Abstract
The return of individual pharmacogenomic research results is increasingly recognized as an ethical obligation, reflecting participants’ right to information and researchers’ duty of care. However, implementing this in low-resource settings poses significant ethical and logistical challenges. We explored stakeholders’ attitudes and challenges to the process of returning individual pharmacogenomics research results in Uganda. Between September 2021 and February 2022, we conducted 54 qualitative interviews. These included five focus group discussions with 30 community representatives across five HIV research institutions, 12 key informant interviews with researchers, and 12 in-depth interviews with ethics committee members. A thematic approach was used to interpret the results. Four themes emerged from this data. These included (i) attitudes towards returning individual pharmacogenomics results to people living with HIV; (ii) social and ethical implications of returning results; (iii) perceived challenges to the return of results; and (iv) proposed recommendations to overcome the challenges. Returning results reflects principles of reciprocity and equity, and is a source of hope to many people living with chronic illnesses. However, stakeholders raised challenges, including low literacy levels and absence of direct translation of genetic terms, which may lead to misinterpretation of the results; depression and anxiety; stigma and discrimination that could result from breach of confidentiality and privacy. Stakeholders highlighted the need to sensitize the public about the role of genes in an individual’s body. Returning pharmacogenomic results to vulnerable populations is ethically important, but must be approached with caution due to implications that may extend beyond the individual. Stakeholders emphasized the need for national guidelines and institutional standard operating procedures to ensure results are communicated safely and sensitively.
Introduction
The increase of genomics and genetics research on the African continent has generated a vast amount of useful information guiding the diagnosis and treatment of infectious diseases, including HIV (Calcagno et al., 2019; Dandara et al., 2019; Mukonzo et al., 2014). Pharmacogenomics, one of the branches of genomics research, provides us with an opportunity to learn about how an individual’s genes influence the way they respond to drugs (Korol et al., 2013a). Genomics and pharmacogenomics research results have similar characteristics. For example, genomics and pharmacogenomics analyses present a vast amount of information (Cassa et al., 2012; Tabor et al., 2011), and are pleiotropic, meaning a single gene can contain information about several phenotypes, which may raise potential incidental findings (Barrio-Hernandez et al., 2023; Meacham et al., 2010). These results are also probabilistic, meaning an association between a gene and a drug may only predict a degree of likelihood of an individual’s response to a specific medication or susceptibility of disease (Dressler, 2012; Nidhi et al., 2023). However, the circumstances in which participants may benefit from genomics and pharmacogenomics research results differ. Genomics results provide an opportunity to learn about the susceptibility to developing a future disease among other components, whereas pharmacogenomics results provide information about an individual’s sensitivity to a given prescribed drug (Korol et al., 2013a). The differences in the clinical utility and actionability of the genomic and pharmacogenomic results is important for discussion in low resource settings. There is an increase in pharmacogenomic research aimed at improving the treatment of HIV through determining the suitable regimen and dosing requirements for an individual to minimize adverse drug reactions (ADRs; Calcagno et al., 2019; Dandara et al., 2019). Antiretroviral drugs have been reported to expose people living with HIV to a number of ADRs, where up to 80% of ADRs in sub-Saharan Africa are related to the use of antiretroviral drugs (Ampadu et al., 2016; Rajman et al., 2017). This information can be useful in both research and clinical settings; and so, several stakeholders have advocated for the return of individual research results to participants. There is a high demand for individual research results from genomic and pharmacogenomic analyses among people living with HIV (Nabukenya et al., 2023; Ralefala et al., 2021). Similarly, genetics professionals and researchers generally agree that medically actionable findings such as pharmacogenomics results should be returned on grounds of beneficence, duty to warn and respect for participants’ decisional capacity (Korol et al., 2013a). Globally, researchers feel that returning individual research results respects participants’ rights to health information and repositions participants as active contributors to research as opposed to being a means of accomplishing research aims (MacNeil and Fernandez, 2006; Sánchez-Izquierdo et al., 2019). Secondly, researchers agree that participants’ autonomy and values are paramount in deciding the types of results to disclose (Grove et al., 2014). However, disclosure of results should be guided by pre-genetic counseling and provision of simple and understandable information when obtaining informed consent (UNCST, 2014; Yu et al., 2014). Furthermore, national ethics guidelines recommend returning research results to participants and research communities, while ensuring protection of their safety (UNCST, 2014).
However, researchers have raised concerns, including: participants’ misunderstanding of genomic information leading to possible emotional harm; the need to provide costly follow up services such as access to medical geneticists or genetic counselors who are currently not available in many low resource countries (Mwaka et al., 2021a); the need to keep current participants’ contact information; and the impact of this resource-intensive activity on valuable research resources (Vaz et al., 2018). Some researchers have also argued that research participants donate their samples for societal benefit and not individual benefit (Crawford et al., 2019a). Furthermore, some researchers argue that they are not obliged to look for meaningful incidental results from genomic datasets. However, if a medically actionable and validated incidental result is noted, it is in the best interests of the research participant to learn about it (Fernandez et al., 2013). While we recognize the bottlenecks of returning individual research results to participants in low resource settings, which may include low literacy levels in participants and strain on limited research resources, there is a growing consensus regarding moral and legal obligations of researchers to return particular results, especially those that are actionable (Vears et al., 2021). In addition, guidelines and practical checklists for the return of individual results from genomic and genetic research have been developed to describe the steps that researchers may undertake to determine whether and how individual results should be fed back results to adult participants (Matimba et al., 2022; Vears et al., 2023).
