Abstract
Neurosensory disorders such as pain and pruritus remain a major health problem greatly impacting the quality of life, and often increasing the risk of mortality. Current pre-clinical models to investigate dysfunction of sensory neurons have shown a limited clinical translation, in part, by failing to mimic the compartmentalized nociceptor anatomy that exhibits a central compartment containing the soma and a peripheral one harboring the axon endings with distinct molecular and cellular environmental composition. Thus, there is a need to validate compartmentalized preclinical neurosensory models for investigating the pathophysiology of peripheral sensory disorders and to test drug candidates. Here, we have addressed this issue and developed a microfluidic-based preclinical nociceptor model and validated it for investigating inflammatory and neuropathic peripheral disorders. We show that this model reproduces the peripheral sensitization and resolution produced by an inflammatory soup and by the chemotherapeutic drug paclitaxel. Furthermore, compartmentalized nociceptor primary cultures were amenable to co-culture with keratinocytes in the axonal compartment. Interaction of axonal endings with keratinocytes modulated neuronal responses, consistent with a crosstalk between both cell types. These findings pave the way towards translational pre-clinical sensory models for skin pathophysiological research and drug development.
Keywords
Introduction
Sensory neurons are pseudo-unipolar cells with their soma located in the dorsal root (DRGs) or trigeminal ganglia (TG) and the axon exhibiting a dual projection, one towards the periphery to innervate the skin or viscera, and the other to the spinal cord to interact with the brain ascending pathway. 1 In-vivo, peripheral endings innervating the skin or viscera are the prime targets of pro-algesic and pruritogenic agents and noxious stimuli coming from the external environment, while the soma and the central terminals are transducers of the neuronal excitability to the brain.2–4 Thus far, nociceptor peripheral endings have been difficult to study due to their small size and peculiar morphology, despite being the main target for painful, inflammatory and pruritogenic mediators. In contrast, because the soma of the primary afferent neurons are readily accessible to experimentation, virtually all in-vitro sensory data have been derived from the cell bodies. These studies assumed that exposure of cell bodies to pro-algesic and pruritogenic agents provide reliable and predictable models of the peripheral endings pathophysiology,5,6 although we are learning that this experimental paradigm has limitations because the main communication in-vivo is afferent (i.e. peripheral-central) i.e. complemented with an efferent function (i.e. axon-reflex vasodilatation). In addition, these in-vitro models have limited the co-culture of sensory neurons with skin cells due to incompatibility of growing conditions. 7 Thus, it is of pivotal importance to correctly study the neural parts in their appropriate environment, and their interactions with and modulation by the innervated peripheral tissue.
In the past 20 years, microfluidic-based compartmentalized neuronal cultures that enable the separation and study the two major parts of a neuron (i.e. soma and axons), have become an emerging technology to investigate neural function and plasticity. 8 The word “microfluidics” refers to devices presenting a series of microchannels connecting two or more chambers. Microchannels range from one to a few hundred micrometers in size, through which microscopic volumes of fluids can be processed and axons can grow. 9 Microfluidic chambers (MFC) facilitate the study of interactions between axonal endings and tissue cells such as keratinocytes and immune cells, under physiological or pathological conditions.10–15 They permit the investigation of neuronal excitability, including action potential frequency and velocities.16,17 Therefore, microfluidic-based, compartmentalized nociceptors primary cultures may provide a suitable preclinical model for investigating the contribution of the neurosensory system to skin pathophysiology and to validate drug candidates that contribute to faster resolution of skin disorders by modulating the neuronal input to the disease.
