Abstract
Chemotherapy-induced peripheral neuropathy (CIPN) is a prevalent and severe side effect affecting cancer patients undergoing paclitaxel treatment. Growing evidence underscores the pivotal role of calcitonin-related peptide (CGRP) in the development of CIPN. Repeated administration of paclitaxel induces alterations in CGRP release from sensory neurons within the dorsal root ganglia (DRG). The density of the CGRP receptor is most prominent in the dorsal horn of the spinal cord, where it overlaps with the distribution of CGRP. However, the impact of chemotherapy treatment on expression of the CGRP receptor in the spinal cord remains unclear, as well as the potential therapeutic benefits of a CGRP receptor antagonist in an animal model of CIPN. Using a mouse model of paclitaxel-induced mechanical hypersensitivity, we show upregulation of
Introduction
Cancer is a global health problem responsible for one in six deaths worldwide.1,2 Currently, despite the advance of targeted treatments and immunotherapy, chemotherapy remains a prevalent approach in cancer patients. While chemotherapeutic agents are used to directly or indirectly inhibit the uncontrolled growth and proliferation of cancer cells, these medications are not selective toward the targets, and also damage normal cells and tissues particularly those that differentiate quickly such as those in the gastrointestinal, skin and hematopoietic systems. 3 Among the many collateral damages that chemotherapy can cause, one of the most severe is the damage to peripheral nerves, which results in the development of chemotherapy-induced peripheral neuropathy (CIPN).4–8 The prevalence of CIPN varies from 10% to 100% when measured the first month after chemotherapy, 9 and is among the highest in the case of taxane-based compounds such as paclitaxel. 10 Symptoms of CIPN include tingling, numbness, touch and cold allodynia and more diffuse pain.11,12 This may result in dose reduction, refusal to continue treatment, psychological distress, neurological impairment, and reduced quality of life. 13 However, very limited options currently exist to treat or prevent CIPN.
The dorsal root ganglia (DRG) contain the cell bodies of peripheral sensory neurons that relay pain from the periphery to the central nervous system.14,15 It is well established that the DRG has an integral and important role in the modulation of peripheral and central sensory processes that include inflammation, somatic pain, and the development of neuropathic pain.16,17 In addition, the primary site of paclitaxel uptake or site of action is along the axons and at the peripheral terminals of sensory neurons such as DRGs. 12 Calcitonin gene-related peptide (CGRP) is a 37 amino-acid neuropeptide, which, along with its receptor, is involved in pain transmission.18–20 CGRP synthesis mainly occurs in the DRG, where the pro-peptide is then cleaved to the active form and stored in large dense-core vesicles at the sensory nerve terminals, where it is commonly co-released with substance P.21–23 The transmission of CGRP signals involves a functional protein complex composed of the calcitonin receptor-like receptor (CALCRL) – a class B G protein-coupled receptor (GPCR) 24 —and receptor activity-modifying protein 1 (RAMP1), a crucial single-transmembrane protein that plays a pivotal role in receptor function by ensuring specificity in ligand binding.25,26 The CGRP receptor is highly concentrated in the spinal cord, particularly the dorsal horns, overlapping with the distribution of CGRP.20,27 Although it is clear that chronic administration of paclitaxel alters CGRP release from sensory neurons in the DRG,12,28,29 the questions remain whether the drug influences CGRP receptor expression in the spinal cord, and what molecular mechanisms drive this compensatory phenomenon may occur.
The status of chromatin organization depends on epigenetic factors, such as DNA methylation and covalent histone modifications that primarily occur on their amino-terminal tails and correlate with open or closed chromatin states.30–33 For example, acetylation of histone H3 loosens DNA-histone interactions and allows the transcriptional machinery to bind and increase transcription. Histone methylation, however, can correlate with either transcriptional activation such as methylation lysine 4 on histone H3 or repression such as methylation of lysine 27 on histone H3. Accumulating studies suggest that altered function of the histone methyltransferase EZH2 and consequently H3K27 methylation levels are critically implicated in the genesis of neuropathic pain.34–37 Previous findings using ChIP-seq assays identified altered patterns of active histone H3 lysine 4 (H3K4) methylation at specific promoters in limbic areas affected in rodent models of neuropathic pain.
