Abstract
While there is a large focus on return of genetic research results, more information is needed regarding participant preferences for the return of non-genetic research results generated from biospecimens that provide unclear clinical benefit. Participants in the Rheumatoid Arthritis Study of the Myocardium (RHYTHM) trial were interviewed over the phone. Many participants expressed the desire to receive both non-genetic and genetic research results that do not have clear clinical actionability. Factors that would affect a participant’s desire to receive non-genetic research results that do not have clear clinical actionability included clinical care considerations, empowerment, and personal preference. Regarding when to return non-genetic research results, there was variability in participants’ preferences, with many recognizing the complexities of the research process and wanting to give researchers enough time to analyze results meaningfully. Participants emphasized flexibility in how to return non-genetic research results and considered other factors such as security and environmental concerns. To assist with comprehension of non-genetic research results, participants had a variety of preferences for the extent of details that they wanted, including contact information for questions, additional explanations in lay and non-medical terms, and guidance on next steps. Our data show both similar and divergent participant preferences for receiving genetic versus non-genetic research results that do not have clear clinical actionability. The variety of preferences regarding what, when, and how non-genetic research results should be returned underscores the ethical imperative to move beyond clinical utility by integrating participant experiences and preferences into research practices to build trust and engagement in the research process.
Introduction
There are many unanswered questions regarding the return of research results derived from biospecimens (Wong et al., 2018). While there is a large focus on return of research results in genetics, there is limited information available regarding return of research results for non-genetic data generated from biospecimens, and to our knowledge, no such information is available in rheumatology. Non-genetic results include individual-level data unrelated to genomic sequencing, which can include biomarkers, imaging, physiological measures, or behavioral assessments (Botkin et al., 2018; National Heart, Lung, and Blood Institute (NHLBI) Working Group, 2005; National Human Genome Research Institute, 2023). Specifically, non-genetic results derived from biospecimens can include biomarker levels (e.g. proteins, hormones), metabolic profiles, environmental toxin exposures, immunologic markers, and tissue-based histological findings, all of which may hold personal or health relevance for participants despite lacking genomic content.
There have been multiple studies investigating participant preferences regarding the return of individual genetic research results (Bollinger et al., 2012, 2014; Murphy et al., 2008), and some evidence for patient experience, attitudes, and beliefs on genetic versus non-genetic medical conditions (Kass et al., 2007; Plantinga et al., 2003). Genetic information is generally considered differently than non-genetic information on the policy, research, and individual levels, so we cannot assume that public preferences for return of genetic research results would apply for non-genetic research results from biospecimens (Bollinger et al., 2014). Genetic and non-genetic results with unclear clinical benefit present ethical challenges around return. While lacking immediate clinical utility, such findings may still hold personal value for participants. Genetic results, such as variants of uncertain significance, often carry implications for familial risk and identity, warranting stricter regulatory oversight and the need for genetic counseling. In contrast, non-genetic findings, such as exploratory biomarkers, are typically less regulated and may be more variable in interpretation. These differences highlight the need for return-of-results frameworks that are tailored to the type of data and the informational needs of participants, not just clinical actionability.
Less is known about participant preferences, attitudes, and beliefs regarding the return of individual non-genetic research results from biospecimens that are acquired via innovative tests that provide unclear clinical benefit. New tests and technologies can produce results of unclear significance that are not obviously clinically actionable. What is considered clinically actionable information can be less apparent with rheumatologic diseases; rheumatology encompasses both common and esoteric diseases with heterogeneous clinical presentations, making the data associated with these conditions sometimes difficult to interpret.
The Rheumatoid Arthritis Study of the Myocardium (RHYTHM) trial provides an opportunity to (1) gather participant preferences regarding return of individual non-genetic research results from biospecimens involving innovative tests that provide unclear clinical benefit and (2) determine if participants’ attitudes and beliefs about return of individual genetic research results vary from their attitudes and beliefs about return of individual non-genetic research results.
Materials and methods
RHYTHM trial
The aims of the RHYTHM trial were (1) to determine if imaging indicators of myocardial pathology at baseline are predictive of longitudinal (adverse) change in measures of left ventricular (LV) structure and function over 4 to 6 years in rheumatoid arthritis (RA) patients without clinical cardiovascular (CV) disease at baseline, (2) to determine if RA therapies reduce myocardial inflammation, and (3) to determine if seroreactivity to citrullinated myocardial antigens is associated with myocardial inflammation and impaired myocardial blood flow reserve (MFR), and whether such seroreactivity predicts pathologic changes in parameters of LV structure and function.
