Abstract
Visceral hypersensitivity is a hallmark feature of irritable bowel syndrome (IBS), yet its underlying mechanisms remain incompletely understood. In the present study, we found that miRNA-let7b5p was downregulated in the spinal cord of IBS model rats induced by neonatal colorectal distension. Concurrently, microglia exhibited a shift toward a pro-inflammatory M1 phenotype and selectively engulfed inhibitory synapses, resulting in impaired GABAergic neuronal function and disruption of the excitatory/inhibitory balance. Intrathecal administration of a miRNA-let7b5p agomir suppressed M1-type microglial activation in the spinal cord, reduced pro-inflammatory cytokine levels, and alleviated visceral hypersensitivity, whereas antagomir treatment induced visceral hypersensitivity in control rats. Mechanistically, MAP3K3 was identified as a direct target of miRNA-let7b5p, and its knockdown recapitulated the protective effects conferred by miRNA upregulation. Collectively, these findings demonstrate that miRNA-let7b5p attenuates IBS-associated visceral hypersensitivity by downregulating MAP3K3, thereby inhibiting spinal microglial activation and restoring GABAergic neuronal function. This study provides novel insights into the pathogenesis of IBS-related visceral hypersensitivity and highlights a potential therapeutic target for drug development.

Highlights
miRNA-let7b5p acts as a pivotal regulator that alleviates visceral hypersensitivity by inhibiting microglial activation and restoring the activity of GABAergic neurons.
Introduction
Chronic abdominal pain is a common clinical condition experienced by patients with irritable bowel syndrome (IBS). 1 Patients typically present with recurrent abdominal pain, abdominal distension, and disturbances in bowel habits. Epidemiological evidence suggests that up to 60% of individuals with IBS experience visceral hypersensitivity, which is a central contributor to disease pathogenesis.2,3 The mechanisms underlying visceral hypersensitivity are complex and may involve both peripheral and central sensitization processes. 4 Central sensitization is characterized by enhanced excitability and synaptic efficacy of neurons within the spinal cord and supraspinal regions of the central nervous system (CNS), thereby amplifying nociceptive transmission and pain perception. 5 As the primary central relay of peripheral nociceptive inputs, the spinal cord plays a crucial role in encoding these signals into long-lasting hypersensitivity. 6
Epigenetic regulation, particularly by microRNAs (miRNAs), plays a critical role in pathological processes including chronic pain.7–10 Dysregulated miRNA expression has been shown to contribute to the onset and amplification of pain signaling, thereby positioning miRNAs as important molecular determinants of nociceptive processing. Targeting specific miRNAs is regarded as a promising strategy to advance the fields of individualized analgesic therapy and precision medicine.11–14 Previous studies in our laboratory found that the spinal non-coding RNA circKcnk9 participates in visceral hypersensitivity in IBS-like rats, and miRNA-let7b5p was identified as a potential downstream effector. 15 The miRNA-let7 family can influence neuroinflammatory responses and nociceptive transmission via microglial activation. 16 Nevertheless, it remains unresolved whether miRNA-let7b5p exerts its effects on visceral hypersensitivity in IBS specifically via modulating microglial activation.
Emerging evidence has highlighted that the activation of microglia is intimately linked to the development of central sensitization in pain pathways.17–19 Microglia, as the resident immune cells of the CNS, are indispensable for the regulation of neuronal networks and the maintenance of homeostatic balance. 20 Upon pathological stimulation, microglia can undergo phenotypic shifts, altering morphology, and producing pro-inflammatory mediators, thereby contributing to central sensitization.21,22 Consistently, our preliminary experimental results showed that spinal microglia in IBS-like rats exhibited marked activation, characterized by hypertrophic cell bodies and a reduction in dendritic branching. Notably, pharmacological suppression of microglial activation markedly attenuated chronic visceral pain in IBS. Furthermore, we observed that miRNA-let7b5p has the capacity to regulate this activation process. Collectively, these findings raise the possibility that miRNA-let7b5p may play a mechanistic role in alleviating IBS-related visceral hypersensitivity by restraining microglial activation, although the underlying molecular pathways remain to be clarified.
The balance between neuronal excitation and inhibition (E/I) is fundamental for normal neural function. 23 Disruption of this balance is increasingly recognized as a hallmark of chronic pain. 24 Hyperactivated microglia can perturb the E/I equilibrium by augmenting the excitability of projection neurons, ultimately facilitating nociceptive transmission. Beyond this, microglia actively shape synaptic plasticity through complement-dependent pathways, in which synaptic elements are tagged, and presynaptic terminals are selectively engulfed.25,26 Nevertheless, it remains unresolved whether spinal microglia in IBS-like rats preferentially target excitatory or inhibitory presynaptic terminals for phagocytosis, thereby leading to an imbalance of E/I dynamics, and whether miRNA-let7b5p is engaged in orchestrating this regulatory mechanism.
This study employed an IBS rat model to elucidate the mechanisms underlying visceral hypersensitivity. We first investigated the functional impact of miRNA-let7b5p on visceral hypersensitivity in IBS-like rats. Next, we focused on the role of spinal microglia in mediating this hypersensitivity, with particular emphasis on the regulatory influence of miRNA-let7b5p on microglial activation and the involvement of its downstream protein targets. Finally, we investigated how microglial activation and miRNA-let7b5p influence excitation and inhibition (E/I) balance to elucidate their potential mechanisms. Collectively, these findings provide new insights into the pathogenesis of IBS-associated visceral hypersensitivity and identify miRNA-let7b5p as a potential therapeutic targe.
Materials and methods
Animals
Adult male Sprague–Dawley rats (body weight range: 180–220 g) were obtained from the Department of Experimental Animal Center, Fujian Medical University. Animal procedures were approved by the Animal Care and Use Committee of Fujian Medical University, and conduct animal experiments in strict accordance with Fujian Medical University’s “Guiding Opinions on Kindness to Laboratory Animals.” Animals were maintained in a specific pathogen-free-grade environment at the animal center. Weaning is typically initiated 21 days after birth. All animals were kept in a greenhouse with a controlled humidity of 65%–77%. They were maintained on a 12 h light/dark cycle. The experiments were approved by the Animal Care and Use Committee of Fujian Medical University. IBS-like rats were established by 60 mmHg colorectal distension (CRD) stimulation once daily during postnatal days 8–14. We examined electromyographic (EMG) magnitude in response to graded strengths of CRD pressures in IBS-like rats and control rats at 6–8 weeks to assess visceral sensitivity. Control rats were subjected to the same procedure, except for colorectal distention (CRD). After stimulation, the rats were housed until they were at least 6 weeks old (about 200 g). To avoid sex and gonadal hormone-related variations, we decided to always use male rats. The number of rats used in each group ranged from 4 to 8, and the selection of experimental animals was conducted using randomization and blinding methods. Specifically, the rats were randomly assigned to different experimental groups, and during the experiment, the investigators were blinded to the group allocation.
