Abstract
Irritable bowel syndrome (IBS) is characterized by chronic visceral pain, but its molecular mechanisms remain controversial, hindering effective treatment. This research is to investigate the role of lncRNA RT1-CE10 in chronic visceral pain associated with IBS and to elucidate the underlying molecular mechanisms. An IBS rat model was developed in rats, and RNA-Seq analysis was conducted to assess lncRNA RT1-CE10 expression. The subcellular localization of lncRNA RT1-CE10 and its co-localization with ATP1a3 in spinal cord neurons were examined. AAV was used to over-express lncRNA RT1-CE10 in the spinal cord to study its effects on ATP1a3 levels and pain response, with knockdown experiments to evaluate the impact of reduced lncRNA RT1-CE10. The RNA-Seq analysis revealed a significant down-regulation of lncRNA RT1-CE10 in IBS rats. The lncRNA was found to be expressed in both the cytoplasm and the nucleus and to co-localize with ATP1a3 in spinal cord neurons. Over- expression of lncRNA RT1-CE10 via AAV-lncRT1-CE10 increased ATP1a3 levels and alleviated visceral pain response, while knockdown of lncRNA RT1-CE10 decreased ATP1a3 levels and enhanced visceral pain response. Additionally, a marked decrease in ATP1a3 expression was observed in the spinal cords of IBS rats. Modulating ATP1a3 expression either through over-expression or knockdown could alleviate or aggravate chronic visceral pain, respectively. LncRNA RT1-CE10, which is lowly expressed in the spinal cord of IBS rats, interacts with ATP1a3 and influences chronic visceral pain. These findings could lead to the development of targeted therapeutic interventions for IBS.

Keywords
Introduction
Chronic functional visceral pain, notably in irritable bowel syndrome (IBS), poses significant clinical challenges due to its multifaceted pathophysiology and the scarcity of efficacious treatments.1,2 The underlying mechanisms of visceral pain, encompassing intricate molecular and cellular processes, remain poorly understood. Emerging research highlights the pivotal role of long non-coding RNAs (lncRNAs) in various biological processes, including pain modulation,3,4 but their specific contributions to chronic functional visceral pain and IBS are not yet fully elucidated.
Several lncRNAs have been identified to be involved in the development of chronic pain by regulating the expression of relevant genes and a range of mechanisms including ion channels, inflammatory pathways, neurotransmitter release, oxidative stress, and neuronal autophagy and apoptosis.5,6 Significantly different lncRNAs were found, for example, in the spinal cord of rats with chronic constriction injury (CCI) of the sciatic nerve, 7 in the spinal cord of rats with peripheral neuropathy, 8 and in the serum of patients with diabetic neuropathic pain (DNP), 9 suggesting that lncRNA mediates neuroinflammation, central sensitization, and chronic pain. In addition, lncRNAs are increased in inflammatory pain rats,10,11 and microarray analyses of bone cancer pain rats have revealed dysregulated expression of lncRNAs in the DRG and the dorsal horn of the spinal cord, which are involved in pain-associated inflammatory and immune pathways.12,13 Recent studies underscore the influence of lncRNAs in pain pathways, with certain lncRNAs like H19, XIST, and MEG3 implicated in diverse pain types, including neuropathic, inflammatory, and cancer pain.14,15 These lncRNAs exert their effects through chromatin remodeling, transcriptional control, and post-transcriptional modifications. 16 For example, HOTAIR modifies gene expression by altering chromatin structure, while MALAT1 influences mRNA stability and translation. 14 Given their regulatory capacity in gene expression, lncRNAs are now considered crucial in neuropathic pain progression, 17 with transcriptomic analyses revealing their involvement post-nerve injury, hinting at their therapeutic potential. 18 Knockdown of lncRNA MRAK159688 mitigated the development of morphine tolerance and partly reversed morphine-induced mechanical hyperalgesia. 19 However, it is unclear whether lncRNAs are involved in chronic visceral pain and its pathogenic mechanisms.
The ATP1A3 gene encodes the α3 subunit of the Na+/K+-ATPase, which plays a key role in the maintenance of the intra- and extracellular electrochemical gradients of Na+and K+, especially in neurons. ATP1A3 plays a key role in human cortical development, 20 and mutations in the gene have been associated with a variety of neurological disorders,21,22 including rapid-onset dystonia-parkinsonism (RDP),23,24 alternating hemiplegia of childhood (AHC),25,26 cerebellar ataxia, areflexia, pes cavus, optic atrophy, sensorineural hearing loss (CAPOS) syndrome, 27 and seizures,28,29 Leads to neurodevelopmental delay, attention deficit, trunk instability, dystonia, or ataxia,24,26,30–33 for which no effective treatment exists. 30 Holm et al. 34 found that ATP1A3 plays a role in the control of spatial learning and memory and that this role is related to GABA transmission.
