Abstract
Background
Irritable bowel syndrome (IBS) is one of the typical representatives of chronic functional visceral pain that lacks effective treatment. Recently, attention has been given to the role of microglia in IBS, particularly the activation of spinal microglia and the subsequent release of Cathepsin S (Cat S), a proteolytic enzyme. However, the specific role of spinal Cat S in IBS remains to be elucidated. The purpose of this study is to investigate the mechanisms underlying the regulation of visceral hypersensitivity in IBS-like rats by Cat S.
Methods
An IBS-like rat model was developed, and visceral sensitivity was tested via the electromyographic (EMG) response to colorectal distention (CRD) and pain threshold. Western blot and immunofluorescence were used to examine the expressions of proteins. The effects of inhibitors or neutralizing antibodies on visceral pain and the downstream molecular expressions were detected. The open-field test was performed to evaluate locomotor activity and anxiety-like behaviors in rats.
Results
We discovered that spinal Cat S was upregulated and colocalized with microglia in IBS-like rats. Treatment with LY3000328, a selective inhibitor of Cat S, dose-dependently down-regulated EMG amplitude and Fractalkine (FKN) expression, indicating that Cat S regulated visceral hypersensitivity via activating FKN in IBS-like rats. Furthermore, the expressions of FKN, CX3CR1, and p-p38 MAPK were elevated in IBS-like rats whereas inhibition of these molecules could alleviate visceral pain. Moreover, pharmacological inhibitor experiments suggested the activation of CX3CR1 by FKN facilitated p38 MAPK phosphorylation, which in turn promoted Cat S expression in IBS-like rats.
Conclusions
Neonatal adverse stimulation might enhance the expression of spinal microglial Cat S, thereby activating the FKN/CX3CR1/p38 MAPK pathway and lead to visceral hypersensitivity in IBS-like rats. As a selective inhibitor of Cat S, LY3000328 could become a potential therapeutic option for IBS.
Introduction
tIrritable bowel syndrome (IBS) is one of the most common gastrointestinal diseases with a prevalence of 15% worldwide. 1 Patients with IBS have symptoms of recurrent abdominal pain, changes in bowel habits, and abdominal distension, which are related to visceral hypersensitivity.2,3 Recently, spinal microglia has been reported to relieve neuropathic pain via insulin-like growth factor 1 (IGF1). 4 Activation of microglia induced a peak of Cathepsin S (Cat S) in the dorsal horn of the ipsilateral spinal cord after injury for 7 days in a rat neuropathic pain. 5 Similar results have been reported that Cat S mRNA levels are up-regulated in the ipsilateral side of the spinal cord 1-day post peripheral nerve injury. 6 These studies gave rise to the concept that Cat S might be a key molecule in microglia-mediated pain processing in neuropathic pain. However, the role of spinal Cat S in chronic functional visceral pain such as IBS remains to be determined.
Cat S is a kind of proteolytic enzyme located in the lysosomes, 7 dendritic cells, macrophages and microglia. Furthermore, Cat S could be released from the intracellular compartment of microglia 8 and promote fractalkine (FKN) cleavage from neuronal membranes to regulate the pain process in the spinal cord of rats. 9 FKN is a transmembrane chemokine that constitutively expressed in neurons of the spinal dorsal horn. Cleaved FKN then binds to its specific receptor microglial CX3CR1 to activate microglia. 10 The pathway of spinal microglia-derived Cat S regulating visceral hypersensitivity appears to be identified clearly in neuropathic pain, which sheds light on us to probe the underlying mechanism of spinal Cat S modulating visceral hypersensitivity in IBS-like rats.
The p38 MAPK signaling pathway has been reported to participate in cell growth, apoptosis and inflammation. 11 Also, activation of p38 MAPK has a direct relationship with many pain-related mediators.12,13 Our previous study showed that spinal phosphorylated p38 MAPK was involved in the central sensitization of chronic visceral pain. 14 In light of the above literature, we wondered whether CX3CR1 and p38 MAPK might be linked in IBS-like rats. Collectively, we focused on whether spinal Cat S was involved in visceral hypersensitivity of IBS-like rats and its underlying mechanism in this study, which could open new insights into the molecular basis of IBS.
Materials and methods
Animals
Neonatal Sprague Dawley (SD) male rats (<5 days old) with their mothers were acquired from the Department of the Experimental Animal Center of Fujian Medical University. The weaning began on the 21st day after birth. After weaning, the litters were raised for 5 weeks with free access to water and food in a quiet room with constant temperature (24–26°C) and humidity (50–60%). All procedures were approved by the Animal Care and Use Committee (SCXK 2016-0006, Fujian Province, China).
