Abstract

Introduction
Irritable bowel syndrome (IBS), known as a functional gastrointestinal disorder, is currently one of the most common gut-brain interaction disorders, with an estimated incidence of more than 10% worldwide. 1 Visceral hypersensitivity, a typical feature of IBS, is observed in up to 60% of patients. 2 The high incidence of visceral hypersensitivity in IBS and its impact on people’s quality of life present a critical unmet need to develop an improved understanding and new clinical treatment.
Chronic exposure to adverse life events especially in the initial stages increases personal susceptibility to IBS disorders.3,4 Accumulating evidence shows that the visceral hypersensitivity in IBS is regulated by multiple epigenetics,5–8 including DNA methylation, histone modifications (acetylation and methylation) and noncoding RNA regulation. N6-methyladenosine (m6A) is the most abundant form of internal RNA modifications, 9 which is installed by m6A methyltransferases, removed by demethylases, and recognized by m6A-binding proteins. 10 Methyltransferase-like 3 (METTL3) is identified as a key catalytic component of m6A “writer” complex and plays critical roles in neuronal development and brain function in mammals. 11 It impacts almost all stages of RNA biogenesis, including RNA transcription, splicing, export, translation, and degradation.12–14 However, how METTL3 is involved in IBS progression remains elusive.
Recent studies demonstrated that dysregulation of circular RNA (circRNAs) was correlated with chronic pain.15,16 In our previous study, the expression of hippocampal circKcnk9, a novel circRNA, was identified that being significantly increased in IBS-like rats. 17 Up to now, the potential mechanisms of hippocampal METTL3 and circKcnk9 in the progression of IBS are still unknown.
In the current work, we found that neonatal colorectal colorectal distention (CRD) leaded to a significant increase in METTL3, but not in METTL14, WTAP, ALKBH5, and FTO in the hippocampus. This increase contributed to CRD-induced visceral hypersensitivity at least in part through writing the m6A in circKcnk9 and elevating the nuclear export of circKcnk9 in the hippocampus in YTHDC1-dependent manner. Herein, we show the new regulatory model of “m6A-circKcnk9” as an important marker and therapeutic target in IBS and provide mechanistic insight for exploring the mechanism of m6A-circKcnK9 in early life stress.
Materials and methods
Animals
Neonatal male Sprague-Dawley rats were obtained from the Department of Experimental Animal Center of Fujian Medical University. The animals were maintained in a specific pathogen-free-grade environment at the animal center. Weaning was typically initiated 21 days after birth. The experiments were approved by the Animal Care and Use Committee of Fujian Medical University. IBS-like rats were established by 60 mmHg CRD stimulation once daily for 1 min during postnatal days 7–14. 18 We examined the electromyographic (EMG) magnitude in response to graded strengths of CRD pressures in control and IBS-like rats at 6–8 weeks to assess visceral sensitivity.
Stereotactic infusion
The rats were anesthetized with isoflurane (2%) and fixed on a stereotactic instrument (Ruiwode Life Science, China). Following routine skin sterilization, a midline scalp incision was made, and local anesthesia was administered with lidocaine. The skull was exposed and cleaned by scraping with 10% hydrogen peroxide. All surgical procedures were performed under aseptic conditions and no infection was detected.
Adeno-associated virus, Lentivirus vectors, siRNA, and delivery route
Lentivirus and AAV (shcircKcnk9 and aavcicKcnk9) vector of production and verification came from BrainVTA (Wuhan, China) were performed as described. 17 AAV-METTL3 came from GeneChem (Shanghai, China), siYTHDC1 were designed and commercially synthesized (CRN2901, Cohesion Bioscience, London, Britain) (Figure S1). siMETTL3 was constructed by GenePharma (Shanghai, China). The sequences of related siRNAs vectors are shown in Table S1. The coordinates of the injection locations were centered at 4.0 mm in anteroposterior plane, 2.5 mm in mediolateral plane, and 2.8 mm in the dorsalventral plane. The rats underwent stereotaxic surgery for double cannula (inner radium 2.8 mm, inner diameter: 0.3 mm, out diameter: 0.48 mm, Ruiwode Life Science, China) implantation and were allowed to rest for at least 7 days.
The control rats received intrahippocampal injection of AAV-METTL3 (1 μL) were allowed to rest for 21 days postoperatively (the timeline of this process is correlated with that of viral production). The IBS-like rats received an intrahippocampal injection of siRNAs respectively and were allowed to rest for 3 days postoperatively.
