Abstract
This study explores how qualitative health researchers navigate the demands of medical research ethics committees in Germany where qualitative research is subject to approval only when it is conducted in medical contexts. We present the results of a grounded theory study to investigate qualitative health researchers’ experiences with procedural ethics and the strategies they adopt to navigate its demands. Our analysis revealed six dimensions of experience and three strategies adopted by researchers to navigate the demands of medical research ethics committees. All participants agreed that research ethics is of high importance in qualitative health research, but strategies to navigate the demands of medical research ethics committees ranged from avoiding, and adapting, to transforming the procedures of ethics review. Based on our findings, we provide recommendations for improving the ethics review of qualitative health research.
Keywords
Introduction
The obligation to obtain approval for medical research from an independent research ethics committee (REC) is enshrined in article 23 of the Declaration of Helsinki of the World Medical Association (2013). Legal and professional regulations and levels of implementation of this ethical requirement vary across the globe. For example, it is a requirement in countries such as Australia, Canada, South Africa, the UK and the US that researchers obtain ethics approval from a REC for all research involving humans, whether it be medical or social science research (Allbutt and Masters, 2010; Cutcliffe and Ramcharan, 2002; Gillam et al., 2009; Guillemin and Gillam, 2004; Haggerty, 2004; Reid et al., 2018; Stevenson et al., 2015). The situation is different in countries such as Germany, Austria and Denmark.
In Germany, research involving humans is subject to approval by a REC located at a medical faculty or a medical professional organisation if it is carried out by a physician or at a medical faculty or hospital, or if the participants are patients. On the other hand, there is no nationwide legal or professional obligation for social scientists working at the faculty of social sciences to obtain approval from an independent REC for research with humans, and specialist social science RECs are relatively uncommon in Germany (Hopf, 2004; RatSWD: Rat Für Sozial-Und Wirtschaftsdaten, 2017; Von Unger et al., 2016). Whether qualitative health research is subject to independent review by a REC located at a medical faculty or medical professional organisation depends primarily on situational factors, such as where (medical hospital or faculty or elsewhere), by whom (physicians or non-physicians) and with whom (patients or nonpatients) the research is carried out, rather than the nature of the research methods or the level of risk involved. Qualitative health researchers in Germany, especially those affiliated with hospitals or medical faculties, face practical difficulties in navigating the ethics review process, as procedures and requirement can vary substantially from project to project.
Guillemin and Gillam (2004) term the full process leading up to obtaining ethics approval of a study from an independent REC ‘procedural ethics’. Procedural ethics encompasses the development of materials like research protocols, participant information sheets, consent forms, and ethics application forms as well as communicating with the responsible REC (Chiumento et al., 2020; Johansen and Frederiksen, 2021). Guillemin and Gillam (2004) contrast procedural ethics with ‘ethics in practice’, which refers to the practice of identifying, reflecting on, and addressing ethical issues during the research process (see also Tolich and Tumilty, 2020; Von Unger, 2021). Although procedural ethics and ethics in practice may seem, superficially, to be distinct components of ethical research, Guillemin and Gillam (2004) argue that there is considerable continuity between them, further enhancing the complexity of navigating these types of research.
While there is extensive literature on the challenges of ethics review of qualitative social science and qualitative health research by RECs, this literature refers primarily to regulations and practices in anglophone countries (Guillemin et al., 2012; Israel, 2015; Pollock, 2012; Stevenson et al., 2015; Van Den Hoonaard, 2011; Van den Hoonaard and Hamilton, 2016). A number of scholars have criticised the development of institutionalised ethics review procedures because these formal procedures can pose barriers to scientifically important and socially valuable qualitative research (Allbutt and Masters, 2010; Cannella and Lincoln, 2018; Haggerty, 2004; McCormack et al., 2012; Stevenson et al., 2015; Thompson and Harper, 2012; Thompson and Russo, 2012; Tilley et al., 2009; Von Unger et al., 2016; Øye et al., 2016). It has been argued that institutionalised ethics review procedures are especially likely to pose barriers to ethnographic and participatory research, as well as research with so-called ‘vulnerable’ individuals (Bell, 2014; Bell et al., 2014; Dingwall, 2008; Israel, 2015; Murphy and Dingwall, 2007; Van den Hoonaard, 2011; Van den Hoonaard and Hamilton, 2016; Øye et al., 2007).
