https://books.google.co.kr/books?id=o_zyAAAAMAAJ&ots=M6fbLbbdVx&lr&pg=PA34#v=onepage&q&f=false | Ceased publishing in 1951 and absorbed by Public Health Reports | No | 87 |
| 4 | Deibert (December 1946) | Untreated syphilis in the male Negro, III: Evidence of cardiovascular abnormalities and other forms of morbidity | Journal of Venereal Disease Information | https://digirepo.nlm.nih.gov/ext/mm/2934097RX11/PDF/2934097RX11.pdf | Ceased publishing in 1951 and absorbed by Public Health Reports | No | 67 |
| 4A | Deibert (June 1947) | Untreated syphilis in the male Negro: III. Evidence of cardiovascular abnormalities and other forms of morbidity: Deibert, A. V., and Bruyere, M. C.: J. Ven. Dis. Inform. 27:301 (Dec.), 1946 (Abstract only) | American Heart Journal | https://doi.org/10.1016/0002-8703(47)90,052–5 | Journal continues without name change | No | Not applicable |
| 5 | Pesare (May 1950) | Untreated syphilis in the male Negro. Observation of abnormalities over sixteen years | American Journal of Syphilis, Gonorrhea, and Venereal Diseases | https://pubmed.ncbi.nlm.nih.gov/15410959/ | Name changed to the Journal of the American Venereal Disease Association in 1974, then to Sexually Transmitted Diseases | No | 65 |
| 5A | Pesare (n.d.) | Untreated Syphilis in the Male Negro. Observations of Abnormalities Over Sixteen Years. PESARE, P. J., BAUER, T. J., and GLEESON, G. A. (1950). Amer. J. Syph., 34, 201. (Abstract only) | British Journal of Venereal Diseases | https://pmc.ncbi.nlm.nih.gov/articles/PMC1053722/pdf/brjvendis00170-0035.pdf. | Name changed to Genitourinary Medicine in 1997, then to Sexually Transmitted Infections in 2007 | No | Not applicable |
| 6 | Rosahn (November 1952) | The adverse influence of syphilitic infection on the longevity of mice and men | AMA Archives of Dermatology and Syphilology | https://doi:10.1001/archderm.1952.01530300003001 | Name changed to AMA Archives of Dermatology in 1955, then to JAMA Dermatology in 2013 | No | 26 |
| 7 | Rivers (April 1953) | Twenty years of follow-up experience in a long-range medical study | Public Health Reports | https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2024012/pdf/pubhealthreporig00184-0037.pdf | Journal continues without name change | No | 88 |
| 8 | Shafer (July 1954) | Untreated syphilis in the male Negro: a prospective study of the effect on life expectancy | Public Health Reports | https://doi.org/10.2307/4588863 | Journal continues without name change | No | 80 |
| 9 | Shafer (July 1954) | Untreated syphilis in the male Negro: a prospective study of the effect on life expectancy | Milbank Memorial Fund Quarterly | https://www.jstor.org/stable/3348281?origin=crossref | Renamed to The Milbank Memorial Fund Quarterly. Health and Society in 1973, and then The Milbank Quarterly in 1986 | NO | |
| 10 | Olansky (July 1954) | Environmental factors in the Tuskegee study of untreated syphilis | Public Health Reports | https://pmc.ncbi.nlm.nih.gov/articles/PMC2024316/ | Journal continues without name change | No | 50 |
| 11 | Peters (February 1955) | Untreated syphilis in the male Negro: Pathologic findings in syphilis and nonsyphilitic patients | Journal of Chronic Disease | https://doi.org/10.1016/0021-9681(55)90,204–6 | Name changed to Journal of Clinical Epidemiology in 1987 | No | 84 |
| 12 | Schuman (November 1955) | Untreated syphilis in the male Negro: background and current status of patients in the Tuskegee study | Journal of Chronic Disease | https://doi.org/10.1016/0021-9681(55)90,153–3 | Name changed to Journal of Clinical Epidemiology in 1987 | No | 104 |
| 13 | Olansky (May 1956) | Untreated syphilis in the male Negro: twenty-two years of serologic observation in a selected syphilis study group | AMA Archives of Dermatology | https://doi.org/10.1001/archderm.1956.01550050094017 | Name changed to JAMA Dermatology in 2013 | NO | 42 |
| 14 | Olansky (August 1956) | Untreated syphilis in the male Negro: X. Twenty years of clinical observation of untreated syphilitic and presumably nonsyphilitic groups | Journal of Chronic Disease | https://doi.org/10.1016/0021-9681(56)90019-4 | Name changed to Journal of Clinical Epidemiology in 1987 | No | 52 |
| 15 | Brown (March 1960) | The Reiter protein antigen test compared with the TPI and other treponemal and nontreponemal antigen technics in the diagnosis of syphilis | Journal of Investigative Dermatology | https://doi.org/10.1038/jid.1960.34 | Journal continues without name change | No | 8 |
| 16 | Simpson (June 1960) | The Reiter protein complement fixation test as a diagnostic aid in syphilis | AMA Achieves of Dermatology | https://doi.org/10.1001/archderm.1960.03730060020003 | Name changed to JAMA Dermatology in 2013 | No | 5 |
| 17 | Hunter (May 1964) | An improved FTA test for syphilis: An absorption procedure (FTA-ABS) | Public Health Reports | https://pmc.ncbi.nlm.nih.gov/articles/PMC1915432/pdf/pubhealthreporig00065-0048.pdf | Journal continues without name change | No | 306 |
| 18 | Rockwell (December 1964) | The Tuskegee study of untreated syphilis: The 30th year of observation | AMA Archives of Internal Medicine | https://doi.org/10.1001/archinte.1964.03860120104011 | Name changed to JAMA Internal Medicine in 2013 | No | 293 |
| 19 | Caldwell (March 1973) | Aortic regurgitation in the Tuskegee study of untreated syphilis | Journal of Chronic Disease | https://doi.org/10.1016/0021-9681(73)90089-1 | Name changed to Journal of Clinical Epidemiology in 1987 | No | 43 |
Ethical considerations
The authors declare that research ethics approval was not required for this study.
Author contributions
M.K. designed and performed the study, analyzed the data, wrote the manuscript, and approved the final version of the manuscript; C.S. designed and performed the study, analyzed the data, wrote the manuscript, but was not able to approve the final version of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
