Abstract
Background:
There is an increased demand for nutrition during pregnancy to improve fetal growth and development. Several dietary interventions have been recommended to pregnant women to meet their maternal needs. Using a larger sample size, we aim to assess the effect of balanced protein–energy supplementation given to pregnant women on birth outcomes.
Objective:
To evaluate the effect of balanced protein–energy supplementation given to pregnant women on birth outcomes.
Design:
Search included PubMed, Cochrane Central, and Embase from initiation till 20 March 2023 to select relevant studies examining perinatal factors associated with balanced protein–energy supplementation. Mean differences (MD) and risk ratios (RRs) with a 95% confidence interval (CI) were calculated using Review Manager.
Data sources and methods:
Randomized controlled trials and observational studies were included. Out of 218,720 studies initially identified, 24 met the inclusion criteria, involving pregnant women who received balanced protein–energy supplementation and reported outcomes related to perinatal death, birthweight, length, or head circumference.
Results:
Meta-analysis of pooled studies revealed that balanced protein supplementation had a significant effect on birthweight (g) (MD: 62.67, CI: 27.92–97.43), low birth weight (LBW) (RR: 0.73, CI: 0.57–0.95), birth length (MD: 0.20, CI: 0.10–0.30), and small-for-gestational-age (SGA) babies (RR: 0.74, CI: 0.59–0.93) and had no significant effect on the birth head circumference (MD: 0.05, CI: −0.09 to 0.20), perinatal death (RR: 0.83, CI: 0.50–1.37), and preterm birth (RR: 0.86, CI: 0.68–1.08).
Conclusion:
Balanced protein–energy supplementation is an effective intervention for birth length, birth weight, risk of LBW, and SGA births, particularly in women who are undernourished.
Plain language summary
Introduction
Fetal mortality, defined as the loss of a fetus during gravidity, remains a significant global health concern. 1 This includes stillbirths (fetal deaths) and neonatal deaths that happen within the first week of life. 2 Approximately 2 million stillbirths occur worldwide each year, but a lot of these mishaps can be avoided through antenatal care of satisfactory quality. 3
During pregnancy, sufficient nutritional intake is indispensable for optimal fetal growth and development. 4 To address this need, various dietary interventions, particularly balanced protein–energy supplementation, have been recommended for pregnant women.5,6 Balanced protein–energy supplementation refers to a nutritional intervention where protein contributes to less than 25% of total energy intake, ensuring sufficient caloric and protein intake without excessive protein load. These supplements aim to improve birth outcomes, especially for undernourished mothers. 7
We aim to see through analysis if balanced protein–energy supplementation can decrease preterm birth, which means giving birth before completing 37 weeks of gravidity. The exact gestational age at which preterm birth is distinct from spontaneous abortion is differentiated by circumstances and areas. The United States has a preterm birth rate of approximately 12%–13%, while in Europe and other developed regions, it ranges from 5% to 9%.8,9 Premature birth poses significant health risks for infants, including conditions such as respiratory distress syndrome, growth irregularities, and lifelong impairments in eyesight and hearing.
Each year around 20 million babies have birth weight below 2500 g, and a substantial proportion, approximately 40%, of these cases occur in developing nations.10,11 Addressing these concerns, a Cochrane review by Ota et al. has indicated that balanced protein–energy supplementation during pregnancy can tremendously enhance both birth weight and birth length. 12 In contrast, recent meta-analyses led by Kramer and Kakuma have revealed no significant effects of protein supplementation that is unbalanced on pregnancy results, encompassing birth weight and gestational age. Among supplements for undernourished pregnant women, balanced protein–energy supplementation offering less than 25% of total energy is recognized as the most beneficial. 6
Prior studies found that giving balanced protein–energy supplementation was associated with birth weight,13 –35 low birth weight (LBW),14,16,17,19,24,25,31,32 birth length,14 –19,21 –25,28,29,31,33,35 birth head circumference,14,16,17,19,22 –25,28,29,33,35 perinatal death,14,15,17 –19,25 –27 small-for-gestational-age (SGA) baby,14,17 –19,24 –28,31,32,35 and preterm birth.14,16 –19,24,26,27,33,35 A systematic review by Ota et al. found there is a suggested likelihood of reduced stillbirths with balanced energy/protein supplementation compared to no supplementation. 36 A previous meta-analysis by Stevens et al. found an association between balanced protein supplementation and birthweight, birth length, and head circumference, but the population consisted of pregnant women from only low and middle-income countries Findings demonstrated a significant increase in birth weight with balanced protein–energy supplementation, while no significant effects were observed on birth length or head circumference. 37 Another meta-analysis by Imdad et al. found an association between balanced protein supplementation and birth weight, risk of lower birth weight, SGA babies, stillbirth, and preterm births. 38 Given the emergence of new studies in this field since the most recent meta-analysis, our objective is to provide an updated analysis, encompassing all relevant research published since 1973.We intend to thoroughly assess the influence of administering well-balanced protein–energy supplements to expectant mothers on birth outcomes.
Methods
Data sources and search strategy