Establishment and evaluation of IBS-like rats
IBS-like model
Neonatal SD male rats were randomly allocated to each group. The IBS-like rat model was established as in our previous studies. 15 Briefly, the litters in IBS-like group received colorectal distention (CRD) stimulation at a pressure of 60 mmHg for 1 min each day during the 8th to the 14th days after birth. The control litters were subjected to the same procedure, except for CRD.
Electromyography
The electromyography (EMG) response to CRD was used to assess visceral hypersensitivity in rats. The rats (8–10 weeks old) were fasted for 18 h prior to the experiment. Under isoflurane anesthesia, a balloon was placed in the rectum of the rat for CRD. A pair of silver bipolar electrodes was inserted into the external oblique abdominal muscle to record the spikes of discharge. The balloon pressure was increased to 40 or 60 mmHg and maintained at that pressure for 10 s at intervals of 4 min. The EMG responses to different degrees of CRD were recorded three times and collected by the RM6240BD system (Chengdu, China). The change in EMG was expressed as a relative percentage change, EMG = (EMGc-EMGb) * 100%/EMGb, where EMGc is the response to CRD and EMGb is the baseline.
Pain threshold
The rats were anesthetized with isoflurane and placed in a 20 × 6 × 8 cm plexiglass box with a latex balloon inserted through the anus. Before testing, the rats were habituated to the environment for about 30 min. The abdominal contractions were observed during pressurization, with an increasing pressure of 5 mmHg each time. When rats exhibited prominent lower abdominal wall lifting from the bottom of the box, the minimum pressure value was used as the pain threshold (PT) at this time. The PT was repeatedly measured 3 times, and the average value was calculated.
Intrathecal catheter implantation and drug administration
The rats were anesthetized with barbitone (0.1 g/kg, i.p.), and a sterile polyethylene catheter (BB31695-PE/1, Scientific Commodities Inc, USA) was planted into the intervertebral space between the L6 and S1. Rats that developed neurological deficits after catheter implantation were euthanized. Besides, the rats were omitted from the experiment if hind paw paralysis did not occur after intrathecal lidocaine injection. 16 The Cat S selective inhibitor (LY3000328, MCE) and the p38 MAPK inhibitor (SB203580, MCE) were dissolved in 1% of DMSO, respectively. Various dosages (1, 3, and 10 μg) of LY3000328 were injected once daily for 3 days. SB203580 (10 μg) was injected once daily for 3 days. FKN (AF537) antibody and rabbit IgG (AB-105-C) were purchased from R&D Systems (Minneapolis, MN, USA). CX3CR1 antibody (TP501) was purchased from Torrey Pines Biolabs (Shanghai, China). Both FKN antibody and CX3CR1 antibody were injected once daily for 3 days. Intrathecal administration was conducted at a rate of 20 μL/min.
Open field test
A grey-walled box (100 × 100 × 60 cm) with a black floor and an open top was prepared in a quiet room. The rats were gently placed on the center floor of the box, and their movements were recorded and tracked using overhead camera for 5 min. The experiment was conducted in an environment with sound insulation, suitable temperature, humidity, and light intensity. After each trial, the box was carefully cleaned to remove any residue that could serve as a clue to the subsequent trial. Rats’ motion tracks were recorded by Yishu Vision tracking system (Shanghai, China). Total distance (cm), average speed (cm/s), central area total distance (cm), and central area total time (s) were analyzed.
Western blotting
Rats were anesthetized with isoflurane. T13-L2/L6-S2 segments of the spinal cord were immediately extracted and kept in liquid nitrogen until needed. Each protein sample (20 μg) was separated by electrophoresis and transferred to the PVDF membrane by electroblotting (Bio-Rad, Hercules, CA, USA). The membranes were incubated overnight at 4°C with primary antibodies (Cat S, 1:200, sc-271619, Santa Cruz, FKN, 1:1000, ab25088, Abcam, CX3CR1, 1:1000, ab8021, Abcam) and then for 2 h at room temperature with HRP-conjugated secondary antibodies (1:10,000, anti-rabbit or anti-mouse). The relative expression of the protein bands was measured from scanned films using GS-700 Imaging Densitometer (Bio-Rad Laboratories) and analyzed by using Image J software.
Immunofluorescence
The rats were perfused after deep anesthesia. The thoracolumbar segment (T13-L2) and lumbosacral segment (L6-S2) of the spinal cord were collected. The tissues were placed in a 4°C refrigerator, fixed with 4% paraformaldehyde for 2 h, and then placed in 20% sucrose overnight. The tissues were uniformly sectioned to a thickness of 20 μm. The tissues were soaked in immunostaining blocking solution and incubated at 37°C for 1 h. After adding the primary antibody (Cat S, 1:20; FKN, 1:100; CX3CR1, 1:100; IBA-1, 1: 10,000; Neuron, 1:1000), the slides were incubated at 4°C in the refrigerator for 48 h. After that, two diluted fluorescent secondary antibody mixtures were added and incubated for 2 h in the dark. Slices were observed with an SP8 confocal microscope (Leica TCS).