There are few studies on ATP1A3 in pain, but based on the role of ATP1A3 for the nervous system and the involvement of lncRNAs in regulating the expression of a variety of disease-associated ion channels, as well as studies suggesting the involvement of lncRNAs and ATP1A3 in mood and cognitive dysfunction,34–36 we hypothesize that ATP1A3 may act as an important downstream molecule of the lncRNA.
Our study focuses on lncRNA RT1-CE10, identified as significantly down-regulated in the spinal cords of IBS rats through transcriptome sequencing. This suggests a possible role in pain modulation. Further research find lncRNA RT1-CE10 can alleviate visceral hypersensitivity by promoting the expression of its RBP ATP1a3 in IBS rats. This comprehensive approach will contribute to our understanding of lncRNAs in chronic visceral pain and pave the way for targeted therapies in IBS.
Materials and methods
Animals
Male Sprague−Dawley rats (<5 days old) were obtained from the Experimental Animal Center of Fujian Medical University (SCXK (Fujian Province) 2016−0006), Fuzhou, China). The animals were randomly allocated to each group. All animals were kept in a greenhouse with a controlled humidity of 65%–77%. They were maintained on a 12 h light/dark cycle. All procedures were approved by the Animal Care and Use Committee. Animal groups were randomized and coded by an independent researcher. Experimental operators and data analysts remained blinded to group assignments throughout the study.
Establishment of chronic functional visceral pain model of IBS
SD rats were modeled on days 8–14 after birth. A 20 mm × 2.5 mm human vascular reconstruction balloon was used to give colorectal distension (CRD) at a pressure of 60 mmHg at a fixed time every day. The balloon was connected to a sphygmomanometer to control the pressure. The balloon was distended once a day for 1 min. The rats were weaned on day 21 after birth and raised in separate cages normally. The control group rats were operated in the same way as the IBS model group rats except that colorectal distension was not given. The rats were raised normally until the sixth week when their body weight reached about 220 g.
Electromyographic (EMG)
To assess the visceral hypersensitivity characteristic of IBS, we replicated the methodology from our preceding work, focusing on capturing the modifications in EMG reactions elicited by CRD at specific pressures (40 mmHg, 60 mmHg). 37 Balloons for distending the descending colon were positioned under isoflurane sedation. To capture abdominal muscle spike activity, silver bipolar electrodes were implanted within the musculature. Distention was achieved via swift balloon inflation for 10 s, followed by a 4-min quiescent interval. An RM6240BD system (Chengdu, China) was utilized for gathering EMG data. Relative percentage change in EMG, calculated as (EMGc − EMGb) * 100%/EMGb, denoted the alteration, with EMGc reflecting the CRD-induced response and EMGb signifying the 20-s baseline mean.
Pain threshold measurement
After the latex balloon was placed as described above, the rat was placed in a 20 cm × 6 cm × 8 cm plexiglass box. The rat could move forward and backward but could not turn around. The experiment was started after the rat was fully adapted for about 30 min. The pressure was increased by 5 mmHg each time. The rat’s lower abdominal wall was obviously lifted off the bottom of the box or obviously contracted and flattened by naked eye. The minimum pressure value at this time was taken as the pain threshold. The CRD expansion pressure range was 0–60 mmHg. The measurement was repeated 3 times, with an interval of 4 min each time, and the average value was calculated (n = 8).
Intrathecal injection
IBS model rats and blank SD rats were catheterized 6 weeks after birth, and intrathecal injections were performed one week after catheterization. IBS model rats were intrathecally injected with lncRT1-CE10-AAV (5.38E+12 vg/ml, Wuhan BrainVTA Co., Ltd.) or lncRT1-CE10-NC (3.42E+12 vg/ml, Wuhan BrainVTA Co., Ltd.), 10 μl/time, once in total; blank SD rats were intrathecally injected with rat shlncRT1-CE10-AAV (5.45E+12 vg/ml, Wuhan BrainVTA Co., Ltd.) or shlncRT1-CE10-NC (5.12E+12 vg/ml, Wuhan BrainVTA Co., Ltd.), 10 μl/time, once in total; IBS model rats were intrathecally injected with ATP1a3-AAV (1.22E+13 vg/ml, Shanghai GeneChem Co., Ltd.) or ATP1a3-NC (3.73E+13 vg/ml, Shanghai GeneChem Co., Ltd.) 10 μl/time, once in total. The virus reacted in SD rats for 21 days; IBS model rats were intrathecally injected with minocycline or saline, 100 μg/20 μl, once a day for seven consecutive days.