The overall hypothesis of RHYTHM was that subclinical myocardial inflammation and microvascular ischemia are prevalent in RA patients and may be mediated in part by antibodies directed against citrullinated myocardial proteins (APCAs), which may promote decline of LV function. The identity and citrullination state of these autoantigens and the relationship of the corresponding APCAs to abnormal myocardial phenotypes was not yet known. The RHYTHM trial used a novel microarray of fractionated myocardial proteins, with identification of the citrullinated myocardial autoantigens by mass spectrometry to determine seroreactivity; therefore, new tools and biomarkers for identifying RA patients at highest risk for progressive myocardial dysfunction may be identified, allowing for earlier intervention.
RA participants enrolled in RHYTHM have been described previously, and were recruited from the rheumatology clinics of Columbia University Medical Center (CUMC) and by referral from local rheumatologists (Amigues et al., 2019). Inclusion criteria were age ⩾18 years and fulfillment of the American College of Rheumatology/European League Against Rheumatism 2010 classification criteria for RA (Aletaha et al., 2010). Exclusion criteria included the following: (1) any prior self-reported physician-diagnosed CV event or procedure, (2) contraindication to pharmacologic stress agents, and (3) active cancer (Amigues et al., 2019).
Interview guide
An interview guide was developed using the existing literature (Bollinger et al., 2012; Murphy et al., 2008) and in consultation with CUMC faculty advisors with interview research experience, to gather information on attitudes and beliefs about participant preferences regarding return of individual research results (not incidental findings) from biospecimens; specifically regarding non-genetic results that do not have clear clinical actionability. Participants were given a definition of what non-genetic results may entail. The interview guide focused on what, when and how data should be returned. Within each of these sections participants were asked about their answers as they related to non-genetic versus genetic research results (see Supplementary Material for the interview guide).
Study population, recruitment, and interview administration
The study sample consisted of 119 RA participants from the RHYTHM trial, and recruitment continued until 20 participants consented to participate. At the time of recruitment, all eligible participants were emailed an information sheet regarding the interview study and the interviewer also called participants to gauge interest in participating. For participants who were interested in participating in the interview, additional information and a consent form for signature were provided through a secure online link, and a consent script at the beginning of the interview was read to participants. An experienced interviewer from the Herbert Irving Comprehensive Cancer Center at CUMC led and recorded all the interviews (AR). Due to limitations during the COVID-19 pandemic, individual interviews were completed over the phone and recorded from December 2020 to February 2021. No detailed field notes were taken during the interviews; however, the interviewer recorded brief notes during each session. Participants were compensated with $20 for their participation. The study was approved by the Institutional Review Board (IRB) at CUMC.
Analysis
We conducted a thematic analysis of interview data from a target sample of 20 participants, using iterative data collection and analysis to monitor for saturation. Interviews were audio-recorded, transcribed verbatim using secure software (CD), anonymized, and transcripts were reviewed alongside the audio files to ensure accuracy (JSK); participants did not review transcripts. One researcher performed initial inductive open coding (JSK). Focused coding was applied to identify the most salient and relevant codes, which were grouped into preliminary categories based on conceptual similarity; discrepancies were discussed until consensus was reached and categories were collaboratively refined into broader themes (JSK, JMB). Pertinent quotes (non-verbatim when appropriate) were used to illustrate key findings. The analysis was reviewed through ongoing collaboration with the full study team. Saturation was confirmed as no new themes emerged in the final interviews. Three interviews lacked audio recordings due to technical issues; however, review of the corresponding notes revealed no new themes.