With the growing advances in genomic and pharmacogenomic research in Uganda, there is need to identify the key challenges that might hinder a safe return of individual research results to people living with a chronic disease such as HIV. Therefore, this study explored stakeholders’ attitudes and perceived challenges to the return of individual research results from pharmacogenomic analyses. We hope the study findings can be used inform the national guidelines for returning individual research results from genomic and genetic analyses.
Methods
Materials and methods
Study design and setting
This was a descriptive study that used a qualitative exploratory approach (Hunter et al., 2019). The study was conducted at the college of one research-intensive university in Uganda and five research institutions affiliated with the university. We considered three of the five accredited college-based research ethics committees (RECs), that had experience with reviewing pharmacogenomic research specifically for HIV treatment. In addition, we included three accredited RECs experienced with reviewing pharmacogenomic research for HIV treatment not affiliated with the college. The research team comprised a social scientist, bioethicists, a medical anthropologist, and medical scientist experienced with conducting and analyzing qualitative data.
Study participants selection
The study inclusion criteria included researchers who were involved in pharmacogenomic research for HIV treatment based at the college or affiliate research institutes. We approached the administration of the research institutions who provided us with the contact details including email addresses of the researchers. A total of 15 researchers were purposively selected based on the inclusion criteria. Of these, 12 consented to participate, while three declined, citing prior commitments. The administrators of the research institutions also provided us with a list of six ethics committees that had reviewed proposals of pharmacogenomics research in HIV treatment. We purposively selected six chairpersons of the RECs and the rest were community representatives on ethics committees. Three of the REC chairpersons preferred to nominate a member of their RECs who was more knowledgeable in the field of study. We also approached the chairpersons of the community representative groups affiliated with the five research institutions, who organized their respective teams and proposed a suitable date and time to hold the focus group discussions. Each group comprised six community representatives This study was conducted between September 2021 and February 2022.
Study procedure
In-depth interviews with researchers and REC members
Considering the restrictions that prevailed during the COVID-19 pandemic, we conducted in-depth interviews virtually. The research team contacted the researchers and REC members who met the inclusion criteria by email that contained a brief description of the study and a request to schedule an appointment for the interview. Expression of interest to participate was recorded by a positive response to the email. Participants provided their consent electronically or by a written email notification of acceptance to participate in this study. Appointments were scheduled and conducted virtually via Zoom, at the convenience of the participants. Interviews were audio recorded and lasted between 30 and 40 minutes.
Deliberative focus group discussions with community representatives
Five focus groups were conducted with community representatives. Some focus groups were conducted in English, while others were in Luganda, the most commonly spoken language in Central Uganda. We conducted focus groups after the national lockdown to minimize the spread of COVID-19 was lifted. We sat in a well-ventilated tent and, all participants and research teams wore face masks throughout the discussions. Prior to the focus groups, each community representative received a consent form document in a language of their choice. At the beginning of each focus group, participants were provided with an educational session on how antiretroviral drugs interact with human genes including a vignette describing a hypothetical scenario of the possible results that could be generated from pharmacogenomic research. We provided a detailed explanation about primary results and incidental findings from pharmacogenomic analysis. The prior educational session helped participants to gain an understanding of pharmacogenomic research and the different kinds of results that could emerge from it. Clarifications were made prior to and during the discussions. Two authors SN and AT moderated the sessions interchangeably and a note-taker was present throughout the discussions. The information disclosed during the focus groups were audio-recorded and lasted about 60–90 minutes.
Both focus group and interview guides were developed from the literature (Delanne et al., 2019; Dressler et al., 2012; Husedzinovic et al., 2015; Mackley et al., 2017; Ochieng et al., 2021; Scheuner et al., 2019; Vears et al., 2021; Wright et al., 2017) and subsequently revised to capture] new emerging topics. The topics of discussion included questions related to their attitudes and perceptions of returning individual research results to participants; the implications, perceived challenges to the return of individual research results; and the recommendations to overcome the challenges. The guides were first piloted on three genomic researchers, two REC members and three HIV peer support members who were excluded from the study. However, their feedback was used to improve the interview and discussion guides, respectively. The research team held debriefing meetings at the end of each interview to identify new perspectives that were not initially captured by the tool. Data were collected until no new information or insights were being revealed from both the interviews and focus groups.
Data analysis
Data were analyzed continuously throughout the study using a thematic approach (Braun and Clarke, 2006; Fereday and Muir-Cochrane, 2006). All audio recordings were transcribed verbatim. Transcripts of the community representatives were translated from Luganda to English. All transcripts were verified for accuracy by reading word by word while listening to the audio recordings for quality checks. This step also helped the authors to familiarize, mark and memo the data. Transcripts were first analyzed separately based on the three participant categories – researchers, REC members and community representatives – to gain a deeper understanding of each category’s perceptions about the subject matter. Three authors (SN, AT, and ESM) selected three transcripts from each participant category for open coding. These scripts were read line by line to generate the first set of codes. Synthesis of codes from the independent reading were iteratively discussed among the three authors and codes of similar ideas were merged. Differences in coding among the independent coders were resolved by consensus. A hierarchal codebook and coding framework was developed for each participant category by the three authors to guide the analysis of the data. The hierarchy of codes were then sorted into categories based on how themes were related and linked. We then deductively generated themes using our pre-existing analytic framework, which we developed from the literature. We also inductively considered new themes that emerged from the transcripts. All the transcripts were then imported into NVivo version 12 (International-Pty-Ltd Q, 2018) and coded by three authors (SN, AT, and EMS). Two authors DK and CW examined the themes for patterns consistency until consensus was achieved. All the authors compared the emergent themes with the existing literature to confirm that the final themes accurately represented the stakeholders’ attitudes and perspectives on the return of individual research results. We also returned some transcripts to the stakeholders to verify whether the data collected were a true reflection of their statements on the subject matter. This ensured that the data can be transferable to similar settings and enhanced the credibility of the study findings (Kaplan et al., 2017; Tindana et al., 2017). The key findings were summarized and the overlapping themes across the three categories of participants emerged as presented in Table 2. The final codebook included the merged themes and codes from all the three categories of participants was continuously refined to establish the themes, presented in the Results section.