Western blotting
Protein sample loading (protein content 30 μg) was separated by electrophoresis and transferred to polyvinylidene fluoride (PVDF) membranes (Invitrogen, USA). Rabbit anti-ATP1a3 primary antibody (1:1000, abcam), rabbit anti-Iba-1 primary antibody (1:2000, abcam), mouse anti-MHC2 primary antibody (1:1000, Santa Cruz), and mouse anti-β-actin primary antibody (1:7000, ABclonal) were added respectively. Horseradish peroxidase (HRP)-labeled goat anti-rabbit secondary antibody (1:2000) and goat anti-mouse secondary antibody (1:1000) were added respectively and incubated at room temperature for 2 h. Immobilon Western HRP reagent A and reagent B were prepared in a 1:1 ratio, dropped onto the PVDF membrane, placed in a molecular imaging instrument for imaging, and the results were observed.
Quantitative real-time PCR
Total RNA was isolated from rat spinal cord thoracolumbar segment (T13-L2) cells. cDNA was reverse transcribed from the sample RNA using Evo M-MLA (AG11707) Reverse Transcription Kit (Accurate Biotechnology, China). cDNA was prepared by using the SYBR Green Pro Taq HS (AG11701) Premixed qPCR kit (Accurate Biotechnology, China) for RT-PCR reactions, and all procedures were performed according to the manufacturer’s protocol. RNA expression was calculated using the ΔΔCt method.
Fluorescence in situ hybridization
RNA localization and quantification were performed using GIMA FISH kits and Exiqon RNA probes. For combined RNA FISH/immunostaining, we first performed RNA FISH followed by immunofluorescence. All steps used diethylpyrocarbonate (DEPC)-treated water to exclude RNA degradation.
RNA immunoprecipitation
The spinal cord tissue was broken by ultrasonic treatment. RNA immunoprecipitation was performed using an RNA-binding protein immunoprecipitation kit (BersinBio Biotechnology Co., Ltd., Guangzhou, Guangdong, China). Input was used as a positive control and IgG as a negative control. The levels of co-precipitated lncRNA RT1-CE10 and ATP1a3 were detected by qPCR.
RNA pull-down assay
The lncRNA RT1-CE10 probe was synthesized, the tissue samples were ultrasonically lysed, RIP buffer and protease inhibitors were added, and the lysate was centrifuged at 15,000 g for 15 min at 4°C to obtain the lysate, which was incubated with the lncRNA RT1-CE10 probe and streptavidin magnetic beads. The proteins of the RNA-protein complex were captured from the magnetic beads. 30 μl of protein samples were taken for LC-MS detection, and 15 μl of protein samples were taken for PAGE-Western blot detection.
Statistical analysis
All quantitative data were expressed as mean ± standard error (mean ± SEM). Regarding the discharge of the external oblique muscle, the percentage increase of the original discharge amplitude of the external oblique muscle under each CRD pressure relative to the basal discharge amplitude was used to represent the discharge amplitude of the external oblique muscle under the corresponding CRD pressure. This method was used to obtain the discharge data of the external oblique muscle, and two-way ANOVA was used. The pain threshold of rats was analyzed by two-way ANOVA. Immunoblotting and qPCR data were analyzed by unpaired t-test (two groups) or one-way ANOVA (three groups). All data were analyzed and pictures were prepared using IBM SPSS Statistics 23 and Graph Pad Prism 8.0.2 statistical software. p < 0.05 was used as the standard for statistically significant differences.
Results
IBS rats shows enhanced visceral nociceptive sensitivity and spinal lncRNA RT1-CE10 is down-regulated in IBS rats
Newborn rats were subjected to colonic distension and the success of establishing an IBS model was evaluated at seven weeks of age. The amplitude of EMG in the IBS group rats was higher significantly compared to the control group both at 40 and 60 mmHg CRD (p < 0.001, Figure 1(a), (b)). Pain threshold of the IBS group rats was significantly decreased (p < 0.001, Figure 1(c)). The HE staining results showed that there was no significant difference in the structure of the colonic gland between two groups (Figure 1(d)), suggesting that visceral nociceptive sensitivity of IBS rats is functional but not structural. These findings suggest that stressful stimulation during the neonatal period may result in persistent visceral hypersensitivity in adulthood, and the chronic functional visceral pain rat model of IBS has been successfully established.

Assessment of visceral hypersensitivity in rats and qPCR validation of lncRNA RT1-CE10 down-regulation in IBS rats. (a) Original representative graph of rat EMG. (b) The statistical chart of the percentage of EMG amplitude over baseline. The formula of calculating EMG to CRD is equal to (CRD responses-baseline)/baseline × 100%. n = 6, ***: p < 0.001 versus control rats, Two-way ANOVA. (c) Statistical chart of pain threshold measured in rats. n = 8, ***p < 0.001 versus control rats, Unpaired t-test. (d) Representative and enlarged images of HE staining of rat colon glands (scale 200/50 µm). (e and f) Volcano plot and heat map of spinal cord dorsal horn lncRNA sequencing. n = 3. (g) qPCR showed the expression of 6 different lncRNAs. lncRNA RT1-CE10 showed significant down-regulation in the spinal cord. n = 6, *p < 0.05, ***p < 0.001, versus control rats, Unpaired t-test. (h) Tissue specificity of lncRNA RT1-CE10 on heart, liver, spleen, lung, kidney, DRG, hippocampus, spinal cord. (i) Expression of spinal lncRNA RT1-CE10 in different pain models. (j) The expression of lncRNA RT1-CE10 decreased significantly from 4 to 10 weeks of age. n = 6, *p < 0.05, ns: p > 0.05 versus control rats, Unpaired t-test.