Cell culture and transfection
PC12 cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM, Gibco/ThermoFisher Scientific, Waltham, MA) containing 5% fetal bovine serum (FBS; Gibco/ThermoFisher Scientific) at 37°C in a humidified incubator with 5% CO2. The plasmids (GenePharma, Shanghai, China, 1 μg/uL) and siRNAs (at a concentration of 50 μM oligonucleotides) were transfected into the PC12 cells with Lipofectamine 3000 (Invitrogen, USA) according to the manufacturer’s protocol.
Electromyography
Electromyography was performed to assess visceral hypersensitivity. Rats (6–8 weeks old) were anesthetized with isoflurane. Prior to the CRD procedure, a glycerol-lubricated balloon was inserted into the rectum. A pair of bipolar electrodes was implanted in the abdominal external oblique musculature of shallow anesthetized rats to detect EMG activity. Under isoflurane superficial anesthesia (2%, 2 L/min), the discharge of the rat ventral oblique muscle was recorded at CRD pressures of 40- and 60- mmHg. The EMG responses to different degrees of CRD were recorded using the RM6240BD system (Chengdu, China). Data were analyzed by averaging the baseline amplitudes. Values over the baseline were used to assess visceral hypersensitivity. 19
Immunofluorescence
Rats were deeply anesthetized with sodium pentobarbital (200 mg/kg, i.p.) and transcardially perfused with 500 mL ice-cold 0.9% NaCl, followed by 750 mL 4% paraformaldehyde. Brains were removed from the skull and placed in 4% paraformaldehyde overnight. After gradient dehydration with 20% and 30% sucrose (Sigma, USA), tissue was embedded with optimal cutting temperature (OCT) compound and sectioned using a cryotome (Leica, Germany).
The sections (20–40 μm) were removed from the −20°C refrigerator for half an hour before use. Phosphate buffer solution (PBS) was used to wash out the OCT. Pap pen was used to circle the tissue. The sections were blocked with immunostaining blocking buffer solution containing 5% goat serum and 0.3% TritonTMX-100 at room temperature for 2 h (or 37°C for 30 min) and then incubated with primary antibodies at 4°C for 24–48 h. Sections were incubated with secondary antibodies at room temperature for 2 h before washing with PBS (3 times/15 min). Immunofluorescence analysis of frozen sections was performed using the primary antibodies described in Table S2.
RNA extraction and quantitative real-time PCR
According to the instructions of the manufactures, total RNAs were extracted using TRIzol reagent (Invitrogen, USA). An Evo-M-MLV reverse transcription kit (Accurate Biology, China) was used to perform reverse transcription of circRNA and mRNA. After reverse transcription, qRT-PCR was performed with a SYBR Green method (Biosune Biotech, Shanghai). Primer sequences were listed in Table S3.
Protein extraction and western blot
Proteins from the hippocampus of rats were extracted using radioimmunoprecipitation assay (RIPA) and protease inhibitor (PMSF), separated by 8% SDS-PAGE, and electro-transferred onto PVDF (Invitrogen, USA) membranes, which were probed with rabbit anti-YTHDC1 (ab122340, 1:1,000, Abcam, USA) and rabbit anti-METTL3 (ab195352, 1:1000, Abcam, USA), and mouse anti-β-actin primary antibody (82,261:1000, Abcam, USA). Horseradish-peroxidase conjugated secondary antibody (1:10,000) was used to incubate the membrane for 8 h. Protein bands were detected using an enhanced chemiluminescence kit (WBKLS0500, Immobilon, Millipore). Furthermore, protein expression level was detected using ImageJ (https://rsb.info.nih.gov/ij/).
RNA fluorescence in situ hybridization
Rat brain slices were prepared as described previously. RNA localization and quantification were determined using a fluorescent in situ hybridization (FISH) kit (GenePharma, Shanghai, China) and an RNA probe from Exiqon (Exiqon Life Sciences, Denmark), according to the manufacturer’s protocol. For combined RNA FISH and immunostaining, we first performed RNA FISH, followed by immunofluorescence. Diethyl pyrocarbonate-treated water was used in all the steps to rule out RNA degradation.
Immunofluorescence and FISH images were captured using NikonE200 microscopes equipped with 405, 488, and 594 lasers. Colocalization between two channels was analyzed using the Fiji plugin Colocalization Threshold. 20
RNA immunoprecipitation assay
The homogenates from rat hippocampus were suspended in the RIP lysis buffer containing protease inhibitor cocktail and RNase inhibitor. The BersinBio RIP Kit or the BersinBio MeRIP m6A Kit (BersinBio Biotech, Guangzhou, China) were used to capture the antigen after the magnetic beads were connected to the m6A antibody or IgG antibody. Following this, RNAs were then extracted, and PCR was employed for verification.