The study was reported following the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines (Supplemental Table 1). 39 Two separate investigators performed a literature search on databases, including PubMed, Cochrane Central, and Embase, spanning from their initiation to March 20, 2023. The search strategy used: (Protein supplements OR protein supplementation) AND (primigravidae OR pregnant women) AND (fetal loss OR perinatal death OR neonatal death OR fetal death OR stillbirth OR fetal mortality OR birth weight OR low birth weight OR birth length OR birth head circumference OR preterm birth OR small for gestational age baby). We initially applied built-in database find randomized controlled trials (RCTs) and observational studies to refine our search results. However, we acknowledge that built-in filters may introduce limitations due to indexing inconsistencies, potentially excluding relevant studies. To mitigate this, we conducted manual screening of search results and references to ensure a comprehensive selection of studies. The keywords used for the literature search included “Protein supplementation,” “Pregnant women,” “Perinatal death,” “Birth weight,” “Birth length,” “Birth head circumference,” “Small for gestational age baby,” and “Preterm birth.” The following MESH terms were included: (fetal loss OR fetal death) (perinatal OR neonatal death). Detailed search strategy in presented in Supplemental Table 2.
Study selection and inclusion criteria
RCTs and observational studies (e.g., cohort study or case–control study) were included. Quasi-randomized trials (cross-over trials), case reports, reviews, study protocols, comments, or posters as they may introduce bias or lack the necessary controls for causal inference were excluded. The inclusion criteria encompassed pregnant women who received balanced protein–energy supplementation, which means supplementation where protein provided <25% of the total energy content. This criterion ensures consistency in the type of supplementation under consideration. Studies were excluded where the primary intervention involved providing diet health education to gravidas to improve their protein consumption, high protein supplementation (where protein provides greater than or equal to 25% of the sum of energy content), isocaloric protein supplementation (where protein replaced an equal amount of non-protein–energy content), or poor-energy nutrition for gravidas who were either overweight or had experienced significant weight gain early in pregnancy. Eligible control included “routine,” and “no intervention” as this would enable meaningful comparison with the intervention group. The main measure of outcome was perinatal death but in the absence of perinatal death data, we used stillbirth, fetal loss, or fetal death as relevant outcomes. We incorporated studies that involved anthropometric measurements of intrauterine growth, such as birth length, birth weight, and birth head circumference. To identify and eliminate duplicate articles from the systematic search, we utilized Endnote (Clarivate Analytics, Thomson Reuters Corporation, Philidelphia, USA).
Data extraction and outcomes
Two reviewers independently gathered appropriate data using a predefined data collection table. Any disparities between the reviewers were resolved by an impartial third reviewer. For all qualified articles, information including the primary author’s name, study design, publication year, sample size, country of origin, and both raw and adjusted data related to fetal outcomes were extracted and recorded in a prearranged data extraction sheet. In cases where data were missing for certain participants, we assumed that the data were missing at random and performed sensitivity analyses to assess the impact on our results. Fetal outcomes such as birth weight, length, head circumference, SGA babies, perinatal death, and preterm birth were included. For the extraction of data on perinatal death studies providing information on fetal loss, fetal death, and/or stillbirth were also considered.
Statistical analysis
Review Manager, version 5.4 (The Nordic Cochrane Centre, The Cochrane Collaboration), for all our analyses, was employed. The summary estimations for the meta-analyses were depicted as relative risk (RR) for categorical data and mean difference (MD) for continuous data, accompanied by 95% confidence intervals (CIs). Statistical significance was determined using a p-value threshold of 0.05. Substantial heterogeneity was observed among the studies, with an I2 value exceeding 50%. To explore potential variations in fetal outcomes based on the study design, we performed a subgroup analysis. This analysis aimed to investigate whether the impact on fetal outcomes differed between undernourished pregnant women and adequately nourished women. Additionally, we performed a leave-1-out sensitivity analysis to examine if any individual study disproportionately influenced the combined results. Furthermore, we performed a sensitivity analysis using adjusted data from studies that provided such data. This analysis allowed us to assess the potential influence of adjusted data on the overall study findings. Funnel plots of key outcomes that showed significance as per analysis were used to explore the publication bias. Two researchers autonomously evaluated the risk of bias in the individual studies with the help of the Cochrane Risk of Bias Tool (RoB 2.0) across several domains: sequence generation, allocation concealment, blinding of personnel, selective reporting, blinding of outcome assessment, handling of incomplete data, and other potential biases. If any discrepancies arose, a third researcher was consulted to reach an understanding. The Risk of Bias assessment was categorized for each domain as either low, moderate, or high. In our analysis, we found that the risk of bias was low. Grade criteria were used to evaluate the quality of evidence. According to this, overall evidence quality was adjudged as “high,” “moderate,” “low,” or “very low.” 40
Results
Baseline characteristics
The initial literature search yielded 218,720 records. After a thorough evaluation of 82 potentially eligible articles, we ultimately included 24 studies in our analysis. The PRISMA flowchart visually illustrates the study selection process (Figure 1). The sample sizes of the included studies ranged from 20 to 1708 participants, with a total of 11,305 participants. Of these, 7171 were categorized as adequately nourished, while 4134 were identified as malnourished. Table 1 presents the baseline characteristics, study design, and assessed outcomes of the included studies. Additionally, Table 2 outlines the reasons for the excluded studies from the analysis.