Electromyographic (EMG)
EMG was performed to assess visceral hypersensitivity. Rats (6–8 weeks old) were anesthetized with isoflurane. Prior to the CRD procedure, a glycerol-lubricated balloon was inserted into the rectum. A pair of bipolar electrodes were implanted in the abdominal external oblique musculature of shallow anesthetized rats to detect EMG activity. Under isoflurane superficial anesthesia, the discharge of the rat ventral oblique muscle was recorded at CRD pressures of 40 and 60 mmHg. EMG responses to different degrees of CRD were recorded using the RM6240BD system (Chengdu, China). The obtained data were analyzed by calculating the mean amplitude relative to the baseline, and values exceeding the baseline were used to assess visceral hypersensitivity.
Protein extraction and western blot
Spinal tissues (T13–L2) were lysed in RIPA buffer (P0013B; Beyotime, China). The proteins were separated by electrophoresis and transferred onto a PVDF membrane. The following antibodies were used: Iba-1 (1:2000, 10937-1-AP; Proteintech), MAP3K3 (1:2000, ab288437; Abcam), VGAT (1:2000, R22768; ZEN BIO), VGLUT2 (1:2000; 310164; ZEN BIO), GAD65/67 (1:2000, R233412; ZEN BIO), β-actin (1:5000, AC004; ABclonal), and GAPDH (1:4000, A19056; ABclonal). Protein bands were detected using an enhanced chemiluminescence reaction. GAPDH was used as an internal control.
RNA extraction and quantitative real-time PCR (qPCR)
Total RNA was extracted using TRIzol (Invitrogen, USA). An Evo-M-MLV reverse transcription kit (Accurate Biology, China) was used to perform reverse transcription for circRNA and mRNA, according to the manufacturer’s instructions. The miRNA first-strand cDNA synthesis kit (Accurate Biology, China) was used to perform reverse transcription for microRNA, according to the manufacturer’s instructions. Nuclear and cytosolic fractions of cells were purified using a nuclear/cytosolic fractionation kit (AM1921; Thermo Fisher), as per the manufacturer’s instructions. After reverse transcription, qRT-PCR was conducted according to the manufacturer’s instructions. Primer sequences were shown in Table 1.
Primer sequences used in fluorescence quantitative PCR experiments.

Immunofluorescence
Rats were deeply anesthetized with Ulatan (0.5 mL/100 g) and perfused transcardially with 500 mL ice-cold 0.9% phosphate buffer (PBS), followed by 750 mL 4% paraformaldehyde. The dorsal horns of the thoracolumbar segments (T13–L2) tissues were collected and placed in formalin overnight. After gradient dehydration with 20% and 30% sucrose (Sigma, USA), tissues were embedded with OCT compound and sectioned using a cryotome (Lecia, Germany). Removed the sections (20–40 μm) from −20 ℃ refrigerator for half an hour before using. We used phosphate buffer solution (PBS) to wash out OCT. Pap Pen was used to circle out the tissue. The sections were blocked with immunostaining blocking buffer solution containing 5% goat serum and 0.3% Triton™ X-100 at room temperature for 2 h (or 37 ℃ for 30 min), and then hybridized for 12 h at 37 °C with the 3′ and 5′-FAM-labeled miRNA-let7b5p probe 5′-AACCACACAACCTACTACCTCA-3′ (1:100; EXIQON). The sections were incubated with primary antibodies against MAP3K3 (1:100, ab109085; Proteintech), GFAP (1:300, 3670; CST), Iba1 (1:10k, 019-19741; Wako), or NeuN (1:500, MAB377; Millipore) overnight at 4 °C. Goat anti-rabbit lgG, 488 (1:300, 150073; Abcam), goat anti-Mouse lgG, 488 (1:300, 150105; Abcam), goat anti-rabbit lgG 594 (1:500, 8889S; CST), or goat anti-mouse lgG, 594 (1:300, 150108; Abcam) was then used to conjugate the secondary antibody at 37 °C for 2 h. Immunofluorescence and FISH images were captured using Lecia SP5 confocal microscopes equipped with 405, 488, and 594 lasers. The contrast of the final images were adjusted using Photoshop (Adobe Systems, Mountain View, CA, USA).
Intrathecal injection
Neuronal hSyn promoter was used to drive gene expression in neuron. rAAV-U6-shRNA (MAP3K3) EGFP-SV40 polyA (shMAP3K3 and AAV-scramble) was designed and synthesized by BrainVTA (Wuhan, China). miRNA-let7b5p agomir and antagomir were obtained from GenePharma Biotech (Shanghai, China). A sterile polyethylene-10 catheter (BB31695-PE/1; Scientific Commodities, Inc., USA) was implanted between the L4 and L5 vertebrae. Animals with numbness or paralysis in their hindlimbs were removed from the trial. Agents were administered 5 days after the surgery. ShRNA was intrathecally injected (10 μl), AAV was intrathecally injected (10 μl), and GFP was observed after 21 days. miRNA-let7b5p agomir and antagomir (20 mM) were intrathecally injected for 3 days.
Luciferase reporter assays
The pmirGLO Dual-Luciferase vector containing wild-type miRNA-let7b5p (miRNA-let7b5p-wt), wild-type MAP3K3 (MAP3K3-wt), miRNA-let7b5p-mutant (miRNA-let7b5p-mut), and MAP3K3-mutant (MAP3K3-mut) were synthesized by GenePharma Biotech (Shanghai, China). The 293T cells were seeded in 96-well plates and co-transfected with miRNA-let7b5p-wt plasmids, miRNA-let7b5p-mut plasmids, MAP3K3-wt or MAP3K3-mut using Lipofectamine 3000. Cell lysates were collected after transfection for 48 h, and the Dual-Luciferase Reporter Assay System (Promega) was used to detect firefly and Renilla luciferase activities.
Statistical analysis
All experiments were randomized. All data were presented as mean ± standard error of the mean. In behavioral tests and molecular biology experiments, two-tailed independent sample t-tests were performed to determine differences between control and IBS-like rats if data satisfied the normal distribution, and if data did not satisfy normal distribution, Wilcoxon correction was performed for two independent samples. Statistical analysis of data from more than two groups was performed using one-way analysis of variance (ANOVA)–LSD-t comparisons if the data were normally distributed. The rank sum test, Kruskal–Wallis H test, and Nemenyi test were performed when the data did not satisfy normal distribution and were more than two groups. In addition, repeated-measures ANOVA was performed to analyze the electrophysiological results. The EMG results were analyzed using a two-way ANOVA. Correlation analysis was performed using a two-tailed Pearson correlation. The data analysis was performed using GraphPad Prism8 and R4.0.3, and a p < 0.05 was considered statistically significant.