One possible explanation for the challenges in the ethics review of qualitative health research, especially in countries with regulatory frameworks similar to those in Germany, is that review procedures of RECs located at medical faculties or medical professional organisations tend to be tailored to quantitative biomedical research. This can result in a mismatch between ethics review criteria and key methodological principles of qualitative research, such as the lack of predefined hypotheses, the openness towards unexpected outcomes, and the iterative nature of the research process (Doyle and Buckley, 2017). Moreover, the risks of qualitative research are less predictable because qualitative researchers exercise less control over their research setting than researchers using experimental and quantitative methods (Bosk and De Vries, 2004; Murphy and Dingwall, 2007).
Other authors have argued that challenges in the ethics review of qualitative research can be explained by features of the ethics review process itself (Gillam et al., 2009; Guillemin and Gillam, 2004; Pollock, 2012). These authors contend that due to insufficient knowledge about qualitative research methodology amongst their members, RECs apply ethics review criteria rigidly and are unable to adapt them to qualitative research approaches (Cutcliffe and Ramcharan, 2002; McCormack et al., 2012; McMurphy et al., 2013; Pollock, 2012).
Still, other authors envisage a more positive role for RECs in the ethics review of qualitative health research. They have contended that the differences between quantitative and qualitative methods and the challenges inherent in the ethics review of qualitative health research have been overstated and that qualitative health research must be subject to ethics review by an independent research committee (Jennings, 2012; Potthoff et al., 2023; Wassenaar and Mamotte, 2012). These authors also proposed models that aim to address the identified challenges and increase the competence of RECs in the review of proposals that include qualitative methodologies.
The main research question for the current interview study was as follows: How do qualitative health researchers navigate the demands of medical RECs in contexts in which qualitative research is subject to approval by a REC only when it is conducted in medical contexts? Qualitative health research in Germany is an illustrative example in this regard. The focus of our interview study was on experiences with medical RECs rather than with RECs in general (including committees located at other faculties). For ease of expression, the term ‘REC’ will be understood as referring to a medical REC. Recommendations for improving the ethics review practice for qualitative health research can be developed based on our findings.
Methods
We followed the methodology of grounded theory according to Strauss and Corbin (1990). Grounded theory is a qualitative methodology that aims to develop middle-range theory from the data rather than simply undertaking descriptive analysis. The interplay of data collection and analysis is a core feature of the methodology; data analysis is initiated once the first data has been collected and the results of the analysis of the first data guides further data collection. While concepts are to be derived from the data, the use of so-called ‘sensitising concepts’ enabled us to make use of our prior literature-based knowledge and personal experience with the ethics review process.
The research team included one researcher with a background in sociology, one in social sciences, and one in philosophy and medical ethics. Two researchers had experience in applying for ethics approval from a REC and one researcher served on a medical REC.
Sampling strategy
We used theoretical sampling to generate the sample so that the nature of the sample was guided by the results from the preliminary data analysis and the provisional theory constructed (Glaser and Strauss, 1967). The initial inclusion criteria were:
being a researcher with expertise in qualitative health research; and
having experience in applying for ethics approval from a medical REC for qualitative health research projects in Germany.
In accordance with the method of theoretical sampling, we determined additional dimensions based on the analysis of the first data collected. The additional dimensions that we identified were professional background, university faculty, and level of experience with ethics applications.
An initial list of potential research participants was made based on the contact details of authors of articles in qualitative health research and referrals from colleagues in our own network. SP screened the profiles of these researchers on their university homepages using the initial inclusion criteria and later also the additional dimensions. Researchers were approached for research participation only if they were likely to meet the inclusion criteria based on the information on their public personal profile. We sent out invitations in sets of three, started data collection and analysis as soon as we secured an interview, and sent out the next set of invitations only after the initial data analysis of the previous set was concluded. In the full process, a total number of 25 potential participants were contacted via email, some of whom distributed the invitation to their colleagues. Of the people we approached directly, three did not fulfil the inclusion criteria, seven declined research participation, nine did not respond and seven agreed to participate.
Although we initially set out to include more participants to cover the additional dimensions (i.e. professional background, university faculty, and level of experience with ethics applications), our working assumption that the nature of the data collected would depend on these factors was not confirmed during the further analysis. Moreover, the data collected from the seven interviews was so rich that we believe we reached theoretical saturation regarding our main research question.
Data collection
Data were collected by means of problem-centred interviews (Witzel, 2000). The problem-centred interview is a semi-structured interview method that aims to elicit the experiences and views of participants about a specific problem or topic. While they are more structured than narrative interviews, problem-centred interviews enable researchers to combine structure and openness by using open questions to prompt free narratives and actively relating these narratives to the problem or topic under discussion (Witzel, 2000). SP conducted the interviews in Germany via zoom or telephone, depending on the preferences of individual participants between June and November 2021. Carrying out face-to-face interviews was not possible due to Covid-related restrictions. The interviews lasted between 22 and 62 minutes excluding pre- and post-conversations, where only one interview lasted 22 minutes and the duration of the other interviews was close to an hour. All interviews were audio-recorded and then transcribed verbatim by FR. The data analysis was carried out using the original German transcripts. The interview excerpts quoted in the article were translated into English with the assistance of a native English speaker. We tried to stay as close as possible to the original German transcripts when translating, but sometimes it was necessary to find idiom and set phrases in the English language with a comparable meaning to ensure comprehensibility while preserving meaning.