RNA pull down assay
RNA pull-down analysis was conducted using the Magnetic RNA-Protein Pull-Down kit (BersinBio Biotech., Guangzhou, China) in accordance with the manufacturer’s protocol. Briefly, tissue was lysed using IP Lysis Buffer for 10 min before ultrasonication and then centrifuged at 13,000×g for 30 min at 4°C to obtain lysates. Hippocampal lysates were co-incubated with nucleic acid-compatible streptavidin magnetic beads combined with biotinylated sense and probe circKcnk9 (BersinBio Biotech., Guangzhou, Guangdong, China). Proteins of the RNA-protein complexes were eluted from the magnetic beads, and YTHDC1 protein expression was examined as aforementioned via Western blotting.
Bioinformatic analysis
Secondary structure modeling was performed using an online tool at the RNA fold web server (https://rna.tbi.univie.ac.at/cgi-bin/RNAWebSuite/RNAfold.cgi). RBPDB (https://rbpdb.ccbr.utoronto.ca/) was used to predict the binding sites of circKcnk9. The m6A modification was predicted by SRAMP (https://www.cuilab.cn/sramp/).
Statistical analysis
All experiments were randomized and blinded. All data are presented as mean ± standard error of the mean. The Student’s t-test (two groups) or one-way ANOVA (more than two groups) was used for the immunoblot, qPCR, and c-Fos immunolabeling data. Besides, one-way ANOVA analyses followed by Dunnett test was performed when appropriate. The EMG results were analyzed using a two-way ANOVA. p-value of < 0.05 was considered statistically significant. GraphPad Prism 8.0 was used for plotting. ImageJ was used to analyze immunostaining images and protein band intensity.
Results
Methyltransferase-like 3, along with m6A methylation, is upregulated in IBS-like rats undergoing neonatal colorectal distension
The IBS-like model was established using neonatal CRD to mimic IBS in clinical cases.
18
Visceral sensitivity was assessed by recording the response of EMG to CRD. The EMG amplitudes at 40 and 60 mmHg both significantly increased in IBS-like rats compared to those in controls, indicating that violent visceral hyperalgesia developed after neonatal CRD (Figure 1(a) and (b)). To investigate whether m6A modification participates in early life stress modulation, we first examined the total m6A level in hippocampal tissue in a neonatal CRD-induced visceral pain model. As shown, the immunofluorescence experiments revealed that the total m6A were significantly increased in hippocampus after CRD treatment (Figure 1(c) and (d)). Since m6A methyltransferases and demethylases are closely associated with m6A methylation,
21
we evaluated METTL3, METTL14 WTAP, FTO and ALKBH5 enrichment in the hippocampus and revelated that METTL3 level was dramatically increased after neonatal CRD (Figure 1(e) and (f)), suggesting a trend consistent with changes in hippocampal m6A. Meanwhile, we found that METTL3 mainly coexisted with NeuN (a specific neuronal marker) and very few METTL3-detectable cells were co-located with glial fibrillary acidic protein (GFAP) (a specific astrocyte marker) or IBA1 (a specific microglial marker), suggesting that METTL3 was predominantly expressed in neurons of the hippocampus (Figure 1(g)). Collectively, our data indicate that METTL3, along with m6A is involved in the development of the visceral hypersensitivity induced by neonatal CRD. Neonatal colorectal distension (CRD)-induced visceral hypersensitivity increases m6A level and METTL3 expression in hippocampus. (a, b) The original graph and statistical chart of EMG amplitude in control and IBS-like rats. N = 9, two-way ANOVA, F = 111.2, ***p < 0.001. (c, d) Immunofluorescence was employed to detect the expression of m6A+ cells. N = 6, two-tailed Student t-test, t = 3.589, *p < 0.05. Scale bar, 250 μm. (e) The relative expression of m6A modification-associated mRNAs in IBS or control rats were evaluated using qRT-PCR. N = 6 per group, *p < 0.05 (two-tailed Student t-test, t = 2.223). (f) The relative expression of METTL3 protein in hippocampus after CRD was evaluated using Western blotting. N = 3, two-tailed Student t-test, t = 2.893,
*
p < 0.05. (g) Double-fluorescent labeling experiment was performed with the respective antibodies to verify the tissue location of METTL3. The indications of green markers were as follows: NeuN indicates neuron, lba1 indicates microglia and GFAP indicates astrocyte. Scale bar, 250 μm. EMG, electromyography; METTL3, methyltransferase-like 3.