Results
Downregulation of spinal miRNA-let7b5p induces visceral hypersensitivity in IBS-like rats
The IBS models were established as previously described, 27 newborn rats were exposed to colorectal distention stimulation at a pressure of 60 mmHg for 1 min from 8 to 14 days after birth (Figure S1). Prior studies from our group suggested that the spinal non-coding RNA circKcnk9 contributes to the development of visceral hypersensitivity in IBS-like rats, potentially through the regulation of downstream miRNAs. 15 Here, quantitative PCR confirmed a significant downregulation of miRNA-let7b5p expression in the spinal cord of IBS-like rats (Figure 1(a)). To further investigate its functional role, we administered miRNA-let7b5p agomir intrathecally to IBS-like rats. This intervention markedly elevated spinal miRNA-let7b5p levels (Figure 1(b)) and significantly attenuated EMG amplitude induced by 40/60 mmHg CRD, indicating an effective reversal of visceral hypersensitivity (Figure 1(d) and (e)). In contrast, intrathecal delivery of miRNA-let7b5p antagomir in control rats resulted in a pronounced reduction of spinal miRNA-let7b5p (Figure 1(c)) and a concomitant increase in EMG amplitude under identical CRD pressures (Figure 1(f) and (g)). Together, these results suggest that downregulation of spinal miRNA-let7b5p is sufficient to induce IBS-like visceral hypersensitivity in otherwise normal rats. To clarify the cellular targets of spinal miRNA-let7b5p, fluorescence in situ hybridization (FISH) was performed to assess its colocalization with microglia, astrocytes, and neurons. The results showed that in IBS-like rats, miRNA-let7b5p exhibited strong colocalization with Iba-1, whereas colocalization with GFAP and NeuN was weak, suggesting that miRNA-let7b5p may primarily exert its effects through microglia (Figure 1(h)).

Downregulated spinal miRNA-let7b5p contributes to visceral hypersensitivity in IBS-like rats and is mainly distributed in microglia: (a) statistical graphs of miRNA-let7b5p content in the dorsal horn of the spinal cord in IBS-like rats; n = 4, unpaired t-test, *p < 0.05, compared with normal rats, (b) statistical graph of mRNA expression of miRNA-let7b5p in the spinal cord after intrathecal injection of miRNA-let7b5p agomir in IBS-like rats; n = 8, unpaired t-test, *p < 0.05, compared with that of the agomirNC group, (c) statistical graph of mRNA expression of miRNA-let7b5p in the spinal cord after intrathecal injection of miRNA-let7b5p antagomir in normal rats; n = 8, unpaired t-test, *p < 0.05, compared with the antagomirNC group, (d, e) representative and statistical graph of the percentage of discharge amplitude versus basal value of the external abdominal oblique muscle at 40/60 mmHg CRD pressure after intrathecal injection of miRNA-let7b5p agomir in IBS-like rats; percentage = (discharge amplitude − basal value)/basal value × 100%; n = 4, two-way ANOVA, *p < 0.05, compared with the agomirNC group, (f, g) representative and statistical graph of the percentage of discharge amplitude versus basal value of the external abdominal oblique muscle at 40/60 mmHg CRD pressure after intrathecal injection of miRNA-let7b5p antagomir in normal rats; percentage = (discharge amplitude − basal value)/basal value × 100%; n = 4, two-way ANOVA, *p < 0.05, compared with the antagomirNC group, and (h) cellular localization of miRNA-let7b5p in the spinal cord dorsal horn of IBS-like rats. Immunofluorescence double labeling was performed to identify the cellular distribution of miRNA-let7b5p (red) using markers for microglia (IBA-1), astrocytes (GFAP), and neurons (NeuN; all green). Representative images and fluorescence intensity overlap profiles demonstrated that miRNA-let7b5p signals exhibited prominent co-localization with IBA-1-positive microglia, whereas minimal overlap was observed with GFAP-positive astrocytes or NeuN-positive neurons. Scale bar: 20 µm.
Upregulation of miRNA-let7b5p reverses spinal microglial activation in IBS-like rats
Further analysis revealed that both protein and mRNA levels of Iba-1 in the spinal dorsal horn were significantly elevated in IBS-like rats compared with controls (Figure 2(a) and (b)). Immunofluorescence demonstrated that microglia in IBS-like rats displayed enlarged somata, reduced branching, a typical amoeboid activated morphology, and markedly enhanced Iba-1 fluorescence intensity (Figure 2(c) and (d)). Activated microglia are known to be classified into pro-inflammatory M1 and anti-inflammatory M2 subtypes. qPCR results showed that mRNA expression of the M1 markers CD68 and CD86 was significantly increased, whereas the M2 markers CD206 and Arg-1 were markedly decreased in the dorsal horn of IBS-like rats, indicating that microglia predominantly exhibit a pro-inflammatory M1 phenotype (Figure 2(e)). Meanwhile, pro-inflammatory cytokines IL-6, TNF-α, and iNOS were significantly upregulated, whereas anti-inflammatory factors IL-10 and Arg-1 were significantly downregulated, further indicating a pro-inflammatory activation profile of microglia (Figure 2(f)).

Activation of spinal cord microglia is involved in visceral hypersensitivity in IBS-like rats: (a, b) representative graphs and statistical graphs of protein and mRNA expression of Iba-1, a marker for microglia in the dorsal horn of the spinal cord of IBS-like rats; n = 8, unpaired t-test, *p < 0.05, compared with normal rats, (c, d) fluorescence representation and number statistics of microglia in the dorsal horn of the spinal cord of IBS-like rats; n = 4, unpaired t-test, *p < 0.05, compared with normal rats. Scale bar is 20 μm, (e) mRNA expression levels of microglia typing markers in the spinal cord of IBS-like rats; n = 8, unpaired t-test, *p < 0.05, compared with normal rats (M1 type: CD68, CD86; M2 type: Arg-1, CD206, same below), (f) mRNA expression levels of inflammatory factors in the spinal cord of IBS-like rats; n = 8, unpaired t-test, *p < 0.05, compared with normal rats, (g, h) representative graphs and statistical graphs of protein and mRNA expression of Iba-1; n = 8, unpaired t-test, *p < 0.05, compared with saline group, (i, j) representative graph of fluorescence of microglia in the dorsal horn of the spinal cord after intraperitoneal administration of the microglia inhibitor minocycline in IBS-like rats; n = 3, unpaired t-test, *p < 0.05, compared with normal rats. Scale bar is 20 μm, (k) mRNA expression levels of microglia typing markers in the spinal cord after intraperitoneal administration of minocycline, a microglia inhibitor, to IBS-like rats; n = 8, unpaired t-test, *p < 0.05, compared with the saline group, (l) mRNA expression levels of inflammatory factors in the spinal cord after intraperitoneal administration of the microglia inhibitor minocycline in IBS-like rats; n = 8, unpaired t-test, *p < 0.05, compared with the saline group, and (m) statistical plot of the percentage of discharge amplitude versus basal value of the external abdominal oblique muscle at 40/60 mmHg CRD pressure after intraperitoneal administration of the microglia inhibitor minocycline in IBS-like rats; percentage = (discharge amplitude − basal value)/basal value × 100%; n = 4, two-way ANOVA, *p < 0.05, compared with saline group.