Immunohistochemistry
Tissue sections were treated with xylene and gradient alcohol. Following this, they were subjected to high pressure-induced antigen retrieval for 2 min at 125°C, and then rinsed 3 times with PBS (for 5 min each). Tissue sections were placed side-by-side in a wet box and incubated with 3% H2O2 solution for 10 min at 37°C. The primary antibody (CX3CR1, 1:400) was added, and incubated in a 4°C refrigerator overnight. Subsequently, reagents 1 and 2 (PV-9001, ZSGB-BIO) were added and incubated at room temperature for 20 min, respectively. Then DAB was used to visualize the staining. Hematoxylin was used to counterstain the sections for approximately 1 min before being washed in distilled water. The tissue sections were sealed with neutral resin after being treated with a gradient of ethanol and xylene.
Statistical analysis
All data experiments were expressed as mean ± SEM. Statistically significant differences were determined by Student’s t-tests or one-way analysis of variance (ANOVA). Image J was used to analyze immunostaining images and protein band intensity. Significance was determined at a level of p < .05. Statistical analysis was performed with SPSS 26.0.
Results
Spinal Cathepsin S of microglial cells was up-regulated in IBS-like rats
As shown in Figure 1(a), an IBS-like rat model was established by neonatal CRD, as our previous study.
17
Visceral sensitivity was assessed by recording the response of EMG to CRD and PT. EMG magnitudes were increased at 40 and 60 mmHg CRD (Figure 1(b)) and PT was decreased (Figure 1(c)) significantly in IBS-like rats, suggesting that stress stimulation in early life could lead to visceral hypersensitivity in adult rats. Cat S has been proposed to induce neuropathic pain through multiple mechanisms.
18
To determine whether Cat S was involved in the development of central sensitization of IBS-like rats, the expression of Cat S was detected in the spinal segments (T13–L2, L6-S2) by Western blot. The result demonstrated that Cat S was increased in the IBS-like rats (Figure 1(d) and (e)). Furthermore, Cat S can be released from microglia.
8
Thus, we speculated spinal Cat S of microglial cells might be up-regulated in IBS-like rats, which was verified by immunofluorescence experiments (Figure 1(f)–(h)). Spinal Cat S of microglial cells was upregulated in IBS-like rats. (a) Experimental design. (b) The statistical chart of EMG amplitude to 40 and 60 mmHg CRD in control and IBS-like rats. N = 8. (c) The pain threshold was markedly decreased in IBS-like rats. N = 8. (d, e) The expression of Cat S in spinal cord in control and IBS-like rats. N = 6. (f) Representative images with immunohistochemical staining of DAPI (blue), Iba-1 (geen), Cat S (red), and merged images for colocalization of Cat S and Iba-1 in spinal dorsal horn of control and IBS-like rats. Scale bar: 100 μm. (g, h) The number of IBA-1+ cells (g) and the number of IBA-1+ and Cat S+ puncta (h) in spinal dorsal horn of control and IBS-like rats. N = 3. Ctrl: Control; IBS: irritable bowel syndrome; Cat S: Cathepsin S; TL: thoracolumbar; LS: lumbosacral; CRD: colorectal distension; EMG: electromyography. **p < .01, ***p < .001.
Spinal treatment of a Cathepsin S inhibitor LY3000328 attenuated visceral hypersensitivity in IBS-like rats
Since the expression of Cat S was up-regulated in IBS-like rats, we also sought to investigate whether spinal treatment of a Cat S inhibitor LY3000328 could affect visceral hypersensitivity. The procedure of drug administration was shown in Figure 2(a) and LY3000328 was administered at doses of 1, 3 and 10 μg, respectively. EMG response to CRD was inhibited by 3 μg and 10 μg LY3000328 significantly but not 1 μg in IBS-like rats, indicating a dose-dependent inhibitory action (Figure 2(b)–(d)). However, LY3000328 treatment did not affect EMG response even at 10 μg in control rats (Figure 2(e)). Additionally, we tested the time effect of 10 μg LY3000328 and found that the inhibitory effect was significant at the 10th min and maintained for at least 90 min after injection in IBS-like rats (Figure 2(f) and (g)). The spinal cord is closely related to locomotor activity.
19
Additionally, Our previous study reported that neonatal CRD resulted in visceral hypersensitivity and anxiety-like behaviors in adult rats.