Manipulation of METTL3 in the hippocampal neurons regulates visceral hypersensitivity and neuronal activity
Given that METTL3 was highly expressed in IBS-like rats induced by neonatal CRD, we explored whether modulation of METTL3 could affect chronic visceral pain. To circumvent siRNA-mediated off-target effects,
22
PC12 cells were used to screen siMETTL3 locus. The PCR results showed that the siMETTL3A (1311) site (-5′ GGCUCAAUAUACCAGUACUTT 3′-) could most effectively inhibit the mRNA expression of METTL3 (Figure 2(a)). Western blot analysis demonstrated that siMETTL3A site markedly downregulated endogenous METTL3 protein expression in PC12 cell lines (Figure S2). Besides, we silenced METTL3 expression by microinjecting its corresponding siMETTL3A into the hippocampal region (CA1 A/P: 4.0 mm, M/L: 2.5 mm, D/V:2.8 mm) and found that METTL3 knockdown (Figure 2(b)) could alleviate visceral hypersensitivity in IBS-like rats (Figure 2(c) and (d)). Knockdown of METTL3 alleviates visceral hypersensitivity in IBS-like rats (a) Validation of siRNA knockdown for METTL3 in PC12 cells. N = 3 per group, one-way ANOVA followed by Dunnett’s test, *p < 0.05, **p < 0.01, ***p < 0.001 vs. siNC. (b) The relative expression of METTL3 protein in IBS-like rats with siMETTL3 was evaluated using Western blotting. N = 3, two-tailed Student t-test, t = 4.416, *p < 0.01. The original graph (c) and statistical chart (d) of EMG at 40 and 60 mmHg CRD in IBS-like rats after microinjected with siMETTL3. N = 9, two-way ANOVA, F = 57.23, ***p < 0.001. CRD, colorectal distension; EMG, electromyography; METTL3, methyltransferase-like 3.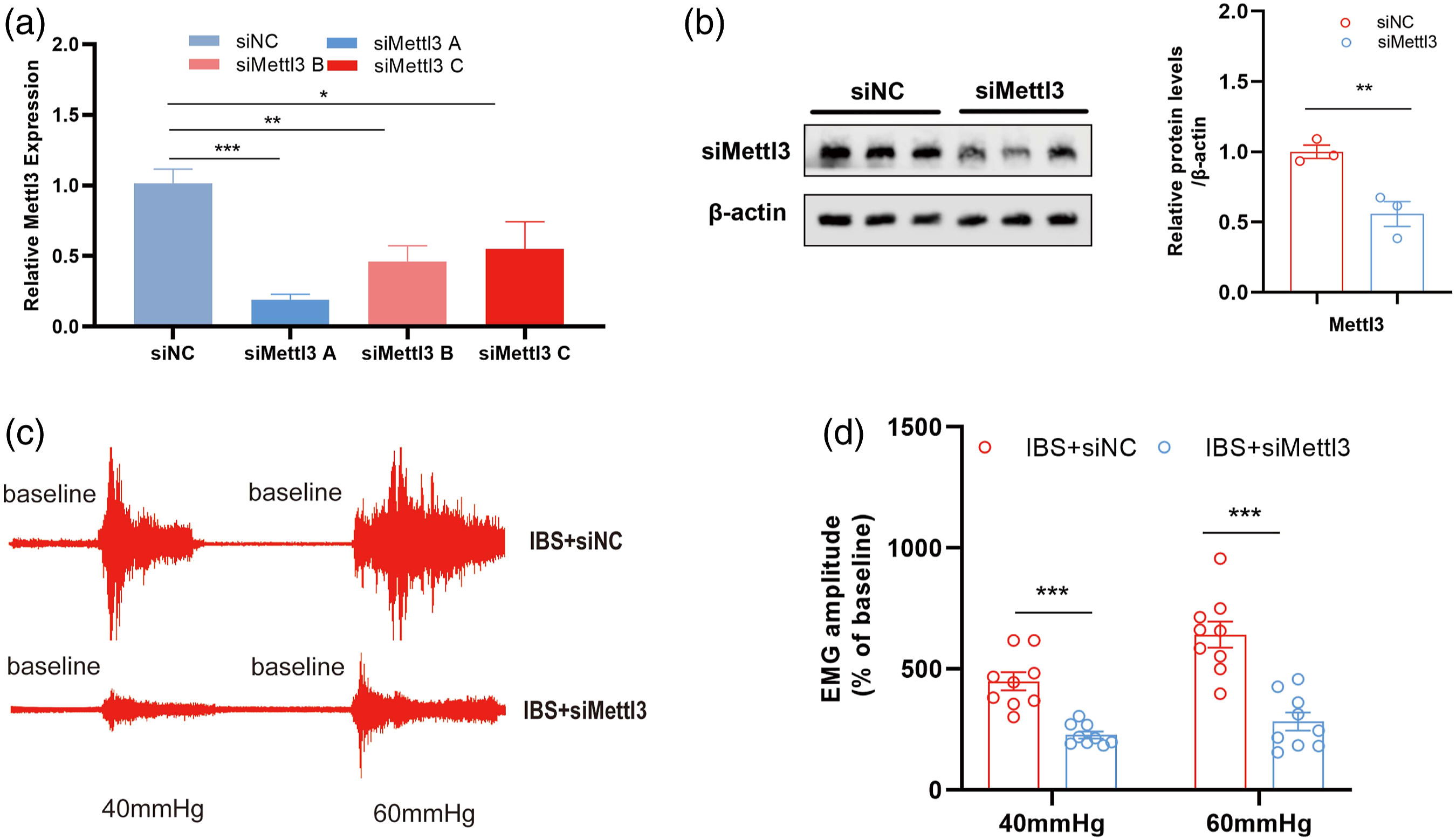
On the other hand, adeno-associated virus (AAV) was microinjected into bilateral CA1 of control rats to overexpress METTL3. Immunofluorescence results showed that the overexpressed METTL3 was mainly distributed along the pyramidal cell layer of hippocampus (Figure 3(a)), and it was significantly co-labeled with m6A (Figure 3(b) and Figure S3). Besides, AAV-METTL3 also could activate the neurons in pyramidal cell layer of hippocampus (Figure 3(c)). Our data indicate that the increased METTL3 may be responsible for an enrichment of m6A sites in pyramidal of neurons. As expected, a marked increase in the level of METTL3 mRNA (Figure 3(d)) and protein (Figure 3(e)) were detected 21 days after stereotactic injection of CA1 compared to AAV-NC treated groups. And compared with the group microinjected with empty-loaded virus, the EMG results showed that the rats in AAV-METTLT3 group had higher visceral sensitivity (Figure 3(f)). Collectively, these results proved our hypothesis that METTL3 was essential for the modulation of hippocampal m6A in IBS-like rats induced by neonatal CRD, and m6A methylation might be critical for the formation of visceral hypersensitivity. Overexpression of