Results
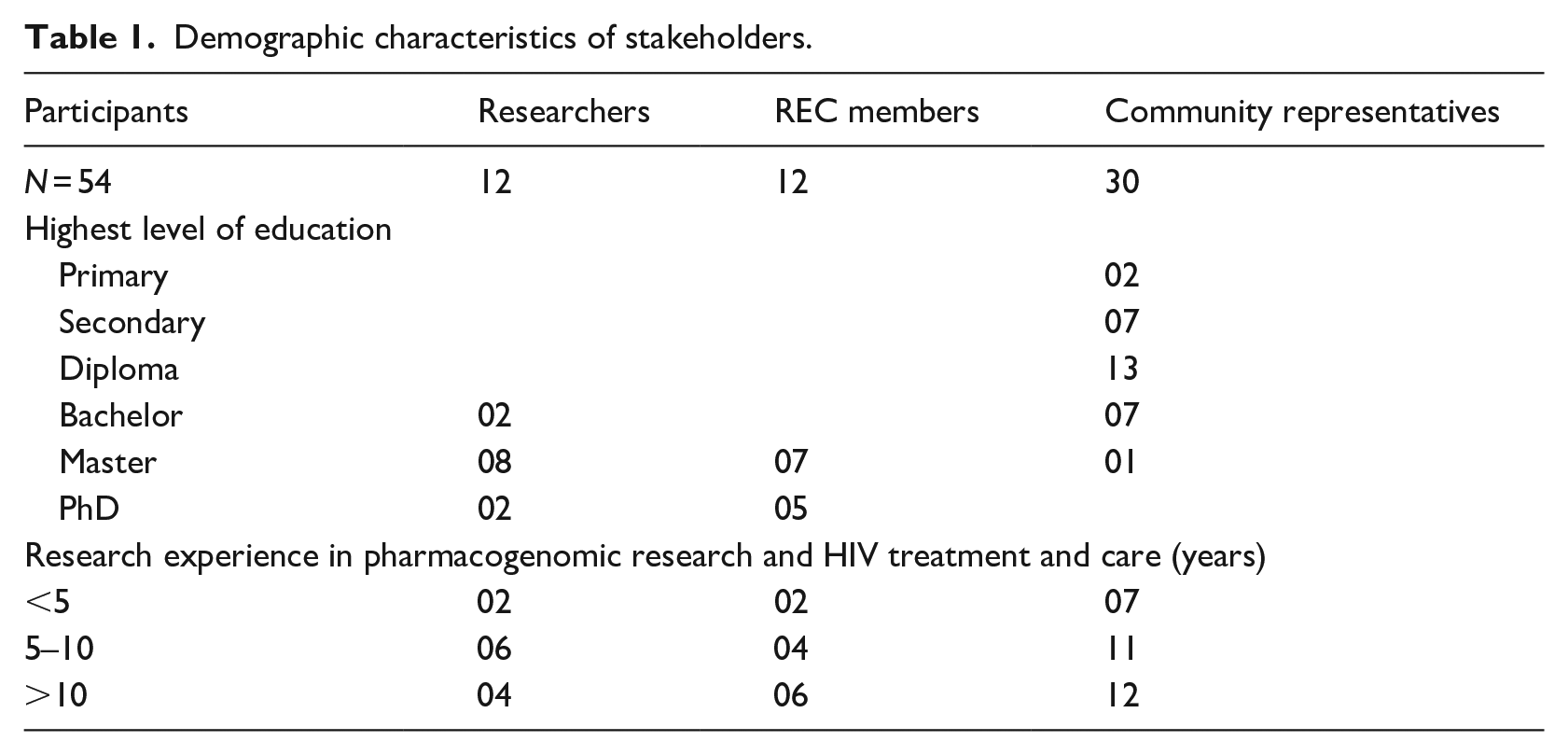
A total of 54 stakeholders participated in this study, including 12 researchers, 12 REC members, and 30 community representatives. The majority of the stakeholders had more than 5 years’ experience in pharmacogenomic research and HIV care and treatment. The demographic characteristics of stakeholders are presented in Table 1. Four themes were generated from the findings are summarized in Table 2.
Demographic characteristics of stakeholders.
A summary of the key findings.

Stakeholders’ attitudes toward the return of individual research results
Interestingly, the majority of the researchers (10) had never returned individual research results to participants. Only two researchers had shared results with participants, but did not provide detailed information since the results were used to assign the participants to a study arm. Similarly, no community representatives had ever been involved in the process of returning individual research results to participants. None of the REC members had ever reviewed a research proposal intending to return individual research results from pharmacogenomic studies to participants.