Emerging research highlights the pivotal role of lncRNAs in pain modulation, therefore we investigated the expressions of lncRNAs using RNA-Seq analysis on the thoracic spine (T13-L2). The results revealed that 75 lncRNAs were upregulated (red) and 52 lncRNAs were down-regulated (blue; Figure 1(e), (f)). Based on the criteria of differential expression with a fold change of greater than 2 and p < 0.05, six lncRNAs were selected for qPCR analysis (Figure 1(g)). Among which, the difference of lncRNA RT1-CE10 between two groups was the most (p < 0.05). Therefore, we selected lncRNA RT1-CE10 as our target molecule to explore its roles in visceral nociceptive sensitivity of IBS rats.
QPCR results showed that lncRNA RT1-CE10 was more expressed in the spinal cord and hippocampus than other tissue (Figure 1(h)). The expression of spinal lncRNA RT1-CE10 in IBS model was significantly decreased (p < 0.05, Figure 1(i)), while that in TNBS and SMIR models was not observed. (p = 0.9077, p = 0.1185, Figure 1(i)). Furthermore, the expression of spinal lncRNA RT1-CE10 significantly decreased from 4 to 10 weeks in IBS rats (p < 0.05, Figure 1(j)). The above results suggest a possible role of lncRNA RT1-CE10 in the pathogenesis of IBS.
Localization of lncRNA RT1-CE10 in the spinal cord
Since lncRNA RT1-CE10 expression was substantially down-regulated in IBS rats, we next conducted co-staining of lncRNA RT1-CE10 with markers for neurons (NeuN), astrocytes (GFAP), and microglia (Iba-1) with FISH to explore the cellular specificity. The results revealed co-labeling of lncRNA RT1-CE10 with neurons, but not with astrocytes or microglia (Figure 2(a)), indicating that lncRNA RT1-CE10 is neuron-specific in the spinal horn. Next, Fish experiments (Figure 2(b), (c)) indicated a significant reduction in the fluorescence intensity of lncRNA RT1-CE10 in the spinal horn of IBS rats (p < 0.05). Furthermore, quantitative analysis found that it was expressed in both the cytoplasm and the nucleus, with more in the cytoplasm than in the nucleus (Figure 2(d), (e)).
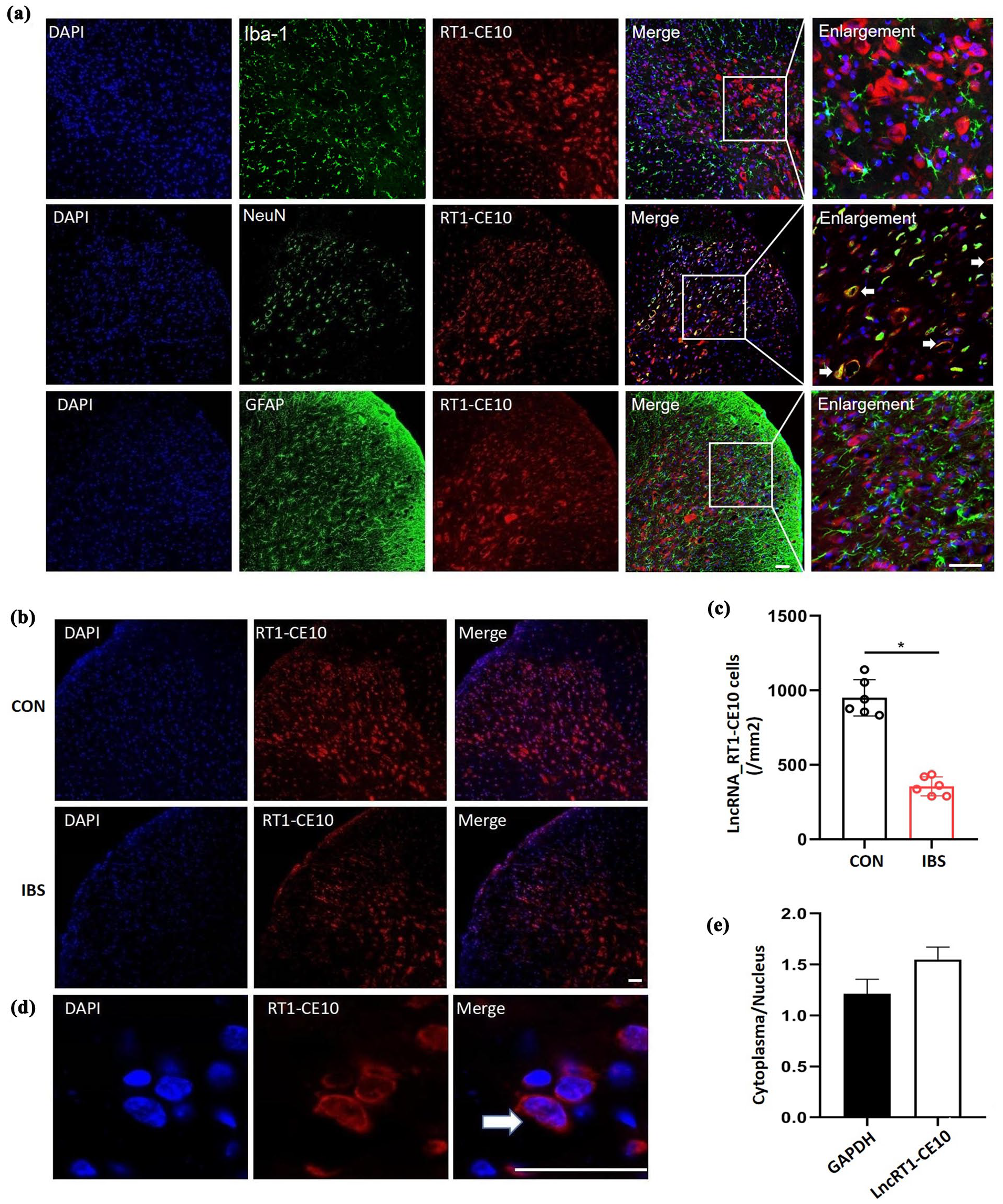