The interview guide included open narrative questions to participants about:
their experience in applying for ethics approval from the responsible REC for qualitative health research projects in Germany;
their experience with challenges in the ethics review practice; and
their suggestions for improving the ethics review practice of qualitative health research.
Data analysis
We used the MAXQDA 2020 Standard software (VERBI Software GmbH, Berlin, Germany) for data analysis. SP and FR began open coding the data after the first data was collected and followed an iterative data analysis process. Each researcher worked on a separate transcript first and then exchanged these transcripts and crosschecked the transcript that the other researcher had worked on to ensure intersubjective comprehensibility. The researchers added, changed and restructured codes while crosschecking, and all changes made were discussed between SP and FR until consensus was reached. The researchers each formulated working assumptions during the analysis, and these were documented using memos. The other researcher provided feedback and expanded on the working assumptions documented in the memos while cross checking. Disagreements were discussed until consensus was reached. Using axial coding in a next step, SP and FR jointly adapted and restructured the initially emerging codes by comparing codes across transcripts and focusing on the data from the perspective of the working assumptions that emerged from the initial analysis. In a final step, SP and FR jointly distilled a middle-range theory from the data using selective coding, resulting in the reconstruction of three strategies for dealing with procedural ethics. All steps of the iterative data analysis were discussed continuously between SP and FR until agreement on the final interpretation was reached. Provisional results of the data analysis were repeatedly discussed with MS to improve intersubjective and interdisciplinary comprehensibility of the analysis.
Ethical considerations
Research participants were informed about the details of the study, both verbally and in writing, and gave their written informed consent before participating in the study. Although the study was not conducted with patients nor in a clinical context, it was subject to approval by the REC because members of the research team were affiliated to the faculty of medicine. The study received ethics approval from the REC of the medical faculty of the Ruhr University Bochum (registration no 20-7133).
Results
Sample characteristics
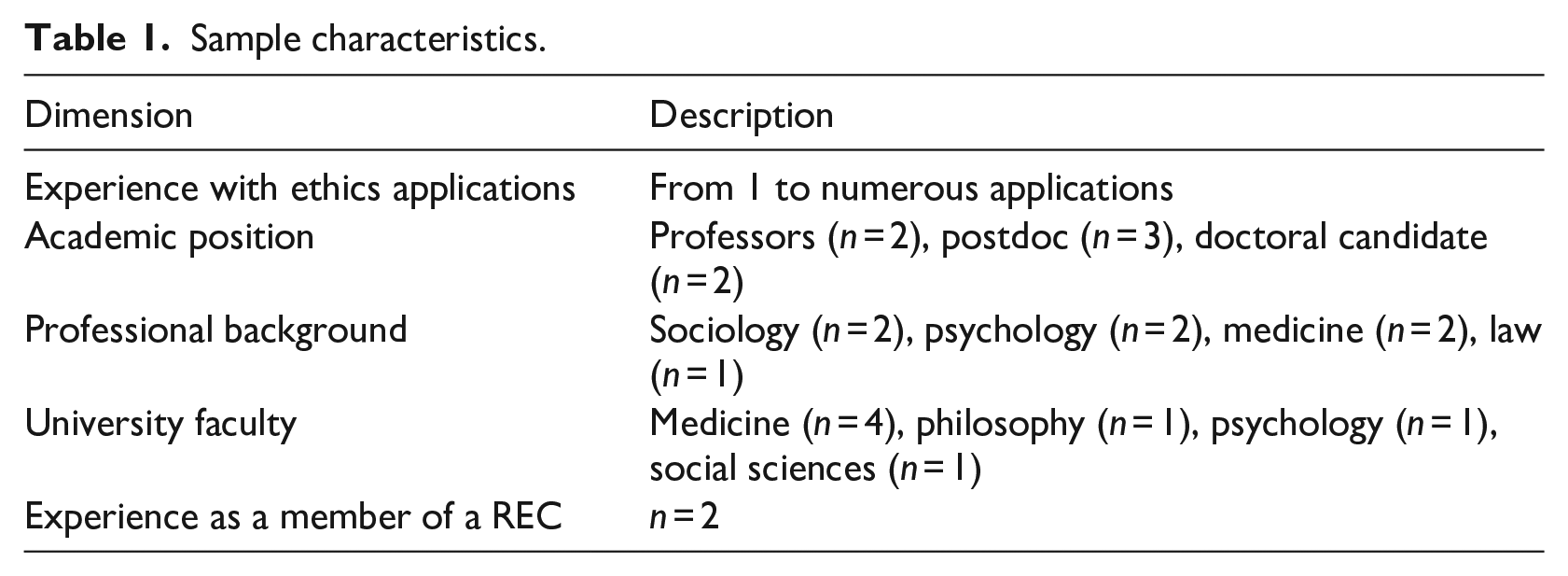
The final sample consisted of seven researchers. Sample characteristics are summarised in Table 1.