Results
Characteristics of participants
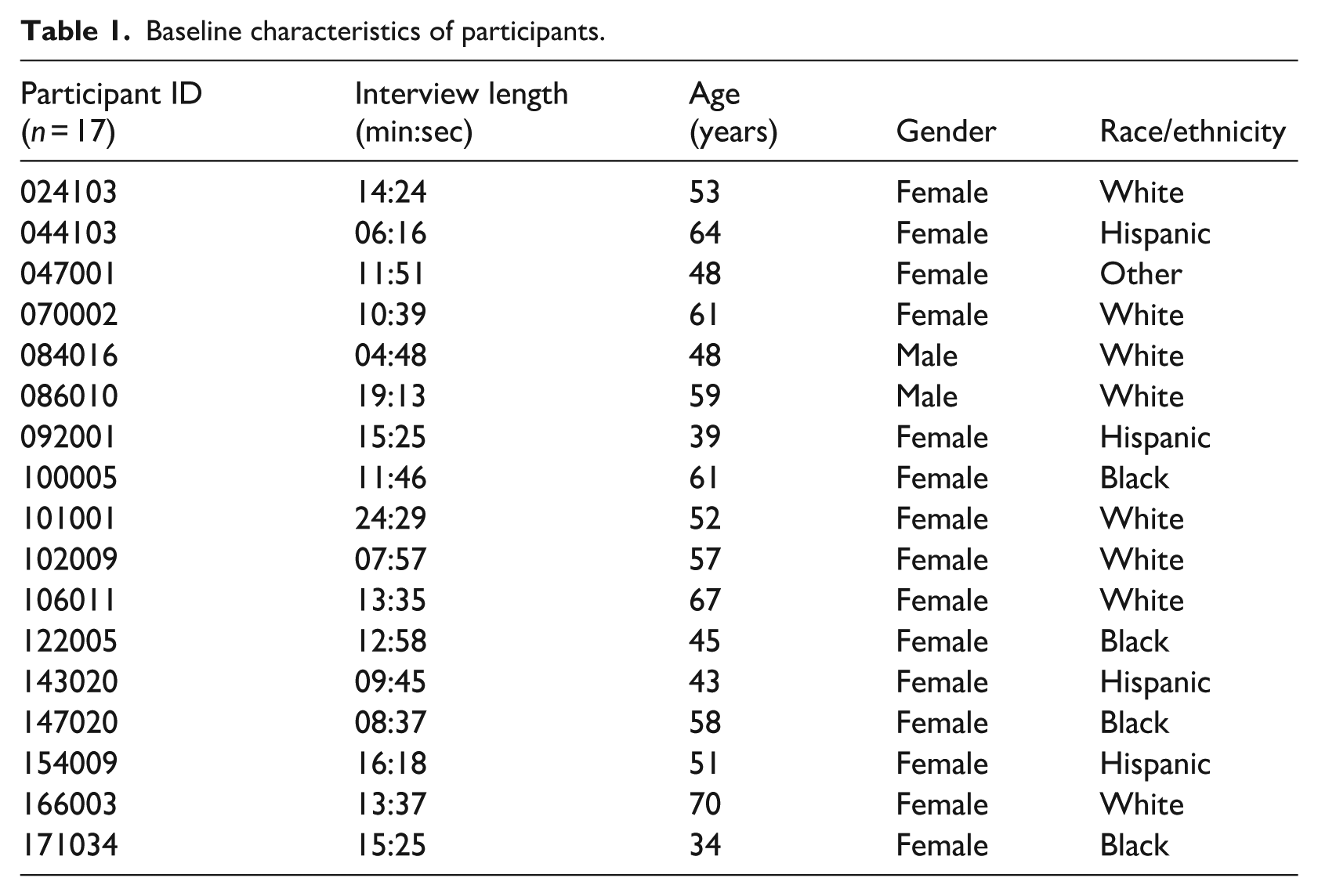
Participants’ characteristics are shown in Table 1; the mean age was 53 (SD = 9.9, range 34–70) and they were mostly female (88%) and White (47%). The study population also included participants who self-identified as Black (23.5%), Hispanic (23.5%), or other (6%). Interviews lasted about 5 to 25 minutes with an average duration of 12 minutes and 46 seconds.
Baseline characteristics of participants.
Receiving non-genetic versus genetic research results
Many participants expressed the desire to receive both non-genetic and genetic research results that do not have clear clinical actionability simply because they would want to have that information. Receiving both non-genetic and genetic results may also help in understanding the cause of their existing medical condition, if it may be from “another family member or great great grandparent, or if it was something with the environment” (147020).