METTL3 induces visceral hypersensitivity and activates hippocampal neurons in control rats. (a) Representative immunofluorescence traces of AAV-METTL3 microinjection in hippocampal CA1. Scale bar, 500 μm. (b) Colocalization of METTL3 and m6A in the rat hippocampus. Scale bar, 500 μm. (c) Immunofluorescence was employed to detect the expression of c-fos in control rats with AAV-NC or AAV-METTL3. The METTL3 RNA (d, N = 4, t = 6.68, ***p < 0.001) and protein (e, N = 3, t = 4.385, *p < 0.05) expression increased in control rats after microinjecting with AAV-NC or AAV-METTL3 (two-tailed Student t-test). (f) The statistical chart of EMG at 40 and 60 mmHg CRD in control rats after microinjected with AAV-NC or AAV-METTL3. N = 9, two-way ANOVA, F = 11.92, *p < 0.05, **p < 0.01. CRD, colorectal distension; METTL3, methyltransferase-like 3.
CircKcnk9 is the major substrate of m6A in IBS rats
More recent studies suggested that m6A modification could mark long non-coding RNA (lncRNA) for processing.23,24 Considering circKcnk9 was significantly associated with IBS in our previous study,
17
we speculated that circKcnk9 might contribute to the development of chronic visceral pain through m6A modification. fluorescent in situ hybridization results showed that circKcnk9 in IBS-like rats significantly co-localized with more c-fos in hippocampus as compared with the controls, indicating that circKcnk9 in hippocampus could activate neurons (Figure 4(a)). Next, the secondary structure of cicKcnk9 was predicted based on the base sequence of circKcnk917,25 (Figure 4(b)). The RNA fold prediction website revealed that circKcnk9 was highly m6A modified (Figure S4), and Figure 4(c) showed the specific structure (GGACA) of the circKcnk9 modification. Further m6A RNA-binding protein immunoprecipitation (MeRIP) experiments confirmed that circKcnk9 and m6A antibodies had binding sites (Figure 4(d)). YTHDC1 binds directly to circKcnk9 as an RNA-Binding-Protein. (a) Combined circKcnk9 and c-fos immunofluorescence staining in the hippocampus of IBS or control rats. Scale bar, 250 μm. (b) Secondary structure prediction plot of circKcnk9. (c) The RNA fold prediction website revealed the m6A motif structure of circKcnk9. (d) MeRIP assays showing the association between circKcnk9 and m6A. N = 3, two-tailed Student t-test, t = 13.56, ***p < 0.001. (e) The RBPDB website revealed RBP of circKcnk9 (combined possibility >80%). (f) FISH assay showing the location of YTHDC1 and circKcnk9 (up). Line scan graphs of YTHDC1 (green) and circKcnk9 (red) fluorescence intensities in the PC12 cells are shown (down). Scale bar, 25 μm. (g) A pulldown assay confirmed the binding of YTHDC1 and circKcnk9 in PC12 cell lines. (h) RIP assays showing the association of YTHDC1 with circKcnk9. IgG antibody served as a control. N = 4, two-tailed Student t-test, t = 15.26, ***p < 0.001.