20
To verify whether the effect of LY3000328 on visceral pain was specific, the open-field test was performed to detect locomotor activity and anxiety-like behaviors before and after the administration of LY3000328 in IBS-like rats. Data analysis showed that LY3000328 did not affect the total distance, the average speed, the central area total distance and the central area total time of IBS-like rats (Figure 2(h)–(k)), suggesting the effect of LY3000328 on visceral pain was specific in IBS-like rats. LY3000328, a selective Cat S inhibitor, attenuated visceral hypersensitivity but did not affect locomtor activity and anxiety-like behaviors in IBS-like rats. (a) Timetable for the drug administration. (b–e) The EMG at the pressure of 40 and 60 mmHg CRD before and after administration of 1, 3 and 10 μg LY3000328 in rats, respectively. N = 6. (f, g) The time course of EMG at 40 and 60 mmHg CRD following treatment with 10 μg LY3000328 in control and IBS-like rats. N = 6. (h–k) The total distance, the average speed, the central area total distance and the central area total time of IBS-like rats in OFT before and after administration of LY3000328. N = 9. Ctrl: Control; IBS: irritable bowel syndrome; CRD: colorectal distension; EMG: electromyography; LY: LY3000328; OFT: open field test; Pre: prior to administration; Post: with LY. *p < .05, **p < .01.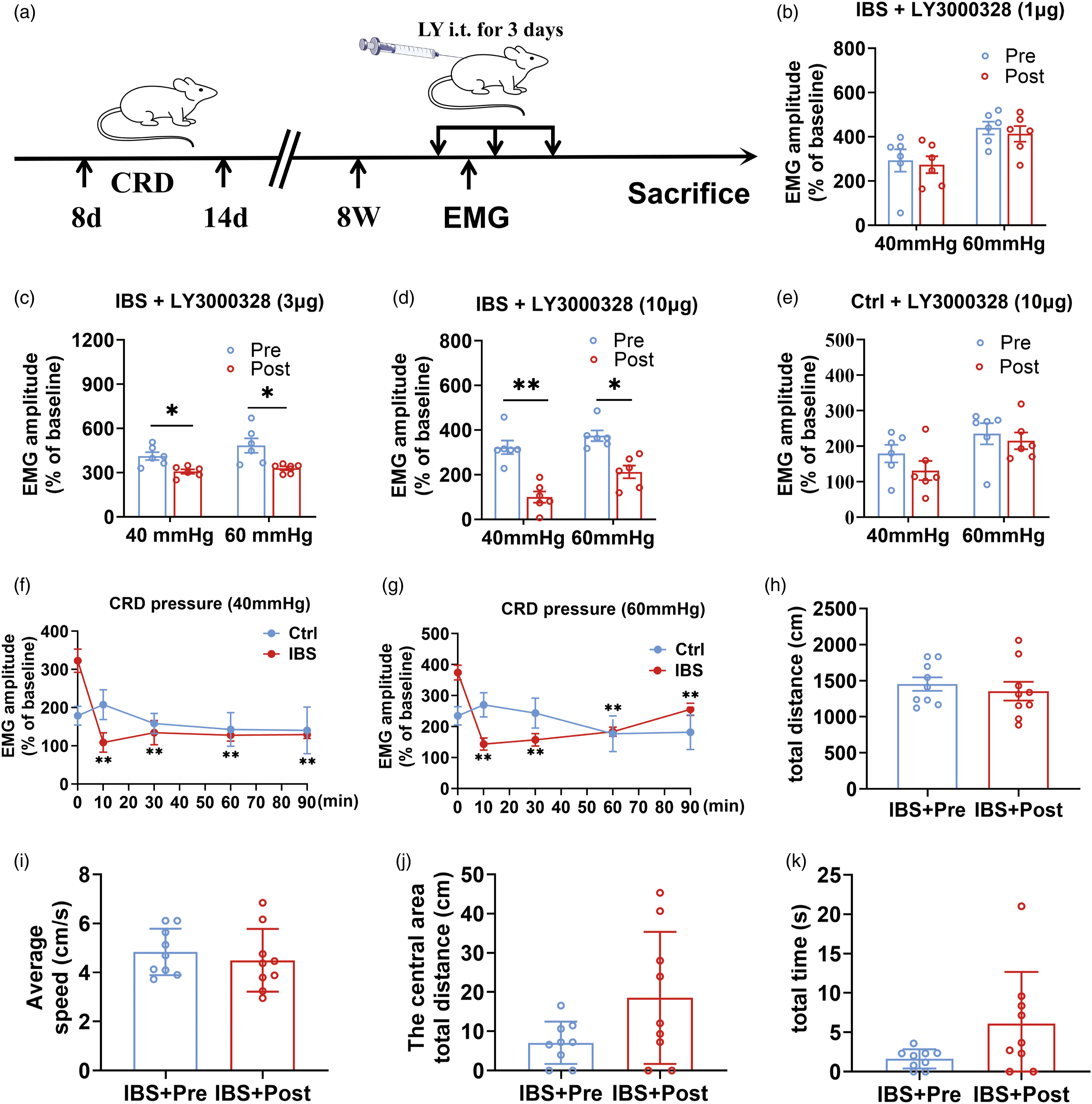
Microglial Cathepsin S regulated visceral pain via neuronal FKN in IBS-like rats
The study of neuropathic pain reported that Cat S was critical for the maintenance of pain via cleavage of FKN.