Sample characteristics.
Overview of results
We developed two main categories from the data analysis and pre-structuring of the data according to the themes of the interview guide:
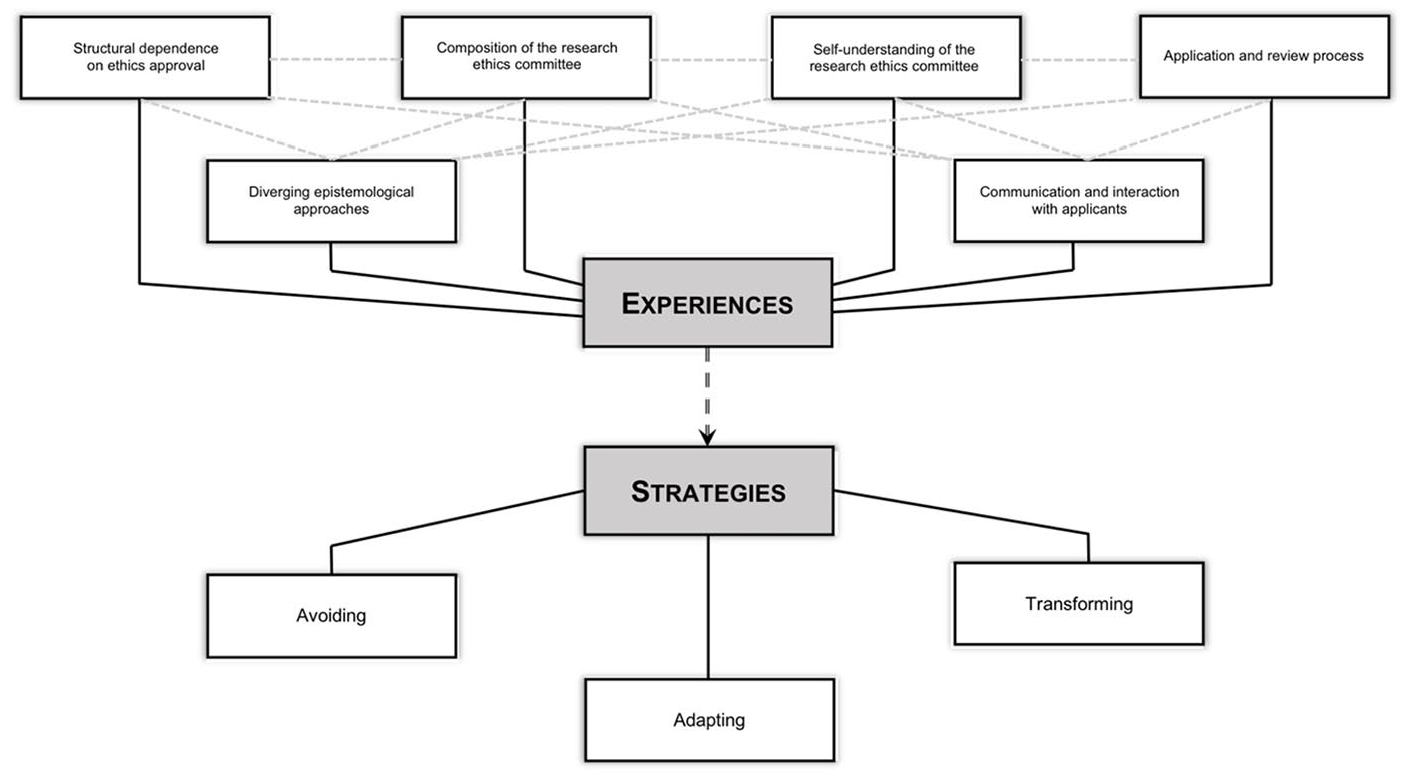
Overview of results.
Dimensions of researchers’ experiences with procedural ethics
This category includes both positive and negative appraisals of experiences of researchers with procedural ethics.