Those who were more comfortable with receiving genetic results mentioned prior experiences in participating in genetic studies, having already thought about the implications of receiving such results. Others emphasized the implications for current and future generations as an important factor for receiving genetic results, including informing their children “so that they can make informed decisions” (102009).
However, others expressed trepidation in receiving genetic results without clinical actionability while recognizing the possible benefits of receiving them, which may include instituting lifestyle changes. One participant highlighted the potential dangers of receiving genetic results without understanding what they could mean and how genetic information could be received or perceived differently than nongenetic results: Genetic information, I think carries a certain weight in people’s minds because it can be used rightly or wrongly for people to convince themselves that they are or are not going to get some condition. . .or that their kids are likely doomed because of some bad gene that we passed on. And if you tell me that a given gene in my DNA exists or is slightly unusual or something without telling you what it means. Most people are going to jump to the worst conclusion. With non-genetic information, I think if you provide me with context, like a reference range for blood work, I think I can make use of that more effectively in understanding where I am and what I need to do and how I get myself ready for whatever it is that I need to worry about in terms of managing my health. And so I just see them as different. I see the harm for misuse of genetic information as being much greater than the harm from routine medical information. (086010)
Privacy and confidentiality were also raised as concerns. One participant was worried that genetic results could have implications on health care coverage, and while they wanted to be informed of genetic results they did not want the health insurance industry to have that information (166003).
Factors affecting return of non-genetic research results
Participants described factors that would affect their desire to receive non-genetic research results that do not have clear clinical actionability. Three themes were identified including (1) Clinical care considerations (2) Empowerment (3) Personal preference.
Clinical care considerations
Participants wanted to have their non-genetic research results back, even if they were not clinically actionable at that time, focusing on the possibility that some intervention or treatment would be possible in the future. The potential benefit of having additional information that may only be available by participating in a research study was also highlighted: “At least I did the test and have some kind of tangible result versus not having done it at all. . .” (171034).
Participants also pointed out the potential to focus on prevention rather than treatment: “I would [want] to know what my risks are and if I could take some precautions to prevent triggering something” (101001). Others noted that they would want to inform their physicians “so they have better information on what’s going on” (147020). Although the information may not be clinically actionable at the time, some idea of prognosis was also desired, for example, “if it could get any worse or what else it could trigger inside of my body” (147020). Another participant felt that they should ultimately benefit from contributing to that research: I think that if there was a benefit that was clear from the study that if you were someone that could benefit from it, you should be at the front of that line. . .The very best that people that are doing this study could do for you. . .inform you that this medicine works and this is what’s happening with it and you can get it here. . .It seems like the people who were involved in the study and giving themselves as guinea pigs, should be rewarded right? (092001)
Empowerment
Access to their non-genetic research results was a form of empowerment for participants. Participants noted that “information is power” (092001), “the more information you have, the better you can take care of your health” (102009), and they “just don’t want somebody else to decide for me what I need to know and what I don’t” (086010). Participants also stated that receiving their non-genetic research results would allow them to “do some research about it” (047001) and “be a little bit more informed and going to the doctor when I need to” (086010).
One participant also expressed concerns with the medical system, and felt that having the information would help maximize and improve the health care received while advocating for themselves: I don’t trust physicians or their PAs or NPs to have the time to go through a full set of test results and recognize something that may not be jumping out at them but might spark a question or concern to the patient. . .that’s not a criticism of doctors, just. . .when the metric is how many patients can you see in an hour? You’re dealing at the very highest level, not getting down into details, so give me the details and I’ll ask questions. (086010)
Personal preference
Many participants shared that their personality traits influenced their desire to receive non-genetic research results, even if they were not clinically actionable, describing themselves as “detailed” and wanting “all the pieces of the puzzle related to my health” (122005) or “curious” (166003). One participant also recognized that preferences were not only closely tied to personality traits but also individual experiences: I feel it’s connected to just your disposition and the type of person you are like. I know for some people they don’t like going to the doctor because they’re afraid they hear something negative even when, for example, they’re already experiencing symptoms or not feeling well or just because of experiences, personal family experiences and connections that make it difficult for them or challenging. . . (122005)
Others also noted that it would not negatively affect them to receive research results, saying “I don’t get upset by stuff like this” and “I don’t get panicky” (092001). Being aware of their research results might also help in life planning: “I think I would rather know. . .say I had. . .a terminal illness. . .the reason I think I would want to know so that I can make whatever arrangements I’m going to make. . .” (106011).