38
Additionally, recent work indicated modulation of expression of the
This study explores the intricate interplay between chemotherapy, pain pathways, and epigenetic modifications, providing valuable insights into strategies for mitigating the adverse effects of cancer treatment. We identify an epigenetic mechanism through which repeated administration of paclitaxel upregulates the expression of the CGRP receptor in the spinal cord. Additionally, we evaluate the effects of the CGRP receptor antagonist BIBN4096 on paclitaxel-induced mechanical and cold hypersensitivity in mice. By targeting specific epigenetic mechanisms and leveraging CGRP receptor antagonists, our findings highlight the potential for developing effective treatments to alleviate CIPN and improve patient outcomes.
Materials and methods
Materials and drug administration
Paclitaxel (Sigma-Aldrich) and BIBN4096 (1-[3,5-dibromo-N-[[4-(1,4-dihydro-2-oxo-3(2H)-quinazolinyl)-1-piperidinyl]carbonyl]-D-tyrosyl-L-lysyl]-4-(4-pyridinyl)-piperazine) (Tocris) stocks were dissolved in DMSO (50 mg/kg) and stored at −20ºC until thawed for use. Paclitaxel was diluted in a vehicle which comprised Kolliphor EL (Sigma-Aldrich, MO)/ethanol/saline (0.9%) in a 1:1:18 ratio, and administered (8 mg/kg) intraperitonally (i.p.) in a total volume of 100 µl on four alternate days (cumulative dose 32 mg/kg). BIBN4096 was diluted saline (0.9%) in a 1:1:18 ratio and administered (2.5 mg/kg and 5 mg/kg, i.p.) in a total volume of 100 µl, informed by existing publications. 39 At these doses, BIBN4096 did not induce any noticeable changes in rearing, grooming, or locomotion (data not shown). The control groups received the same vehicle containing DMSO instead of the active compound. All other chemicals were obtained from standard sources.
Mice and paclitaxel-induced peripheral neuropathy
Adult male C57BL/6J mice (12 weeks) were purchased from Jackson Laboratories. Mice were housed in groups of three to five in 500 cm2 Plexiglas cages (Allentown) in the animal care facilities (22 °C, 12-h light-dark cycle) with a
Mechanical sensibility using the von Frey testing
Mechanical sensibility was determined using the Von Frey filament test, as previously reported.40,41 Briefly, mice were given 30 min to acclimate to individual Plexiglass cages with mesh metal flooring prior to testing (Bioseb-PVF, Bioseb, Chaville, France). Paw withdrawal thresholds were measured using a range of calibrated von Frey filaments (Stoelting Co, Wood Dale, IL) with logarithmically incremental force from 0.166 to 5.88 expressed as dsLog10 of [10-pound force in milligram] that were applied to the hind paws of the mouse. If a paw withdrawal response was not observed (paw withdrawal, licking, shaking, jumping) to a tested filament, a thicker/heavier gram filament was tested. Once a withdrawal response was recorded, the next weaker filament was tested. Each filament was applied vertically against the center of the hind paw with sufficient force to cause slight bending to the filament that was then held for 2–3 s. The stimulation of the same intensity was repeated three times at intervals of a few seconds. For experiments with the CGRP receptor antagonist, mechanical hypersensitivity was evaluated 7 days after the final cumulative dose of paclitaxel at 1h, 3h, 6h, and 24h following administration (i.p) of BIBN4096 (2.5 or 5 mg/kg), or vehicle. Mechanical hypersensitivity experiments shown in Figure 1(b) and Figure 7(a) were conducted by different experimenters, which may account for the variation in baseline mechanical threshold observed between these two cohorts of mice. 42

Paclitaxel-induced mechanical hypersensitivity in mice. (a) Timeline of the experimental design. Mice received (i.p.) paclitaxel (8 mg/kg) or vehicle on four alternative days (days 1, 3, 5 and 7; cumulative dose 32 mg/kg). Mechanical hypersensitivity with the Von Frey test was tested at days 1, 4, 7, 10 and 15. (b) Treatment with paclitaxel induced mechanical allodynia in mice (time F[4,112] = 5.64,
Cold sensitivity using acetone
Cold sensitivity was evaluated using the acetone test, as previously reported. 41 Mice were given 30 min to acclimate to individual Plexiglass cages with mesh metal flooring prior to testing (Bioseb-PVF, Bioseb, Chaville, France). A 10 µl volume of acetone (Sigma-Aldrich, MO, USA) was applied to the central surface area of each hind paw using a pipette. The duration of licking, shaking, or lifting of the hind paw was recorded in a 1 min period. For experiments with the CGRP receptor antagonist, cold sensitivity was evaluated 7 days after the final cumulative dose of paclitaxel at 1, 3, 6, and 24 h following administration (i.p) of BIBN4096 (2.5 or 5 mg/kg), or vehicle.