Flowchart of the systematic review process.
Summary of the included studies in the systematic review (N = 24).

OMH: Oklahoma Memorial Hospital; WIC: women, infants, and children; SGA: small-for-gestational-age; BMI: body mass index; LBW: low birth weight; RCT: randomized controlled trial; SD: standard deviation; CSB: corn soya blend.
Excluded studies characteristics.

Quality and risk of bias assessment
According to the quality evaluation of the studies that were included (Supplemental Figure 1), 1 study has a low risk of bias, 1 study has a moderate risk, and 22 studies have a high risk.
Results of meta-analysis
The combined findings from the studies regarding birthweight (g) reveal a statistically significant moderate impact of supplementation (MD: 62.67, CI: 27.92–97.43) (Figure 2). Notably, there was substantial heterogeneity of 81%, prompting the utilization of random-effects models. In one of these studies, despite the fact that the authors classify the supplement as balanced, it had a higher percentage of protein-based energy (>25%). 28 Upon excluding this study, although slightly diminished, the impact of supplementation was still statistically significant (MD: 61.07, CI: 26.12–96.02, I²: 82%) (Supplemental Figure 2). Furthermore, in the intervention group of two studies, balanced protein–energy supplements were administered alongside micronutrient supplements, with micronutrient supplements also administered to the control group. 21 Excluding these studies increased the effect of supplementation (MD: 69.02, CI: 33.60–104.44, I²: 78%) (Supplemental Figure 3). According to a stratified analysis based on the nutritional status of mothers, the effect of protein supplementation was significant in both malnourished (MD: 96.17, CI: 53.00–139.34, I2: 70%) and adequately nourished (MD: 36.16, CI: 9.60–62.73, I2: 30%) women (Figure 2).

Analysis of comparison: balanced protein–energy supplementation versus control, outcome: birth weight.
According to data on LBW incidence (birthweight <2500 g), balanced protein–energy supplementation reduced LBW by 27% (RR: 0.73, CI: 0.57–0.95) (Figure 3). Since there was heterogeneity in the pooled data (I2: 52%), random-effect models were applied.

Analysis of comparison: balanced protein–energy supplementation versus control, outcome: low birth weight (<2500).
Data from studies on birth length (cm) indicate that supplementation had a significant moderate effect on birth length (MD: 0.20, CI: 0.10–0.30, I²: 12%) (Figure 4). One of these studies was excluded because the intervention supplement contained more energy on protein (>25%) despite the fact that the authors saw that the supplement had a modest but significant effect of supplementation (MD: 0.19, CI: 0.10–0.28, I²: 0%) (Supplemental Figure 4). 28 An analysis of the subgroups depicted that the effect of protein supplementation was significant in only adequately nourished women (MD: 0.23, CI: 0.11–0.35, I²: 16%) and not in malnourished women (MD: 0.09, CI: −0.08 to 0.27, I²: 0%) (Figure 4).