21
We, therefore, set out to search for the role of FKN and the relationship of Cat S with FKN in IBS-like rats. Immunofluorescence imagines showed FKN fluorescence intensity was enhanced significantly in the spinal dorsal horn of IBS-like rats, and FKN was colocalized with neurons (Figure 3(a) and (b)). Then, we examined whether spinal FKN expression was affected by the intrathecal administration of LY3000328 to inhibit Cat S in IBS-like rats. Western blot results showed the treatment of both 3 μg and 10 μg LY3000328 down-regulated the expression of spinal FKN in IBS-like rats (Figure 3(c)–(f)). Furthermore, FKN expression was elevated in spinal dorsal horn of IBS-like rats (Figure 3(g) and (h)), which was consistent with the immunofluorescence result. Moreover, we assessed the possible effect of an FKN-neutralizing antibody on EMG in IBS-like rats. Intrathecal administration of an FKN-neutralizing antibody could relieve visceral nociception sensitivity in IBS-like rats (Figure 3(i) and (j)). These results reveal the critical role of microglial Cat S in the pathogenesis of chronic visceral pain via neuronal FKN. Microglial Cat S regulated visceral pain via neuronal FKN in IBS-like rats. (a, b) Representative images of neurons (red) and FKN (green) coexpression in spinal dorsal horn (a) and coexpression puncta (b) of control and IBS-like rats. The solid line box indicates the observation area of the spinal dorsal horn under low magnification. Scale bar, 100 μm. N = 3. (c–f) The expression of FKN declined in the spinal cord of IBS-like rats after treatment with 3 or 10 μg LY3000328. N = 6. (g, h) The expression of FKN in spinal cord in control and IBS-like rats. N = 6. (i, j) The EMG at 40 and 60 mmHg CRD before and after administration of 1 μg IgG (i) or 1 μg FKN Ab (j) in IBS-like rats. N = 6. Ctrl: Control; IBS: irritable bowel syndrome; CRD: colorectal distension; EMG: electromyography; TL: thoracolumbar, LS: lumbosacral; Pre: prior to administration; Post: with FKN -neutralizing antibody. *p < .05, **p < .01, ***p < .001.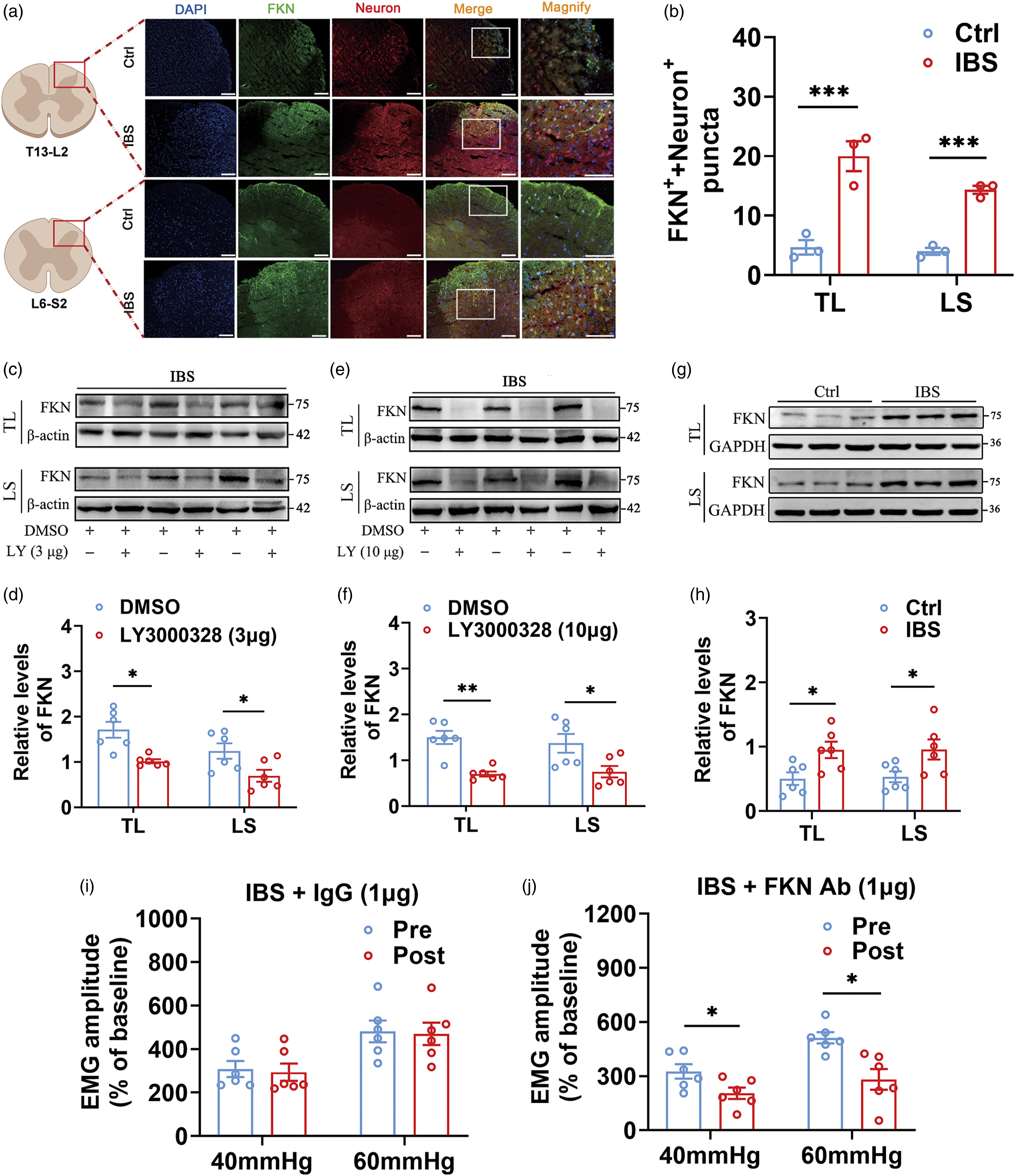
Activation of CX3CR1 by FKN enhanced the activity of p38 MAPK in IBS-like rats
CX3CR1 is a unique receptor for FKN and is mainly expressed in microglial cells.
22
