Abstract
This study aimed to explore stakeholders’ perspectives on the ethical considerations for returning individual pharmacogenomics research results to people living with HIV. A qualitative approach to investigation involved five focus group discussions with 30 Community representatives, 12 key informant interviews with researchers, and 12 in-depth interviews with research ethics committee members. In total, 54 stakeholders who were involved in pharmacogenomics research and HIV treatment and care contributed to the data collection between September 2021 and February 2022. The study explored five prominent themes: (i) defining the nature of research results to return to participants; (ii) preparing research participants to receive their results; (iii) obtaining informed consent for the return of results; (iv) opinions on health personnel to return the results to participants; and (v) opinions on how research results should be communicated to participants. Respondents identified various strategies for the return of individual results with minimal ethical risks including the setting up of a diverse and independent committee to undertake a risk-benefit assessment based on local context; ongoing discussions about the possible kinds of results and their implications throughout the study; and employing genetic counsellors to communicate results to participants. The strategies identified in this study should be further studied and independently verified.
Keywords
Introduction
The introduction of antiretroviral treatment in the mid-1990s transformed
Pharmacogenomics research results can be generated by genomic and genetic analyses (Korol et al., 2013). Several stakeholders, including genetics professionals and researchers, and members of research ethics committees (RECs), advocate for returning the clinically significant results of genetic tests (Beskow and O'Rourke, 2015; Fernandez et al., 2013; Lázaro-Muñoz et al., 2021). Proponents of returning results argue that withholding relevant health-related information denies participants the opportunity to make informed decisions regarding their lives, and undermines their autonomy (Virani and Longstaff, 2015). Globally, studies have reported a high demand for individual genomic and pharmacogenomic results from the public and research participants (Byrjalsen et al., 2018; Facio et al., 2013; Matshabane et al., 2022; Nabukenya et al., 2023; Ralefala et al., 2021; Sanderson et al., 2016; Sundby et al., 2017).
Individual genomic and pharmacogenomic results can be categorized as primary results, secondary results and incidental findings (Ortiz-Osorno et al., 2015). Primary results have been defined as the findings responding to a well-defined research question, while secondary findings are often referred to as results that are not associated with the primary research question, but are intentionally sought (Darnell et al., 2016). On the other hand, incidental findings are any results discovered unintentionally and not related to the primary research question, but may have significant implications for the health or wellbeing of the participant and family members (Middleton et al., 2016). A recent study exploring the factors that influence participants’ preferences and their reasons for wanting to receive individual results from pharmacogenomic research among PLHIV in Uganda indicated that 98% wanted to receive all their primary results, while 66% wanted to receive both primary results and incidental findings (Nabukenya et al., 2023). Generally, PLHIV consider the return of their results as a way of respecting their right to information, valuing their contribution to the success of research, and an opportunity to learn more about their health status (Nabukenya et al., 2023; Ralefala et al., 2021).
Nonetheless, results from pharmacogenomic research have been associated with several ethical and social harms including discrimination (Sarwar, 2023), and loss of privacy and confidentiality (Kisiangani et al., 2022). Specific to communities infected with and affected by HIV, many PLHIV suffer depression (Abas et al., 2018; Bernard et al., 2017), despair (Dela Cruz et al., 2020), distress and fear (dela Cruz et al., 2016) after receiving results that reveal HIV infection. In addition, PLHIV suffer stigma (Nabunya et al., 2020; Oke et al., 2019), social exclusion (Dahlui et al., 2015), reduced chances of finding a marriage partner (Jürgensen et al., 2012), and fear of abandonment, divorce and domestic violence (Ndayala and Ngige, 2020; Sweeney et al., 2015). Most communities affected by HIV in sub Saharan Africa are characterized by low socio-economic status (Bernard et al., 2017) and low literacy levels, which exposes them to the risks of poor understanding and misinterpretation of pharmacogenomics research information (Nabukenya, 2019).