Localization of spinal lncRNA RT1-CE10. (a) FISH and IF showed that lncRNA RT1-CE10 (red) is expressed mainly in neurons (green), but not in astrocytes (green) and microglia (green) of the T13 spinal cord. (b and c) FISH showed lncRNA RT1-CE10 was decrease in spinal dorsal horn of IBS rats. (d and e) qPCR and FISH showed that lncRNA RT1-CE10 expressed in both the nuclear and cytoplasm of cultured spinal neurons. n = 6, *p < 0.05, versus control rats, Unpaired t-test. Scale bar = 50 μm.
lncRNA RT1-CE10 plays roles in the visceral pain hypersensitivity of IBS rats
To further verify the effect of lncRNA RT1-CE10 on IBS, AAV-lncRT1-CE10 was intrathecal injected to over-express lncRT1-CE10 (Figure 3(a)). While AAV-ShlncRT1-CE10 was intrathecal injected to knockdown lncRT1-CE10 (Figure 3(b)). The EMG results indicated spinal lncRT1-CE10 over-expression relieved visceral pain response (p < 0.05, Figure 3(c), (d)) while lncRT1-CE10 knock-down enhanced visceral pain response (p < 0.05, Figure 3(e), (f)), suggesting the role of lncRNA RT1-CE10 in the visceral pain hypersensitivity of IBS rats.

Involvement of lncRNA RT1-CE10 in the regulation of chronic visceral pain in IBS rats. (a) Intrathecal injection of AAVlncRNA RT1-CE10 in IBS rats, qPCR detection of lncRNA RT1-CE10 over-expression efficiency. (b) Intrathecal injection of shlncRNA RT1-CE10 in control, qPCR detection of lncRNA RT1-CE10 knockout efficiency. (c and d) Raw representative and statistical plots of lower external oblique muscle firing at 40 and 60 mmHg CRD stimulation after intrathecal injection of AAVlncRNA RT1-CE10 in IBS rats. n = 8, *p < 0.05, versus IBS-AAV-NC rats, Two-way ANOVA. (e and f) Raw representative and statistical graphs of lower external oblique muscle firing at 40 and 60 mmHg CRD stimulation after intrathecal injection of shlncRNA RT1-CE10 in control. n = 8, *p < 0.05, versus CON-AAV-scramble rats, Two-way ANOVA.
lncRNA RT1-CE10 plays roles in chronic functional visceral pain by binding ATP1a3
To explore the mechanism of lncRNA RT1-CE10 in IBS, an RNA pull-down assay was performed using the lncRNA RT1-CE10 biotin probe to identify proteins that may interact with it. The silver-stained gel blocks obtained from the assay were subjected to mass spectrometry analysis, which identified five proteins with significant PSM values: Lrpprc, PC, ATP1a1, and ATP1a3 (Figure 4(a)). Further experiments of qPCR revealed these proteins showed differential expression between the IBS and control groups, especially ATP1a3 (Figure 4(b)).

