Abstract
In Jordan, research ethics committees exist in most health settings. However, little is known about Jordanian public views regarding the ethics of clinical research. This study aimed to evaluate Jordanian public understanding and attitudes about ethics in clinical research. A questionnaire was used to collect information that included demographics, public knowledge, and attitudes towards ethics in clinical research. It was administered via face-to-face interviews in two major cities in Jordan from 1st June to 15th August 2017. Of the 2315 respondents, 2.33% were found to have poor knowledge, 22.16% had fair knowledge, and 75.51% had good knowledge of ethics in clinical research. Furthermore, 75.81% of respondents had positive attitudes towards ethics in research. However, only 45.23% reported that they trust clinical researchers in Jordan. Even though a large majority of respondents were aware of key features of research ethics, efforts are needed to address negative perceptions and knowledge deficits.
Background
Research that involves human participants should be guided by essential ethical values to protect participants’ welfare and ensure that rights are maintained (Normile, 2008; WMA, 2013). For clinical research, there are well developed ethical requirements that are accepted and applied worldwide (Abdur Rab and Mamdouh, 2004; Mathur and Swaminathan, 2018; Normile, 2008; Vallotton, 2010; WMA, 2013). Jordan was the first Arab nation to legislate for clinical trials and is now one of the leading clinical trial locations with regulatory settings in the Arab World. There are 22 approved and licensed Research Ethics Committees (REC) in Jordan, which are located in academic and private settings, and hospitals (Al-Omari and Al-Hussaini, 2017; Shafout and Al Mahrouq, 2014). The total number of clinical studies submitted for approval between 2005 and 2013 were as follows: 1471 bioavailability/ bioequivalence, 47 Phase I, 42 Phase III, 29 Phase IV, and 17 Phase II clinical trials (Arouri et al., 2015). Despite this, little is known about Jordanian public understanding and perceptions regarding the ethical aspects of clinical research.
Previous studies conducted in Jordan focused on faculty members and healthcare workers. For instance, one study found that Jordanian faculty members had a good understanding of REC functions but lack awareness of their specific tasks (Ayoub et al., 2019). Another study reported that more than 50% of resident doctors had a basic knowledge of REC requirements but had minimal knowledge of major ethical guidelines (Al Demour et al., 2019). A further related study evaluated Jordanian parents’ knowledge and views of medical research with children. Around 72% of parents were found to be knowledgeable about the ethical aspects of research with children and 76.3% were willing to allow their children to participate in research (Almomani et al., 2020).
It is important that the general public have at least a basic understanding of ethics in research to foster public accountability in research (Longstaff and Burgess, 2010). Understanding of clinical research can have an impact upon recruitment; higher levels of awareness of CTs has been associated with greater willingness to participate (Lim et al., 2017). Furthermore, lack of understanding and awareness can render potential participants vulnerable to exploitation in research (Cook et al., 2018). The aim of this study was to investigate understanding and attitudes of the general public about ethics in clinical research. It is the first of its kind to be conducted in Jordan.
Methods
The method of choice was a cross-sectional survey of members of the Jordanian public. The data collection period ran from 1st June to 15th August 2017. Participants were recruited via convenience sampling and the questionnaires completed via face-to-face interviews with the researchers. Adults, aged 18 years or older, were approached in public places (e.g. malls, parks, transport stations, coffee shops) in two major cities in Jordan (Irbid and Amman).
Items for the questionnaire were formulated following a literature review of related studies. The researchers reviewed a draft of the questionnaire for content and face validity and it was piloted on a sample of 50 people to collect feedback and make revisions. The final questionnaire included 38 items and took around 10 minutes to complete. Data from the pilot sample were excluded from the final analysis.
The questionnaire was initially written in the English language. It was translated into Arabic and subsequently translated back into English to check for any inconsistencies. Two independent researchers conducted the forward and backward translation process between Arabic and English. The Arabic version of the questionnaire was used for data collection.
The 38-items in the questionnaire were structured around the following four domains:
Part I. Demographics of participants (e.g. age, occupation, educational level and previous participation in clinical research).
Part II. Public knowledge of ethics in clinical research: Knowledge scores were based upon ‘correct’, ‘incorrect’, and ‘do not know’ responses for 12 questions.
Part III. Attitudes of the public towards clinical research: Responses to attitudes questions (n = 7) were collected using a 5-point Likert Scale (strongly disagree, disagree, uncertain, agree and strongly agree).
Part IV: Public perception of unethical research: This section focused on vulnerable populations (e.g. children, pregnant women) and other research practices. The questions were administered as ‘yes’ or ‘no’ questions.
Regarding knowledge scores, each correct response was scored as 1. The total score ranged from 0-12. Scores of 0–5 were classified as ‘poor knowledge’, scores of 6–9 as ‘fair knowledge’, and scores of 10-12 as ‘good knowledge’. The seven attitudes items were scored on a 5-point Likert Scale from 1 (strongly disagree) to 5 (strongly agree) except item 4, which was reversed. The potential total score ranged from 7 to 35. The cut-off for the attitude score was 28 (⩾80% of overall score); attitude scores of less than 28 were considered a ‘negative attitude’, while attitude scores of ⩾ 28 indicated a ‘positive attitude’.