Analysis of comparison: balanced protein–energy supplementation versus control, outcome: birth length.
Regarding birth head circumference (cm), protein supplementation had no significant effect overall (MD: 0.05, CI: −0.09 to 0.20, I²: 67%) (Figure 5). Subgroup analysis revealed no significant changes in birth head circumference for malnourished (MD: 0.08, CI: −0.16 to 0.33, I²: 35%) and adequately nourished women (MD: 0.05, CI: −0.13 to 0.23, I²: 75%) (Figure 6). However, one study, where the intervention included supplemental foods made from animal sources and locally grown dark green leafy vegetables, showed a significant effect on birth head circumference when excluded (MD: 0.11, CI: 0.01–0.21, I²: 33%) (Supplemental Figure 5). 16

Analysis of comparison: balanced protein–energy supplementation versus control, outcome: birth head circumference.

Subgroup analysis of balanced protein–energy supplementation in adequately nourished women versus malnourished women. Outcome: birth head circumference.
Regarding perinatal death, protein supplementation did not impact the risk (RR : 0.83, CI: 0.50–1.37, I²: 62%) (Figure 7). Even after excluding a study involving corn soya blend (CSB) supplementation, there was no significant difference (RR: 0.73, CI: 0.48–1.08, I²: 34%) (Supplemental Figure 6). 19 Subgroup analysis revealed a 44% decrease in the risk of perinatal deaths in malnourished women (RR: 0.56, CI: 0.32–0.99, I²: 0%), while no significant effect was noted in adequately nourished women (RR: 1.09, CI: 0.56–2.10, I²: 22%) (Figure 8).

Analysis of comparison: balanced protein–energy supplementation versus control, outcome: perinatal death.

Subgroup analysis of balanced protein–energy supplementation in adequately nourished women versus malnourished women. Outcome: perinatal death.
In the pooled results, the intervention group had a 26% lower risk of SGA babies than the control group (RR: 0.74, CI: 0.59–0.93, I²: 87%) (Figure 9). Excluding a study that included fortified formula milk from the analysis reduced the risk reduction to 16% (RR: 0.84, CI: 0.76–0.92, I²: 16%) (Supplemental Figure 7). 31 A subgroup analysis revealed a 40% risk reduction in SGA babies (birthweights below the 10th percentile for babies of the same gestational age) for malnourished women (RR: 0.60, CI: 0.41–0.87, I²: 87%), while no significant effect was observed for adequately nourished women (RR: 0.91, CI: 0.83–1.00, I²: 0%) (Figure 10).

Analysis of comparison: balanced protein–energy supplementation versus control, outcome: small-for-gestational-age baby.