ATP1a3 functioned as an RBP of lncRNA RT1-CE10. (a)The lncRNA RT1-CE10 biotin probe was used to detect its possible binding protein by RNA pull down. (b) Target protein was screened from mass spectrometry results. n = 6, *: p < 0.05, versus control rats, Unpaired t-test. (c) lncRNA RT1-CE10 biotin probe co-precipitates with ATP1a3. (d) ATP1a3 antibodies can be enriched for lncRNA RT1-CE10. n = 3, *p < 0.05, versus control rats, Unpaired t-test. (e) Expression levels of ATP1a3 mRNA in dorsal horn of spinal cord in rats. n = 6, *p < 0.05, versus IBS-AA-NC rats, One-Way ANOVA. (f) Original graph and statistical graph of the expression level of ATP1a3 protein in the dorsal horn of spinal cord in rats. n = 6, *p < 0.05, versus control rats, Unpaired t-test. (g) ATP1a3 mRNA expression level after intrathecal injection of AAVlncRNA RT1-CE10 in IBS rats. n = 6, *p<0.05, versus IBS-AA-NC rats, One-Way ANOVA. (h and i) Original graph and statistical graph of ATP1a3 protein expression level after intrathecal injection of AAVlncRNA RT1-CE10 in IBS rats. n = 6, *p < 0.05, versus IBS-AA-NC rats, Unpaired t-test. (j) The expression level of ATP1a3 mRNA in control after intrathecal injection of shlncRNA RT1-CE10. n = 6, *p < 0.05, versus CON-AAV-NC rats, One-way ANOVA. (k and l) Original graph and statistical graph of ATP1a3 protein expression level after intrathecal injection of shlncRNA RT1-CE10 in control group. n = 6, *p < 0.05, versus CON-AAV-NC rats, Unpaired t-test.
Thus, it was hypothesized that lncRNA RT1-CE10 may bind to ATP1a3 to exert its effects. This speculation was confirmed by the co-precipitation of ATP1a3 protein with the lncRNA RT1-CE10 probe (Figure 4(c)) and the enrichment of lncRNA RT1-CE10 by the ATP1a3 antibody in a reverse RIP experiment (p < 0.05, Figure 4(d)).
QPCR results demonstrated that spinal ATP1a3 expression reduced in IBS rats, which was further verified by western blot experiment (Figure 4(e), (f)). ATP1a3 level was significantly increased after intrathecal injection of the adenovirus over-expressing lncRNA RT1-CE10 in IBS rats (p < 0.05, Figure 4(g)–(i)). In contrast, ATP1a3 level was significantly decreased after adenovirus knocking down lncRNA RT1-CE10 in control rats (p < 0.05, Figure 4(j)–(l)). These results further suggest ATP1a3 is the downstream of lncRNA RT1-CE10. It may bind to lncRNA RT1-CE10 and then play a role in IBS chronic visceral pain.
Localization of ATP1a3 in the spinal cord and its role in visceral pain of IBS
lncRNA RT1-CE10 is neuron-specific in the spinal horn, and ATP1a3 is proved to serve as a binding protein for lncRNA RT1-CE10. Therefore, we speculate that the cellular localization of ATP1a3 might be neuron-specific. The double-labeling immunohistochemical fluorescence staining experiments indicated that ATP1a3 was co-localized with neurons but not with astrocytes and microglia (Figure 5(a)). In addition, co-stained results revealed that ATP1a3 and lncRNA RT1-CE10 were co-expressed in neurons (Figure 5(b)).

Localization of ATP1a3 in the spinal cord and spinal ATP1a3 relieves visceral pain in IBS rats. (a) ATP1a3 (green) is expressed in neurons (red) but not in microglia, astrocytes (red). (b) Spinal cord ATP1a3 co-expression with lncRNA RT1-CE10. n = 3. Scale bar = 50 μm. (DAPI: nucleus, NeuN: neuronal marker, Iba-1: microglial marker, GFAP: astrocytic marker). (c) Expression levels of ATP1a3 mRNA in dorsal horn of spinal cord after intrathecal injection of AAV-ATP1a3 in IBS rats. n = 6, *p < 0.05, versus IBS-AAV-NC rats, One-Way ANOVA. (d and e) Original and statistical graphs of ATP1a3 protein expression after intrathecal injection of AAV-ATP1a3 in IBS rats. n = 6, *p < 0.05, versus IBS-AAV-NC rats, Unpaired t-test. (f and g) Raw representative and statistical plots of the inferior external oblique muscle firing at 40 and 60 mmHg CRD stimulation following intrathecal injection of AAV-ATP1a3 in IBS rats. n = 6, *p < 0.05, versus IBS-AAV-NC rats, Two-way ANOVA.