Since spinal FKN plays a vital role in IBS-like rats, the question naturally arises as to whether CX3CR1 expression is dysregulated in IBS-like rats. Immunohistochemical staining verified that the expression of CX3CR1 was up-regulated in IBS-like rats (Figure 4(a) and (b)), which was further demonstrated by Western blot results (Figure 4(c)). Interestingly, intrathecally injection of FKN-neutralizing antibody down-regulated CX3CR1 expression only in IBS-like rats but not in controls (Figure 4(d) and (e)). On this basis, we further blocked CX3CR1 function in IBS-like rats using different doses of CX3CR1-neutralizing antibody (3 μg, 6 μg, and 12 μg) intrathecally. We observed the changes in EMG responses before and after administration. The results showed that the magnitude of EMG was significantly reduced by 6 μg and 12 μg CX3CR1-neutralizing antibody but not by 3 μg CX3CR1-neutralizing antibody or 12 μg IgG (Figure 4(f)–(i)). The blocking effect of CX3CR1-neutralizing antibody to EMG was dose-dependent in IBS-like rats (Figure 4(j)). Our previous study has shown that phosphorylated p38 MAPK is involved in the central sensitization of chronic visceral pain.
14
We wondered whether there was a possible link between CX3CR1 and p38 MAPK in IBS-like rats. To this end, we examined the effect of CX3CR1-neutralizing antibody on the phosphorylation level of p38 MAPK in the spinal cord of IBS-like rats. The results suggested that CX3CR1-neutralizing antibody could down-regulated the phosphorylation level of p38 MAPK (Figure 4(k) and (l)), providing support that CX3CR1 activation might enhance p38 MAPK activity in IBS-like rats. Activation of CX3CR1 by FKN enhanced the activity of p38 MAPK in IBS-like rats. (a, b) CX3CR1 was highly up-regulated in IBS-like rats by immunohistochemistry. Scare bar, 200 μm. N = 3. (c) The expression of CX3CR1 in the spinal cord of control and IBS-like rats. N = 6. (d, e) The expression of CX3CR1 in spinal cord of control (D) or IBS-like rats (e) after treatment with FKN Ab compared with IgG. N = 3. (f–i) The EMG at 40 and 60 mmHg CRD in the IBS-like rats before and after administration of 12 μg IgG, 3 μg, 6 μg and 12 μg CX3CR1 Ab, respectively. N = 6. (j) The inhibitory rate of EMG to CRD after treatment with 3, 6 and 12 μg CX3CR1 Ab, respectively. N = 5. (k, l) The expression of p-p38 MAPK in the spinal cord after treatment with CX3CR1 Ab compared with IgG in IBS-like rats. N = 3. Ctrl: Control; IBS: irritable bowel syndrome; CRD: colorectal distension; EMG: electromyography; TL: thoracolumbar; LS: lumbosacral; Pre: prior to administration; Post: with CX3CR1-neutralizing antibody or IgG; Ab:antibody. *p < .05, **p < .01, ***p < .001.