In our study, we generate the genetic results, specifically to determine the participants’ NAT2 acetylator status. . .. For fast or intermediate metabolizers, we give them a higher dose of 10mg/kg and the slow metabolizers get the standard dose of 5mg/kg. So, we tell them their status but still most of them do not understand what that means. But we don’t necessarily provide serious details about the results, because the study was not necessarily designed that way. . . They [the results] were helpful in the allocation of participants to a given study arm at enrollment. . .. (R 10)
Despite stakeholders’ limited experience, they generally had a positive attitude toward returning individual research results from pharmacogenomic analyses. They mentioned six reasons why they considered the process of returning individual research results important, including upholding reciprocity, equity, and justice; an opportunity to provide additional support and care; a source of hope; and an ethical requirement.
(1). Reciprocity: Most stakeholders (52/54) believed that returning meaningful and actionable results is one way of appreciating participants’ contribution toward the research project’s success. Community representatives believed that providing feedback prevents participants from feeling that they are being used by researchers to achieve research objectives with little regard to their needs. They also reported that returning individual research results promotes mutual benefits and overcomes “research fatigue” where participants are often involved in numerous studies without getting feedback from investigators. Researchers believed that the return of individual research results is an opportunity for participants to understand why they may respond to the same drugs differently.
One of the ways of appreciating participants’ efforts is by returning these results and clearly explain to them why they are given different drugs, yet they are all suffering from the same disease. . . this is very common in our HIV clinic. Sometimes, it happens to some couples attending the HIV clinic, we have cases where each of them receives a different regimen. We have a lot more to explain to them. . .. (R 8)
(2). Source of hope: Three community representatives contended that some information is powerful and can be used to positively influence the lives of research participants. They indicated that some people living with HIV have battled with the disease for many years and information about how their genes respond to antiretroviral medication could encourage and motivate them to continue adhering to their treatment plans despite their social and economic challenges.
You know people living with HIV experience many challenges in their lives and need as much hope and positive attitude to cope with these life challenges. . . Just telling them that you know, in this study you were part of revealed that the ARVs [antiretroviral drugs] you are taking are rightful for your genetic type and you should faithfully continue taking your medication as directed by your doctor is already good news to them. . .. (CR 11)
(3). Equity: The idea of promoting equity when returning individual research results was emphasized by researchers. Researchers believed that participants should be provided with information about individual variances in response to medication and drug dosages. They pointed out that the results from pharmacogenomic research can be used to optimize drug dosing and also guide policy makers in procuring the appropriate antiretroviral drugs that are more effective with minimal adverse reactions and side effects to the Ugandan population.
These results provide very important evidence to various stakeholders. . . First, participants need to appreciate that we are all different, that’s why we may respond differently to the same drugs even when we are suffering from the same disease. . .. Secondly, policymakers need this evidence to plan better for the populations they serve. These results will help them [policy makers] know which antiretroviral drugs they need to procure, rather than procuring one type of regimen to fit the whole population. . . (R 5)
(4). Offering additional support and care: Researchers and community representatives, particularly emphasized the need to provide additional support and care to participants. Specific to the incidental findings that could arise from pharmacogenomic analyses, three researchers mentioned that returning individual research results to participants requires providing additional information on the next steps of action. They felt that to some extent, they would approach their colleagues at the referral facilities to smooth the participants’ referral process, especially if an incidental finding reported a health condition that would be best treated in another specialized hospital.
Sometimes incidental findings may present conditions that cannot be well managed by our HIV clinics. This will necessitate referring the participant to another facility, which may be a government hospital. You know the systems there are very different, and our participants may need help. . .. (R 3)
Six of the community representatives emphasized the necessity for offering social care before and after returning individual research results to participants. They mentioned that they would like to offer ongoing counseling services and home visits to encourage participants to take their medication especially for participants who may require drug and dosage adjustments of their antiretroviral drugs, as one of them highlighted below: I would feel happy to see participants live a positive life after receiving these results. I would like to even go an extra mile to visit them at their homes and maybe see if they may need anything that I can offer and how they may be coping up with adjusting to their new medication. I would like them not to feel bad about the results, but feel encouraged to look forward to other beautiful days ahead. . . (CR 28)
(5). Justice: The aspect of justice was emphasized by the REC members when returning individual research results to participants, especially for those who wish to receive them. They asserted that these results belong to participants, and it is only fair to let them learn about their results. They however pointed out that participants should be empowered with relevant information packaged in a way that facilitates informed decisions on the choices of the kind of results they wish to receive.
When planning to return these results, researchers should provide enough information to the participants including the costs of the most effective alternative treatment regimen based on the participant’s genetic type. Then the participants should decide which results they are willing to receive or not. It is not good to withhold this information from participants because we think that they are poor and will not be able to afford the best medication. . . (REC 6).
(6). Ethical requirement: Two researchers felt obliged to return individual research results because it is a requirement from the national research regulatory body, the Uganda National Council for Science and Technology (UNCST).
For me, I might certainly feel obliged to return these results because it is an ethical requirement by UNCST that researchers may return results to participants and research communities. . . (R 6)
Ethical and social implications of returning individual research results
Stakeholders acknowledged several ethical and social implications of returning individual research results during the discussions. Researchers, for instance, posited that feedback of results is key in building and strengthening participant-researcher relationships. Two of the researchers explained that preparing participants to receive these results often involves several interactions, which encourages free information sharing of participants’ preferences, values, and expectation. Thus, creating a relationship between the researcher and participant that facilitates shared decision making.