The increased demand for individual genomic and pharmacogenomic research results has sparked the need to develop locally contextualized guidelines for returning individual research results to participants (Kisiangani et al., 2022; Mwaka et al., 2021). Several guidelines have described when and how different kinds of results from genomic analyses should be returned (Burke et al., 2013; Green et al., 2013; Matimba et al., 2016). The 2007 National Institutes of Health (NIH) data sharing policy on genome-wide association suggests that contributing institutions and institutional review boards (IRBs) may wish to establish policies to determine when it is appropriate to return individual findings from research studies (Shurin and Science, 2008). The Human Heredity and Health in Africa (H3Africa) guidelines for the return of individual genetic research findings recommend the return of validated, clinically significant and medically actionable (with a proven therapeutic or preventive intervention) results from genomic studies. Nonetheless, studies from sub-Saharan Africa have reported inadequate, unclear, or an absence of national guidelines for the returning of genomics research results, yet the ethical, legal, and social implications of these results do not only affect participants but may also affect family members and research communities (Chanda-Kapata et al., 2015; de Vries et al., 2017; Staunton and de Vries, 2020). Researchers in sub-Saharan Africa have reported several ethical considerations for the returning of individual genomic and genetics results, including the need for community engagement, genetics counselling services and determining appropriate methods of communicating genetics results to participants (Matimba et al., 2022; Ochieng et al., 2022). However, there is limited literature on how individual results from pharmacogenomics research should be safely returned to people living HIV, a chronic disease that is still associated with many ethical and social issues. Therefore, this study explored stakeholder perspectives on the ethical considerations for the returning of individual pharmacogenomics research results to PLHIV. We hope that findings from this study can inform the development and refinement of institutional and national guidelines.
Methods
Study design
We adopted a qualitative exploratory approach (Creswell, 2018) that employed focus group discussions, key informant interviews, and in-depth interviews to understand what different stakeholders regarded as key ethical issues for consideration in returning individual pharmacogenomics research results to PLHIV. Data was collected between September 2021 and February 2022.
Study setting
We conducted this study at Makerere University College of Health Sciences (MakCHS) and its affiliate research institutions located on Mulago hill, Kampala, Uganda. MakCHS is one of the nine constituent colleges of Makerere University, with vast experience in HIV/AIDS research, including pharmacogenomics. MakCHS has five independent research ethics committees (RECs) out of the 33 RECs accredited by the Uganda National Council for Science and Technology. However, only three RECs had experience in reviewing pharmacogenomics research specifically for HIV treatment. The study team sought to include three additional accredited RECs that are not affiliated with MakCHS but have prior experience in reviewing pharmacogenomics research in HIV treatment. We also considered five renowned HIV research institutions that are affiliated with MakCHS and have established community representative groups.
Participants
We enrolled 54 stakeholders who were researchers, members of RECs, and community representatives. We included stakeholders with prior experience in pharmacogenomic research for HIV treatment from MakCHS and affiliated research institutions. We purposively selected 15 researchers with the above characteristics. Of these, 12 consented to participate, while three declined, citing inadequate time to participate in this study. We also purposively selected 12 REC members with experience in reviewing proposals for pharmacogenomic research in HIV treatment. Six were REC chairpersons and the rest community representatives on RECs. Three of the REC chairpersons preferred to nominate a member of their RECs who was more knowledgeable in the field of study. We also purposively selected 30 community representatives from five HIV-renowned research institutions affiliated to MakCHS who provide input into study design and ensure that research strategies acknowledge and respect the cultural values of communities and research participants.
Research team
The data collection team comprised three research staff. SN, one research assistant with prior experience in conducting interviews of genomic research, and a note taker. All authors (MES, DK, AT, IM, CW, and SN) performed data analysis and interpretation throughout the study. All the authors had worked on a genomic research project and had experience analysing and interpreting qualitative genomics research data.
In-depth and key informant interviews
The researchers and REC members were contacted by email and interviews were scheduled for those who provided their consent to participate. Interviews were conducted virtually (via zoom) due to the COVID-19 pandemic, whose mitigation measures restricted face-to-face interactions. SN and one research assistant moderated the interviews interchangeably, and a note-taker was present throughout the discussions. With the help of an interview guide, we asked open-ended questions. All interviews were conducted in English and audio recorded. On average, interviews lasted between 45 and 60 minutes.
Focus group discussions
We contacted the respective leaders of the community advisory boards (CABs) and peer support groups who organized their respective group members to select an appropriate date, time, and venue to hold the focus group discussions. A follow-up call was made the day before the discussion to confirm availability. On arrival, one research assistant and SN explained the details of the study and sought written consent in either English or Luganda, the commonly spoken local language in Central Uganda. We employed deliberative focus group discussions (dFGDs) for community representatives, which are often used to educate participants about the topic of interest prior to the focus group discussion. Deliberative FGDs enable participants to gain a better understanding of the subject matter and promote the provision of quality data from informed opinions (Rothwell et al., 2016). We used dFGDs because pharmacogenomic research is still a new concept in Uganda and it is not well understood by most community stakeholders. We conducted five dFGDs, each with six (6) participants. Before the discussions, the research assistant provided a brief overview of how antiretroviral drugs interact with human genes. A vignette describing a hypothetical scenario of possible outcomes from pharmacogenomics research with the categorization of findings into primary results and incidental findings followed this. With the help of a dFGD guide, open-ended questions were presented to the participants to explore their views on how individual results from pharmacogenomics research should be returned to PLHIV. Clarifications were offered prior to and during the discussions. SN and one research assistant moderated interchangeably and a note-taker was present throughout the discussions. The dFGDs were conducted in a well-ventilated tent after lifting the national lockdown during the COVID-19 pandemic. The dFGDs were audio-recorded and they took about 60–90 minutes.