Enhanced activity of p38 MAPK up-regulated Cathepsin S expression in IBS-like rats
Given the link between CX3CR1 and p38 MAPK, it is possible that inhibiting the activity of p38 MAPK would ameliorate pain hypersensitivity in IBS-like rats. Consequently, 10 μg SB203580 (a selective p38 MAPK inhibitor) attenuated EMG responses to 40 and 60 mmHg CRD in IBS-like rats but not in controls, and the inhibitory effect was significant at the 10th min and sustained for at least 60 min after intrathecal injection in IBS-like rats (Figure 5(a)–(d)). Up to this point, our results implied that spinal Cat S of microglial cells was up-regulated and that activation of microglial CX3CR1 by FKN enhanced p38 MAPK activity in IBS-like rats. Along these lines, we reasoned that enhanced phosphorylation level of p38 MAPK might modulate the expression of microglial Cat S in IBS-like rats. With regards to this, the effect of 10 μg SB203580 on Cat S expression was examined in IBS-like rats. Western blot showed inhibiting p38 MAPK activity by intrathecal use of SB203580 down-regulated Cat S expression in IBS-like rats (Figure 5(e)–(h)). Altogether, our data support that microglial Cat S-mediated FKN/CX3CR1/p38 MAPK signaling amplifies visceral sensitivity in IBS-like rats (Figure 5(i)). Enhanced activity of p38 MAPK up-regulated Cat S expression in IBS-like rats. (a, b) The time course of EMG at 40 and 60 mmHg CRD following treatment with 10 μg SB203580 in rats, respectively. N = 6. (c, d) The EMG at 40 and 60 mmHg CRD in IBS-like rats before and after administration of DMSO or 10 μg SB203580, respectively (once a day for three consecutive days). N = 6. (e–h) The expression of Cat S in spinal cord of IBS-like rats before and after administration of DMSO or 10 μg SB203580, respectively. N = 6. (i) Spinal Cat S acts as a medium for microglial communication, promoting visceral hypersensitivity through regulation of the FKN/CX3CR1/p38 MAPK pathway. Left: representation of IBS state. Right: inhibitors relieved visceral hypersensitivity in IBS rats. Figure created using BioRender.com. Pre: prior to administration; Post: with SB203580 or DMSO; SB: SB203580. **p < .01.
Discussion
In the current study, we sought to explore the role of spinal microglial Cat S in regulating visceral pain hypersensitivity in IBS-like rats. Our results showed that Cat S coexisted with microglia, and the expressions of Cat S and microglia were increased in IBS-like rats. Inhibiting the activity of spinal Cat S ameliorated visceral hypersensitivity and down-regulated FKN expression without affecting the locomotor activity and anxiety-like behaviors. Furthermore, The enhancement of neuronal FKN participated in the process of visceral hypersensitivity by activating microglial CX3CR1 and then facilitating the phosphorylation of p38 MAPK in IBS-like rats. The signaling pathway of microglial Cat S was also verified by behavioral experiments that inhibition of FKN, CX3CR1 or p38 MAPK could alleviate visceral pain in IBS-like rats.