The Jordan University of Science and Technology Institutional Review Board (IRB) committee approved the study protocol (IRB number 19/106/2017). Verbal informed consent was recorded after explaining the study to participants.
Statistical analysis
Participants’ demographic characteristics were described using frequency distribution for categorical variables and mean and standard deviation for continuous variables. The demographic characteristics of respondents were compared between the three knowledge groups (poor, fair, and good) using Chi-square test. The analysis of variance (ANOVA) test was used to examine the differences of attitude scores based on demographics. Post-hoc Tukey HSD (Honestly Significant Difference) test was conducted to determine the difference within groups. A P-value of less than or equal to 0.05 was considered statistically significant. All statistical analyses were conducted using JMP software version 10.0 (SAS Institute, Cary, NC, USA).
Results
Demographics and general information
Table 1 provides an overview of the demographic characteristics of the respondents in this investigation. Of note are that a large proportion (39.44%) were aged thirty years or less and only a small proportion (8%) of respondents were older than 60 years of age. Almost two-thirds (64.1%) were graduates with bachelor’s degree and most (76.54%) were currently working. Of those who worked, 35 worked in academia, 265 worked in a medical field, and three worked in research. When asked if they had previously participated in clinical research, only 1.17% responded in the affirmative.
Demographic characteristics of study respondents (n = 2315).
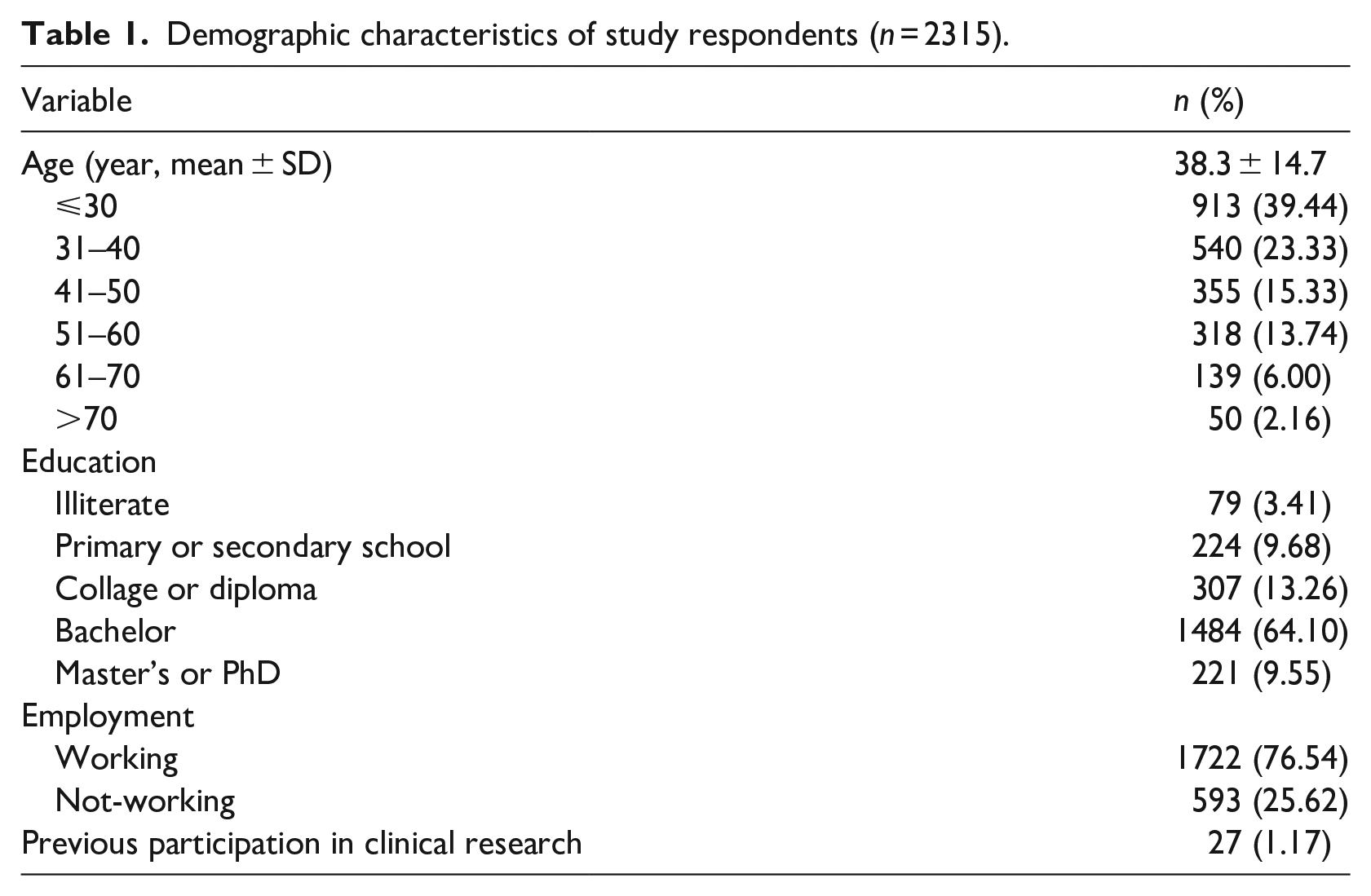
Knowledge of ethics in clinical research
Table 2 reveals responses of study participants to the twelve questions that sought to assess their knowledge of ethics in clinical research. The vast majority of respondents had knowledge of the need for respecting the rights and welfare of participants (96.2%); the need for voluntarism in research (91.84%); the role of the research ethics committee (87.9%); the need for privacy (88.8%); the nature (86.2%) and need for (83.33) informed consent. However, only just over half understood that clinical research is different from clinical treatment (53.04%) and that a participant has a right to withdraw from research at any time (52.27%).