Here, we investigated whether microfluidic-based compartmentalized nociceptor cultures are suitable models for investigating the pro-algesic and neuropathic sensitization and resolution phases upon their exposure to pro-algesic compounds. We also examined the suitability of MFCs to study the interaction between axonal endings and keratinocytes in co-culture. For this task, we exposed axonal ends to an inflammatory soup or to the chemotherapeutic drug paclitaxel, as representative models of two peripheral disorders. Inflammatory mediators (IMs) have a broad effect on sensory neurons excitability by decreasing the activation threshold of ion channels involved in action potential generation and propagation. Examples of IMs include extracellular protons, arachidonic acid, serotonin, nerve growth factor (NGF), prostaglandins (PG), bradykinin (BK), adenosine triphosphate (ATP) and histamine, among others.18,19 Most IMs increased the activity of Transient Receptor Potential (TRP channels), by promoting their surface expression and/or facilitating their gating.20,21 Inflammatory potentiation of thermoTRP channels underlies the hypersensitivity of inflamed tissues.22,23 Mechanistically, the role of thermoTRPs to inflammatory pain has been performed using non-compartmentalized nociceptor cultures where soma and axons have been exposed to IMs, as opposed to the native conditions where only peripherally expressed receptors are affected by pro-algesic compounds. Similarly, chemotherapeutic drugs such as paclitaxel affect peripheral ends provoking a very disturbing and disabling sensory condition referred to as chemotherapy-induced peripheral neuropathy (CIPN), that may lead to dose reduction or even cessation of treatment.24–26 Rodent models have shown that paclitaxel increases nociceptor excitability by a variety of mechanisms, including increased neuronal electrogenicity, axonal transport, and axonal degeneration.24,27 We have reported that paclitaxel directly affects nociceptor function by altering the expression and function of Nav, Kv and thermoTRP channels. 28 However, these studies have been performed in non-compartmentalized chambers were soma and endings were exposed to the drug, thus questioning whether this model provide reliable translational information on the peripheral mechanisms underlying paclitaxel neuropathy. Hence, MFC compartmentalization provides a more translational strategy to investigate the pathophysiological sensitization of axonal terminals. We used this approach and report that compartmentalized MFCs are a valuable tool to investigate peripheral nociceptor sensitization along with the crosstalk between peripheral ends and cutaneous cells.
Material and methods
Animals and ethical statement
All procedures were approved by the Institutional Animal and Ethical Committee of the Miguel Hernández University of Elche, in accordance with the guidelines of the Economic European Community, the National Institutes of Health, and the Committee for Research and Ethical Issues of the International Association for the Study of Pain.
Cell cultures
Primary cultures of DRG and TG sensory neurons were carried out as previously described.20,29,30 Neurons were seeded in the soma compartment of microfluidic chambers (Millipore devices “AXIS™: Axon Investigation System”). The procedure is detailed in supplementary methods.
Scanning electron microscope
At DIV (days in vitro) six of co-culture of DRGs and keratinocytes, the cells were fixed with 2.5% glutaraldehyde (EMS) for 45 min maintaining fluidic isolation between soma and keratinocytes. Afterwards, cells were washed with dH2O and dehydrated with serial washes of ethanol. 31 Liquid was removed, MFCs were carefully detached from the slide and dried at room temperature for at least 24 h. The slides were coated with chromium and analyzed by the SIGMA 300 VP (Zeiss) field emission scanning electron microscope.
Calcium imaging
Ca2+ imaging was used to evaluate the functionality of the sensory neurons and their modulation by the keratinocytes, inflammatory soup and paclitaxel. For these measurements, neuronal activators were added to the axonal chamber, and Ca2+ influx monitored in the soma compartment. Methodological details are described in supplementary methods. Analysis of percentage of cells responding to each stimulus was performed considering crossing neurons (Did’ and KCl positive) as the total number of cells. The soma KCl response was considered as the maximal Ca2+ signal and was used to normalize the size of the responses. An increase in fluorescence was considered a response if it was higher than 10 times the noise of the baseline before adding the stimuli to the axonal compartment or 20 times before the addition of 40 mM KCl in the soma compartment.
Statistical analysis
All data were tested for normal distribution with D'Agostino & Pearson omnibus normality test. Data with normal distribution such as percentage of responses in DRGs or in DRGs co-cultured with keratinocytes were analyzed by two-way ANOVA, Sidak’s post-hoc test or with unpaired t test (axonal length quantification). All the other data had non-normal distribution and were analyzed by two-tailed Mann-Whitney U or by Kruskal-Wallis test with Dunn's multiple comparisons test in the case of comparing more than two groups at the same time. Statistical analysis was performed using GraphPad Prism 9.4.1 (GraphPad Software Inc.). Throughout the manuscript, data are represented as bars with mean ± SEM while percentage of co-response to two stimuli are expressed as mean values in quantitative Venn diagrams.