The roles of microglia in chronic pain have come into focus recently. In neuropathic pain, microglia have been found to mediate pain processing.23,24 “Glial cell activation” is becoming an important mechanism in the pathogenesis of chronic pain. 25 However, the molecules by which microglia regulate chronic pain are controversial. Cat S, a proteolytic enzyme, distributed in dendritic cells, macrophages, and microglia, 7 can be released from the intracellular compartments. 26 It has been implicated in inflammatory processes and pain, which cleaves extracellular matrix proteins and is involved in antigen processing and presentation. Literature has reported that spinal Cat S from microglia is involved in acute and chronic neuralgia. 27 However, the role of spinal Cat S in IBS remains unknown. IBS-like rats were induced by neonatal CRD as our previous study. 28 Though we are aware that chronic functional visceral pain is different from chronic neuralgia, we wondered whether spinal Cat S was involved in visceral hypersensitivity of IBS. To answer this question, a battery of experiments has been done in this study. Immunofluorescence and Western blot analyses were conducted, which provided compelling evidence to suggest that spinal Cat S expressed by microglia was elevated in IBS-like rats. Furthermore, visceral sensitivity was measured in IBS-like rats after the administration of LY3000328 (a selective inhibitor of Cat S). The results indicated that LY3000328 had a dose-dependent effect on visceral sensitivity, with the most significant response observed at a dosage of 10 μg. Interestingly, the effects of LY3000328 on visceral sensitivity appeared to be acute, with inhibition being noticeable at the 10th minute after injection and persisting for at least 90 min. To obtain stable results regarding protein expression, protein levels were measured 3 days after injection. Surprisingly, we found that LY3000328 ameliorated visceral hypersensitivity by inhibiting the full-length form of FKN, rather than the cleaved form in the spinal cord, which might be due to its effects on Cat S to decrease FKN synthesis.
Recent evidence suggested that LY3000328 could alleviate myocardial ischemia/reperfusion (I/R) injury by suppressing inflammation and apoptosis. 29 Furthermore, LY3000328 has been reported to reduce nociception in cancer mice generated with two human tongue oral cancer cell lines. 30 Despite the promising initial results of Cat S inhibitors in treating aortic aneurysm and arthritis, their use for chronic visceral pain is currently limited.31,32 In our study, intrathecal injection of LY3000328 has been shown to reduce visceral sensitivity in animal models of IBS. This rapid onset of inhibition may be due to the enzyme’s proteolytic activity being inhibited by the Cat S inhibitor. Furthermore, LY3000328 may decrease the release of pain mediators from immune cells or reduce the activation of microglia in the spinal cord. These findings suggest that LY3000328 may be a valuable alternative for treating chronic IBS. Interestingly, intrathecal treatment of LY3000328 did not affect anxiety-like behaviors of IBS-like rats, which might point toward two potentially different mechanisms of visceral hypersensitivity and anxiety of IBS.
In the context of neuropathic pain, Cat S acts as a soluble active molecule to activate the p38 MAPK pathway by cleaving FKN located on neurons, which binds to the receptor CX3CR1 in microglia.33,34 Though the pathway by which spinal microglia-derived Cat S regulates pain hypersensitivity seems to be identified in neuropathic pain, the molecular mechanism of Cat S in chronic visceral pain has not been studied. In follow-up experiments, we set out to search for the underlying mechanism of spinal Cat S regulating visceral hypersensitivity of IBS. We utilized immunofluorescence images and Western blot analysis to investigate the expression of spinal FKN, CX3CR1 and p-p38 MAPK in IBS-like rats. Our findings revealed an increase in the expression levels of these molecules in IBS-like rats. Additionally, we conducted behavioral experiments and found that inhibiting FKN, CX3CR1 and p38 MAPK, respectively, could alleviate visceral pain in IBS-like rats. Most importantly, we uncovered the interrelationship of upstream and downstream among spinal Cat S/FKN/CX3CR1/p38 MAPK/Cat S using receptor inhibitors and neutralizing antibodies, combined with Western blot. We obsedved that this signaling pathway forms a closed loop, which might result in positive feedback and magnify visceral pain in IBS (Figure 5(i)). These results have been supported by various studies demonstrating the role of microglia-derived Cat S in promoting FKN cleavage from neuronal membranes to regulate neuropathic pain in the spinal cord of rats. 9 Futhermore, p38 MAPK, a serine/threonine protein kinase, is activated by various stimuli, including cytokines, growth factors, and stress. And it is involved in the regulation of various cellular processes, including inflammation and pain. 35 The phosphorylation of p38 MAPK by different inflammatory factors and stress stimulations activates it to participate in pain development.36,37 Taken together, these findings highlight the importance of spinal Cat S signaling for the development of pain hypersensitivity.
In conclusion, neonatal CRD might induce release of spinal microglia derived Cat S, then through activating the signaling pathway of FKN/CX3CR1/p38 MAPK/Cat S to magnify visceral pain in IBS-like rats. As a selective inhibitor of Cat S, LY3000328 could become a valuable alternative to treat IBS.
Footnotes
Author Contributions
Conception and design: P.S., W.L. and J.G.; Development of methodology: Y.-X.W. and Y.H.; Data curation: Y.T.; Writing, review and/or revision of the manuscript: P.S., W.L. Y.C., A.-Q.C. and C.L.; Study supervision: C.L.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the National Natural Science Foundation of China (81471138), Natural Science Foundation of Fujian Province (2021J01675), Fujian Science and Technology Innovation Joint Fund (2018Y9069).
Data availability
All data are available in the main text or supplementary materials.