Public knowledge of ethics in clinical research.
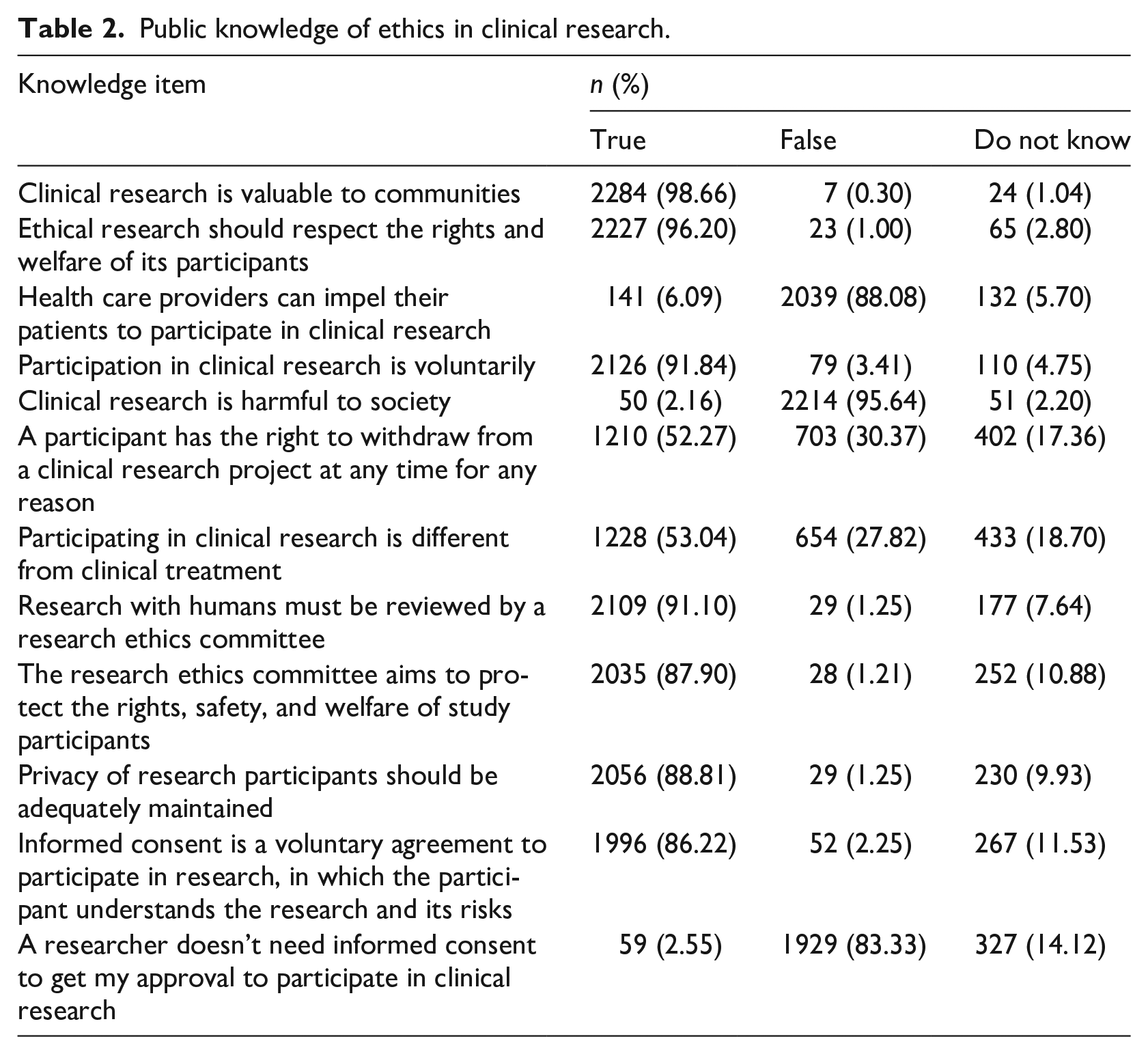
Knowledge of ethics in research was categorized as follows: 54 respondents (2.33%) had poor, 513 (22.16%) had fair, and 1748 (75.51%) had good knowledge. Interestingly, 468 respondents (20.22%) achieved a 100% knowledge score. Among these respondents, the mean age was 38.1 ± 14.3 and 77.8% had Bachelor, Master’s, or Ph.D.