RBPDB website was used to screened out RNA-Binding-Protein (RBP) (combined possibility >80%). The results showed that circKcnk9 has 15 predicted RBPs on the premise of binding probability greater than 80% (Table S4). YTHDC1, as the only m6A-associated protein among them, had a 100% binding probability to circKcnk9 (Figure 4(e)). Line scan graphs of YTHDC1 (green) and circKcnk9 (red) fluorescence showed both of them were colocalized in the nucleus (Figure 4(f)). To this end, we performed RNA pull-down assays and verified YTHDC1 as a circKcnk9-binding protein (Figure 4(g)). Further RIP experiments demonstrated enrichment of circKcnk9 in complexes precipitated with antibody against YTHDC1 compared to those precipitated with control IgG (Figure 4(h)).
YTHDC1 modulates visceral hypersensitivity through regulating the nuclear export of circKcnk9 in an m6A-dependent manner
As YTHDC1 is essential for the nuclear export of m6A methylated mRNAs,
26
we hypothesized that circKcnk9 m6 A modification sites combined with YTHDC1 could promote circKcnk9 nuclear translocation in IBS-like rats. First, we constructed an overexpress plasmid (YTHDC1-WT) and synthesized its siRNA (siYTHDC1-A, siYTHDC1-B and siYTHDC1-C). To search the direct target of YTHDC1, we carried out an siRNA screening. qRT-PCR results showed that YTHDC1-WT resulted in a significant elevation of basal YTHDC1 level in PC12 cells, whereas treating with siYTHDC1-B had an opposite effect (Figure 5(a)). Fluorescent in situ hybridization experiments confirmed that the increased nuclear staining of circKcnk9 caused by siYTHDC1 was recovered by overexpression of YTHDC1-WT but not YTHDC1-NC (Figure 5(b)). Second, we sought to explore whether the expression of YTHDC1 was altered by circKcnk9. Adeno-associated virus was microinjected into CA1 of control rats to overexpress circKcnk9 while shcircKcnk9 was microinjected into CA1 of IBS-like rats to knockdown circKcnk9. In our study, circKcnk9 overexpression of AAV-circKcnk9 by stereotactic injection was increased the protein expression of YTHDC1 in controls (Figure 5(c)). In contrast, IBS-like rats with CA1 shcircKcnk9 showed a decrease in the expression of YTHDC1 (Figure 5(d)). These above results indicated that YTHDC1 could indeed expedite the nuclear export of m6A-modified circKcnk9, and furthemore, circKcnk9 could also affect the expression of YTHDC1. YTHDC1 promotes circKcnk9 nuclear translocation. (a) Validation of knockdown or overexpression for YTHDC1 in PC12 cells. N = 3 per group (one-way ANOVA followed by Dunnett’s test, *p < 0.05 vs. siNC; two-tailed Student t-test, t = 9.644, ***p < 0.001 vs. YTHDC1-NC). (b) RNA-FISH showing that the increased nuclear staining of circKcnk9 (red) caused by siYTHDC1 was rescued by YTHDC1-WT but not YTHDC1-NC. Scale bar, 25 μm. (c, d) The relative expression of YTHDC1 protein after CA1 treatment of AAV-circKcnk9 in control (C, N = 3, t = 5.104, **p < 0.01) or sh-circKcnk9 in IBS-like rats (D, N = 4, t = 2.477, p = 0.05) were evaluated using Western blotting (two-tailed Student t-test). IBS, irritable bowel syndrome.