Research instruments
Both dFGD and interview guides were developed from the literature (Bollinger et al., 2012; Fernandez et al., 2013; Godard et al., 2010; Haga et al., 2011; Heaney et al., 2010; Klitzman et al., 2013; Lohn et al., 2013; Ramoni et al., 2013) and subsequently revised to capture new emerging questions. The discussion included questions related to the nature of results to be returned to participants, how to prepare participants to receive their results, how to obtain informed consent for receiving individual results and appropriate methods for conveying individual results to participants. The interview guides were all piloted with relevant personnel prior to use and feedback was used to improve the guides. For example, some revisions were made to the dFGD guide that was translated into Luganda to represent the community representatives’ meaning of genomic terms. The research team held debriefing meetings at the end of each interview to identify new perspectives that were not initially captured by the tool. Data were collected until no new insights resulted.
Data analysis
All audio recordings were transcribed verbatim. Transcripts from members of the community representatives were translated from Luganda (one of most frequently spoken) to English. Transcripts of both Luganda and English languages were verified for accuracy by reading word by word while listening to the audio recordings for quality checks and spelling errors. This step also helped the authors to familiarize, mark and memo the data. The data were analysed throughout the study using a thematic approach (Braun and Clarke, 2006; Fereday and Muir-Cochrane, 2006). Transcripts were first analysed separately for the three categories of the participants, namely researchers, REC members and community representatives to gain a deeper understanding of each category’s perceptions about the topic. Three authors (SN, AT and ESM) selected three transcripts from each category of participants for open coding. These scripts were read line by line to generate the first set of codes. Syntheses of codes from the independent reading were iteratively discussed among the three authors and codes with similar meaning were merged. Differences in coding among the independent coders were resolved by consensus. A hierarchal codebook and coding framework was developed for each participant category by the three authors to guide the analysis of the data. The hierarchies of codes were then sorted into categories based on how themes were related and linked. We then deductively generated themes using our pre-existing analytic framework, which we developed from the literature about the ethical considerations for returning individual results from pharmacogenomic and genomic research. We also inductively considered new themes that emerged from the transcripts. All the transcripts were then imported into Nvivo version 12 (International-Pty-Ltd Q, 2018) and coded by three authors (SN, AT, and ESM). Three authors DK, IM and CW examined the themes for patterns until consensus was achieved on the final themes. All the authors compared the emergent themes with the existing literature to confirm that the final themes represented the stakeholders’ perspectives accurately. The key findings were summarized and overlapping themes across the three categories of participants were merged as presented in Table 2. The final codebook included the merged themes and codes from all the three categories of participants.
Regarding research reflexivity, the research team are aware that it is impossible to remain entirely neutral throughout. We acknowledge our potential biases based on the prior knowledge about the research institutions where we recruited the research stakeholders and the existing relationships between the interviewees and the research team. However, we tried to engage in listening from the interviewees’ perspective.
Ethical considerations
This study was reviewed and approved by the Makerere University School of Biomedical Sciences Higher Degrees and Research Ethics Committee (SBS- 855) and the Uganda National Council for Science and Technology (SS 735ES). All participants were assured of confidentiality. We sought permission from the institutions’ Heads/ Directors of the Research Departments of the five HIV research institutions prior to conducting the focus group discussions with the community stakeholders.
Results
Respondents’ characteristics
The demographic characteristics of respondents are presented in Table 1. A total of 54 participants participated in this study. The majority were above 35 years.
Respondents’ demographic characteristics.