Results
In-vitro compartmentalized axonal sensory endings are functional
DRGs neurons were seeded in MFCs and a physical and a chemical gradient was established between the compartments to direct axonal growth through microchannels (Figures 1(a)–(e), Table 1). After six DIV, 53 ± 14% neurons (n= 482) extended axons as revealed by Did’ labeling (Figure 1(f)), and the axonal endings expressed mRNA of neuronal markers (Figure S1). We observed that 50% of these neurons exhibited Ca2+ influx in the soma and triggered electrical activity upon stimulation of axonal endings with 40 mM KCl (ax. KCl) (Figures 2(a) and (b)). Exposure of axonal endings to thermoTRP agonists such as capsaicin, menthol or allyl isothiocyanate (AITC) also evoked action potentials (Figure 3). These chemicals are respectively agonists of the thermoTRP channels TRPV1, TRPM8 and TRPA1.32–34 Among the Did’ labelled neurons, 12.1% responded to 100 nM capsaicin, 9.8% to 100 µM menthol, 7.9% to 100 µM AITC (Figure 2(b)), 1.4% were activated by capsaicin and menthol, 1.6% by capsaicin and AITC, and 3.7% by menthol and AITC (Figure S2). The size of the Ca2+ responses was similar for all activating stimuli (Figure 2(c)). As expected, direct stimulation of the neurons cultured on glass coverslips produced higher percentage of response (Figures 2(d) and (e)) and 2-fold larger Ca2+ responses (Figure 2(f)). No functional differences were observed between DRG sensory neurons from females and males (Figure S2, Tables S4 and S5). Trigeminal sensory neurons were also amenable to culture in MFCs and exhibited a similar functionality than their DRG counterparts (Figure 4). Mouse DRGs cultured in microfluidic chambers (a) DRGs cultured in MFC at DIV 0 loaded with Fluo-4. Scale bar = 100 μm. (b) DRGs cultured in MFC at DIV six loaded with Did’. Scale bar= 100 μm. (c) MFC with 400 μL PBS in the upper compartment and 200 μL blue-coloured PBS in the lower compartment, at time 0 h or 24 h at 37°C (d) Schematic upper view of MFC without cells or with cells and developed axons. (e) Schematic side view of MFC with cells. (f) DRGs cultured in MFC at DIV 6. Merge image of transmitted light and Did’ staining (red). Scale bar = 100 μm. Media composition for DRGs microfluidic culture. Activity of mouse DRGs cultured in MFCs, non-compartmentalized and co-cultured with human keratinocytes. (a)–(c) Calcium imaging recordings of DRGs cultured in MFC. (a) Representative fluorescent signals recorded in the soma of Did+ neurons stimulated in the axonal compartment. Y-axis represents fluorescence normalized with respect to that evoked by KCl in the soma compartment (KCl). (b) Percentage of response of Did+ neurons normalized with respect to the response evoked by KCl+ in the soma. (c) Size of the response normalized with respect to the response evoked by KCl+ in the soma. Data are as mean ± SEM, every dot is a cell. N = 4 animals, two males and two females; n ≥ 2 coverslips per animal; total number of crossing neurons=747. (d)–(f) Calcium imaging measurements of non-compartmentalized DRGs. (d) Representative fluorescent signals of responsive neurons. Y-axis represents fluorescence normalized to that evoked by KCl. (e) Percentage of responses. (f) Size of the response normalized to KCl. Data are mean ± SEM. N = 4 mice, two males and two females. n ≥ 2 coverslips per animal. Total number of neurons= 5498. (g)–(i) Scanning electron microscope pictures of the axonal compartment of co-cultured DRGs with human keratinocytes. The dashed line indicates the end of the microchannels. The white square indicates high magnification views of the selected area. Scale bars: 100 μm (g), 10 μm and 500 nm (h), and 10 and 2 μm (i). (j)–(l) Calcium imaging measurements of co-cultured DRGs with keratinocytes. (j) Representative fluorescent signals of responsive neurons. (k) Percentage of response of Did+ neurons normalized with respect to the response evoked by KCl+ in the soma. Mean ± SEM. (l) Size of the response normalized with respect to the response evoked by KCl+ in the soma. Data are mean ± SEM, every dot is a cell. DRGs-keratinocytes co-culture: N = 4 animals, two males and two females; n = 2 coverslips per animal; total number of crossing neurons = 298. mDRGs cultured in microfluidic chambers coupled to a MEA chip. (a) Picture of cells at DIV 0 (b) Picture of cells at DIV 6 (c) Spikes obtained from mDRGs stimulated with AITC 100 μM, menthol 100 μM, capsaicin 200 nM or KCl 40 mM. Trigeminal culture in microfluidic chambers (a) TG cultured in microfluidic chambers at DIV six loaded with Fluo-4. Scale bar = 100 μm. (b) Representative traces of neurons responding to capsaicin, menthol, AITC or axonal KCl. N = 4 mice, two males and two females. n ≥ 2 MFC per animal. Total number of crossing neurons= 740. (c) Quantification of the percentage of response to capsaicin, menthol, AITC or axonal KCl normalized to KCl response. (d) Quantification of the size of the response normalized to KCl response. Data are represented as scatter dot plot, mean ± SEM. (e) Quantification of the percentage of response to capsaicin and menthol, menthol and AITC or capsaicin and AITC normalized to KCl response. Data are represented as quantitative Venn Diagram.