We always try to build a friendly relationship with our participants so that they can freely share any information they want us to know. . ..and so returning these results could help strengthen our bond and trust. . .. (R 10)
Similarly, two of the community representatives described how returning results may improve participants’ quality of healthcare. They argued that pharmacogenomic research results might prompt researchers and clinicians to determine the appropriate drug and dosage requirements or recommend measures to prevent adverse effects. They opined that this information may also help participants prepare and plan for their future health seeking behavior. This was also reaffirmed by one researcher who mentioned that returning individual research results is a step toward the translation of pharmacogenomic research outputs into clinical care.
You see. . . we need to see the long- term goal of pharmacogenomics research, especially for people living with chronic illness. I think if we share these results with participants and the policymakers, it will be a step to improve HIV treatment in our clinical care (R 6)
Conversely, stakeholders were aware of the several risks that could arise from returning individual research results. They mentioned the risk of societal stigmatization, discrimination and domestic violence if participants’ results are exposed to third parties.
This information especially the incidental findings should be well guarded and restricted to only the participants and the researcher. . .because once that information gets to be known by any other people, trust me, the participant will not remain the same. Many things can happen especially if the participant is a woman. She can even lose her marriage or she might be chased away by her in-laws. . .. (CR 5)
Perceived challenges to the return of individual research results
In addition to the risks that could arise from the process of returning individual research results, stakeholders also mentioned several perceived challenges to the process of returning these results. The challenges raised were categorized as participant-related, research team-related, study specific, and challenges at institutional and national levels.
Participant-related challenges
Respondents raised concerns about low genetic literacy levels among the local population, misconceptions about the role of genes to only determining paternity which have been associated with limited communities’ sensitization on genetic and genomic research, inability to afford or access more efficacious drugs, and difficulties in navigating the procedures at referral health facilities.
Almost all respondents (51) expressed concerns about the possibility of participants’ misunderstanding and misinterpreting their results. Four researchers noted that pharmacogenomic terms are quite complex to understand, even for the educated. They further noted the absence of direct translation of genomic terms in most local languages in Uganda. Six REC members expressed worry about losing the scientific meaning of genomic information during the translation of consent documents into local languages.
I worry that when there is no direction of genetic terms in local languages, the descriptions of these terms will be left at the discretion of the translator. I find this tricky. . .. because there is a possibility of losing the scientific meaning of the genetic terms, depending on how the translator interprets these terms (REC 2) Many people in our communities only think about paternity when you talk about DNA or genes. They think you want to know who the real father of their children are. . . and this can cause panic in our communities. Many of them don’t know that genes can even tell us how individuals’ bodies respond to different doses of drugs. . .. (CR 5)
Three researchers also expressed concerns about some participants’ inability to afford or access more efficacious drugs matching their genetic profile, and the associated psychosocial implications. They for example, noted that informing participants with low income about the existence of more efficacious drugs than their current regimen might lead to anxiety and loss of hope. Some participants may consequently stop taking their current antiviral treatment and resort to herbal remedies, a decision that may be more harmful to their health.
Some of our clients [People living with HIV] are really very poor and cannot even afford a balanced diet daily. What is still helping them to survive is because the Government provides free ARVs [antiretroviral drugs]. That is why they do not have any choice but to take what is available. Such people may lose hope for the future knowing that they can take something better, but because they are poor, they have to take low class drugs (R 5)
Similarly, two community representatives also expressed concerns about navigating the procedure of receiving care and treatment after referral to government hospitals. They expressed worry that participants might face difficulties in establishing new relationships with healthcare workers at the referral health facilities.
It takes time to build a relationship with the clinicians. . . and sometimes it depends on how kind the health worker is to patients. . . Many of the health workers in Government hospitals are very tired after seeing many patients and by the time it’s your turn, they are not that friendly. . . so you can’t freely open up to them as we do here at the IDI clinic. (CR 5).
Researcher-related challenges
The context where these studies are undertaken may pose several challenges to researchers. Five researchers pointed out the difficulty in determining the appropriate language to use when communicating pharmacogenomics research results, especially in central Uganda, where the society is multi-ethnic. They also worried about the possibility of losing scientific meaning when simplifying or describing genomics terms that have no direct translations in local languages.
You see. . ., we usually try as much as possible to break down these words [genomic terms] into simpler words and try to provide a description of these words, but using many words which can still be confusing. I feel language is a big issue to deeply think about when it comes to genomics research. (R 2)
Nearly all researchers (11) acknowledged that they had limited or no experience in returning individual research results from pharmacogenomic analyses to participants. However, they felt that careful consideration of ethical and social issues needed to be made before engaging research participants in this regard.
With my limited experience in communicating individual results from genomics analyses, I worry about how to balance returning only clinically actionable results and leaving out other results, yet the participant might be interested in learning about all his/ her results. How can I explain to them that both results [clinically actionable and non-clinically actionable results] are important but then return only some? I find this quite complex. . .. (R 12)
Two other researchers also expressed concerns about challenges in tracing and updating participants’ contact information. They noted that some participants do not have personal cell phones while others might change location, thus, making it difficult to contact them when the results are available.
Some participants keep changing their telephone numbers and where they stay. And since these results are sometimes available when the study ended several months ago, it might be hard to trace these participants with incorrect telephone numbers or even when they shift to another location (R 1)
Study-related challenges
Researchers were particularly concerned about the resources required for returning individual research results, as many studies often do not have a budget for this activity.