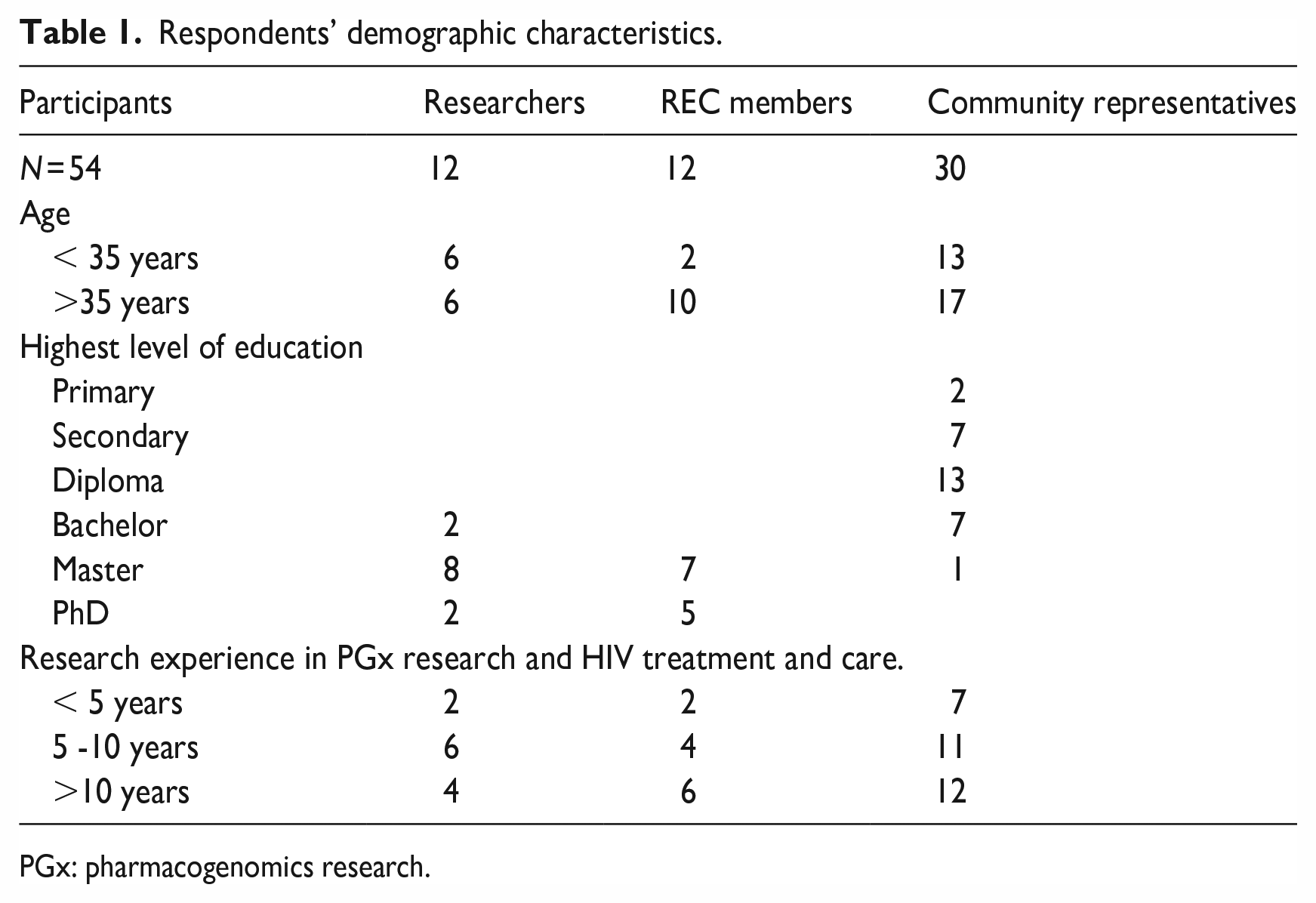
PGx: pharmacogenomics research.
Five key themes emerged from this data as shown in Table 2. These included defining the nature of results to be returned to participants; preparing research participants to receive their results; obtaining informed consent for receiving of research results, opinion on who should return the results to participants; and opinion on how research results should be communicated to participants.
Emergent themes.