To verify the role of microglial activation in visceral hypersensitivity, IBS-like rats were treated with minocycline (Mino). The results showed that minocycline significantly inhibited spinal Iba-1 expression and microglial activation (Figure 2(g)–(j)), reduced the proportion of M1 microglia while increasing M2 cells (Figure 2(k)), and correspondingly decreased levels of pro-inflammatory cytokines IL-6 and TNF-α (Figure 2(l)). EMG recordings demonstrated that minocycline significantly reduced the abdominal external oblique muscle responses under 40/60 mmHg CRD stimulation (Figure 2(m)), suggesting that inhibition of microglial activation effectively alleviates visceral hypersensitivity in IBS rats.
On this basis, the regulatory effect of miRNA-let7b5p on microglia was examined. The results indicated that intrathecal injection of miRNA-let7b5p agomir significantly downregulated spinal Iba-1 expression in IBS rats (Figure 3(a)–(d)) and decreased the levels of M1 pro-inflammatory microglia and associated inflammatory factors (Figure 3(e) and (f)). In contrast, intrathecal injection of miRNA-let7b5p antagomir produced opposite effects (Figure 3(g)–(l)). Collectively, these results indicate that miRNA-let7b5p plays a key role in the occurrence and regulation of visceral hypersensitivity in IBS by modulating spinal microglial activation.

miRNAlet7b5p inhibits microglia activation in the spinal cord of IBS-like rats: (a) statistical graphs of protein expression of microglia marker Iba-1 in the dorsal horn of the spinal cord after intrathecal injection of miRNA-let7b5p agomir in IBS-like rats; n = 8, unpaired t-test, *p < 0.05, compared with the agomirNC group, (b, c) representative and statistical graph of fluorescence of spinal cord dorsal horn microglia after intrathecal injection of miRNA-let7b5p agomir in IBS-like rats; n = 3, unpaired t-test, *p < 0.05, compared with the agomirNC group. Scale bar is 20 μm, (d) statistical graphs of mRNA expression of microglia marker Iba-1 in the dorsal horn of the spinal cord after intrathecal injection of miRNA-let7b5p agomir in IBS-like rats; n = 8, unpaired t-test, *p < 0.05, compared with the agomirNC group, (e) mRNA expression levels of microglia typing markers in the spinal cord after intrathecal injection of miRNA-let7b5p agomir in IBS-like rats; n = 6, unpaired t-test, *p < 0.05, compared with the agomirNC group, (f) mRNA expression levels of inflammatory factors in the spinal cord after intrathecal injection of miRNA-let7b5p agomir in IBS-like rats; n = 6, unpaired t-test, *p < 0.05, compared with the agomirNC group, (g) statistical graph of protein expression of miRNA-let7b5p in the spinal cord after intrathecal injection of miRNA-let7b5p antagomir in normal rats; n = 8, unpaired t-test, *p < 0.05, compared with the antagomirNC group, (h, i) representative and statistical graph of spinal cord dorsal horn microglia after intrathecal injection of miRNA-let7b5p antagomir in normal rats; n = 3, unpaired t-test, *p < 0.05, compared with the antagomirNC group. Scale bar is 20 μm, (j) statistical graph of mRNA expression of miRNA-let7b5p in the spinal cord after intrathecal injection of miRNA-let7b5p antagomir in normal rats; n = 8, unpaired t-test, *p < 0.05, compared with the antagomirNC group, (k) mRNA expression levels of microglia typing markers in the spinal cord after intrathecal injection of miRNA-let7b5p antagomir in normal rats; n = 6, unpaired t-test, *p < 0.05, compared with the antagomirNC group, and (l) mRNA expression levels of inflammatory factors in the spinal cord after intrathecal injection of miRNA-let7b5p antagomir in normal rats; n = 6, unpaired t-test, *p < 0.05, compared with the antagomirNC group.
miRNA-let7b5p exerts regulatory effects through the MAP3K3 signaling pathway
To elucidate the molecular mechanism of miRNA-let7b5p, TargetScan bioinformatic analysis and dual-luciferase reporter assays were performed, revealing MAP3K3 as a downstream target of miRNA-let7b5p (Figure 4(a) and (b)). Intrathecal injection of miRNA-let7b5p agomir significantly reduced MAP3K3 levels (Figure 4(c) and (d)), whereas miRNA-let7b5p antagomir markedly upregulated MAP3K3 expression (Figure 4(e) and (f)).