Tissue collection
Mice were sacrificed by cervical dislocation. Tissue samples from the entire lumbar spinal cord and DRG were collected as previously reported,40,43 and stored at −80ºC until use.
RNA-seq and differential analysis
RNA was purified from lumbar spinal cord samples (see above) using RNeasy mini kit (Qiagen). Total RNA (5 µg) was used for mRNA library construction following instructions of Illumina RNA sample kit. All libraries were sequenced on an Illumina HiSeq 2500 platform. Sequencing data were processed using the Nextflow rnaseq v.3.14.0 pipeline, the mouse mm10 reference genome, and the STAR-RSEM gene quantification method. RNA-seq counts were analyzed for differential expression using the edgeR v.4.2.0 R package.
44
Real-time quantitative PCR
Quantitative real-time PCR (RT-qPCR) assays were carried out as previously described. 48 Primer pairs sequences are listed in Supplemental Table S1.
Chromatin immunoprecipitation assay
Chromatin immunoprecipitation (ChIP) experiments were performed as previously reported48,49 with minor modifications using MAGnify Chromatin Immunoprecipitation System (Invitrogen). Briefly, 2 µl of either 1 mg/ml rabbit anti-H3K4me2 antibody (Active Motif 39141), rabbit anti-MLL2 antibody (Abcam ab224156), or rabbit negative control IgG (Millipore PP64B) antibodies were conjugated to beads (Protein A/G Dynabeads, Invitrogen) and tested following manufacturer’s instructions. Primer pairs sequences are listed in Supplemental Table S2.
Statistical analysis
Statistical analyses were performed with GraphPad Prism software version 9. No statistical methods were used to predetermine sample sizes, but our sample sizes are similar to those reported in our previous publications. Animals were randomly allocated into the different experimental groups. Experimenters were blind to experimental conditions throughout. Datapoints were excluded based on previously established criterion and were set to ± 2 s.d. from the group mean. Statistical significance of experiments involving three or more groups and two or more treatments was assessed by two-way ANOVA followed by Bonferroni’s multiple comparison test. Statistical significance of experiments involving time courses was assessed by two-way repeated measures ANOVA followed by Bonferroni’s multiple comparison test. Statistical significance of experiments involving three or more groups was assessed by one-way ANOVA followed by Bonferroni’s multiple comparison test. Statistical significance of experiments involving three or more comparisons in two groups was assessed by multiple unpaired
Results
Paclitaxel induces mechanical hypersensitivity in mice
We first evaluated whether paclitaxel induced mechanical hypersensitivity at different time points (Figure 1(a)). Assessment of mechanical hypersensitivity using the Von Frey filaments test demonstrated a significant decrease in the paw withdrawal threshold in paclitaxel-treated mice compared to control animals at day 7 (
Paclitaxel upregulates Calcrl mRNA in the lumbar spinal cord
To evaluate the consequences of paclitaxel exposure, we performed poly(A)+ RNA sequencing in lumbar spinal cord samples from mice treated with paclitaxel versus vehicle. Mice that received paclitaxel showed a list of 26 differentially expressed genes (DEGs) in comparison to vehicle-treated mice (Figure 2 and Supplemental Table S3). Among these genes are those previously identified as key regulators of neuropathic pain, such as

Comparative heat map displaying genes differentially expressed in the spinal cord of mice. (a) Mice received (i.p.) paclitaxel (8 mg/kg) or vehicle on four alternative days (days 1, 3, 5 and 7; cumulative dose 32 mg/kg), and were sacrificed on day 15 for tissue sample collection. (n = 5 mice per library).