Some participants highlighted a researcher’s obligation to disclose and participants’ right to their own data: “. . .they should let you know. . .and then it’s up to you if you want to do something more with that information” (047001) and “if it’s my information. . .if there’s an option to send it or not, I’m always going to pick yes, it is mine. My name, from my blood. . .” (154009).
When non-genetic data should be returned
Some participants gave various time frames that they would feel comfortable with, ranging from “a few weeks” (092001) to “a year” (086010), but at the same time were more general including wanting to know “as soon as possible” (092001, 044013), “while you could access them” (143020), “it all depends on what the ramifications of the information is” (092001) and “as quickly as somebody can tell me what they mean” (086010). These more general comments seemed to reflect participants’ efforts to respect the research process and the time needed to interpret and make conclusions about the data: I wouldn’t put a time on that because. . .whenever the results are all ready I feel the researcher or research organization, whoever [is] in charge, they’ll send it out whenever they are finished, completed. And I wouldn’t put a time limit on that. I wouldn’t be in a rush to get it, but also whenever they finish whatever they have to do about it, and they send it. (106011)
How to return non-genetic research results
Participants described how they would want non-genetic research results returned to them, which included details on reporting method and report content and comprehension.
Reporting method
Participants had a variety of preferences for how non-genetic research results should be shared with them, which included reports being mailed, emailed and/or provided another way electronically (i.e. online portal), with an option to discuss results with study staff or a study physician. For potentially more serious research results, a phone call initiated by study staff may be preferred: “. . .if there was something extremely negative, or that might make me uneasy. . .I would prefer that someone called me with the results for discussion specifically about what those results are, the impact, and then what are my next steps” (122005). Some participants wanted more flexibility and resources to help them learn more about their research results: Give me results in a form that I can easily read or print out. Provide me with an option to get more information. Whether it’s through links they give me more information about X, Y or Z. Leading up to the opportunity to talk to the doctor or study team, who can take 20 minutes and answer questions or tell me how to interpret the results that you’re sending me. . . (086010)
Other factors in reporting method preference included security and environmental concerns: “I’m not sure I feel comfortable putting it in the computer, so probably a hard copy, mailed” (044013) and “[paper is] becoming obsolete and plus a waste of resources” (047001). Others cited their experience with online patient portals and were very clearly for or against it: The good thing about when you sign into a portal is that, let’s say you lost the email, the information is kept there. . .if you need to go back, you can go back. . .the good thing about the portal is that. . .you have the doctor or the research team or whoever you want to speak to. You don’t necessarily even have to correspond over the phone, you could just email them. . . (171034) . . .just generally, I think patient portals suck. . .Patient portals have become a crutch for doctors to use, so they don’t have to talk to patients. . .I feel so strongly about that, that if I were a participant in a research study and you said go to the patient portal to get your results, that would be the last time I would participate. I think they are devoid of information, and they are impersonal, and I think for somebody who takes the time. . .to come to participate in a study to send them to a patient portal, to me is, it’s insulting. (086010)
The potential impact on insurance coverage or access also seemed to be implicated as a concern for including research results in a patient portal used for clinical care, implying that having a portal separate from the electronic medical record and a choice in specifying how research results would be received was favored.
Report content and comprehension
Participants had a range of preferences for the extent of details that they wanted in the research results report. One participant shared: “. . .just sending me a bunch of numbers or. . .a result tied to the name of attached without some explanation of what that test was and what it was looking for. I don’t find that particularly helpful” (086010). Participants felt that it would be helpful to include a “reference range” (044013), “if I had something terrible. . .some helpful information or pamphlets” (101001), “if there’s something I could change in my diet, in my living” (154009), “a summary. . .or if there was anything in particular that I had to know about, highlighted” (084016), guidance for “next steps” (122005), if follow up with a physician is recommended and “contact information that if you had any questions” (106011). Receiving more information about the research was also noted, “something that puts my mind in the perspective of why the research is being done, or what they are looking for” (143020).