Most studies do not a budget for returning these results to participants, even if we all appreciate their [pharmacogenomics research results] relevance to patient care. As you know, these activities are tedious and costly, and require a dedicated team, which needs to be well facilitated. . . (R 10)
Moreover, researchers highlighted the numerous consent forms participants have to read and sign before enrollment. They cited the main study consent form, the consent form for the collection and storage of biological samples, and another consent form for genetic testing. They surmised that the numerous consent forms have a lot of information that might confuse participants and make it difficult for them to understand the research information adequately.
I feel the separation of these consent forms makes it hard for both us [researcher] and the research participants to have a meaningful discussion. First of all, the information is too much and when translated into local languages, one can have over 8 pages for one set of consent form. . . That’s already too much for our semi-literate or even illiterate population. . . (R 5)
Institutional-related challenges
Stakeholders highlighted several institutional-related challenges to the result feedback process. They cited the absence of policies and standard operating procedures to guide the process of returning individual research results. They also pointed out that by the time the results are ready, the research staff involved in the participant recruitment activities might have moved on to other opportunities. As such, the available staff may not be competent enough to respond to participants’ concerns. They also expressed concern about the lack of professional genetic counselors.
Usually, us [researchers] who recruit the participants into these studies may not be available to give back the results to participants. Most of our contracts run up to when data collection is completed. So even if we have created good rapport with the participants, we may not be around to answer their questions since our contracts would have expired and we are in search for other job opportunities. . . (R 9) . . .. most research institutions where genomics research is being conducted have no full-time employed genetic counselors, yet these people [genetic counselors] are very instrumental in such institutions especially, when it comes to helping participants digest the implications of these results [pharmacogenomics results] and plan how to move forward (R 7).
Inadequate infrastructure and national level policies
Stakeholders expressed concerns about the limited and inadequate infrastructure, the absence of national guidance for the return of individual research results, and limited interest from the Ministry of Health, policymakers, and the public.
The lack of capacity and infrastructure to perform some biological sample processing and genomic analysis with improved technological techniques was highlighted as the main reason for sample exportation and contribution to prolonged waiting period for research results.
In most cases, we don’t have adequate laboratories to perform some high technological assays, for example, some genomics analyses. So, we have to ship samples to developed countries with such facilities. But the process of shipping samples is sometimes bureaucratic and takes several months to get back the results. . .. Most times participants might even have forgotten about the purpose of the study by the time the results are ready. . . (R 1)
In addition, the absence of national research guidelines on how results from genetic and genomic can be safely returned to participants with minimal ethical issues was emphasized by three REC members.
As you know, the circumstance in which genomics results are returned are not the same as one returning a malaria result. A lot has to be considered when thinking about returning these results. Therefore, without clear guidance from our regulators, it becomes hard to avoid other ethical and social issues that might arise from these results. (REC 12)
Stakeholder recommendations to the perceived challenges
After mentioning the above challenges, the stakeholders were asked to propose the recommendations that could mitigate some of the highlighted challenges. They recommended the need for community sensitization and education, utilizing community engagement approaches to actively engage the research communities, and capacity building of research staff in genetic counseling.
(a) Community sensitization and education
Stakeholders proposed different methods of promoting community sensitization and education to enhance research participants’ understanding of the implications of the results.
Researchers suggested the use of education and communication materials such as videos and flip charts when describing the role of an individual’s genes in metabolizing the drugs taken by the participants. They believed that the visual materials create a lasting imagination that may enhance participants’ understanding of pharmacogenomic research information. Additionally, two community representatives suggested the use of social media platforms to share videos with participants. They felt that such platforms could help participants revisit study-related information at their leisure time for reflection.
Communication technologies have greatly improved these days. Many people now have smartphones where they can watch very educative videos. E.g., if a researcher can share a short video on WhatsApp describing the key things of this kind of research, people will be able to revisit the video over and over again to understand better the research information (CR 30).
One researcher suggested using group education as another way of promoting participants’ understanding of pharmacogenomic research information including results.
I would think about holding group discussions among the potential participants who may be attending the ART [antiretroviral treatment] clinic on a given day. For example, I could plan a session on one Tuesday, the day when many patients come in, then I give them some information about the study and encourage them to ask questions. This way, some other potential participants can learn from others’ questions but can ask more questions when they choose to join the study (R 2) We need to have more open discussions to sensitize our research communities. . . for example, these discussions can be held on radio or TV [television] talk shows. Our people need to have a good understanding of the roles genes play during metabolization of ARVs [antiretroviral treatment] for example. . . (R6)
Researchers suggested having ongoing conversations about the role of pharmacogenomic research with participants on their study visits to improve their understanding. Researchers might also utilize the study visits to update participants’ contact details.
Genetic information is complex and difficult to understand. Participants can’t understand everything about this kind of research on day one, moreover, in less than 30 minutes when you are consenting them. Therefore, I suggest that the research teams spare at least 30 minutes on each participants’ study visit to go over the role of genes in pharmacogenomics research and the purpose of this study. . . (R 1)
Community representatives (03) proposed that the government through the Ministry of Health should come up with strategies or programs to educate communities about the different functions of genes in the human body Many people in our communities do not know that genes play several roles in our bodies beyond just informing us about one’s paternity lineage. The MoH [Ministry of Health] could come up with some programs to educate people about the functions of genes in one’s body. (CR 15)
(b) Community engagement
Two REC members articulated the need to actively engage key community leaders and community influencers before, during, and after returning the results to participants. They felt that community representatives may provide insightful opinions on the appropriate language and tools for communicating the results to participants.