Gene set enrichment analysis of DEGs in spinal cord samples of paclitaxel-treated mice and controls. (a,b) KEGG pathways (a), and ontology gene sets (b) from the Molecular Signatures Database (MSigDB).
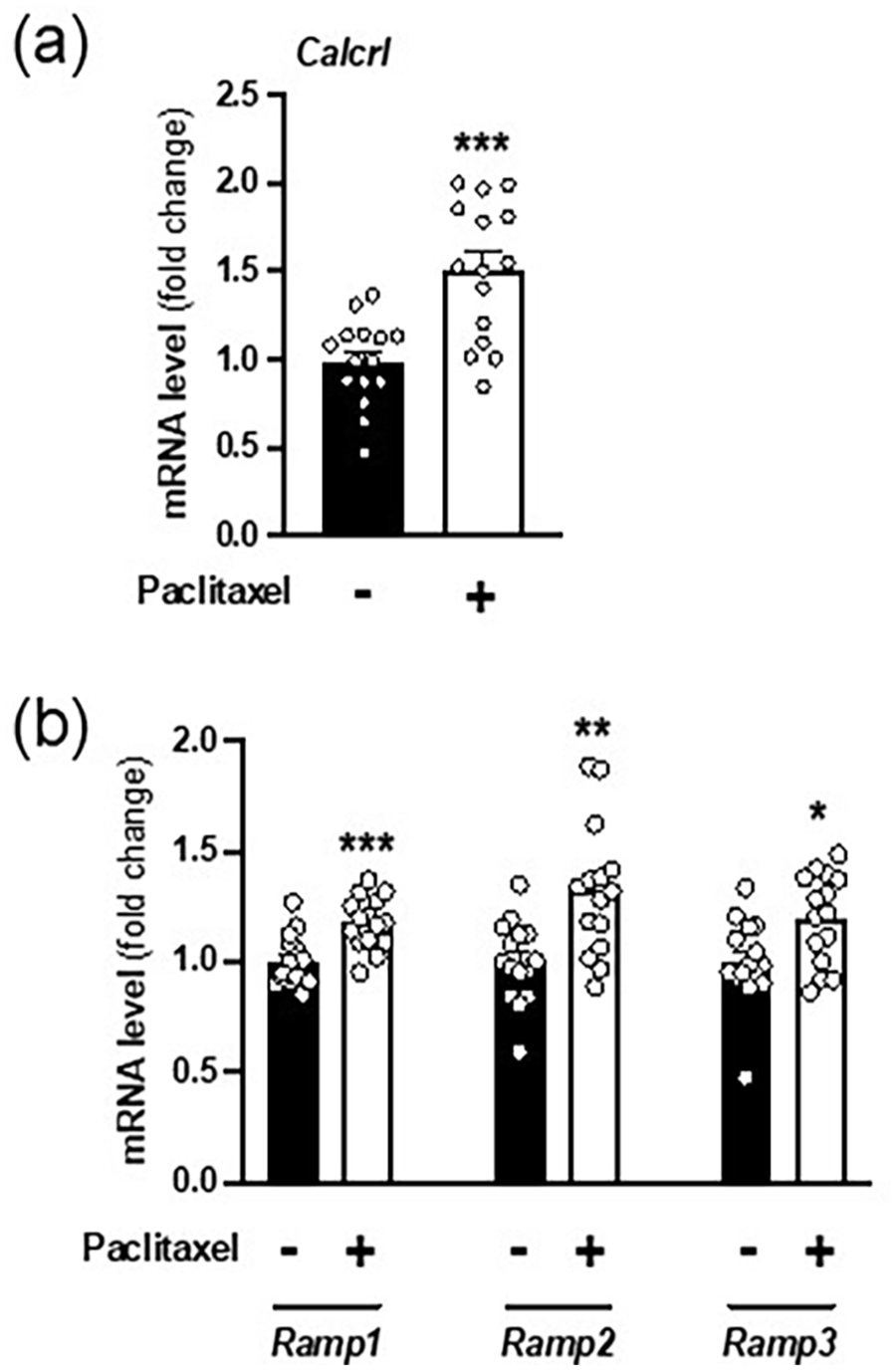
Upregulation of

Exposure to paclitaxel does not affect expression of neuropeptides in the spinal cord or the DRG. (a and b) Mice received (i.p.) paclitaxel (8 mg/kg) or vehicle on four alternative days (days 1, 3, 5 and 7; cumulative dose 32 mg/kg), and sacrificed on day 15 for tissue sample collection. (a) Expression of
Paclitaxel induces permissive histone modifications at the Calcrl promoter via MLL2
As mentioned above, previous research using ChIP-seq assays revealed changes in the methylation patters of H3K4 at specific promoters within limbic regions involved in rodent models of neuropathic pain.
38
We therefore profiled numerous lysine methyltransferases (KMTs) that are known to control H3K4 methylation.
51
Only

Epigenetic alterations at the
Pharmacological inhibition of the CGRP receptor mitigates hypersensitivity
We next tested whether the CGRP receptor antagonist BIBN4096 would reduce mechanical and cold hypersensitivity in paclitaxel-treated male mice. As above, repeated administration of paclitaxel induced mechanical allodynia in mice at day 15 (Figure 5(a)). Factorial analysis revealed significant impact of BIBN4096 treatment on paclitaxel-induced mechanical hypersensitivity (F[20, 175] = 6.9,

Effect of BIBN4096 on paclitaxel-induced mechanical and cold hypersensitivity. (a-b) Mice received (i.p.) paclitaxel (8 mg/kg) or vehicle on four alternative days (days 1, 3, 5 and 7; cumulative dose 32 mg/kg). Mechanical hypersensitivity with the Von Frey test (a) and cold hypersensitivity with the acetone test (b) were tested 7 days after the last cumulative dose of paclitaxel at 1, 3, 6 and lastly 24 h, at different at different time points after administration of BIBN4096, or vehicle. Reversal of mechanical (
Discussion
In our study, we observed upregulation of
Several studies have demonstrated an association between alterations in histone modifications and gene expression in the development of neuropathic pain. For instance, a recent study on cells treated with lipopolysaccharide (a characteristic component of Gram-negative bacteria and powerful mediator of systemic inflammation
65