Subgroup analysis of balanced protein–energy supplementation in adequately nourished women versus malnourished women. Outcome: small-for-gestational-age baby.
Pooled results for preterm birth (<37 weeks) showed that between the groups, there was not a significant difference in the risk of preterm birth (RR: 0.86, CI: 0.68–1.08, I²: 43%) (Figure 11). Subgroup analysis indicated no significant impact on the risk of preterm birth for malnourished women (RR: 0.85, CI: 0.66–1.10, I²: 0%) and adequately nourished women (RR: 0.83, CI: 0.58–1.21, I²: 62%) (Figure 12). Removing a study that involved CSB from the analysis did not change the insignificance of the risk of preterm birth in the intervention group as opposed to the control group (RR: 0.91, CI: 0.75–1.11, I²: 23%) (Supplemental Figure 8). 19 The table on “summary of findings” using the GRADE criteria is shown in Table 3.
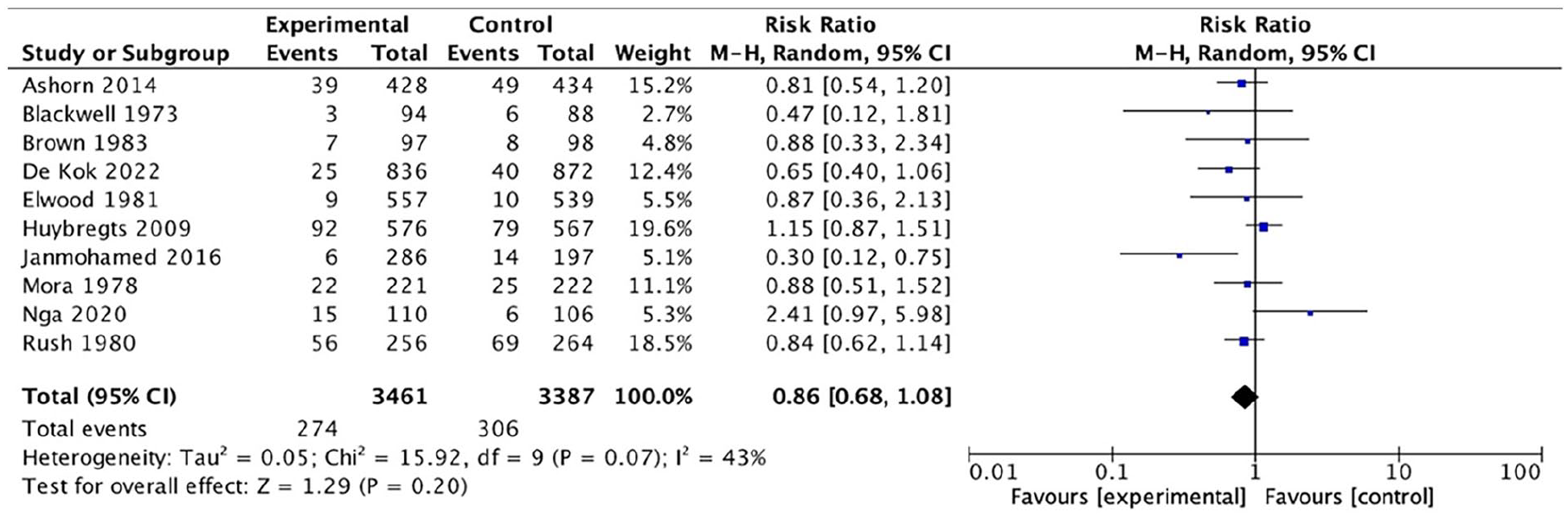
Analysis of comparison: balanced protein–energy supplementation versus control, outcome: preterm birth.

Subgroup analysis of balanced protein–energy supplementation in adequately nourished women versus malnourished women. Outcome: preterm birth.
GRADE criteria for quality evidence and pooled analysis.