Next, we further evaluated the role of YTHDC1 in chronic visceral hypersensitivity and found YTHDC1 was high expressed in the hippocampus of IBS-like rats (Figure 6(a)). Thereafter, we observed CA1 siYTHDC1-B was able to reduce the protein expression of YTHDC1 in hippocampus in IBS-like rats (Figure 6(b)). Meanwhile, the EMG results also showed that CA1 siYTHDC1-B treatment alleviated visceral hypersensitivity in IBS-like rats (Figure 6(c)). Interestingly, FISH results showed circKcnk9 was largely colocalized with METTL3 in hippocampus (Figure 6(d)), which implied that circKcnk9 (binding with YTHDC1) modified by m6A might exert its biological role through METTL3 in the hippocampal pyramidal cell. As expected, we also found YTHDC1 was decreased after knockdown METTL3 in either IBS rats (Figure 6(e)) or PC12 cell lines (Figure S5), whereas it was increased after overexpression of METTL3 in control rats (Figure 6(f)). METTL3 modulates IBS through mediation of YTHDC1. (a) The relative expression of YTHDC1 protein in control and IBS-like rats was evaluated using Western blotting. N = 3, two-tailed Student t-test, t = 7.450, **p < 0.01. (b) The relative expression of YTHDC1 protein in IBS-like rats after microinjected with siNC or siYTHDC1 was evaluated using Western blotting. N = 4, two-tailed Student t-test, t = 7.450, t = 3.162, *p < 0.05. (c)The statistical chart of EMG at 40 and 60 mmHg CRD in IBS-like rats microinjected with siNC or siYTHDC1. N = 8, two-way ANOVA, F = 28.38, ***p < 0.001. (d) FISH assay showing the location of METTL3 and circKcnk9. Scale bar, 250 μm. (e, f) The relative expression of YTHDC1 in IBS-like rats with siMETTL3 or siNC (E, N = 3, t = 7.045, **p < 0.01) and in control rats after CA1 treatment with AAV-METTL3 or AAV-NC (F, N = 3, t = 4.184, **p < 0.01) were evaluated using Western blotting (two-tailed Student t-test). CRD, colorectal distension; IBS, irritable bowel syndrome; METTL3, methyltransferase-like 3.
As revealed from these results, YTHDC1 modulated visceral hypersensitivity through regulating the nuclear export of circKcnk9 in an m6A-dependent manner.
Discussion
Visceral hypersensitivity in response to intestinal inflammation in IBS has been studied for several decades,27, 28 but the mechanism of how its multiple layers of epigenetic modifications (noncoding RNA, histone modification, DNA methylation) lead to visceral hypersensitivity is severely unclear. In this study, we provided the first evidence that neonatal CRD leaded to high level of m6A through an increase of METTL3 in the hippocampal neurons. This increase correlated with gain of m6A sites in circKcnk9 and a nuclear export of circKcnk9 in the hippocampus, which leaded to visceral hypersensitivity. By contrast, blocking this increase reversed gain of m6A sites in circKcnk9, nuclear export of circKcnk9 in the hippocampal neurons, and alleviated visceral hypersensitivity in IBS-like rats. Hippocampal METTL3 likely contributed to visceral hypersensitivity through enriching m6A-sites of circKcnk9 in the hippocampus. We have presented a number of findings demonstrating the significance of m6A modification in circKcnk9 nuclear export through its RBP (YTHDC1) in IBS.
Similar to other epigenetic modifications,29–31 m6A modification is dynamic and reversible, which is established mainly by the methyltransferases (METTL3, METTL14, and WTAP) and removed by demethylases (FTO and ALKBH5). In the light of the literature that hippocampal m6A might be involved in the neuronal development such as synapse function and synaptic plasticity.32–34 And our previous research demonstrated that long-term potentiation (LTP) was enhanced in the hippocampal CA1 of IBS-like rats. 35 Thus, we compared the expression of several genes about m6A in the hippocampus between control and IBS-like rats. Interestingly, in IBS, we found no changes in hippocampal METTL14, WATP, FTO and ALKBH5 except for METTL3. Besides, METTL3 was largely colocalized with neurons but not microglia or astrocytes, which was consistent with the studies that m6A were abundantly expressed in neurons. 36 Apparently, our findings suggested that hippocampal METTL3 was likely involved in the neuronal activity in IBS. It is well known that METTL3 plays multiple roles in biological processes, including cell cycle progression, cell proliferation, cell apoptosis, cell migration and invasion, cell differentiation and inflammatory response.37–39 In addition, depleting METTL3 in hippocampal mouse reduced memory consolidation ability, whereas overexpression of METTL3 could enhance long-term memory consolidation.32,34 In this study, inhibition of hippocampal METTL3, along with m6A, alleviated visceral hypersensitivity in IBS-like rats. Overexpression of METTL3 facilitated pain hypersensitivity symptom in controls. Notably, our results are consistent with the findings of a previous study, which demonstrated that inhibiting METTL3 rescued the dysregulation of spinal m6A and alleviated inflammatory pain. By contrast, upregulation of spinal METTL3 could cause pain behaviors and spinal neuronal sensitization. 40