Structural dependence on ethics approval
An important dimension of experience that we revealed from the data is the structural dependence of researchers on ethics approval. Participants addressed the requirements of international journals, typically from the Anglo-American area, that all submitted articles reporting results from studies with humans have ethics approval from an independent REC. This requirement sometimes contradicted the advice and guidance of the local REC, which did not require ethics review for the study. This lack of consistency between the requirements for ethics approval of international journals and local RECs can lead to uncertain situations for researchers.
This kind of structural dependency relates to the requirements not only of international journals but also of national and international funding agencies. Some agencies require ethics approval by the local REC at the time at which a funding application for research with human participants is made. The situation becomes more pressing for researchers if the time frame for obtaining ethics approval is longer. One participant emphasised this kind of structural dependency of researchers by attributing a ‘position of power’ to RECs:
This research participant attributes a position of power to RECs, not merely because they can decide whether a study is carried out, but also because their procedures and time frames can indirectly dictate whether or not researchers can submit a funding application and fulfil their obligations to project partners on the proposal.
Diverging epistemological approaches
Another dimension of experience with procedural ethics is the variety of epistemological approaches – such as social constructivism and positivism – among members of RECs and applicants. Positivistic epistemological assumptions are reflected in the specialisation of medical RECs’ assessment of clinical research and medical trials that use a quantitative research design. Participants emphasised that epistemological assumptions go hand in hand with judgements about scientific quality and suitable research methods, and these assumptions result in judgements about what is good science, and which methodological approach is legitimate.
Problems for qualitative researchers can arise when RECs evaluate qualitative studies based on quality criteria for quantitative research methods.
Further assumptions about research methods and quality criteria are also reflected in unsuitable guidance materials and templates provided by RECs for qualitative studies. RECs often expect a well-defined hypothesis, a sample size calculation, and a fixed research protocol in advance. However, establishing these things before study commencement is at odds with the idea of open qualitative study designs, as a participant noted:
Composition of the REC
The composition of a REC is a further dimension of qualitative researchers’ experience with the practice of ethics review. Participants emphasised how individual members of a REC shape the committee’s understanding of the scientific quality and practise of ethics review. They thus assigned special importance to the inclusion in RECs of members with knowledge and experience of qualitative research.
In addition, participants mentioned the personal values of individual committee members as a factor influencing the ethics review process. The personal values of individual members can play a decisive role in determining which research is considered ethically sensitive and which is not. This can lead to inconsistencies between REC members.
Another participant ascribed a particularly pronounced sensitivity to ethical issues to medical RECs in light of the long history of research ethics in the field of medicine.
Opposing the view that medical RECs are not suitable for the ethics review of qualitative research, this participant contended that, in virtue of the long history of research ethics in clinical trials, RECs at medical faculties may be better at identifying potential ethical issues in qualitative research than research committees in other faculties. The combination of medical experience and experience in RECs can thus also be a positive factor in the ethics review of qualitative research.
Self-understanding of the REC
We identified the self-understanding of the REC as another dimension of experience revealing two main types: the REC as an advisory or the REC as a supervisory body.
Formal written feedback and the lack of personal exchange with applicants were seen as typical for a REC that perceives itself as a supervisory body.
Participants sometimes missed the advisory function of RECs, and they saw this function as being replaced by supervision, standardisation, and bureaucracy. Some participants underlined that RECs could offer their special ethical expertise to researchers instead of monitoring them. They wished to make use of the ethical and legal expertise in RECs because they encountered challenges in their research which they were unable to resolve by themselves. A concrete example of such a challenge is data management.
Participants considered face-to-face exchanges as an important element of the advisory role. One participant emphasised that face-to-face exchanges offer a better opportunity to discuss and address ethically problematic issues.
Communication and interaction with applicants
The experience of our participants was that the self-image of a REC affects how it communicates and interacts with applicants before and during the review process. One participant referred to the tone of voice used by RECs. The tone of voice was seen as reflecting the level of engagement of committee members with the details of the research project during the ethics review process.
Excerpts like this one suggest that researchers can interpret feedback from the REC in different ways depending on the committee’s tone of voice when providing feedback. The style of communication also signals whether communication is at eye level and in an advisory capacity, or whether it is hierarchical, in a supervisory capacity.
Some participants pointed out that the expectations of the RECs were not transparent. Because of this lack of transparency, they felt insecure about how to present their research project when they were invited to give a face-to-face presentations.