Associations were found between, age, education, occupation and research ethics knowledge, as shown in Table 3. Age categories older than forty years had higher percentages of good knowledge (p < 0.0001). In addition, as might be expected, most respondents (84.60%) with graduate education (Master’s or Ph.D.) had good knowledge scores.
Associations of research ethics knowledge with demographics.
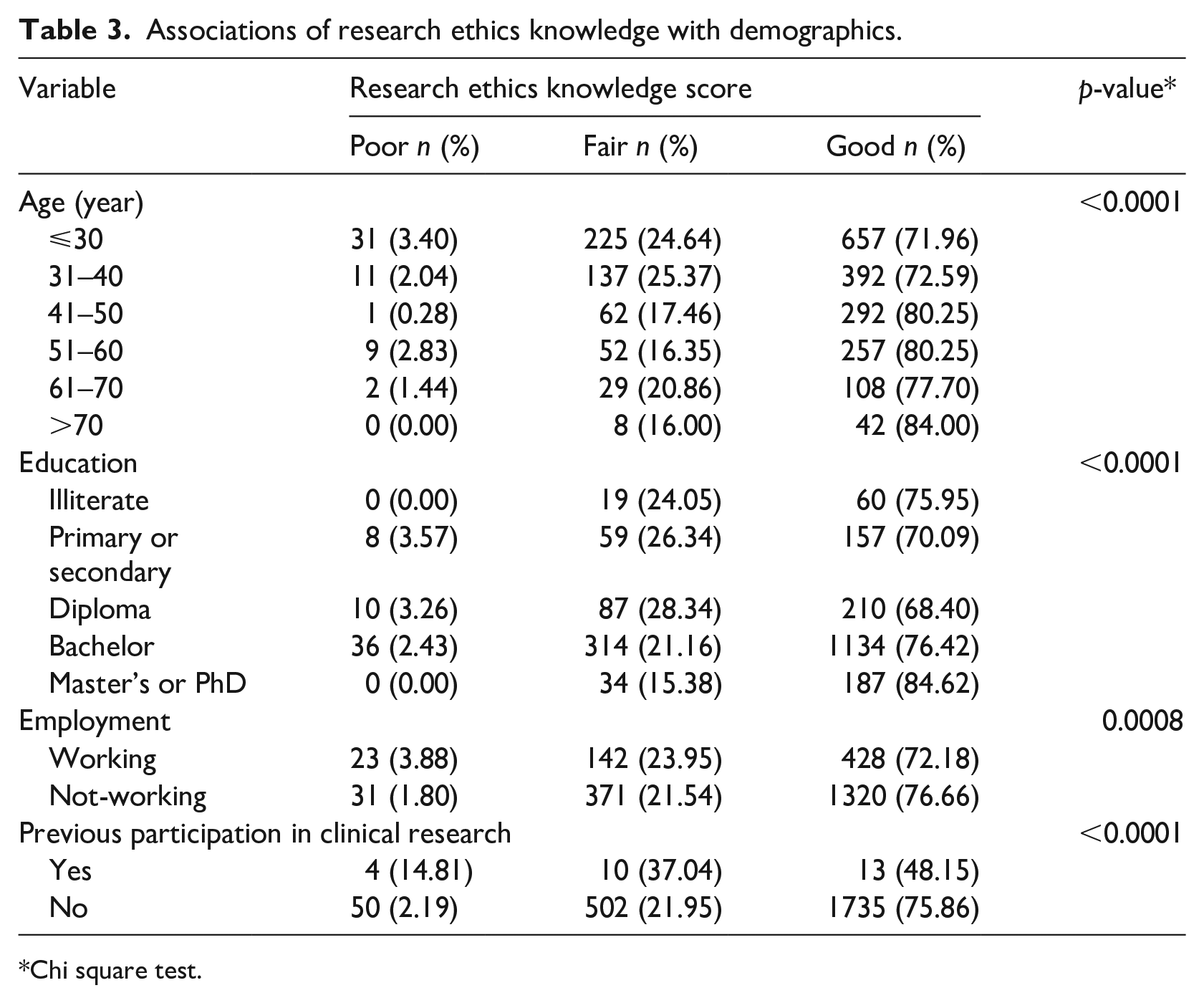
Chi square test.