miRNAlet7b5p is involved in the inhibition of microglia activation and visceral hypersensitivity in IBS-like rats through down-regulation of MAP3K3: (a) Targetscan software predicts the downstream target protein MAP3K3 of miRNA-let7b5p, (b) dual luciferase reporter results indicate that the relative luciferase reporter activity of let7b5p mimics co-transfected with MAP3K3 wild type is significantly lower than that of the mutant type, **p < 0.01, compared with the normal group, (c, d) representative and statistical graphs of protein and mRNA expression of MAP3K3 in the dorsal horn of the spinal cord of IBS-like rats after intrathecal injection of miRNA-let7b5p agomir; n = 8, unpaired t-test, *p < 0.05, compared with the agomirNC group, (e, f) representative and statistical graphs of protein and mRNA expression of MAP3K3 in the dorsal horn of the spinal cord after intrathecal injection of miRNA-let7b5p antagomir in normal rats; n = 8, unpaired t-test, *p < 0.05, compared with the antagomirNC group, (g, h) representative plots and statistics of protein and mRNA expression of MAP3K3 in the dorsal horn of the spinal cord of IBS rats; n = 8, unpaired t-test, *p < 0.05, compared with normal rats, (i) statistical graph of mRNA expression of MAP3K3 in the spinal cord after intrathecal injection of shMAP3K3 in IBS-like rats; n = 8, unpaired t-test, *p < 0.05, compared with the scra group, (j) statistical graph of the percentage of discharge amplitude versus basal value in the external abdominal oblique muscle at 40/60 mmHg CRD pressure after intrathecal injection of shMAP3K3 in IBS rats; percentage = (discharge amplitude − basal value)/basal value × 100%; n = 4, two-way ANOVA, *p < 0.05, compared with the scra group, (k, l) representative and statistical graph of microglia in the dorsal horn of the spinal cord after intrathecal injection of shMAP3K3 in IBS rats; n = 3, unpaired t-test, *p < 0.05, compared with the scra group. Scale bar is 20 μm, (m) representative and statistical graphs of Iba-1 mRNA expression, a marker of spinal cord dorsal horn microglia, after intrathecal injection of shMAP3K3 in IBS rats; n = 7, unpaired t-test, *p < 0.05, compared with the scra group, (n) mRNA expression levels of spinal cord microglia typing markers after intrathecal injection of shMAP3K3 in IBS-like rats; n = 7, unpaired t-test, *p < 0.05, compared with the scra group, and (o) mRNA expression levels of inflammatory factors in the spinal cord after intrathecal injection of shMAP3K3 in IBS-like rats; n = 7, unpaired t-test, *p < 0.05, compared with the scra group.
Whether MAP3K3 is altered in the spinal cord of IBS-like rats remained to be determined. Western blot and qPCR analyses demonstrated that MAP3K3 expression in the spinal cord of IBS-like rats was significantly higher than that in normal controls (Figure 4(g) and (h)). Moreover, intrathecal administration of shMAP3K3 effectively suppressed spinal MAP3K3 expression (Figure 4(i)) and significantly reduced EMG amplitude under 40/60 mmHg CRD stimulation in IBS-like rats (Figure 4(j)). qPCR and immunofluorescence results showed that shMAP3K3 intervention decreased Iba-1 expression and microglial activation (Figure 4(k)–(m)), reduced the proportion of M1 pro-inflammatory microglia, and downregulated pro-inflammatory cytokine expression (Figure 4(n) and (o)).
Collectively, these results indicate that miRNA-let7b5p may inhibit spinal microglial activation in IBS-like rats and attenuate visceral hypersensitivity by downregulating its target gene MAP3K3.
Activation of spinal microglia suppresses inhibitory neuron activity, leading to visceral hypersensitivity in IBS-like rats
Microglia, as the primary immune cells in the central nervous system, can influence neuronal activity through synaptic phagocytosis; however, their role in IBS remains unclear. In this study, dual immunofluorescence labeling was used to analyze the colocalization of CD68 (a microglial phagocytosis marker) with VGAT (an inhibitory presynaptic marker) and VGLUT2 (an excitatory presynaptic marker) in the spinal dorsal horn. The results showed that in IBS-like rats, colocalization of CD68 with VGAT was significantly enhanced, whereas colocalization with VGLUT2 was minimal. Fluorescence quantification further confirmed that CD68+VGAT+ signals in the spinal cord of IBS-like rats were markedly higher than CD68+VGLUT2+ signals, suggesting that activated microglia preferentially phagocytose inhibitory presynaptic terminals (Figure 5(a) and (b)). Additionally, compared with controls, the fluorescent area of CD68+VGAT+ in the dorsal horn of IBS-like rats was significantly increased (Figure 5(c) and (d)).
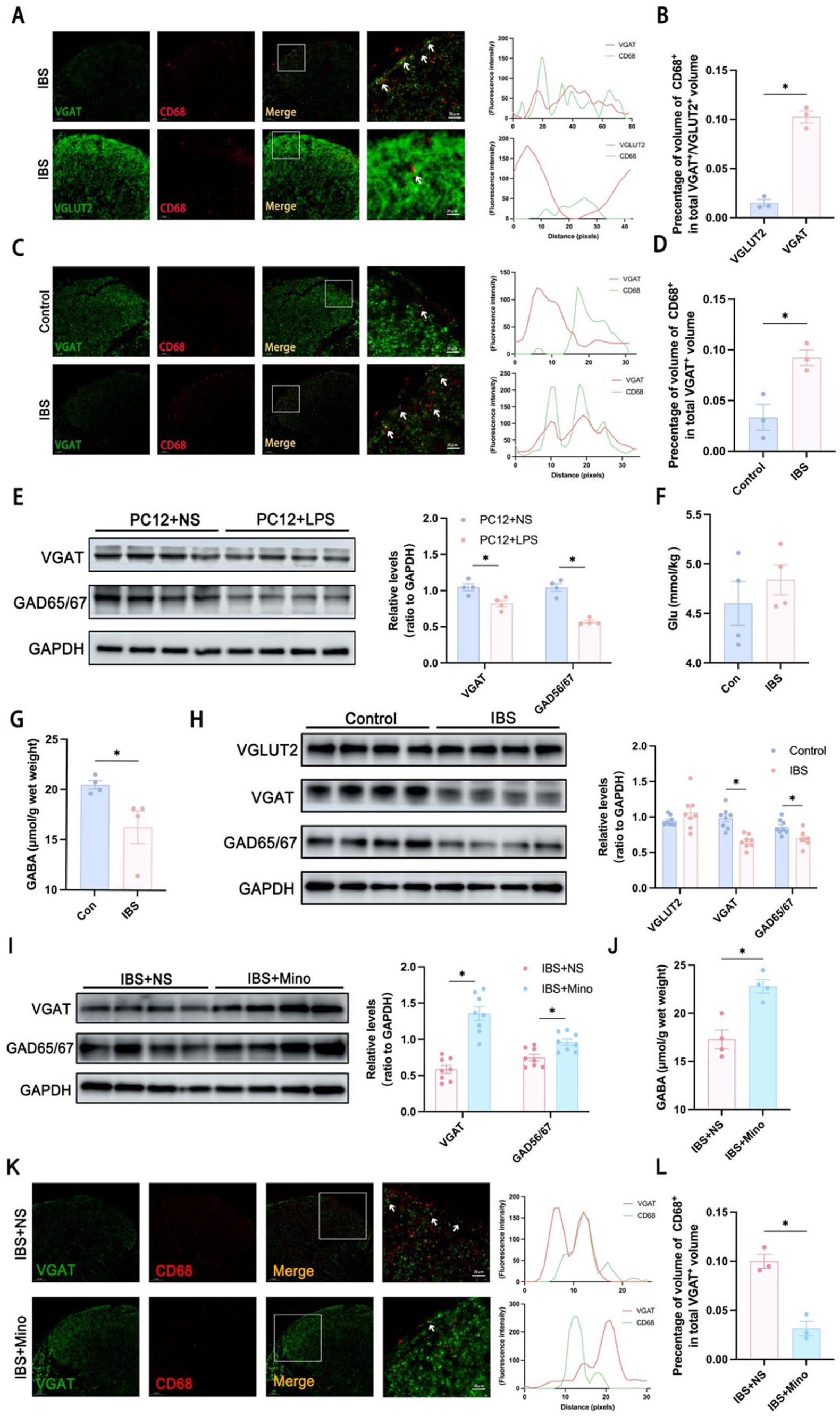