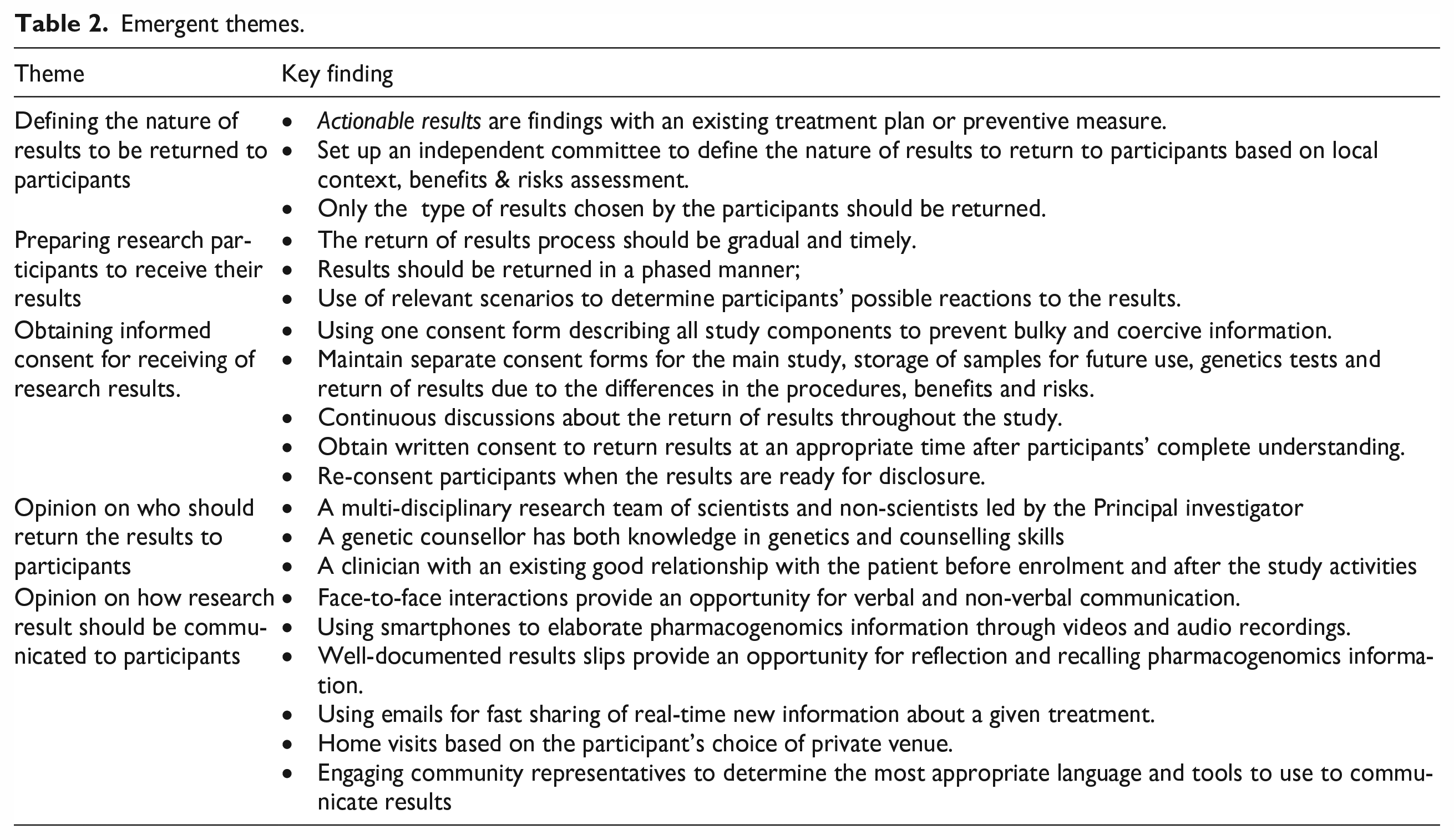
In the following, the identified themes are elucidated further with illustrative quotes from the researchers (R), REC members (REC) and community representatives (CR).
Defining the nature of results to be returned to participants
It is imperative to define the nature of the results to be returned to participants. Therefore, we asked respondents to describe the kind of results that should be returned to participants. The majority (50) of the respondents enunciated that returning the primary results of pharmacogenomics research to participants is important. They defined primary results as those addressing the research questions approved by RECs, specifically related to how genes interact with HIV drugs or determining the appropriate dosage. In addition, most respondents considered clinically actionable incidental findings equally essential for return to participants. Researchers defined actionable results as findings with an existing treatment plan or preventive measure.
Community representatives mentioned several factors to consider when defining the nature of results to be returned to participants. They suggested setting up a committee comprised of scientists and non-scientists to assess the magnitude of harm that may arise from returning the results. They also emphasized the need to define the actionability of results on a case-by-case basis.
Three researchers emphasized the need to consider the local context when determining the actionability of the research results. They mentioned that some drugs that have been proven effective to an individual might not be available in Uganda and inaccessible to the participant. In some instances, the most effective drug for an individual may be available in Uganda but not affordable.
Researchers enunciated that certain results currently described as ‘not clinically actionable’ might become actionable in the near future. Therefore, they suggested that such results should be kept in bio repositories or data banks for future research but not discarded.
Nearly all respondents (52) agreed that participants’ choices and interests should determine the nature of the results to be returned. However, two REC members emphasized that participants should be provided with adequate information about the implications of the results but advised caution.