Attitudes towards the ethics of clinical research
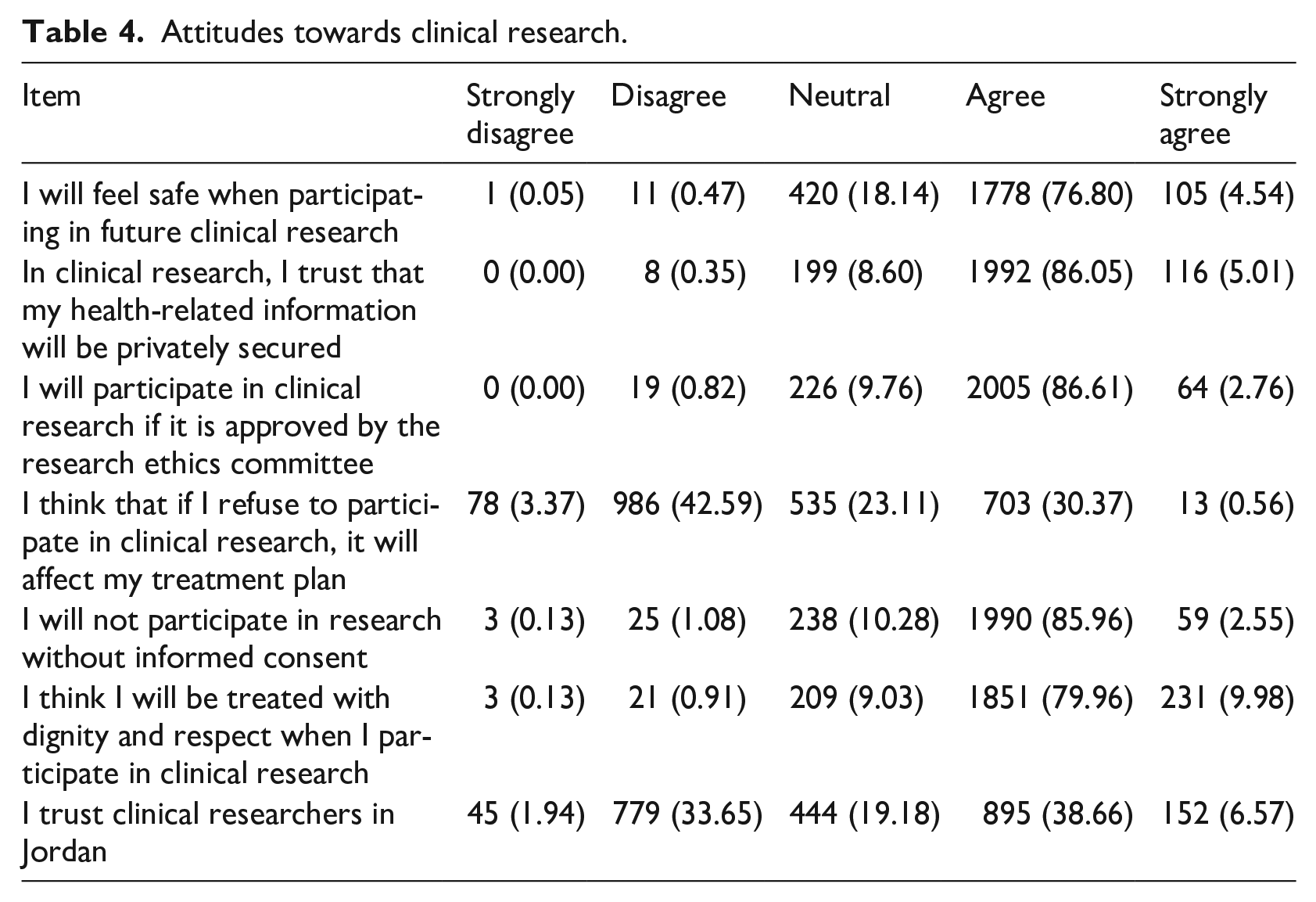
Responses to the attitudes statements on the questionnaire are summarized in Table 4. Overall, the attitudes of respondents towards research ethics were positive as 1755 respondents (75.81%) had an attitude score of 28 or more (out of 35), while 560 respondents (24.19%) had an attitude score less than 28 (negative attitude). Around 81.34% of respondents reported they would feel safe participating in research. In addition, 86.05% of respondents reported trust in the research team to maintain their data privacy. However, only 45.23% indicated that they trust clinical researchers in Jordan.
Attitudes towards clinical research.
In terms of attitudes towards research, a statistically significant difference was found among age groups, in particular, between age groups 31-40 and 41-50 (mean scores 25.72 and 26.20, respectively). Moreover, there was a statistically significant difference in the mean attitude score among educational levels (p = 0.0131). According to the Post-hoc Tukey HSD test, respondents with postgraduate education (Master’s or Ph.D.) had a higher mean attitude score than respondents with primary or secondary education (26.19 vs 25.61). The results of this analysis are depicted in Table 5.
Effect of demographic characteristics on public attitude score of ethics in research (n = 2315).