Selective phagocytosis of VGAT by spinal microglia in IBS-like rats and downregulation of inhibitory neuron activity: (a, b) immunofluorescence double labeling was performed to examine the spatial association of the inhibitory presynaptic marker VGAT and the excitatory presynaptic marker VGLUT2 (green) with the microglial phagocytosis marker CD68 (red). Representative images, fluorescence intensity overlap profiles, and quantitative analyses demonstrated that VGAT exhibited prominent co-localization with CD68 in the dorsal horn of the spinal cord in IBS-like rats, whereas minimal overlap was observed between VGLUT2 and CD68, suggesting a preferential microglial engulfment of inhibitory synaptic components, n = 3, unpaired t-test, *p < 0.05, scale bar: 20 µm, (c, d) immunofluorescence double labeling was performed to examine the spatial association of VGAT (green) with CD68 (red). Representative images, fluorescence intensity overlap profiles, and quantitative analyses demonstrated that an increased VGAT–CD68 co-localization was observed in the IBS-like model group compared with controls. n = 3, unpaired t-test, *p < 0.05, compared with normal rats. Scale bar is 20 μm, (e) statistical graph of the content of the excitatory neurotransmitter Glu in the dorsal horn of the spinal cord of IBS-like rats; n = 4, unpaired t-test, *p < 0.05, compared with normal rats, (f) statistical plot of the content of inhibitory neurotransmitter GABA in the dorsal horn of the spinal cord of IBS-like rats; n = 4, unpaired t-test, *p < 0.05, compared with normal rats, (g) representative and statistical graphs of protein expression of VGAT and GAD65/67 in the dorsal horn of the spinal cord of PC12 + LPS cells; n = 4, unpaired t-test, *p < 0.05, compared with PC12 + NS cells, (h) representative and statistical graphs of protein expression of neurotransmitter transporter proteins VGLUT2, VGAT, and GABA synthase GAD65/67 in the dorsal horn of the spinal cord of IBS-like rats; n = 8, unpaired t-test, *p < 0.05, compared with normal rats, (i) representative and statistical graphs of protein expression of neurotransmitter transporter proteins VGAT and GAD65/67 in the dorsal horn of the spinal cord after intraperitoneal administration of the microglia inhibitor minocycline in IBS-like rats; n = 8, unpaired t-test, *p < 0.05, compared with the saline group, (j) statistical graph of the content of inhibitory neurotransmitter GABA in the dorsal horn of the spinal cord after intraperitoneal administration of minocycline, a microglia inhibitor, in IBS-like rats; n = 4, unpaired t-test, *p < 0.05, compared with the saline group, and (k, l) immunofluorescence double labeling was performed to examine the spatial association of VGAT (green) with CD68 (red). Representative images, fluorescence intensity overlap profiles, and quantitative analyses demonstrated that VGAT–CD68 co-localization was significantly reduced in the IBS-like rats treated with minocycline compared with the saline-treated IBS model group, n = 3, unpaired t-test, *p < 0.05, compared with the saline group. Scale bar is 20 μm.
Moreover, in vitro co-culture experiments demonstrated that when LPS-activated BV2 microglial conditioned medium was applied to PC12 neuronal cultures, the expression of VGAT and GAD65/67 in PC12 cells was significantly reduced (Figure 5(e)). These results suggest that microglia may exert regulatory effects by selectively phagocytosing inhibitory synapses.
To investigate whether the balance between excitatory and inhibitory neuronal activity is disrupted in the spinal cord of IBS-like rats, ELISA was performed to measure neurotransmitter levels. The results revealed that GABA levels in the dorsal horn were significantly decreased, whereas GLU levels showed no significant change (Figure 5(f) and (g)). Western blot analysis showed that the protein levels of VGAT and the GABA-synthesizing enzymes GAD65/67 were significantly downregulated, while VGLUT2 expression remained unchanged (Figure 5(h)). Inhibition of microglial activation by minocycline significantly increased spinal VGAT and GAD65/67 expression as well as GABA content (Figure 5(i) and (j)) and markedly reduced the CD68+VGAT+ fluorescent area (Figure 5(k) and (l)). These results indicate that activated microglia in the dorsal horn may selectively suppress inhibitory neuronal activity, contributing to visceral hypersensitivity in IBS rats.
Activation of spinal microglia in IBS-like rats participates in visceral hypersensitivity by downregulating inhibitory neuronal activity. To examine the role of miRNA-let7b5p in this process, ELISA and Western blot analyses were performed. Upregulation of spinal miRNA-let7b5p in IBS-like rats significantly increased levels of GABA, VGAT, and GAD65/67 (Figure 6(a) and (b)), whereas inhibition of miRNA-let7b5p expression in normal rats significantly reduced GABA, VGAT, and GAD65/67 levels (Figure 6(c) and (d)). Dual immunofluorescence analysis showed that upregulation of miRNA-let7b5p markedly decreased the CD68+VGAT+ fluorescent area (Figure 6(e) and (f)), whereas inhibition of miRNA-let7b5p significantly increased CD68+VGAT+ area (Figure 6(g) and (h)). Collectively, these results suggest that miRNA-let7b5p may alleviate IBS-associated visceral hypersensitivity by inhibiting microglial activation, thereby enhancing inhibitory neuronal function.