Preparing participants to receive their individual results
Nearly all respondents (48) opined that the process of returning pharmacogenomics research results is complex and requires researchers, research participants and institutions to prepare adequately. They suggested that the feedback process should be gradual and involve ongoing discussions with participants throughout the study. Respondents further indicated that participants should be given ample time to reflect on the information provided before deciding the kinds of results they would like to receive.
In addition, REC members (03) suggested that the research team should release results in a phased manner, with positive (good) results shared first. They also highlighted the need for verification of the results before returning them.
On the other hand, six researchers emphasized the need to update participants’ contact information and study records regularly to ensure the results are returned to the rightful participant.
One community representative suggested using relevant scenarios that present negative and positive situations to determine the participants’ possible reactions after learning about these results. This approach might help researchers determine the appropriate time and method to release the results.
The majority (50) of the respondents agreed that genetic counselling is a key and paramount component in the process of returning results. They considered genetic counselling as an opportunity to explain the implications of sharing pharmacogenomics research results with participants.
Obtaining informed consent for receiving pharmacogenomics research results
Prospective seeking of participants’ consent on whether to return their results or not demonstrates respect for their autonomy. One researcher proposed combining information about the possible results and details of other study procedures in one consent document. All this information should be provided to the potential participant at enrolment.
However, some community representatives and REC members opposed incorporating information about the study procedures, storing human samples for future use, and receipt of results in one consent form. They believed this information, specifically about the procedures, risks, and benefits, differs across each process; one consent form may not provide adequate information for the participant to make an informed choice.
Respondents had varying views about the appropriate time for obtaining informed consent for returning individual pharmacogenomic research results. Whereas some researchers (04) felt that discussions about the return of results should start at enrolment, some community representatives felt that it was too early because participants may not have fully comprehended all information about the study procedures. In addition, some participants may think it is a prerequisite to consent to feedback of results before joining a study. Majority of the community representatives (26) thought that the research team should seek consent on one of the subsequent study visits.
The majority of REC members (09) indicated that the consent process should be ongoing. They mentioned that researchers should summarize the possible categories of results generated from the analyses at the time of enrolment and provide more details about these results on subsequent study visits. Nearly all respondents (51) agreed that participants should be re-consented to receive the results when they are eventually available
Opinions on who should return the results
Determining who should provide feedback on the results was also considered important. Five REC members said that a multi-disciplinary team of scientists and non-scientists would be best positioned to provide feedback on the results. They suggested that the team should be headed by the principal investigator, whom they felt was the most senior and knowledgeable.
The majority of respondents (47) felt that genetic counsellors were the most suited health providers to return these results, while two researchers considered clinicians as more suitable to return these results.
Some community representatives (02) suggested training of social workers who are already working among communities infected and affected by HIV about how to return individual pharmacogenomic research results safely to research participants.
Opinions on how research results should be communicated to participants
How results should be communicated was of paramount concern to the respondents. Nearly all (48) respondents preferred face-to-face interaction when returning results to participants. They suggested that participants should be contacted to meet with the researchers at the health facility once the results are ready. Respondents suggested that face-to-face interactions facilitate a two-way discussion of the results where answers to participants’ questions are provided in real-time. Further, face-to-face interactions also provide an opportunity for verbal and non-verbal communication.
The majority (52) of respondents said that using telephones to communicate pharmacogenomics-related results might not be appropriate. They mentioned that it might be challenging to explain sensitive information via telephone since it is a “one-way” direction of communication. However, two community representatives believed the research team could use smartphone social media platforms to elaborate pharmacogenomics information through videos and audio recordings.
One researcher suggested using well-documented results slips or reports, especially among the educated research participants. The researcher indicated that the results slips could be used for further reflection and recall of the explanations about the implications of the results provided to the participant. However, two community representatives expressed fear of breach of confidentiality if a friend or family member gets hold of the result slip.
The use of emails was another method of communication that was mentioned by another researcher.
Two community representatives suggested that the research team might consider home visits to return results to research participants. They mentioned that participants should choose a private place and an appropriate time to receive one or two research team members.