In order to assess the efficacy of ATP1a3 over-expression, the mRNA and protein expression levels of ATP1a3 in the spinal cords of IBS rats that received intrathecal injections of an ATP1a3 over-expression adenovirus were examined, respectively. The results demonstrated that ATP1a3 level was elevated after receiving the over-expressed ATP1a3 adenovirus (p < 0.05, Figure 5(c)–(e)). Subsequently, the effect of ATP1a3 on chronic functional visceral pain was investigated by evaluating the change in EMG amplitude. The results reveal that the EMG amplitude is significantly lower in rats over-expressing ATP1a3 (p < 0.05, Figure 5(f), (g)). These results suggest that the intervention of over-expression of spinal ATP1a3 has the potential to alleviate chronic functional visceral pain in IBS rats.
Discussion
This study established an irritable bowel syndrome (IBS) model in rats by successfully inducing rectal distension in the neonatal period. The rats exhibited pain sensitization, and a neuron-specific lncRNA, lncRNA RT1-CE10, was found to be down-regulated in the spinal cord dorsal horn of IBS rats. Furthermore, we discovered that ATP1a3, an RNA-binding protein, plays a role in the function of lncRNA RT1-CE10 in neurons. We observed co-expression of ATP1a3 and lncRNA RT1-CE10 in neurons and lncRNA RT1-CE10 can increase the expression of ATP1a3. High expression of ATP1a3 in the spinal cord could alleviate chronic functional visceral hypersensitivity in IBS rats.
lncRNA is a type of non-coding RNA with a length of more than 200 nucleotides, which does not have the ability to encode proteins. Many studies have found that lncRNAs are widely involved in various biological processes such as cell cycle control, apoptosis, differentiation, epigenetics, synaptic plasticity, and negative emotional behavior.38,39 The expression of lncRNAs in various types of tissues was found to be more tissue-specific than mRNAs, suggesting that lncRNAs are closely related to the functional specificity of tissues. 40 Studies have shown that abnormally expressed lncRNAs in different tissues are associated with various neurological disorders, and are widely expressed in central and peripheral tissues, such as the hippocampus, 41 PFC, 42 and DRG. 43 Our research has shown that the down-regulation of lncRNA RT1-CE10 in the spinal cord dorsal horn of IBS rats may be associated with chronic functional visceral hypersensitivity. A large number of lncRNAs have been identified in pain-related regions of the nervous system (e.g. DRG and spinal cord) in mice, rats, and humans. 44 Their expression profiles are related to specific neuroanatomical regions, cell types or subcellular compartments, indicating their potential functional roles in the nervous system. 44 Transcription and translation changes in gene expression in the dorsal root ganglia (DRG), spinal dorsal horn, and pain-related brain regions are believed to be involved in the development of neuropathic pain after peripheral nerve injury. 45 In our study, we established an IBS rat model and found that lncRNA RT1-CE10 was down-regulated in the spinal cord of IBS rats at 4–10 weeks, which coincided with the duration of visceral pain sensitivity, and was distributed in the nuclei and cytoplasm of spinal dorsal horn neurons. The lncRNA RT1-CE10 in the spinal cord of IBS rats was tissue-specific, model-specific. RNA sequencing and qPCR validation in our research demonstrated that lncRNA RT1-CE10 was specifically downregulated in the IBS model, while no significant changes were observed in the inflammatory pain and somatic nerve injury models, suggesting that its role may be more restricted to visceral pain pathways. However, since the spinal dorsal horn serves as a shared processing hub for both visceral and somatic signals, we hypothesize that the lncRNA RT1-CE10 may preferentially regulate visceral pain, but its potential impact on other types of pain requires further validation.
Our findings are consistent with growing evidence that lncRNAs play important roles in embryonic development, cancer, inflammation, and neurological diseases. These molecules, despite not encoding proteins, have been shown to regulate gene expression and are implicated in various human diseases, including pain modulation.46–50 Research has shown that lncRNAs are involved in the modulation of pain pathways. Studies have highlighted the involvement of lncRNAs in different types of pain, such as neuropathic pain, inflammatory pain, bone cancer pain, and chronic pain.51–56 A large number of lncRNAs exhibit differential expression in neuropathic pain models, such as chronic constriction injury (CCI) and spinal nerve ligation (SNL), and are involved in the development and maintenance of pain.57,58 There is massive gene dysregulation in both protein-coding and noncoding genes in chronic pain.59,60 These findings suggest that lncRNAs may play critical roles in the molecular mechanisms underlying pain sensitivity and chronic pain states. Different lncRNAs may play different roles in pain. In our research, the down-regulation of lncRNA RT1-CE10 may be associated with chronic functional visceral hypersensitivity in IBS rats. To investigate how lncRNA RT1-CE10 plays a role in IBS, we further explored its binding proteins.