I have observed that some words translated by the professional linguists differ from how those words are interpreted in some communities. Therefore, I would suggest that researchers engage community representatives early enough before the results are ready. These people [community representatives] should read the translated consent forms to confirm that the translated language matches with the kind of language that communities will easily relate with, especially the youth. . .. (REC 5)
(c) Capacity building for example training research staff in genetic counseling
Researchers suggested that research institutions conducting genomics and pharmacogenomics research should employ full-time genetic counselors. In addition, they contended that research institutions should have flexible contracts with some research team members to facilitate the smooth return of individual research results. They also recommended capacity building for genetic counseling.
Research institutions may consider employing full-time genetic counsellors to offer counselling services to participants and families. These people play a very important role in genetic research to support participants even when the research team may not be available. . . (R 3) The government should put in place well developed laboratories where genetics analyses can be performed and validated, and where samples can be re-analysed in case participants or families are not satisfied with the results. . . (R 5)
(d) Developing appropriate ethical and legal frameworks for the return of individual results.
Researchers suggested that the administration of research institutions should develop policies that guide the process of returning individual results. For example, the standard operating procedures and policies should guide researchers on how to negotiate with funders during study budget development to accommodate the activities involved in the process of returning individual actionable results from genomics analyses.
Even if we agree that it is important to return these results to participants, if there are no funds for these activities, nothing can be done. . .. And sometimes researchers are not the best people to negotiate with funders about sparing funds for returning these results. I wish the institutional heads could help researchers on negotiating with funders. . .. (R 5)
Two REC members articulated the need to develop standard operating procedures and frameworks for returning individual results of genomic and genetic analyses.
Genomics and genetics research is quite still new in Uganda and so many researchers might not know how the results should be returned to individual participants. So, if institutions can come up with SOPs [standard operating procedures] describing a step by step process. . ., this might ease the researchers’ work. . . (REC 9)
Several REC members (09) suggested that national regulators of research involving human participants should develop guidelines describing the steps researchers should undertake when returning results to participants to minimize possible social and ethical challenges.
The UNCST should come up a well-researched step by step guideline on how researchers can safely return these results back to participants. . . Remember, these results might have serious ethical, legal and social implications that could harm communities and the participants. . .. (REC 7)
Discussion
This study explored stakeholder attitudes and perceived challenges to the return of individual research results to people living with HIV. Pharmacogenomic research results may be used by researchers and clinicians to guide the appropriate treatment regimen, dosing requirements and minimize adverse drug reactions, especially among people living with HIV, whose wellness and survival is mainly dependent on antiretroviral treatment (Eriksen et al., 2020). Generally, respondents felt that they were obliged to return these results because of the potential benefits to the research participants, despite limited experience on how results should be returned. Withholding health-related information from research participants, especially clinically actionable results, hinders further consultation with experts in search for better treatment or preventive measures, hence deterioration of health. REC members emphasized the aspect of promoting justice; community representatives considered the return of individual research results as a source of hope to people living with a chronic illness and a way of upholding the reciprocity; while researchers believed that the return of individual research results from pharmacogenomic testing would motivate research participants and encourage them to adhere to their medication, hence achieving better health outcomes. Findings from this study are consistent with prior studies where researchers felt that returning genetic results was a way of valuing and respecting participants’ contribution to research (Ralefala et al., 2020).
However, stakeholders highlighted possible risks, challenges and barriers to returning individual research results. They reported the possibility of breaching participants’ confidentiality and privacy, which could result into societal and self-stigma, discrimination, and domestic violence. Similar risks have been reported in the literature from low-resource settings (Kisiangani et al., 2022; Ndayala and Ngige, 2020; Oke et al., 2019; Sarwar, 2023). Several research communities have developed strategies to overcome such risks. For example, the national and international guidelines recommend de-identifying data during data collection, analysis and publishing participant related information (Lee et al., 2020; Raman et al., 2023). While this practice may protect participants’ privacy, the process of returning individual results necessitates clear identification of the participants to provide the results to the right owner. Therefore, researchers should often restrict access of participants’ information to only authorized staff.
Researchers reported having limited experience in returning individual research results because their studies were not designed to feedback individual research results to participants. The national ethics guidelines generally recommend returning results to participants while protecting their rights and safety, so there is need to develop frameworks that guide the feedback process. Researchers also pointed out that they had no access to professional genetic counseling services. The absence of genetic counselors in Uganda has been reported by other researchers (Mwaka et al., 2021a; Ochieng et al., 2022). Genetic counseling is key in promoting full understanding of the individual research results and their implications, prior to participants’ selection of the types of results they wish to receive. We also highlight the necessity of building capacity for genetic counseling in Uganda.