Wording of the results report was also emphasized, with a focus on wording that is “simple, easy to read and understand” (084016), “not so technical” (143020), and a “simplified explanation” in “lay” and “non-medical terms” (106011). One participant who is a nurse stated: “I would probably not prefer doctor language, but layperson language. . .doctor level, in layperson language, if there is such an animal” (044013). Another nurse participant highlighted the importance of considering baseline medical knowledge and other potential barriers to understanding research results: . . .depending on how med savvy they are, they might need the definition of what the actual tests tells you. . .someone can say that they’re a doctor, but they could be a dentist, they could be a vet. . . And then some people also, depending on their knowledge level, whether English is their first language, there’s language barriers. . .those all have to be considered when you’re writing the material. . . (171034)
Some participants wanted a detailed report because "it might be something that might be helpful for my primary care to know or my rheumatologist” (122005). A physician participant also felt that research results should be shared with a participant’s physician to help in interpreting the results. Others indicated the benefits of speaking with the study physician or someone closely involved in the research, since they would have the most knowledge to effectively interpret and explain research results, while understanding the potential limitations of time or resources. Some participants had less of a desire to speak to the study physician: “. . .if the study doctor isn’t someone that can give me things in layman’s terms or will become irritated or frustrated. . .” (122005) and “. . .we don’t know what it [the research results] means. . .or there is nothing I could do. I don’t know. Maybe I’m not too interested in talking to the doctor” (154009).
Ultimately, evaluating participants’ comprehension of their research results may be advantageous: “. . .if there was some kind of questionnaire or something developed after the results were disseminated. In terms of gauging their understanding. . .and then asking them if there’s any questions that they have” (171034).
Discussion
With the evolving advancements in healthcare, we must consider new ethical paradigms that will ensure ethical research. There is a clear ethical motive to notify participants of individual research results that are clinically relevant and/or actionable (Edwards et al., 2012; Klitzman et al., 2013; Ramoni et al., 2013). Consensus groups have also determined that medical actionability should be used to decide if a researcher should return results to a participant (National Bioethics Advisory Commission, 1999). However, new tests and technologies can produce results of unclear significance that are not obviously clinically actionable.
It has become increasingly important to consider a research participant’s preference to receive individual research results and cultivating understanding of those results (Botkin et al., 2018). Participants have also expressed the desire for a less paternalistic approach, and to be able to make the autonomous choice of receiving their results (Cole et al., 2015). Our participants highlighted the need for choice regarding which results are returned, how, and when, emphasizing the ethical duty to embed preference-sensitive mechanisms within the consent process.
Our study advances the ethical discourse on returning individual research results, focusing on non-genetic, non-clinically actionable findings. Participants expressed a strong interest in accessing these results, motivated by values of autonomy, transparency, and empowerment. These insights underscore the ethical imperative to move beyond clinical utility and to integrate participant experiences and preferences into research practices.
Significant changes in federal regulations have allowed people to have greater access to their test results. Since 2014, the Health Insurance Portability and Accountability Act (HIPAA) privacy rule has granted individuals the right to receive a copy of their test results from HIPAA covered laboratories (HIPPA Administrative Simplification. Regulation Text. 45 CFR Parts 160., 2013). In 2017, revisions to the Common Rule further required researchers to disclose whether they will release clinically relevant results, including individual research results, to participants (Federal Policy for the Protection of Human Subjects, 2017). However, the Clinical Laboratory Improvement Amendments (CLIA) of 1988 prohibit the release of individual research results obtained from non-CLIA certified laboratories (Research Testing and Clinical Laboratory Improvement Amendments of 1988 (CLIA) Regulations, 2014). Researchers may use non-CLIA laboratories to analyze biospecimens using innovative methods. In the RHYTHM study, novel microarrays from a non-CLIA certified lab were used to identify antibodies directed against APCAs, serving as an example regarding this tension between returning research results and regulations.
Regulatory barriers, particularly those related to non-CLIA-certified laboratories, complicate the return of results despite their perceived personal significance. Clinical laboratories are subject to rigorous certification ensuring result accuracy and reliability for medical decision-making, whereas research laboratories often lack comparable oversight, raising concerns about the validity and clinical relevance of their findings. However, research labs are often exploring the initial proof and relevance of molecules to a disease process, so there may be no validation possible.