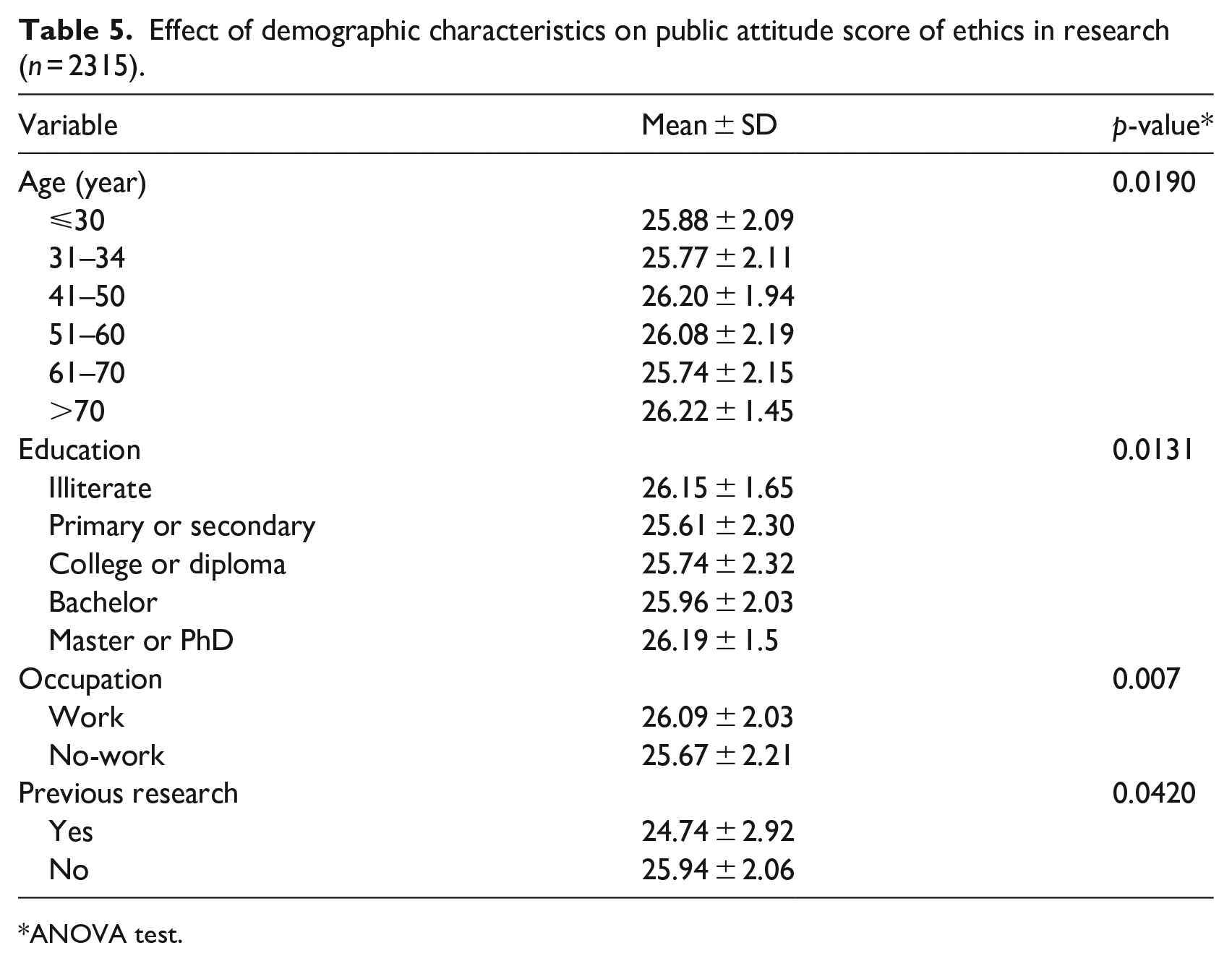
ANOVA test.
Perspectives on unethical research
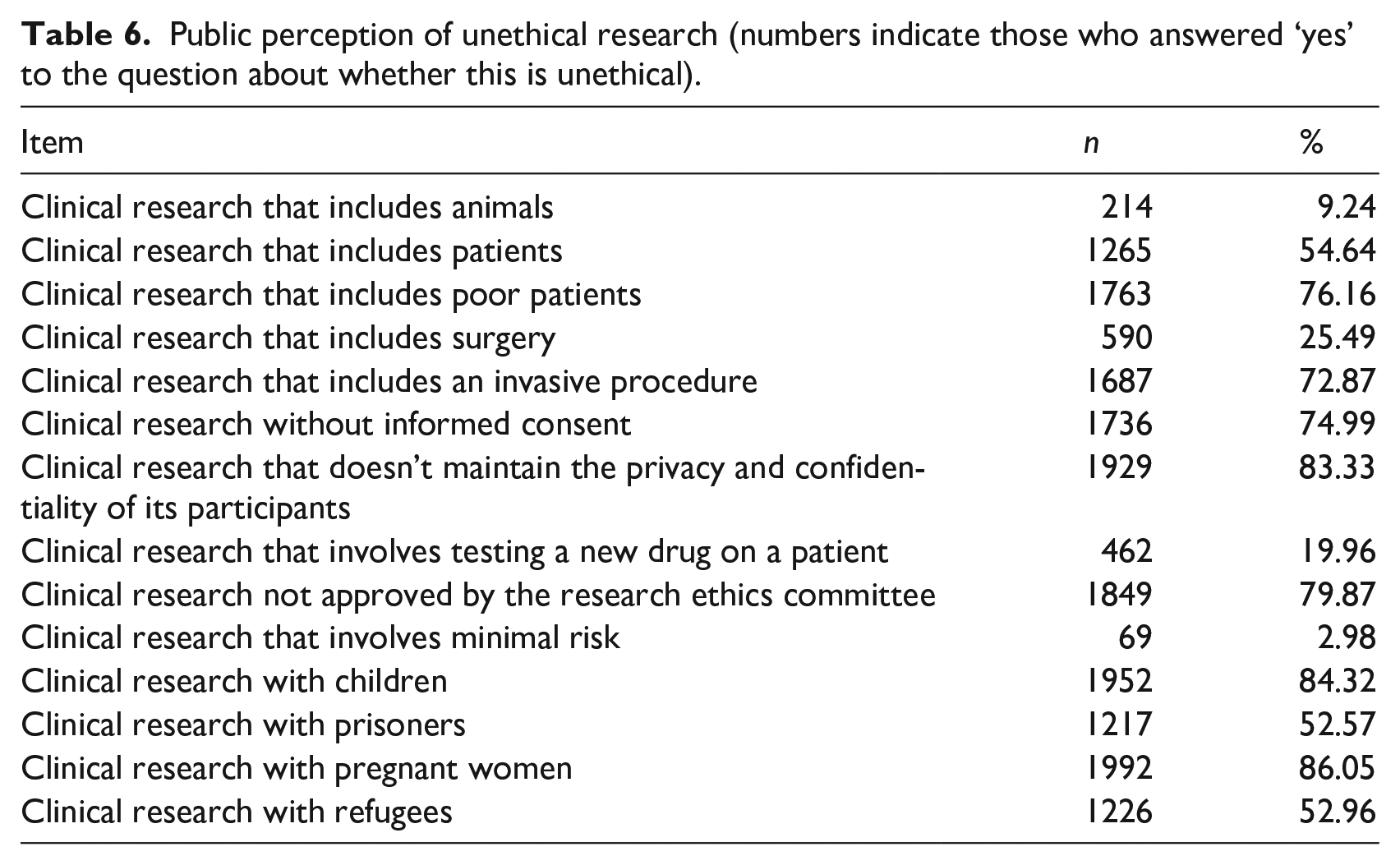
When respondents were asked about unethical clinical research, research with pregnant women was considered the most unethical practice (86.05%), followed by research with children (84.32%), research that does not maintain privacy (83.33%), and research not approved by the ethics committee (79.87%). All responses are shown in Table 6.
Public perception of unethical research (numbers indicate those who answered ‘yes’ to the question about whether this is unethical).
Discussion
This study aimed to assess the knowledge and attitudes of the Jordanian public about clinical research ethics. Overall, 75% of respondents were found to have good knowledge and a positive attitude towards clinical research/ethics. This finding was very similar to a study with Jordanian parents about ethical issues associated with children’s’ participation in medical research (Almomani et al., 2020).
Knowledge
The findings indicate that Jordanian public knowledge of research ethics is comparable to the little that we know from other countries. For instance, 92% of our participants believe that participation in clinical research is entirely voluntarily which is slightly higher than the figure of 85% that was found in an Indian study (Burt et al., 2013).
It is encouraging to note that the vast majority of Jordanian respondents (96%) stated that that ethical research should respect participants’ rights and welfare. However, only 52% believed that a participant has the right withdraw from a study at any time indicating that they may not know precisely what those rights are.
Also, of some concern is that only half of the participants could differentiate between clinical research and clinical treatment. The phenomenon of therapeutic misconception (TM) is defined by the National Bioethics Advisory Commission (NBAC) as ‘the belief that a clinical trial is designed not to gather evidence for the purpose of contributing scientific information, but to be useful for the particular patient’ (NBAC, 2001). TM occurs when participants confuse their roles as research participants and their roles as patients receiving medical care (McCormick, 2018). This can result in an inability to understand both advantages and disadvantages of participation, which poses a threat to the validity of informed consent. Moreover, study participants may view the researchers as their clinicians and the request to participate in a study as a recommendation (Silverman, 2011). Hence, in order to avoid TM, great care must be taken in clinical research to