Stakeholders raised concerns about the process of obtaining informed consent for return of individual research results. For example, having numerous consent forms creates bulky and unreadable information; especially in low-resource settings where poor literacy and reading inabilities are rife (Bukini et al., 2020; Dwyer et al., 2021), yet many participants receive genomics-related information for the first time when participating in genomics and genetics studies. Too much information, coupled with numerous consent forms and limited time for discussion, contribute to a poor understanding of genomics and genetics research and is sometimes considered coercive (Bukini et al., 2020; Tindana et al., 2012). In addition, there is a possibility of misinterpretation the results due to low literacy levels, misconceptions about the role of genes to only determining paternity, the complex nature of the genomic and pharmacogenomic terms coupled with absence of direct translation of such terms in many local languages in Uganda (Amayoa et al., 2022; Mwaka et al., 2021a). Recent studies have reported poor understanding of informed consent for pharmacogenomics and genomics research in Uganda (Amayoa et al., 2022; Nabukenya et al., 2022). Inadequate understanding of pharmacogenomics research procedures makes it difficult for participants to make informed decisions when selecting the different kinds of results they would prefer to receive. Therefore, there is a need for a comprehensive description of the results dissemination procedure to ensure that participants are provided with adequate and understandable information to balance their expectations. Members of the RECs emphasized the need to provide participants with simple and understandable information, thus empowering them to make informed decisions. Sweeney and colleagues also encouraged the feedback of results to research participants without researchers’ fears and prejudice of their literacy levels or economic status (Sweeney et al., 2015). Therefore, stakeholders suggested sensitizing communities about the role of genetic and genomic research, and use of simple and understandable language. Furthermore, translation of consent forms from English to local languages are often done by professional language experts registered as certified translation companies. However, sometimes translating scientific concepts into local languages can distort the meaning. Therefore, it is important to involve community representatives when translating research-related documents to guide researchers on the appropriate terms to use when communicating pharmacogenomic research results to participants. Researchers should also consider sharing information through educative multi-media tools: for example, audio-visual materials through platforms such as smartphones to improve participants’ comprehension. However, the use of multi-media may be a challenge to individuals without smartphones or those with poor internet connectivity. Therefore, we also encourage continuous discussions about returning individual research results between researchers and participants during study visits.
Stakeholders were concerned about participants’ difficulty with adjusting to the referral health facilities systems, especially at government health facilities. A number of government facilities are burdened by the high number of patients admitted and attending the outpatient clinic daily, limited human resources, medical supplies, drugs, and space to accommodate all patients’ needs (Musoke et al., 2014; Nabukeera, 2016). However, in the spirit of continued care for participants, researchers and other stakeholders are willing to offer both additional care and support, even after the closure of the research project. Their actions are in affirmation with the principle of reciprocity and solidarity, whose ultimate goal is promoting good health of research participants. Researchers suggested the possibility of contacting their fellow colleagues at referral health facilities to help participants navigate through the difficult systems of referred health facilities. In addition, research institutions should develop a standard operating procedure (SOP) for referral of participants to other health facilities. The SOP should provide detailed criteria for referral, identify a health facility, identify the specialists, and a list of supporting documents for referral.
Furthermore, the process of returning individual research results requires dedicated funds to meet costs of verification of laboratory results, tracing participants, linkage of participants to care, genetic counseling services, and other activities. Given these challenges, opponents of returning individual research results believe that the limited resources could be used to benefit a bigger population than a few individuals (Crawford et al., 2019a). However, limited resources should not be a primary reason for not returning individual research results from genomic and genetic analyses (Ewuoso et al., 2024); rather researchers should plan for these resources at the proposal development stage to maximize individual benefits of research to participants. Therefore, researchers should include a budget for the return of individual research results during the grant application preparations if they anticipate any clinically significant genomic findings.
Study limitations
Genomic research is relatively new to Uganda and is often difficult to understand especially among communities with low literacy levels. The community representatives might not have fully understood the pharmacogenomic concepts used in this study given the limited time in the dFGDs. However, additional information and a vignette were used to provide an illustrative description of pharmacogenomic research during the focus groups. Although using a vignette might have not accurately represented the truth about stakeholders’ attitudes and perceived challenges toward the return of individual research results from the community representatives, we recommend further research on the challenges and possible solutions to promote a safe return of results with minimal ethical and social issues that might arise from returning individual research results to participants.
We also acknowledge the potential sources of bias from the research team. During the interviews and focus groups, the research team was aware of the biases and it was difficult not to relate to our experiences because of our interests in the ethics of individual research results. However, we tried to overcome this challenge through protocol training and ensuring that the interviews and focus groups were moderated by an experienced team that was relatively unknown to the majority of the participants.
Conclusion
Overall, respondents felt it is important to return individual research results to vulnerable groups of people such as people living with HIV. These results provide an opportunity to improve participants’ quality of life and respect participants’ autonomy. However, there is need for caution when returning individual research results whose implications do not only affect an individual but might extend to their families and communities. Due to the low literacy levels and misinformation about the genes and DNA among research communities, we recommend further research on strategies to sensitize the public about the role of genes in the individuals’ bodies. We also recommend building capacity for genetic counselors in Uganda and other sub-Saharan countries where genomics and genetics research is being conducted. Lastly, we recommend researchers to plan for adequate resources that are required for the return of individual research results, especially if they anticipate clinically significant findings.
Footnotes
Acknowledgements
The authors would like to acknowledge the assistance offered by the Faculty at Berman Institute of Bioethics, Johns Hopkins University and the Faculty of Makerere University International Bioethics who contributed to the study design and interpretation of the results. Waitt C is funded by Wellcome Trust Clinical Research Career Development Fellowship. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Ethical considerations
This study was reviewed and approved by the Makerere University School of Biomedical Sciences Higher Degrees and Research Ethics Committee (SBS-855) and the Uganda National Council for Science and Technology (SS 735ES).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Fogarty International Center of the National Institute of Health through the Makerere University International Bioethics Research Training Program Grant Number D43TW010892.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