) reported an enhanced recruitment of H3K4me2 at the promoter of genes of proinflammatory mediators such as
The MLL family of methyltransferases comprises several proteins that catalyze the transfer of methyl groups (mono-, di-, or tri-) to the fourth residue of histone 3 protein (H3K4), resulting in RNA polymerase II-mediated gene regulation.53,69 Among these proteins, we observed a significant upregulation of MLL2, also known as KMT2B. A recent study found that paclitaxel increases the activation of the protein arginine methyltransferase 5 (PRMT5) in the DRG leading to increased transcription of the
It is essential to remark the potential limitations of our study using mouse preclinical models. First, the use of a chemotherapy-only animal model may not fully replicate the clinical scenario, as cancer patients frequently experience pain resulting from both the tumor and the treatment. However, by employing a chemotherapy-only model, we can directly attribute the observed changes to paclitaxel. Additionally, our study exclusively included male mice. Research has demonstrated the female sex hormones can influence CGRP expression, 71 which in turn affects pain responses and treatment efficacy. Further, a growing body of evidence suggest that the role for CGRP in rodent pain models is sex-specific manner.71,72 Future investigations should incorporate female mice to capture a boarder spectrum of biological variability and enhance the development of effective pain treatments. Neuropathic pain is a multifaceted phenomenon that includes stimulus-evoked responses, spontaneous ongoing pain, and operant behaviors affected by pain. 73 While we focused on evoked-stimulus measures – specifically mechanical and cold hypersensitivity – this emphasis should not diminish the significance or our findings; rather, they should be interpreted within the context of these particular experiments. Finally, we selected the lumbar spinal cord for our investigations due to its vital role in the central nociceptive circuit and its relevance to stimulus-evoked measures. This CNS region also exhibits high expression levels of the CGRP receptor. 18 Future studies should extend their focus to include different subregions of the spinal cord as well as CNS areas such as the ventral thalamus, somatosensory cortex, and limbic system, to gain a more comprehensive understanding of pain mechanism.