This interview excerpt shows that a face-to-face meeting does not necessarily change the interaction between the REC and individual researchers for the better. Regardless of whether the communication is in writing or verbal, the manner of communication is important to researchers’ experience of the ethics review process. Participants found it important, for example, that they are being listened to, that the criteria against which their study is evaluated are transparent, and that the feedback they receive is constructive and professional.
Application and review process
Participants reported a variety of experiences including the review procedure and the duration, and financial costs of the application. We categorised these experiences as belonging to the dimension of experience application and review process. Some participants reported that the time frame for the ethics review process is too long.
Others reported that, at their institution, expedited review procedures were in place for research with no more than minimal risks and burdens for research participants. One participant pointed to an expedited checklist procedure for this type of research that was in place at the local REC, and which shortened the application process.
Some participants reported that their RECs require a quite substantial fee to be paid upon submission of an ethics application. This posed an obstacle, especially for qualitative researchers who were not or do not want to be part of larger research groups with a high budget. One participant noted that the fees attached to an ethics application pose a hurdle to innovation in research and emphasised that an expedited application and review process free of costs offers the opportunity for independent research.
Researchers’ strategies to negotiate procedural ethics
We reconstructed from the data a mid-range theory including three types of strategy that qualitative researchers adopt to navigate procedural ethics. These are the
Avoiding procedural ethics
The strategy of avoiding procedural ethics involves making strategic changes to the research design and protocol to circumvent the obligation to obtain ethics approval. The regulatory framework in Germany does not require researchers to submit their research to the local REC for ethics review if the research is not carried out by physicians, at a medical faculty or hospital, and does not involve patients. Frustrating experiences with procedural ethics on the part of applicants (e.g. rejection of an application without adequate justification) can lead researchers to adopt the strategy of avoiding procedural ethics by adapting their research in such a way that they can conduct it within legal and professional limits, but without ethics approval by a REC. For example, a participant reported a situation in which the researchers switched from finding patients to community volunteers as prospective participants to circumvent the medical REC, which was the only REC available at the university. In the case at hand, the study was subject to ethics approval by the medical REC because the initial approach to finding research participants was via a hospital.
The research team was able to adopt the strategy of avoidance because none of the team members were medical professionals or affiliated with a medical faculty or hospital, and the team ultimately decided not to enrol patients. The interview excerpt demonstrates the lack of transparency regarding which research studies require ethics review and which do not, and why.
Although the strategy of avoiding procedural ethics removes obstacles to carrying out qualitative health research, it creates uncertainty regarding the dissemination of results. This is because most international journals require that all reported studies have received ethics approval from an independent REC. Given the lack of agreement between the German regulatory framework and the requirements of international journals, the aforementioned lack of transparency can lead to situations in which researchers see the need for ethics approval only upon submission of the research report to an international journal. This can lead researchers to submit their research to the local REC retrospectively.
We interpreted the practice of applying for retrospective ethics approval for research merely for dissemination purposes as part of the strategy of avoidance because it is a practice that is fundamentally at odds with the purposes of ethics review. Researchers’ desire to avoid having to apply for ethics review retrospectively can lead them to adopt the second strategy of negotiating procedural ethics, which we have identified as
Adapting to procedural ethics
The strategy of adapting to procedural ethics involves fulfilling the formal requirements for ethics review without a strong commitment to ethical reflection upon the research design and process. Although the strategy is predominantly tactical, it does not imply that researchers who adopt the strategy consider research ethics in general as unimportant. The strategy of adapting to procedural ethics grants researchers the assurance that they can carry out their research while avoiding uncertainties about the dissemination of their results in international journals. Reflecting on the time when they requested retrospective ethics approval, the participant continued to tell us the following:
This might not lead to the improvement of the ethical quality of research, as researchers may adopt a pragmatic attitude and submit applications for ethics review in a merely precautionary and defensive manner.
Submitting precautionary ethics applications is a way of dealing with bureaucratic and procedural requirements rather than a way of improving the ethical quality of research. This is why we conceptualise this strategy as adapting to procedural ethics. This does not imply, however, that those who adopt the strategy of adaptation consider research ethics to be unimportant. Rather, qualitative researchers who adopt this strategy often do not expect helpful advice from the local REC or are trying to avoid becoming stuck between the requirements of the local REC and those of the international journals upon which they are structurally dependent.
In this context, qualitative researchers sometimes try to get advice informally about what can be done to improve the chance that one’s research projects is approved by the REC. Colleagues who are members of the REC provide a gateway to this information, as the following quote highlights:
We interpreted this use of personal contacts with members of the REC for informal preliminary assessment and advice as a form of adaptation if it is merely a pragmatic strategy to improve one’s chances of success in getting ethics approval of one’s research project rather than a genuine research ethics consultation. This informal preliminary assessment and advice can also be categorised as belonging to the third type of strategy – that of transformation – if the main concern is to improve the ethical quality of the research.