This distinction creates an ethical tension between honoring participant autonomy and protecting against potential harm from uncertain or non-validated results. To address this, researchers should transparently communicate these limitations during informed consent, provide clear guidance on how to interpret research results, and consider offering confirmatory testing in certified labs when appropriate. Future policy should reconcile these constraints with ethical obligations, especially as research increasingly intersects with clinical care.
Although it has been argued that genetic information is not significantly different from other medical information (Beckwith and Alper, 1998; Gostin and Hodge, 1999), it has generally been treated differently on multiple levels. On a policy level, state laws presume that genetic information requires special protection (Annas, 2001). On a research level, a significant amount of attention has been placed on participant preferences regarding the return of individual genetic information (Bollinger et al., 2012, 2014; Murphy et al., 2008). On an individual level, there has been a strong interest in personal genetic information as evidenced by the popularity of direct-to-consumer testing. Research participants are also highly interested in receiving individual genetic research results (Goodman et al., 2018; Vears et al., 2021). However, while many prefer to receive all genetic results (Bollinger et al., 2014; Hoell et al., 2020), when given a choice, some want only some or none at all (Hoell et al., 2020). Participants also express interest in receiving non-genetic research results (Purvis et al., 2017), but they tend to prefer genetic research results—particularly those related to medication response and disease risk (Wilkins et al., 2019)—over non-genetic findings such as heart imaging (Sayeed et al., 2021). Participants of genomic research can also perceive their genetic data as “riskier” than other types of medical data (Ruiz-Canela et al., 2011).
It has been shown that participants’ perceived value of well understood research results is higher than for results of unknown significance (Angal et al., 2023), and most people prefer receiving well-validated individual genetic research results for serious, actionable diseases (Bollinger et al., 2014), which is consistent with existing recommendations (Bookman et al., 2006; Fabsitz et al., 2010; Jarvik et al., 2014; National Bioethics Advisory Commission, 1999). However, many participants value genetic results with no definite clinical use (Allen et al., 2014; Bollinger et al., 2012; Meulenkamp et al., 2010; Murphy et al., 2008), suggesting that return of results should not be limited to those considered clinically actionable (Yu et al., 2014). Our participants had a strong preference to receive their research results, whether genetic or non-genetic, even if they were of unclear clinical benefit or actionability. However, some participants expressed specific concerns about receiving genetic research results. These included implications for current and future generations, misinterpreting results to assume the worst outcome, privacy and confidentiality, and potential effects on insurance coverage.
The three major factors that would affect our participants’ decision to receive non-genetic research results that do not have clear clinical actionability included: (1) Clinical care considerations (2) Empowerment and (3) Personal preference. Similarly, a prior study found that participant expectations for receiving research results included inspiring a change in behavior to improve one’s health, feeling empowered to do something about the results, and providing recommendations if there was a major issue that needed follow up (Sayeed et al., 2021).
Variability in participant preferences, as demonstrated in our study, supports the belief that researchers should offer to share research findings though different modalities, allowing participants to choose from a list of preferred reporting methods at the time of consent (Purvis et al., 2017). When considering the return of individual genetic results, receiving detailed reports has been considered more valuable than providing staff to discuss results (Bollinger et al., 2014). Our participants also preferred detailed reports for non-genetic research results, specifically to share with their primary care provider or specialists to help them interpret their data, similar to those receiving genetic research results (Allen et al., 2014; Richmond et al., 2024). However, it has been shown that participant satisfaction was highest when research results were communicated though a meeting with a researcher (Long et al., 2016), and many of our participants appreciated having this option.
Participants in our study raised several ethical considerations regarding the return of individual non-genetic, non-clinically actionable research results. These included trust (or distrust) in medicine, a participant’s right to data, a researcher’s obligation to return research results, and giving back to participants. Other participants have expressed that receiving research results is valuable and that such disclosure would enhance their trust in researchers (Wilkins et al., 2019). One could argue that participants have a right to individual research results, even if they are not clinically actionable (Fernandez et al., 2004; Shalowitz and Miller, 2005). Most research participants believe they have a right to their data (Purvis et al., 2017) and that researchers are obligated to return individual research results (Bollinger et al., 2012; Meulenkamp et al., 2010). Sharing such information can foster participant engagement in the research process and, as observed in our study, empower participants to take a more active role in their clinical care. Many individuals feel that receipt of individual research results serves both as an incentive to participate (Goodman et al., 2017) and as a form of compensation (Bollinger et al., 2012).