RCT: randomized controlled trial; CI: confidence interval.
Publication bias
For birth weight (g), birth length (cm), SGA (birthweights below the 10th percentile for babies of the same gestational age), and LBW (birthweight <2500 g), the publication bias funnel plots are displayed in Supplemental Figures 9 to 12. No discernible publication bias was found across all of the results as the funnel plot was adjudged to be fairly symmetrical.
Discussion
Our meta-analysis has deduced that the overall effects of protein supplementation on fetal outcomes are positive. The administration of protein supplementation during pregnancy was observed to have beneficial effects on weight and length at birth, as well as a decreased probability of LBW. Moreover, in the subgroup of malnourished women, SGA and perinatal deaths both experienced significant declines.
In our study, the impact of protein supplementation on birth weight and the occurrence of LBW showed positive findings. It adds to the growing body of evidence supporting the benefits of protein supplementation during pregnancy. The observed increase in birth weight aligns with previous findings and underscores the crucial role of adequate protein intake for optimal fetal growth. For instance, a recent systematic review and meta-analysis by Laura Pimpin also reported a positive association between protein supplementation and increased birth weight, although the magnitude of the effect may vary because they used animal protein supplementation. 53 According to the combined data, adding protein–energy to the diet had a moderately notable impact on the birth weight. Neonates whose mothers received protein–energy supplementation had an average birth weight difference of 62.67 g from those whose mothers did not. These findings are comparable with a meta-analysis by Imdad and Bhutta for birth weight which also suggests that prenatal protein–energy supplementation can result in a modest but significant rise in birth weight. 38 We did a sensitivity analysis for this outcome by removing Girija et al., after which the reduction in birth weight caused by protein–energy supplementation was 2.5%. 28 The results imply that a supplement with a greater amount of protein-derived energy might help birth weight when opposed to balanced protein energy supplements. Nevertheless, recent evidence in Ota et al.’s meta-analysis contradicts the previous findings, which showed that a higher risk of having SGA babies has been associated with high-protein supplementation. 12 About LBW, our study revealed that when LBW prevalence was supplemented with a balanced protein–energy ratio, it decreased by 27% in the intervention group as opposed to the control group. Similar to our findings, a RCT also concluded that providing women with prenatal protein-calorie nutrition helps in decreasing the number of babies with low weight at birth and increasing the average birth weight of their newborn. 27 This aligns with the broader public health focus on improving maternal nutrition to enhance infant health, as discussed in a recent WHO report on maternal nutrition and infant health. 54
Our meta-analysis looked at how adding protein to the diet affected head circumference and birth length. The findings imply that prenatal protein–energy supplementation has a moderately noticeable effect on birth length, but not on birth head circumference. Specifically, the infants’ mean birth length differences of intervention and control groups differ by 0.20 cm. In contrast, the MD was only 0.05 cm for birth head circumference which was not statistically significant. While we did not find a significant effect on head circumference, future research should explore other measures of neurodevelopment to understand the full impact of protein supplementation on fetal growth and development. 55 A subgroup analysis that took into account the nutritional status of mothers found that in women who received adequate nutrition, protein supplementation had a positive effect on the length of the baby at birth. However, neither the adequately fed nor the undernourished had a discernible impact on the birth head circumference. These results support those of Ota et al.’s Cochrane review which revealed that, while in terms of length at birth or head circumference, there was no significant statistical difference, balanced protein supplementation was linked to a substantial rise in weight at birth. 12
Our research also investigated the effect of protein dietary supplements on birth outcomes which include perinatal death, SGA babies, and births occurring before term. When we pooled data from eight studies, the risk of perinatal mortality between the control group and the intervention group did not differ significantly, according to our findings. However, a noteworthy observation emerged when we specifically analyzed malnourished women—protein supplement intake had a link with a remarkable 44% decrease in perinatal mortality risk among this subgroup. This result underscores the urgent need for effective strategies to improve maternal nutrition, especially in low- and middle-income countries. 56
Regarding SGA births, our overall analysis indicated a 26% decrease in risk within the intervention group compared to controls. Intriguingly, the subgroup analysis targeting malnourished women revealed an even more substantial 40% risk reduction for SGA infants. This disparity underscores the impact of nutritional status, as the combined results, encompassing both adequately nourished and malnourished women, failed to reveal such a pronounced effect.
In terms of preterm births, our study did not find any significant reduction in risk across the intervention, adequately nourished, or malnourished groups. Our findings are in line with previous research conducted by Kramer and Kakuma, which also demonstrated reduced perinatal mortality and fewer SGA births but no significant alteration in preterm birth rates. 6
Limitations
Our study’s strength lies in its diverse inclusion of RCTs from various global income levels. However, limitations include inconsistencies in participant demographics, supplement characteristics, control measures, outcomes, and timing of supplementation. Socioeconomic differences among study populations, coupled with varying dietary habits, lifestyles, supplement types and doses, compliance rates, and genetic factors, contribute to this variability. High heterogeneity in our results is attributed to methodological and sample size differences among the included studies. Additionally, the use of built-in filters during the literature search may have introduced some biases in the selection of studies, limiting the comprehensiveness of the search strategy. Furthermore, although an a priori protocol registration was not conducted, we believe that our systematic approach to the study provides reliable and valuable insights into the topic.
Conclusion
This meta-analysis investigated protein–energy supplementation’s impact on birth outcomes in expectant mothers. Results showed increased length at birth as well as a 27% lower risk of LBW. A 26% decrease in the risk of SGA babies was observed. No notable changes were found in head circumference or preterm births. However, malnourished women experienced more significant benefits in birth weight, perinatal death, and SGA births with balanced protein–energy supplementation. In conclusion, this strategy lowers the possibility of having a bad pregnancy outcome, particularly in undernourished females.
Supplemental Material
sj-docx-1-whe-10.1177_17455057251335366 – Supplemental material for Effect of balanced protein–energy supplementation given to pregnant women on birth outcomes: A systematic review and meta-analysis
Supplemental material, sj-docx-1-whe-10.1177_17455057251335366 for Effect of balanced protein–energy supplementation given to pregnant women on birth outcomes: A systematic review and meta-analysis by Aliha Iftikhar, Hafsa Azam, Mariam Ahmed, Aliza Asad, Amber Noorani, Maaha Shabbir and Kanza Aftab in Women’s Health
Footnotes
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data underlying the results are available as part of the article and no additional source data are required.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