lncRNA can participate in regulating the stability of mRNA by directly binding to mRNA or acting as a molecular bait for RNA-binding proteins, and participate in cellular life activities such as post-transcriptional regulation of mRNA, cell signal transduction, and protein activity.61–63 In the adult mouse brain, Bottger et al. 64 found that ATP1a3 isoforms were highly expressed in the striatum, pallidum, thalamic floor, substantia nigra, thalamus, cerebellum, erythrocyte, nucleus accumbens, pontine reticularis capitis nucleus, and the hippocampus, and that they co-localized mainly with GABAergic neurons. Allocco et al. 65 demonstrated that ATP1A3 was expressed alongside Sox2, a marker for neural stem cells, which underscores its relevance in neuronal differentiation and development. Sweadner et al. 66 noted that ATP1A3 is expressed in various neuronal types, including Purkinje cells and other cerebellar neurons, further supporting the notion of its widespread presence in the nervous system. Our immunofluorescence results also showed that ATP1a3 co-labeled with the neuronal marker NeuN.
The ATP1a3 gene encodes the a3 subunit of the Na+/K+ ATPase pump of the neuronal plasma membrane. 67 Na+/K+-ATPase is a protein complex that is key to electrical excitability, neurotransmitter transport, volume regulation, and other important cellular functions.23,64 Genetic dysfunction of cation pumps has been associated with human neurological diseases. 68 Changes in ion homeostasis are known to affect pain signaling pathways. 69 Moreover, the interaction between ATP1A3 and other signaling pathways, such as those involving chemokines, has been shown to play a role in modulating pain responses, as indicated by studies on chemokine signaling in spinal cord neurons. 70 Mutations in ATP1a3 cause the rare movement disorder rapid-onset dystonia-parkinsonism (RDP). 71 We predicted and verified the binding of lncRNA RT1-CE10 to RBP ATP1a3 by RNA pull-down and mass spectrometry analysis. RNA fluorescence in situ hybridization experiments also showed that lncRNA RT1-CE10 co-localized with RBP ATP1a3. Research indicates that ATP1A3 is integral to maintaining ionic homeostasis in neurons, which is essential for proper neuronal function and pain modulation. 72 It’s been proven the involvement of TRPV1, TRPC4 ion channel in visceral pain sensation, emphasizing the importance of ion transport mechanisms in the development of chronic visceral pain.73,74 Therefore, we speculate that ATP1a3 plays a role in chronic functional visceral pain as the RBP of lncRNA RT1-CE10. We further found that spinal ATP1a3 can relieve chronic visceral pain in IBS. The correlation between ATP1a3 expression and visceral pain sensitivity further supports the hypothesis that ion transporters play a crucial role in the pathophysiology of IBS, as suggested by earlier studies on chronic pain and neurological disorders.75,76
The findings underscore the importance of exploring the roles of lncRNAs in pain regulation, particularly in the context of chronic pain conditions like visceral pain. Targeting lncRNAs could offer novel therapeutic strategies for managing pain conditions, including neuropathic pain and potentially visceral pain associated with disorders like IBS. Additionally, the results of our study imply that ATP1a3 over-expression may serve as a potential therapeutic strategy for the treatment of chronic functional visceral pain in IBS rats. Further research is necessary to fully understand the underlying mechanism and to determine the generalizability of our findings to other animal models and to human patients.
Conclusion
In conclusion, this study identifies lncRNA RT1-CE10 as a key regulator in chronic functional visceral pain, particularly in IBS. We found that lncRNA RT1-CE10 is down-regulated in the spinal cord of IBS rats and that its overexpression alleviates pain hypersensitivity. ATP1a3, identified as an RNA-binding protein and potential therapeutic target. Our findings suggest that targeting lncRNAs and related ion transporters may offer novel therapeutic strategies for IBS and other pain conditions. Further research is needed to fully understand the mechanisms and clinical implications.
Footnotes
CRediT authorship contribution statement
Ying Tang: Writing – Original draft, did the experiments, wrote the paper. Zihan Liu: Writing – original draft, did the experiments, wrote the paper. Xianhe Wu: Data curation, analyzed data. Zhengqing He: Did the experiments. Fan Yang: Did the experiments. Huiqin Chen: Data curation, analyzed data. Yu Chen: Scientifically revised the manuscript. Qibin Zheng: Scientifically revised the manuscript. Yang Huang: Designed the study, scientifically revised the manuscript. Aiqin Chen: Scientifically revised the manuscript. Chun Lin: Supervision, designed and supervised the study, writing – review & editing. All authors contributed to and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Natural Science Foundation of China (82471229), the Natural Science Foundation of Fujian Province (2023J01305), the Natural Science Foundation of Fujian Province (2021J01675), the Natural Science Foundation of Fujian Province (2020J01990), Fujian Provincial Finance Special Fund (22SCZZX020), and Fujian Science and Technology Joint Innovation Fund (2021Y9172).
Data availability statement
The original contributions presented in the study are included in the article, and further inquiries can be directed to the corresponding authors.