ensure that consent is appropriately informed (Miller and Joffe, 2006). Some researchers suggest that a shortened form of informed consent with straightforward language can minimize TM and improve participants’ understanding (Silverman, 2011). Additionally, efforts to educate participants about the differences between clinical studies and clinical treatment before seeking informed consent can result in a significant decrease in TM (Christopher et al., 2017).
Of course, it would be unreasonable to expect all members of the public to have full knowledge and understanding of the ethical aspects of clinical research prior to participation in a study. Indeed, that is exactly why we have clear requirements and protocols for informed consent. Informed consent procedures must ensure that all relevant information is provided, that the participant understands this information and that participation in research is a voluntary act (Dankar et al., 2019). The question of to what extent do we need greater public awareness of research ethics and how much we can rely upon the informed consent process is one that requires further investigation.
Attitudes
Jordanian public attitudes towards research are positive overall (75% had a positive score). Around 81.34% of respondents reported they would feel safe if they participated in research. Similar findings emerged from a study in the United States in 2017 (n = 12,427) which found that 90% of the general population considered participation in research to be safe (Anderson et al., 2018). In our study, 89.94% believe they would be treated with dignity and respect as participants in research. This figure is similar to a study situated in England (n = 1014) which indicated 82% of respondents were confident they would be treated with dignity and respect if they were asked by their doctor to take part in health research (Hunn, 2013). Further, 89.37% of participants agreed/strongly agreed that they would participate in future research if first approved by a REC. This figure is again comparable to the study conducted in England (83%).
Around 91% of our respondents trust that their health-related information will be privately secured, slightly higher than in the Hunn (2013) study where 80% of respondents reported they would be confident that their data would be held securely (Hunn, 2013). Our result is also much higher than has been reported in India where Sridharan et al. (2016) found that only 45.2% of participants believed privacy is protected in research, and Burt et al. (2013) found that only 54.1% of participants agreed that confidentiality of research participants is adequately protected.