Transforming procedural ethics
The strategy of transforming procedural ethics involves attempts by researchers to improve the ethical quality of their own research as well as the quality of ethics review. Like the other two strategies mentioned earlier, this strategy also emerged in response to frustrating experiences with the ethics review process and targets both the REC and the applicants themselves; applicants need to write better ethics applications for qualitative studies, and REC members need more expertise in qualitative research for an adequate review. Participants were self-critical and their strategies aimed at improvement of ethical reflection in qualitative health research on the part of both RECs and applicants. One concrete proposal was to submit high-quality ethics applications for qualitative studies, including a comprehensive and clear presentation of the qualitative approach and methods. In this way, applicants compensate for a lack of knowledge about qualitative research amongst members of a REC and pursue a strategy of adaptation. At the same time, a strategy of transformation is gradually emerging as qualitative researchers implicitly enlighten members of RECs about the value and usefulness of qualitative research approaches. Qualitative and quantitative methods serve different purposes, and both have their own scientific legitimation.
Participants also referred to the importance of a stronger anchoring of ethical reflection in teaching and supervision to achieve qualitative research of good methodological and ethical quality. A greater sensitivity to ethical issues in research can be achieved amongst students and early-career scientists through teaching and supervision. This should include knowledge of the principles of research ethics and procedural ethics as well as education about research ethics of practice, which is particularly important in qualitative research.
Another strategy of transformation is the change of perspective from a qualitative researcher and applicant to a member of a REC. One participant accepted an invitation to become a member of a REC and described the experiences associated with this change of perspective as follows:
The participant here described how she was able to see many of the ethical issues in qualitative research only after she became a member of the REC, and she expressed the concern that members with primarily medical expertise might fail to identify these issues (e.g. because they might assume that qualitative research is ethically unproblematic because it is not invasive). The participant underlined once more that change should work in two ways: RECs should become more sensitive to the particularities of qualitative research designs, and qualitative health researchers should acquire the competence to state their aims, methods, and ways of addressing ethical issues with more clarity. Transformation should occur in both directions.
Discussion
Our results reveal six different dimensions of experience of qualitative health researchers with procedural ethics and three strategies to deal with it: avoidance, adaptation, and transformation. Although our findings relate to qualitative health research, some of the findings may have a broader meaning and be valid for other types of qualitative research as well. Which of the three strategies was adopted by researchers depended primarily on their experiences with the local REC. The importance of experiences with the local REC for the strategy adopted by researchers is reflected in five of the six dimensions of experience, namely the composition of the REC, its epistemological approach, its self-understanding, its style of communication, and the structure of its processes and procedures. Experiences like these have been reported in the literature, for example, in relation to the quality of communication (Brown et al., 2020; Guillemin et al., 2012), differing epistemological approaches (Van den Hoonaard, 2011), and lack of knowledge, not only about qualitative methods but also about emerging technologies such as big data (Ferretti et al., 2022) and social media (Sellers et al., 2020).
One dimension of experience that influenced researchers’ strategies was not dependent on researchers’ experiences with the local REC, namely the structural dependence of researchers on ethics approval. This experience was due to the discrepancies between the requirements of international journals and funding agencies, promoting the strategy of adapting to procedural ethics. The adaptive strategy gives researchers assurance that they can carry out their research and disseminate the results.
Despite the differences in the legal regulations and practical implementation of the ethics review of qualitative health research between Germany and Anglophone countries, there are various similarities between qualitative researchers’ responses to procedural ethics. Pragmatism in dealing with procedural ethics, as reflected in our strategy of adaptation, for example, was also described by Van den Hoonaard (2011), who conducted an extended ethnographic study on ethics review practices relating to qualitative social science research in the US and Canada. Van den Hoonaard (2011) examined strategies that researchers adopt to deal with the demands of research ethics boards, which range ‘from total avoidance to full compliance’ (p. 17). Like Van den Hoonaard, we understand the avoidance of procedural ethics as a pragmatic response of researchers to their experiences with the local REC rather than an ethical orientation of researchers, as Taylor and Patterson (2020) understand it. At the same time, Van den Hoonaard’s (2011) strategy of avoidance encompasses many forms of avoidance that are not reflected in our results, such as researchers opting for off-campus contract research not subject to ethical review, or using anecdotal accounts as data (p. 201). This discrepancy between findings can be explained by differences in the ‘loopholes’ in the applying regulatory frameworks.