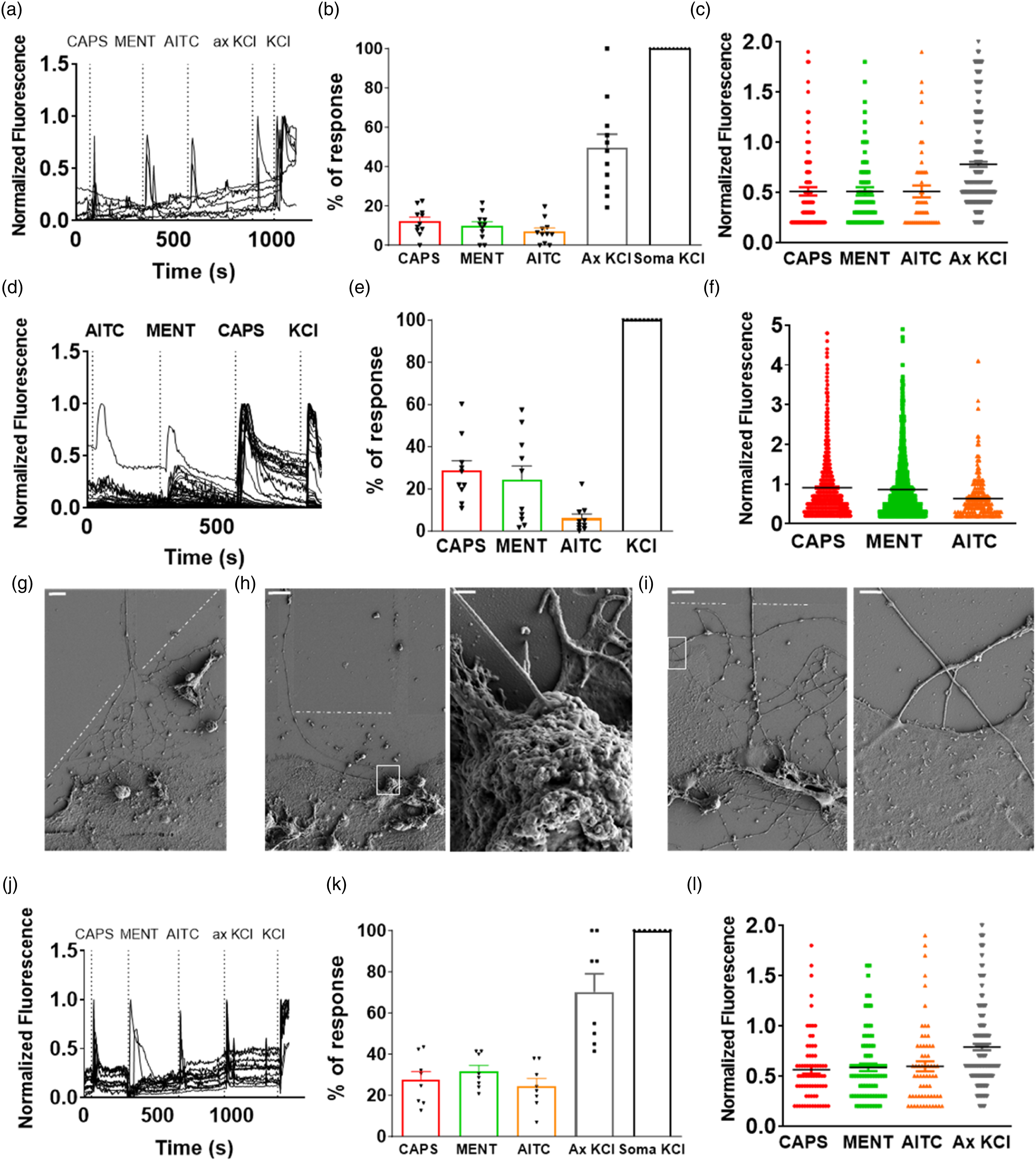


Keratinocytes interact with axonal sensory endings and modulate their activity
Co-culturing axonal sensory endings with keratinocytes (HaCaT cells), in the presence of 100 ng/mL nerve growth factor (NGF) and 100 ng/mL glial derived neurotrophic factor (GDNF) and the absence of 5-fluoro-2’-deoxyuridine to maintain keratinocyte viability (Table S1, Figure S3), resulted in a strong physical interaction between sensory endings and keratinocytes as revealed by field emission scanning electron microscopy (Figure 2(g)). Electron microscopy images show that peripheral ends form synapse-like contacts with HaCaT cells (Figure 2(h)) or by-pass a particular cell to interact with another as it occurs in the epidermis (Figure 2(i)). Under these conditions up to 70% of neurons extended axons through the microchannels.