miRNAlet7b5p enhances inhibitory neuronal activity: (a) statistical graph of the content of inhibitory neurotransmitter GABA in the dorsal horn of the spinal cord after intrathecal injection of miRNA-let7b5p agomir in IBS-like rats; n = 4, unpaired t-test, *p < 0.05, compared with that of the agomirNC group, (b) representative and statistical graphs of protein expression of neurotransmitter transporter proteins VGLUT2, VGAT, and GABA synthase GAD65/67 in the dorsal horn of the spinal cord after intrathecal injection of miRNA-let7b5p agomir in IBS-like rats; n = 4, unpaired t-test, *p < 0.05, compared with the agomirNC group, (c) statistical graph of the content of inhibitory neurotransmitter GABA in the dorsal horn of the spinal cord after intrathecal injection of miRNA-let7b5p antagomir in normal rats; n = 4, unpaired t-test, *p < 0.05, compared with the antagomirNC group, (d) representative and statistical graphs of protein expression of neurotransmitter transporter protein VGAT and GABA synthase GAD65/67 in the dorsal horn of the spinal cord after intrathecal injection of miRNA-let7b5p antagomir in normal rats; n = 4, unpaired t-test, *p < 0.05, compared with the antagomirNC group, (e, f) immunofluorescence double labeling was performed to examine the spatial association of VGAT (green) with CD68 (red). Representative images, fluorescence intensity overlap profiles, and quantitative analyses demonstrated that VGAT–CD68 co-localization was significantly reduced in the IBS-like rats treated with agomir compared with the agomirNC group; n = 3, unpaired t-test, *p < 0.05, compared with the agomirNC group. Scale bar is 20 μm, and (g, h) representative images, fluorescence intensity overlap profiles, and quantitative analyses demonstrated that an increased VGAT–CD68 co-localization was observed in the normal rats treated with antagomir compared with the antagomirNC group; n = 3, unpaired t-test, *p < 0.05, compared with the antagomirNC group. Scale bar is 20 μm.
Discussion
Our study reveals a complex interplay among miRNA-let7b5p, spinal microglial activation, and neuronal synaptic plasticity in the context of IBS, providing new insights into the mechanisms underlying visceral hypersensitivity and potential therapeutic strategies. Our results demonstrate that in IBS-like rats, miRNA-let7b5p participates in visceral hypersensitivity by inhibiting microglial activation, whereas activated microglia suppress the activity of GABAergic neurons, disrupting the excitation/inhibition (E/I) balance within spinal circuits and thereby exacerbating chronic visceral pain. These findings highlight the role of miRNA-let7b5p and microglial activation in IBS-associated visceral hypersensitivity and suggest potential novel therapeutic targets for IBS.
In recent years, epigenetic regulatory mechanisms have attracted increasing attention for their roles in the development and progression of chronic pain. Among these mechanisms, multiple epigenetic layers—including miRNAs, DNA methylation, and histone modifications—are thought to cooperatively contribute to the fine-tuned regulation of pain-related gene expression. 28 miRNAs exert regulatory functions in disease by binding to the 3′ untranslated regions (3′UTRs) of target mRNAs to inhibit translation or promote degradation. 29 Previous studies have demonstrated that miRNAs can modulate pain signaling through regulation of cytokine release, glial activation, and synaptic plasticity. 30 For example, in chronic inflammatory pain models, expression of let-7 family members is downregulated, and restoring their levels significantly attenuates pain behaviors. 16 In Alzheimer’s disease, miR-25802 alleviates neuroinflammation by inhibiting the TLF4/NF-κB pathway 31 ; miR-124 promotes microglial polarization toward an anti-inflammatory M2 phenotype via C/EBPα inhibition, thereby reducing injury-associated neuroinflammation 32 ; in neuropathic pain, miR-223 suppresses IL-1β release by targeting the NLRP3 inflammasome, mitigating microglia-driven sensitization. 33 In addition to miRNAs, histone modifications and DNA methylation also play important roles in the regulation of pain. Ni et al. reported that, in rodent models of bone cancer pain, specific histone modifications cooperate with the transcription factor Sp1 to promote the expression of the pain-related gene GPR160, highlighting the critical contribution of chromatin-level regulation to persistent pain sensitization.34,35 Together with systematic investigations into the epigenetic mechanisms of bone cancer pain, these findings further emphasize the importance of epigenetic regulation in chronic pain conditions. In line with this epigenetic framework, our study demonstrate that the downregulation of spinal miRNA-let7b5p contributes to visceral hypersensitivity in IBS-like rats. This observation suggests that the types of miRNAs involved in modulating pain sensitivity, as well as their directions of action, may vary substantially under different pathological conditions. Upregulation of miRNA-let7b5p expression in the spinal cord effectively suppresses microglial activation and the release of pro-inflammatory cytokines, thereby alleviating visceral hyperalgesia in IBS-like rats. Taken together, these results further highlight the pivotal regulatory role of miRNAs in controlling microglial activation and the development of pain sensitization.
Microglia, as the principal immune cells in the central nervous system, play a pivotal role in the initiation and maintenance of chronic pain. In our study, neonatal CRD-induced IBS-like rats exhibited M1-polarized microglia in the spinal cord, accompanied by elevated expression of pro-inflammatory mediators such as IL-6, TNF-α, and iNOS. Pharmacological inhibition of microglial activation significantly attenuated visceral hypersensitivity, suggesting that microglial activation is instrumental in modulating visceral pain. These observations align with previous evidence supporting the role of microglial activation in chronic pain pathogenesis.36,37 For instance, in neuropathic pain, microglia amplify nociceptive signaling in the spinal cord through BDNF release 38 ; in cancer pain models, M1/M2 microglial polarization contributes to pain maintenance, and suppression of M1 polarization reduces pro-inflammatory cytokine release and mitigates cancer pain severity.39,40 Similarly, Yuan et al. demonstrated that microglia-mediated C1q/C3-CR3 signaling and subsequent Cathepsin S release play a key role in visceral hypersensitivity in IBS models. 41 Collectively, these findings suggest that persistent microglial activation may underlie long-term visceral hypersensitivity in IBS. Furthermore, microglial states may be shaped by early-life stress. Zhang et al. reported that early-life stress induces lasting microglial phenotypic changes, biasing toward a pro-inflammatory state and affecting synaptic pruning in adulthood. 42 This provides a critical clue for our study, indicating that neonatal CRD stimulation may prime SD rat spinal microglia for heightened reactivity in adulthood, resulting in sustained visceral hypersensitivity. Overall, these findings emphasize the importance of microglial function as a potential therapeutic target for chronic pain and suggest that early intervention may prevent maladaptive sensitization.