Despite the positive attitude of most, and that more than 80% would feel safe participating in research, only 45.23% reported that they trust clinical researchers in Jordan. Other studies reveal mixed findings on this subject. Burt et al. (2013) reported that most participants in their Indian survey trusted academics (81.2%) and physicians (63.6%). In Tunisia, around 80% of patients would agree to participation in medical studies, given the importance of engaging in enhancing health care (Bouida et al., 2016). The primary cause for rejection was cited as risk of harm (66%); rejection due to mistrust was cited by 21%. There is concern that lack of public trust could undermine medical research by reducing both willingness to participate and confidence in the results (Hall et al., 2006). Lack of trust by participants can contribute to underrepresentation by certain groups, yet, the role and significance of trust in research is seldom addressed (Guillemin et al., 2018). In their Australian qualitative study, Guillemin et al. (2018) found that people are more likely to place their trust in institutions rather than researchers as individuals, and that the relationships participants had with researchers were based on the researchers’ associations with a university. Kerasidou (2017) has a similar message, recommending that for trust in biomedical research, institutions need to promote their trustworthiness. They can do this, suggests Kerasidou, by fostering and encouraging ethical conduct and ethical research through educational programs, having dedicated ethics teams, and through engagement to help promote the social value of research.
From their survey of 355 New York City residents, Smirnoff et al. (2018) identified four domains of trust/mistrust in medical research: general trustworthiness; perceptions of discrimination; perceptions of deception; and perceptions of exploitation. Each of these domains was associated with different demographic variables. While these domains may also reflect issues of trust/mistrust in the Jordanian population, this cannot be assumed.
Unethical research
Regarding unethical research, the vast majority of respondents deemed research with pregnant women (86.05%), children (84.32%) and poor patients (76.16%) to be unethical. Just over half of respondents also have concerns about the involvement of prisoners (52.57%) and refugees (52.96%). From these findings it can be deduced that the respondents have concerns about the involvement of participants who are perceived as being vulnerable in some respect, perhaps to risks of harm, coercion or duress. However, given the binary nature of these questions (‘yes’ or ‘no’), the basis of these opinions cannot be determined. Furthermore, the involvement of vulnerable populations in research is a matter of some contention. While ethics guidelines are clearly needed to prevent exploitation or harm of vulnerable populations in research (Bracken-Roche et al., 2017), their exclusion also denies them a voice in research. Furthermore, there is evidence of benefit for vulnerable participants and a willingness to participate if given the opportunity; exclusion from research could result in further marginalization (Alexander et al., 2018).
Study limitations
The primary limitation of this study stems from the use of a closed-ended survey tool. While this can be helpful in terms of ease of application, speed of application, and response rate, some responses lack the depth of information needed for interpretation. For instance, the fact that more than half of our respondents believe it is unethical to include patients in clinical research is an unusual finding but there is no qualitative data to explain why people might hold this belief. Additionally, closed-ended questions can ‘lead’ the respondents to answer in specific ways. Lastly, the study was only conducted in two Jordanian cities, which might affect the generalizability of the results.
Conclusion and recommendations for future study
Limitations notwithstanding, this survey makes a significant contribution to our comprehension of general public understanding and attitudes towards research ethics from a Jordanian perspective.
The study has revealed that most of the Jordanian public respondents have good knowledge of the primary ethical issues in clinical research including the need for informed consent and for ethics approval. However, there are some obvious gaps which require further investigation. These include, the difference between clinical treatment and clinical research and the right to withdraw from clinical studies.
Attitudes of the Jordanian public towards clinical research were found to be generally positive across all domains apart from trust in clinical researchers. This relatively low level of trust in researchers requires further qualitative investigation to reveal the reasons behind this viewpoint.
Finally, further qualitative investigation is also needed to aid understanding of why the Jordanian public might perceive the inclusion of certain groups in research (e.g. children, pregnant women, patients) as being unethical.
Footnotes
Acknowledgements
The authors are grateful to the research participants who volunteered to participate in the study survey.
Authors’ contributions
MA, SA, KA, SAD and AR conceived and designed the study. MA, SA and KA implemented the study. MA, SAD and AR analyzed the data. MA had primary responsibility for final content. All authors participated in writing, read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Work on this project was supported by grant # 5R25TW010023-02 from the Fogatary International Center of the U.S. National Institutes of Health on behalf of the Research Ethics Program in Jordan.
All articles in Research Ethics are published as open access. There are no submission charges and no Article Processing Charges as these are fully funded by institutions through Knowledge Unlatched, resulting in no direct charge to authors. For more information about Knowledge Unlatched please see here: ![]() .
.
Ethics approval and consent to participate
Written informed consent was obtained from eligible selected participants before conducting any study procedures. The conduct of the survey was approved by the Institutional Review Board of King Abdullah University Hospital affiliated to Jordan University of Science and Technology.
Consent for publication
Not applicable.
Availability supporting data
The dataset supporting the conclusion of this article can be accessed by writing an email to the corresponding author.