Discussion
The return of individual results from genomic analyses is a complex procedure that requires careful planning about the kind of results to be returned and the resources available. Most researchers in Uganda do not return individual results from genomics analyses (Rutakumwa et al., 2020), and the absence of national and institutional guidelines significantly impacts the choice of genetic and genomic results, when, and how they should be feedback to research participants (Mwaka et al., 2021). Findings from our study were generally favourable towards returning individual results to research participants. Respondents shared their perspectives on how the process of returning individual research results should be conducted based on their vast experience in genomic research, and HIV treatment and care. Respondents felt that the kind of results to be returned should be clearly defined before sharing them with participants. They also agreed that clinically actionable results should be returned to participants on the grounds of beneficence. They defined clinically actionable results as those with a clearly defined treatment or preventive plan. Our study findings are consistent with prior studies where genetics professionals and researchers generally agreed to return clinically actionable pharmacogenomics research results (Korol et al., 2013; Lázaro-Muñoz et al., 2021). However, several factors should be considered when defining actionable results, especially among people suffering a chronic illness like HIV/AIDS in low-resource settings. For example, for several decades, many PLHIV have been accessing antiretroviral treatment cheaply or freely from the Government of Uganda (Commission UACJKUA, 2015; Spacek et al., 2006; Ssewamala et al., 2023). The introduction of pharmacogenomic research aimed at optimizing HIV treatment may present information about approved and efficacious antiretroviral drugs with minimal adverse drug reactions tailored to an individual’s genetic makeup. However, these antiretroviral drugs might not be accessible or affordable to all participants, which could lead to psychological harm due to their economic inability to access such drugs. Nonetheless, bioethicists have argued that a patient’s economic status should not be reason to deny them information (Sullivan and Berkman, 2018). Patients should be provided with all the information that can be useful to improve their quality of life. However, it is also important to note that some therapeutic or treatment methods may not be culturally accepted in some communities.
People living with HIV who receive care and treatment from well-established HIV clinics and research institutions can acquire a vast amount of information about HIV and other health conditions through participating in research studies and other services such as routine counselling. Knowledge empowerment has enabled some to overcome health-related challenges and express a wish for all their individual results from pharmacogenomics and genomics analyses, including results that are not clinically significant (Nabukenya et al., 2023; Ralefala et al., 2021). However, as mentioned by two REC members, ‘information can be harmful’; researchers need to balance respect for individual autonomy with the potential harms and benefits that can arise from sharing all results with participants. Setting up an independent committee comprising both scientists and non-scientists to help guide the researchers’ decisions about the kind of results to be returned and in accordance with participants’ beliefs and values may be necessary.
Results should be disclosed in a language that facilitates easy understanding and interpretation. This could be enhanced by leveraging existing relationships between the research team members and participants receiving care and treatment in the various HIV research institutions. Specific to pharmacogenomic research, research team members ought to hold continuous discussions about the possible kinds of results that could be returned and the ethical, social, and legal implications. Such discussions increase transparency, health literacy and trust between the researchers and participants (Edwards et al., 2018; Shalowitz and Miller, 2008). They can be facilitated by use of relevant life scenarios and other educative materials, such as visual aids to enhance participants’ comprehension of pharmacogenomics research information. Similar studies have also highlighted the need to use such materials to promote participants’ complete understanding of genomics and genetics studies (Ochieng et al., 2022; Wilczewski, 2020).
Information delivery by the research team to the participants is often guided by an approved informed consent document, where key components such as the purpose of the study, study procedures, benefits, and risks are described. The Uganda National Council for Science and Technology (UNCST, 2014) guidelines recommend administering separate consent forms for the main study activities, storage of samples for future use and genetics testing (as a secondary objective). However, the guidelines are silent on the process of consent for the return of individual results. Some researchers felt that separate consent forms create bulky and unreadable information, especially in low-resource settings often characterized with poor literacy and reading inabilities (Bukini et al., 2020; Dwyer et al., 2021). Many participants receive genomics-related information for the first time when participating in genomics and genetics studies, yet researchers often spend little time with participants when obtaining written informed consent at enrolment. Thus, too much information, coupled with numerous consent forms and limited time for discussion, contribute to a poor understanding of genomics and genetics research and is sometimes considered coercive (Bukini et al., 2020; Tindana et al., 2012). Community representatives and REC members certainly did not favour incorporating information about the study procedures, storing human samples for future use, and receipt of results in one consent form. They believed this information differs across each process, specifically the procedures, risks, and benefits. Therefore, having one solid consent form may limit provision of adequate information and restrict participants’ autonomy. Community representatives recommended using precise and simple language when conveying genetic and genomic information to research participants across the different consent forms. The process of information delivery should also be gradual and not rushed. This way, researchers may improve participants’ understanding of genetics and genomics information to make informed choices about their desired results. REC members emphasized the need to inform participants about the possibility of returning individual results at enrolment. Further discussions about the different kinds of results should be held on subsequent study visits. Respondents also emphasized the need to re-consent participants once the results are ready for sharing. Pharmacogenomics and genomics results may be generated several months or years after the closure of study activities. This is because these analyses must be performed and validated by certified laboratories, which are often unavailable in Uganda. Some participants might need to be reminded of the purpose of the study or might have changed their minds about receiving certain kinds of results.