Our study suggests that mitigating pain sensitivity using a mouse model of CIPN can be achieved by targeting the CGRP receptor with a pharmacological antagonist. Additionally, we propose that epigenetic modifications, such as an increase in K3K4me2 mediated via MLL2, may be implicated in the upregulation of the
Supplemental Material
sj-docx-1-mpx-10.1177_17448069251314857 – Supplemental material for Upregulation of the neuropeptide receptor calcitonin receptor-like in the spinal cord via MLL2 in a mouse model of paclitaxel-induced peripheral neuropathy
Supplemental material, sj-docx-1-mpx-10.1177_17448069251314857 for Upregulation of the neuropeptide receptor calcitonin receptor-like in the spinal cord via MLL2 in a mouse model of paclitaxel-induced peripheral neuropathy by Salvador Sierra, Sara M Herz, Doan On, Mikhail G Dozmorov, M Imad Damaj and Javier Gonzalez-Maeso in Molecular Pain
Supplemental Material
sj-xlsx-2-mpx-10.1177_17448069251314857 – Supplemental material for Upregulation of the neuropeptide receptor calcitonin receptor-like in the spinal cord via MLL2 in a mouse model of paclitaxel-induced peripheral neuropathy
Supplemental material, sj-xlsx-2-mpx-10.1177_17448069251314857 for Upregulation of the neuropeptide receptor calcitonin receptor-like in the spinal cord via MLL2 in a mouse model of paclitaxel-induced peripheral neuropathy by Salvador Sierra, Sara M Herz, Doan On, Mikhail G Dozmorov, M Imad Damaj and Javier Gonzalez-Maeso in Molecular Pain
Supplemental Material
sj-xlsx-3-mpx-10.1177_17448069251314857 – Supplemental material for Upregulation of the neuropeptide receptor calcitonin receptor-like in the spinal cord via MLL2 in a mouse model of paclitaxel-induced peripheral neuropathy
Supplemental material, sj-xlsx-3-mpx-10.1177_17448069251314857 for Upregulation of the neuropeptide receptor calcitonin receptor-like in the spinal cord via MLL2 in a mouse model of paclitaxel-induced peripheral neuropathy by Salvador Sierra, Sara M Herz, Doan On, Mikhail G Dozmorov, M Imad Damaj and Javier Gonzalez-Maeso in Molecular Pain
Footnotes
Authors contributions
S.S., I.M.D., and J.G.-M. conceived and designed experiments. S.S., I.M.D., and J.G.-M. analyzed the data and wrote the manuscript. S.M.H., supervised by I.M.D., conducted behavior assays. D.O., supervised by J.G.-M., helped with gene expression assays. M.G.D. conducted transcriptomic data analysis. J.G.-M. supervised the research and obtained funding. All authors reviewed and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.G.-M. has as sponsor research contract with Terran Biosciences.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH R01MH084894 to J.G.-M.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