Stimulation of the axonal compartment with 40 mM KCl resulted in Ca2+ transients in the soma of 70.2% of neurons (Figures 2(j) and (k)). Further analysis revealed that 27.4% of Did´-labelled neurons responded to 100 nM capsaicin, 31.5% to 100 μM menthol, 24.4% to 100 μM AITC (Figure 2(l)), 5% to capsaicin and menthol, 6% to menthol and AITC and 4.4% to capsaicin and AITC (Figure S3). Notably, a higher percentage of neurons responded to capsaicin, menthol and AITC than in the absence of keratinocytes (Figure 2(b), Figure S3, Table S6) and this effect were not due to the medium change in the axonal compartment with respect to the DRGs MFC culture (Table S7). MFC-based DRG cultures and of neurons co-cultured with keratinocytes were exposed to the same NGF and GDNF concentrations to have a comparable neurotrophic effect (Table 1, Table S1). Nonetheless, as human keratinocytes secrete NGF, 35 we cannot exclude that secreted NGF affects neuronal endings length, excitability and thermoTRPs expression.36,37 The magnitude of the normalized responses was not significantly altered by the presence of the HaCaT cells. These results indicate that keratinocytes increased the number of peripheral ends expressing functional thermoTRPs as well as exhibiting K+-dependent excitability, suggesting a paracrine increase in axonal growth through microchannels along with a modulation of peripheral endings functionality.
Inflammatory sensitization and resolution of peripheral terminals
Next, we set to validate the microfluidic nociceptor culture as a model to study inflammatory sensitization. We exposed axonal endings to an inflammatory soup or vehicle at DIV five for 24 h (Table S2), and neuronal activity was recorded at DIV six and DIV 8 (0 h and 48 h post insult) to investigate inflammatory sensitization and its resolution. Note that inflammatory mediators reversibly reduced axonal density (Figure S4). Functionally, axonal endings appear to be more active after inflammatory sensitization (Figures. 5(a)–(C)), as reflected by the 2-fold increment in the percentage of neurons responding to 100 nM capsaicin, 100 µM menthol, and 100 µM AITC (Figure 5(d)). This sensitization was reversible, resolving 48 h post-inflammatory insult (DIV 8, Figure 5(d)). Notice that the percentage of neurons exposed to vehicle responding to capsaicin remained constant up to DIV 8, whereas a significant decrease was observed for the responses to menthol, AITC and KCl (Figure 5(d)). This reduction in responses may reflect the neural aging that intriguingly affected less to TRPV1 sensory neurons. The inflammatory soup increased the responses to capsaicin and menthol from 1.8% to 9%, to capsaicin and AITC from 1.6% to 8.7% and to menthol and AITC from 3.7% to 9.8% after 24 h exposure (Figures 5(e) and (f)). This increment in dual responses resolved to basal level 48 h after inflammatory exposure (DIV8) (Figures 5(g) and (h)). Effect of inflammatory mediators (IM) on DRGs nerve endings. (a) Treatment and assays timeline. Axons were treated with inflammatory mediators or vehicle at DIV five and recorded at DIV six or DIV8. (b)–(c) Representative traces of responsive neurons. Y axes represents fluorescence normalized to somal KCl response. (d) Percentage of response normalized to Did+ and somal KCl responsive cells. Mean ± SEM. Statistical analysis two-tailed Mann-Whitney U. (e)–(h) Responses to capsaicin and menthol, menthol and AITC or capsaicin and AITC normalized to KCl response in the soma. (i) Size of the response normalized to somal KCl response. Mean ± SEM, every dot represents a cell. Statistical analysis two-tailed Mann-Whitney U. N=6 animals, three males and three females; n ≥ 3 coverslips per animal; total number of crossing neurons=3018.