Communicating individual pharmacogenomic research results to participants requires a knowledgeable team experienced in explaining genomics terms and the mechanism of action between one’s genes and drugs to achieve adequate understanding. Research institutions conducting genomic research need to employ genetic counsellors with knowledge and skills in returning individual pharmacogenomic research results appropriately. However, Uganda and most sub-Saharan countries lack this professional cadre (Tata et al., 2020). Therefore, research institutions need to build capacity for genetic counsellors to ensure participants’ comprehension and utility of their results. In the absence of genetic counsellors, respondents suggested training social workers who are already working among communities infected and affected by HIV on how to return the individual pharmacogenomic research results safely to PLHIV and minimize the ethical and social challenges that might arise.
Most respondents recommended face-to-face discussions between the researcher and the participants as the most appropriate and effective method of communicating these results. Face-to-face discussions facilitate open and free information sharing and provide real-time answers to participants’ questions and concerns. Other studies have also reported the relevance of face-to-face discussions between researchers and participants for understanding genetics information comprehensively (Williams et al., 2018). With the increasing use of modern technologies (such as smartphones), researchers can share educative materials such as videos and pictures to describe and simplify pharmacogenomic scientific terms. In addition, engaging community representatives throughout the research project will enable researchers to learn about the most appropriate tools and language for communicating individual pharmacogenomic research results effectively.
Conclusion
Returning individual pharmacogenomic results can be a complex process, presenting ethical, legal, and social challenges. Findings from this study contribute to our understanding of these challenges and how they might be addressed. The various stakeholders in this study highlighted a number of particular actions for consideration including the following:
First, respondents highlighted the need to set up an independent committee with varying expertise and backgrounds to assess the risks and benefits of returning individual results to participants. Second, a genetic counsellor should communicate results simply and in an accessible manner, preferably. Third, community representatives should be engaged throughout the study to provide insightful information about the language and tools to communicate the results effectively.
Lastly, we recommend building capacity for genetic counsellors in Uganda and other sub-Saharan countries where genomic and genetic research is conducted. We also recommend further research to explore ethical considerations for returning individual results of genomic and genetic analyses in contribution to institutional and national guidelines.
Supplemental Material
sj-docx-1-rea-10.1177_17470161231207739 – Supplemental material for ‘It is a complex process, but it’s very important to return these results to participants’. Stakeholders’ perspectives on the ethical considerations for returning individual pharmacogenomics research results to people living with HIV
Supplemental material, sj-docx-1-rea-10.1177_17470161231207739 for ‘It is a complex process, but it’s very important to return these results to participants’. Stakeholders’ perspectives on the ethical considerations for returning individual pharmacogenomics research results to people living with HIV by Sylvia Nabukenya, David Kyaddondo, Adelline Twimukye, Ian Guyton Munabi, Catriona Waitt and Erisa S Mwaka in Research Ethics
Supplemental Material
sj-docx-2-rea-10.1177_17470161231207739 – Supplemental material for ‘It is a complex process, but it’s very important to return these results to participants’. Stakeholders’ perspectives on the ethical considerations for returning individual pharmacogenomics research results to people living with HIV
Supplemental material, sj-docx-2-rea-10.1177_17470161231207739 for ‘It is a complex process, but it’s very important to return these results to participants’. Stakeholders’ perspectives on the ethical considerations for returning individual pharmacogenomics research results to people living with HIV by Sylvia Nabukenya, David Kyaddondo, Adelline Twimukye, Ian Guyton Munabi, Catriona Waitt and Erisa S Mwaka in Research Ethics
Footnotes
Acknowledgements
The authors would like to acknowledge the assistance offered by the Faculty at Berman Institute of Bioethics, Johns Hopkins University and the Faculty of Makerere University International Bioethics who contributed to the study design and interpretation of the results. Waitt C is funded by Wellcome Trust Clinical Research Career Development Fellowship. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:The Fogarty International Center of the National Institute of Health through the Makerere University International Bioethics Research Training Program Grant Number D43TW010892.
Ethics approval information
This study was reviewed and approved by the Makerere University School of Biomedical Sciences Higher Degrees and Research Ethics Committee (SBS- 855) and the Uganda National Council for Science and Technology (SS 735ES).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