Taylor and Patterson (2020) identified three types of responses of qualitative sociologists to REC inquiries in Canada: ‘active engagement, apparent compliance, and overt opposition’ (p. 162). While active engagement is comparable to the strategy of transformation and apparent compliance comparable to the strategy of adaptation, we did not find a strategy comparable to overt opposition. One possible explanation of this is that our sample was not heterogeneous enough for attitudes of overt opposition to emerge. It was not practically possible to actively seek out additional research participants displaying this attitude overtly, as such public displays of opposition towards ethical research procedures are rare. More plausible to us is that qualitative research outside of the health context is not generally subject to ethics review in Germany. As a consequence of this, overt opposition might not be necessary for those who can avoid difficulties by using other strategies. Our analysis suggested, moreover, that qualitative health researchers who cannot avoid procedural ethics because they are physicians or work at a medical faculty are less inclined to oppose procedural ethics. Our sample included researchers who were socialised, at least to some degree, in medicine, and the institutionalisation of ethics review has a long history in this discipline in comparison to the relatively new developments in social science research (Wassenaar and Mamotte, 2012). This can also account for the fact that we did not find extreme responses like overt opposition.
Taylor and Patterson (2020) conclude from their analysis that the type of strategy adopted ‘depends partially on social structural factors such as gender, age, and institutional membership that determine what sources of power are available to individual academics’ (p. 162). Our study did not confirm this finding. Although our method does not allow us to define conclusive associations, the three strategies were adopted across the dimensions of the sample, notably experience with ethics applications, academic position, professional background, and faculty affiliation. Our analysis indicated, moreover, that researchers opt for a specific strategy depending on their experiences with their local REC, rather than their ethical orientation.
A potential limitation of our study is its relatively small sample size. Finding suitable participants was difficult due to the narrow inclusion criteria of our study. The potential sensitivity of the research topic, in combination with the precarity of academic jobs and careers, could also have played a role in our lower response rates. Given the explorative nature of the study, the findings might not describe the experiences and strategies of qualitative health researchers in navigating the demands of RECs in an exhaustive way. On the other hand, the data collected in seven interviews was already so rich that we did not expect to find additional experiences or strategies by carrying out more interviews. The focus of our study was on the experiences of qualitative health researchers with medical RECs in Germany. More research is needed to explore the experiences of qualitative health researchers working in Germany with RECs at other faculties. The applicability of our findings to other national contexts is limited by the fact that the German regulatory framework is rather unique. At the same time, this can also be considered a strength of our study. Given the ample evidence available on the ethics review of qualitative health research in English-speaking countries, our findings usefully complement the available evidence base.
Conclusions and recommendations
Our study provides important insights into the experiences of qualitative health researchers with RECs in Germany and the strategies they adopt to navigate the demands of procedural ethics. Findings from our study can inform attempts to improve the quality of the ethics review of qualitative health research. They can also inform training modules for members of RECs to raise awareness regarding the challenges faced by qualitative health researchers. The following suggestions for improvement of the ethics review process for qualitative health research can be derived from our findings:
RECs and qualitative health researchers should communicate with each other in respectful and appreciative ways.
Qualitative health researchers should have the opportunity to receive advice from the REC before they apply for ethics approval.
RECs should display transparency regarding the requirements, expectations, and evaluation criteria for qualitative health research.
RECs should make available templates tailored to qualitative research, including queries to tease out potential ethical challenges.
RECs should include members with expertise in qualitative methods.
Members of RECs should be informed about the particularities of qualitative research.
A module on research ethics should be integrated in the curriculum of social science studies and the training of qualitative health researchers.
It remains to be investigated whether these suggestions for improvement can count on consensus among all relevant stakeholders, notably members of RECs.
Footnotes
Acknowledgements
We thank Carla Scheytt for her helpful comments on an earlier draft of the manuscript and we thank all the people who took the time to participate in our study.
Availability of data and materials
We cannot share research data publicly as individual privacy could be compromised, but research data are available from the corresponding author on reasonable request.
Funding
All articles in Research Ethics are published as open access. There are no submission charges and no Article Processing Charges as these are fully funded by institutions through Knowledge Unlatched, resulting in no direct charge to authors. For more information about Knowledge Unlatched please see here: ![]() This research is supported by a FoRUM grant from the Medical Faculty of the Ruhr University Bochum (grant number F982-2020).
This research is supported by a FoRUM grant from the Medical Faculty of the Ruhr University Bochum (grant number F982-2020).
Ethical approval and consent to participate
The study has received ethics approval from the Ethics Committee of the Medical Faculty of the Ruhr University Bochum, Germany, on 4 February 2021, registration no. 20-7133. All participants provided written informed consent prior to enrolment in the study.