It has been shown that m6A can also modulate the expression of circRNAs like other RNA molecules through regulating their generation, stability, or degradation. 41 Considering that our previous studies 17 have demonstrated that circKcnk9, a novel circRNA, was found to adsorb MiR-124-3p in the cytoplasm to induce visceral hypersensitivity in IBS-like rats, it is important to trace the changes of m6A modification associated with circKcnk9 in IBS-like rats. Recently, YTHDC1 and its homologous complex have attracted increasing attention. Most of the studies have been carried out in m6A modifying RNA processing are mainly based on the limited information about the role of its cytoplasmic binding partners YTHDF1 and YTHDF2 in the regulation of mRNA translation or mRNA stability.42–44 In addition, according to Xu et al. the crystal structures of the YTH domain of YTHDC1, a couple of YTH domain families and its complex with an m6A-associated RNA have been found out. The structural analyses, as well as a transcriptome-broad identification of YTHDC1-binding sites and biochemical experiments data demonstrated the particular mode of m6A-YTH binding and the preferential recognition of the m6A sequences by YTHDC1. 45 In our study, circKcnk9 was found to have only one m6A-associated binding protein (YTHDC1) predicted with bioinformatics techniques, which was consistent with RIP and RNA pulldown data. Circular RNAs have mainly been reported to be localized in the cytoplasm.46–48 Some studies have confirmed that the m6A modification on circRNAs might modulate their nuclear exportation.47,49 For this reason, the systems regulating nuclear-cytoplasmic circKcnk9 export should be explored. We hypothesized that YTHDC1 modulated visceral hypersensitivity through regulating the nuclear export of circKcnk9 in m6A-dependent manner.
To confirm this conjecture, we carried out in vitro experiments for the first time. Fluorescent in situ hybridization data suggested that the increased nuclear staining of circKcnk9 caused by siYTHDC1 was recovered by overexpression of YTHDC1-WT but not YTHDC1-NC in PC12 cells. Next, AAV (aavcircKcnk9) was microinjected into the CA1 of control rats to overexpress circKcnk9, whereas shcircKcnk9 was microinjected into the CA1 of IBS-like rats to knockdown circKcnk9. The Western blot indicated circKcnk9 overexpression induced high expression of YTHDC1 protein in the controls. In contrast, IBS-like rats with CA1 shcircKcnk9 showed a decrease in YTHDC1 protein level. Besides, we also knocked down and overexpressed METTL3 in the hippocampus of IBS-like and control rats, respectively. The expression trend of YTHDC1 protein was found to be consistent with the above results. Then, we microinjected siYTHDC1 into the CA1 region of IBS-like rats and found that visceral sensitivity was reduced compared with controls. These data confirmed our speculation.
In summary, our study demonstrated that METTL3 modulated visceral hypersensitivity through regulating the nuclear export of circKcnk9 in YTHDC1-dependent manner. These findings revealed a novel epigenetic mechanism in visceral hypersensitivity and might contribute to the future development of potential targeted interventions for IBS through m6A-circKcnk9 modification.
Supplemental Material
Supplemental Material - Methyltransferase-like 3 modulates visceral hypersensitivity through regulating the nuclear export of circKcnk9 in YTHDC1-dependent manner
Supplemental Material for Methyltransferase-like 3 modulates visceral hypersensitivity through regulating the nuclear export of circKcnk9 in YTHDC1-dependent manner by Wei Lin, Yuan Liu, Yifei Zhou, Mengying Lin, Congxu Liu, Ying Tang, Bin Wu and Chun Lin in Molecular Pain
Supplemental Material
Supplemental Material - Methyltransferase-like 3 modulates visceral hypersensitivity through regulating the nuclear export of circKcnk9 in YTHDC1-dependent manner
Supplemental Material for Methyltransferase-like 3 modulates visceral hypersensitivity through regulating the nuclear export of circKcnk9 in YTHDC1-dependent manner by Wei Lin, Yuan Liu, Yifei Zhou, Mengying Lin, Congxu Liu, Ying Tang, Bin Wu and Chun Lin in Molecular Pain
Footnotes
Acknowledgements
We thank the Public Technology Service Center of Fujian Medical University (China) for providing technical support and experimental platforms.
Author contributions
C.L. as the primary and corresponding author supervised all experiments and funded the work; W.L. and Y.L. wrote the first draft and designed the research; Y.L. performed the bioinformatic analysis; Y.L., W.L., Y.-F. Z. M.-Y. L, C.-X. L performed the experiments and analyzed data. C.L., Y.T., B.W. and W.L revised the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the We would like to thank the Foundation of Science and Technology of Fujian Province (2018Y9069), Natural Science Foundation of Fujian Province (2021J01675, 2020J01608), the funders played no role in the study design or implementation.
Supplemental Material
Supplemental material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