A precautionary strategy would support the comprehensive disclosure of individual research results, accompanied by suitable protective measures to prevent, reduce or manage potential risks to participants (Resnik, 2011). However, the ethical justifications for a generalized duty to return research results have been questioned, one argument being that not all research results will have implications that are considered “exceptional” (Miller et al., 2008). Rather than returning only actionable results, considering a “return of value” may be another approach, focusing on participants’ perceived value of different types of results and considering what participants find most meaningful and potentially beneficial (Wilkins et al., 2019). The counterargument is that the purpose of research is to provide generalizable knowledge rather than individual benefit. Individuals often assume they will certainly benefit from participating in research, highlighting concerns that returning individual results will trigger the therapeutic misconception (Burke et al., 2014).
Other major considerations in returning non-genetic research results include provoking anxiety, distress, and unnecessary medical workups or treatments; researchers have expressed some of these concerns in returning research findings to participants (Purvis et al., 2020; Shalowitz and Miller, 2008), including incidental findings (Cole et al., 2015; Kang et al., 2024). However, our participants did not emphasize these concerns when receiving non-genetic research results, perhaps because our interviews focused on hypothetical results that did not have definitive clinical actionability. Return of genetic research results also does not seem to be associated with substantial psychological harm (Vears et al., 2021).
Our study has some limitations. Interview studies are susceptible to interviewer and social desirability bias, leading participants to not share their true opinions. Due to technical issues, notes rather than transcripts were used for three interviews, which may have limited the data captured. This could have led to missed divergent insights and a premature conclusion that data saturation was reached. Additionally, while thematic analysis allows for rich interpretation, it is inherently subjective and shaped by researcher perspectives, despite efforts to ensure analytic rigor through collaborative coding and review. As with most interview-based research, our findings may also be influenced by self-selection bias; the answers of those who did not participate may have provided additional and diverging themes. Our sample was predominantly female, reflecting both the higher prevalence of RA in women and the demographics of the RHYTHM study, from which 82% of participants were female (Amigues et al., 2019); women may also have been more comfortable discussing personal views. Compared to the broader RHYTHM cohort, our sample included a higher proportion of White and Black participants, with fewer Hispanic participants. This may have influenced the range of perspectives captured, particularly regarding cultural or community-specific experiences. Our demographic data does not include other factors that might affect preferences and would be important to investigate further; it has been shown that preferences for different types of research results differs between socioeconomic groups (Richmond et al., 2024; Wilkins et al., 2019).
Significance and future directions
Recommendations and regulations require investigators to communicate whether, under what circumstances, and how individual research results, especially from biospecimens, will be offered and returned. Our data show both similar and divergent preferences for receiving genetic versus non-genetic research results that do not have clear clinical actionability. Given the variety of preferences regarding what, when, and how non-genetic research results should be returned, it is important to ascertain participant preferences to build trust and confidence in the research process.
We propose a participant-centered framework for returning individual research results focusing on five key guidelines: (1) proactively assessing and respecting participant preferences throughout the consent process and the study; (2) ensuring transparency about result nature, limitations, and implications; (3) providing meaningful participant choice regarding receiving results; (4) navigating regulatory challenges while advocating for policy reform; and (5) promoting equitable and inclusive processes, particularly for underrepresented or marginalized populations. By centering autonomy, transparency, empowerment, and reciprocity, this approach goes beyond narrow clinical utility paradigms and fosters ethically responsible research that builds trust and engagement.
Supplemental Material
sj-docx-1-rea-10.1177_17470161251395759 – Supplemental material for Participant preferences for the return of non-genetic research results: An interview study of participants from a rheumatoid arthritis trial
Supplemental material, sj-docx-1-rea-10.1177_17470161251395759 for Participant preferences for the return of non-genetic research results: An interview study of participants from a rheumatoid arthritis trial by Jane S. Kang, Andria Reyes, Christopher Depender and Joan M. Bathon in Research Ethics
Footnotes
Acknowledgements
The authors would like to thank all the participants who participated in RHYTHM and contributed data to this publication.
Ethical considerations
This study was approved by the Columbia University Institutional Review Board (protocol no. AAAS6601) on February 20, 2020.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health (3R01AR050026-14S1).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