Microglia regulate neural network function through multiple mechanisms, including synaptic phagocytosis, cytokine release, and secretion of neurotrophic factors.43,44 In neuropathic pain, activated microglia can phagocytose synaptic structures, leading to local circuit remodeling and altered nociceptive transmission45,46; during acute restraint stress, microglia selectively phagocytose dendritic spines of GABAergic neurons, weakening inhibitory transmission and enhancing excitatory activity. 47 These results collectively support the critical role of microglia in regulating central E/I balance. Beyond suppression of inhibitory circuits, microglia can also directly enhance excitatory neuronal activity through secretion of BDNF, TNF-α, IL-1β, and other factors.48,49 In our study, although microglia were activated and secreted pro-inflammatory cytokines, excitatory synaptic proteins and neurotransmitters showed no significant changes. Fluorescence imaging further indicated that microglia selectively phagocytosed inhibitory synapses, a phenomenon that may be specific to the IBS model. Similar mechanisms have been reported in other diseases: in neuropathic pain, microglial upregulation of P2X4 receptors concurrently weakens GABAergic inhibition 50 ; in Alzheimer’s disease, microglia-mediated synaptic pruning selectively impairs inhibitory connections, leading to circuit hyperexcitability and cognitive decline51,52; in epilepsy, microglial cytokine release enhances excitatory synaptic plasticity, exacerbating seizure activity. 53 Additionally, Yuan et al. reported that microglia influence amygdalar plasticity and participate in emotional regulation in anxiety models.41,54 Accumulating evidence suggests that multiple molecular mechanisms may jointly contribute to the selective recognition of inhibitory synapses by microglia. Among these, the complement system has been identified as a key pathway mediating synaptic tagging and phagocytosis, with the C1q/C3–CR3 axis playing a prominent role in aberrant synaptic pruning across various central nervous system disease models. 55 Furthermore, the CX3CL1/CX3CR1 signaling pathway is critically involved in microglia–neuron interactions and the maintenance of synaptic homeostasis. Neuron-derived CX3CL1 can fine-tune microglial phagocytic activity and inflammatory phenotypes through its receptor CX3CR1.56,57 Our results show that in the spinal cord of IBS-like rats, activated microglia selectively phagocytose inhibitory synapses, accompanied by downregulation of vesicular GABA transporter (VGAT) and glutamate decarboxylase (GAD), thereby impairing inhibitory neuronal function. Disruption of E/I balance and amplification of nociceptive signaling may be a key driver of visceral hypersensitivity in IBS. However, the precise molecular pathways by which microglia achieve selective recognition of inhibitory synapses under IBS-like conditions remain to be further elucidated.
Finally, this study found that miRNA-let7b5p alleviates visceral hypersensitivity through downregulation of MAP3K3. MAP3K3 is a key upstream regulator of the MAPK signaling pathway and can activate ERK1/2, JNK, and p38 pathways, thereby modulating cytokine expression and apoptosis. 58 In the present study, MAP3K3 was identified as a direct target of miRNA-let7b5p, and its knockdown phenocopied the effects of miRNA-let7b5p agomir in suppressing spinal microglial activation and alleviating visceral hypersensitivity, supporting a critical role for the miRNA-let7b5p–MAP3K3 axis in IBS-related visceral pain. However, the activation of downstream MAPK signaling pathways, including ERK, p38, and JNK phosphorylation was not examined. 59 Future studies assessing p-ERK/ERK, p-p38/p38, and p-JNK/JNK in spinal tissues will be essential to further define the MAP3K3-dependent signaling mechanisms underlying chronic visceral pain. Notably, miRNAs also play roles in synaptic regulation. For example, miRNA-429 enhances synaptic insertion of AMPA receptor GluA1 by inhibiting MKP-1, thereby promoting excitatory synaptic plasticity.60–62 Our results indicate that upregulation of miRNA-let7b5p not only suppresses microglial activation but also enhances inhibitory neuronal function, alleviating visceral hypersensitivity. These findings suggest that miRNA-let7b5p may serve as a specific therapeutic target for visceral pain in IBS.
In summary, this study elucidates the mechanistic interplay among miRNA-let7b5p, spinal microglial activation, and synaptic plasticity in IBS-associated visceral hypersensitivity. Several limitations of the present study should be acknowledged. A limitation of this study is that neuronal synaptic plasticity was investigated only at the molecular level, without electrophysiological validation. Future studies integrating electrophysiology and complementary approaches will further clarify how microglial activation influences synaptic dynamics. In addition, only male animals were used to minimize variability related to sex hormone fluctuations. However, given the higher prevalence of IBS in women and the established role of sex hormones in modulating pain perception and microglial function, this design may limit the generalizability and translational relevance of our findings. Future studies should validate the miRNA-let7b5p–MAP3K3–microglia–inhibitory synapse regulatory axis in female models and explore potential sex-dependent mechanisms. In addition, although miRNA-let7b5p is predominantly localized to spinal microglia, its direct effects on neurons or astrocytes have not been examined and warrant further investigation using cell type–specific approaches. Given the specificity and stability of miRNAs, they hold substantial clinical translational potential as therapeutic targets. Currently, miRNA-based interventions, including mimics and antisense oligonucleotides, have advanced in oncology and neurodegenerative diseases. Therefore, exploring miRNA-let7b5p-based interventions may open new avenues for the clinical management of IBS and related disorders.
Conclusion
miRNA-let7b5p alleviates visceral hypersensitivity by inhibiting the activation of spinal microglia in IBS-like rats.
Supplemental Material
sj-docx-1-mpx-10.1177_17448069261430202 – Supplemental material for miRNA-let7b5p alleviates visceral hypersensitivity by inhibiting the activation of spinal microglial in male IBS-like rats
Supplemental material, sj-docx-1-mpx-10.1177_17448069261430202 for miRNA-let7b5p alleviates visceral hypersensitivity by inhibiting the activation of spinal microglial in male IBS-like rats by Xianhe Wu, Ying Tang, Zhengqing He, Fan Yang, Yiqian Liu, Qianli Zhang, Aiqin Chen, Yu Chen and Chun Lin in Molecular Pain
Footnotes
Acknowledgements
We would like to thank the Foundation of National Natural Science Foundation of China (82471229), Science and Technology of Fujian Province (2021Y9172), Natural Science Foundation of Fujian Province (22SCZZX020). The funders played no role in the study design or implementation. In addition, we thank the Public Technology Service Center of Fujian Medical University (China) for providing technical support and experimental platforms.
Author contributions
Xianhe Wu: draft preparation, experiment design, in vivo experiments, and data analysis. Ying Tang and Zhengqing He: in vitro experiments and data analysis. Fan Yang: IBS model construct. Yiqian Liu and Qianli Zhang: data analysis. Aiqin Chen, Yu Chen, and Chun Lin: experiment design, supervision, editing, and manuscript writing.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors have read and approved the final version of the manuscript. They have agreed to the submission of the manuscript to molecular pain. The authors affirm that the manuscript is original, has not been published previously, and is not under consideration for publication elsewhere. Each author has made substantial contributions to the work and agrees to be accountable for all aspects of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Natural Science Foundation of China (82471229), Fujian Provincial Finance Special Fund (22SCZZX020), and Fujian Science and Technology Joint Innovation Fund (2021Y9172).
Ethical considerations
We confirm that the ethical approval has been obtained. The animal experiments in this study were reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of Fujian Medical University (approval no. IACUC FJMU 2024-Y-0581).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