The size of the capsaicin response was significantly augmented by exposure to the inflammatory soup for 24 h, resolving 48 h after removal of the soup (Figure 5(i)). A similar increment was observed for AITC, but not for menthol (Figure 5(i)). KCl responses did not show a significant change after 24 h exposure to the inflammatory soup, but we observed that at DIV eight sensory terminals that were incubated with the inflammatory soup exhibited higher KCl responses than those exposed to the vehicle (Figure 5(i)). Akin to the number of neurons responding to KCl at DIV 8 (Figure 5(d)), we also observed a significant reduction in the magnitude of the KCl-evoked Ca2+ transients incubated with vehicle (Figure 5(i)), most likely due to a reduction of responding neurons due to aging of the primary culture.
Neuropathic sensitization and resolution of peripheral ends
Exposure of nociceptor primary cultures to paclitaxel for 24 h produced a significant increase in neuronal activity that peaked 48 h after removal of the chemotherapeutic drug and mitigated 96 h after exposure.
28
Here, we questioned if a similar sensitization-resolution occurs when the drug is applied onto axonal endings. To investigate the impact of the drug on the peripheral ends, we exposed them to 1 µM paclitaxel (PTX) or vehicle (DMSO 0.04%) for 24 h (DIV6, Table S3). PTX was removed, and neuronal functionality was recorded 48 (DIV8) and 96 h (DIV10) after drug removal (Figure 6(a)). As exhibited in Figure S8, paclitaxel reversibly reduced axonal density, being the strongest effect just after treatment and fully resolving 96 h post-treatment. Effect of paclitaxel 1 µM on mDRGs cultured in microfluidic chambers. (a) Treatment and assays timeline (b)–(c) Representative traces of responsive neurons. Y axes represents fluorescence normalized to somal KCl response. (d) Percentage of response normalized to Did+ and somal KCl+ responsive cells. Mean ± SEM. (e)–(h) Responses to capsaicin and menthol, menthol and AITC or capsaicin and AITC normalized to somal KCl response. (i) Size of the response normalized to KCl. Mean ± SEM, every dot represents a cell. Statistical analysis two-tailed Mann-Whitney U. N = 5 animals, three males and two females; n ≥ 2 coverslips per animal; total number of crossing neurons = 1638.
Functional analysis revealed that 40 mM KCl in the axonal compartment evoked activity in 20% of neurons at DIV 8 and 10. Paclitaxel did not significantly affect the percentage of functional neurons, activated by capsaicin, menthol, AITC and KCl (Figures 6(b)–(d)). Similarly, terminals responding to two agonists was mildly affected by the chemotherapeutic drug (Figures 6(e)–(H)). In marked contrast, we observed a significant increase in the size of capsaicin responses at DIV eight that resolved at DIV 10 in agreement with previous results (Figure 6(i)). 28 Menthol responses show a tendency to increase 48 h after paclitaxel exposure but did not reach statistical significance. Nonetheless, the cellular distribution suggests the presence of two menthol sensitive subpopulations that deserves further investigation. Higher responses for AITC were observed 96 h (DIV 10) after paclitaxel incubation. Overall, these results substantiate a peripheral action of paclitaxel sensitizing neural endings, consistent with the sensory symptoms provoked by the chemotherapeutic drug.
Conclusions and perspectives
Our findings substantiate that compartmentalized sensory neurons in microfluidic chambers are suitable models to investigate the interaction of the sensory endings with dermal cells, as well as to study the mechanisms involved in their peripheral sensitization either by pro-inflammatory and neuropathic drugs. A salient contribution of our study is that sensory neurons from DRGs and TGs can be successfully cultured in MFCs for up to 10 DIV, allowing to investigate nociceptor potentiation and its resolution. Furthermore, we established that axonal endings in these cultures serve as models for studying free nociceptive nerve endings and their modulation by trophic and pro-algesic agents. Our findings reinforce and build upon the existing body of knowledge regarding the viability and functionality of sensory neurons in MFCs, highlighting their relevance as a translational model for investigating peripheral nociceptive mechanisms underlying sensitization and desensitization.14,38,39
Moreover, co-cultures of nerve endings and keratinocytes represent a proof of concept for investigating the interaction and cross-talk of axonal ends with peripheral cells and tissues. We found that keratinocytes modulate axonal function, most likely by releasing growth factors such as NGF that affect axonal excitability. 35 Thus, compartmentalized cultures provide a strategy to investigate the contribution of nociceptive endings in peripheral neuropathies that lead to cutaneous and visceral pain. In this regard, our inflammation and neuropathic models demonstrate that the axonal compartment can be sensitized by manipulating the environmental composition mimicking an inflammatory soup or the presence of a chemotherapeutic drug. We found that both conditions increase the activity of the peripheral ends by enhancing the functionality of thermoTRP channels leading to a higher excitability. Thus, the effect of IMs and chemotherapeutic drugs on DRG function can be primarily assigned to a peripheral action. MFC-based cultures also permit to investigate the effect of the milieu composition on the integrity/size of the axonal endings, that is if compounds produce axonal retraction. Not least, MCFs allow also to evaluate axonal transport to and from the periphery, 27 and to test the effect of peripherally acting drugs. 40
Compartmentalized culture methods present numerous advantages, such as being able to expose axons to a different fluid and cellular environment than cell bodies, which is useful for studies of trophic, ionic, and pharmacological regulation. Specifically, in the case of nociceptors, it is of vital importance to separate the soma from the nerve endings because of their peculiar anatomy in-vivo. In fact, whereas only the peripheral terminal of the nociceptor will respond to environmental stimuli, both the peripheral and central terminals can be targeted by endogenous molecules.5,8 Furthermore, distal axons can be removed performing an axotomy, and subsequently regenerated, which permits many useful approaches to the study of axon growth and regeneration after damage.8,14
These models hold great promise for the future of sensory neurons and pain investigation and open to the possibility of culturing induced sensory neurons (iSNs) derived from human cells. Using iSNs could reduce the use of animals and avoid problems of unreliability and low translationality of animal experimentation.41,42 Culturing these cells in microfluidic chambers would increase the representativity of the model making it more similar to the in-vivo morphology of sensory neurons.
Supplemental Material
Supplemental Material - Compartmentalized primary cultures of dorsal root ganglion neurons to model peripheral pathophysiological conditions
Supplemental Material for Compartmentalized primary cultures of dorsal root ganglion neurons to model peripheral pathophysiological conditions by Simona Giorgi, Angela Lamberti, Laura Butrón, Olivia Gross-Amat, David Alarcón-Alarcón, Enrique Rodríguez-Cañas, Asia Fernández-Carvajal and Antonio Ferrer-Montiel in Molecular Pain
Footnotes
Author contributions
S.G. conceived the idea, performed all the experiments, wrote and revised the manuscript; A.L. performed DRGs culture and paclitaxel treatment; L.B. performed DRGs culture; O.G. wrote and revised the manuscript; D.A.A. performed DRG and TG culture and calcium imaging; E.R.C contributed to samples preparation and took electron microscope pictures; A.F.C. and A.F.M. supervised the project and data, and revised the manuscript. All authors provided critical feedback and helped improve the research’s quality.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the S.G. was financed by Generalitat Valenciana [GRISOLIAP/2019/094 44/19]; A.L. received funding from the European Union's Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie Actions [No 956477]; Spanish Ministry of Science, Innovation and Universities financed L.B. [MCI-PID2021-126423OB-C21]; O.G. received funding from Generalitat Valenciana [GVA-PROMETEO/2021/031]; D.A.A. was financed by the Spanish Ministry of Science, Innovation and Universities [AF2015-66275-C2-1-R]; E.R.C. was financed by the Spanish Ministry of Science, Innovation and Universities [PTA2018-015394-I]. The FESEM equipment was funded by Generalitat Valenciana [GVA-IDIFEDER_2018/020]; Financial support of the Spanish Ministry of Science, Innovation and Universities [RTI2018-097189-B-C21] and Miguel Hernández University of Elche [UMH-PAR2019] was granted to to A.F.C. and A.F.M.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
