Abstract
We used functional MRI and a longitudinal design to investigate the brain mechanisms in a previously reported estrogen-dependent visceral hypersensitivity model. We hypothesized that noxious visceral stimulation would be associated with activation of the insula, anterior cingulate cortex, and amygdala, and that estrogen-dependent, stress-induced visceral hypersensitivity would both enhance activation of these regions and recruit activation of other brain areas mediating affect and reward processing. Ovariectomized rats were treated with estrogen (17 β-estradiol, E2) or vehicle (n = 5 per group) and scanned in a 7T MRI at three different time points: pre-stress (baseline), 2 days post-stress, and 18 days post-stress. Stress was induced via a forced-swim paradigm. In a separate group of ovariectomized rats, E2 treatment induced visceral hypersensitivity at the 2 days post-stress time point, and this hypersensitivity returned to baseline at the 18 days post-stress time point. Vehicle-treated rats show no hypersensitivity following stress. During the MRI scans, rats were exposed to noxious colorectal distention. Across groups and time points, noxious visceral stimulation led to activations in the insula, anterior cingulate, and left amygdala, parabrachial nuclei, and cerebellum. A group-by-time interaction was seen in the right amygdala, ventral striatum-pallidum, cerebellum, hippocampus, mediodorsal thalamus, and pontine nuclei. Closer inspection of the data revealed that vehicle-treated rats showed consistent activations and deactivations across time, whereas estrogen-treated animals showed minimal deactivation with noxious visceral stimulation. This unexpected finding suggests that E2 may dramatically alter visceral nociceptive processing in the brain following an acute stressor. This study is the first to examine estrogen-stress dependent interactions in response to noxious visceral stimulation using functional MRI. Future studies that include other control groups and larger sample sizes are needed to fully understand the interactions between sex hormones, stress, and noxious stimulation on brain activity.
Keywords
Background
Psychological factors including stress and/or anxiety can increase pain sensitivity. Many patients with functional pain disorders, including irritable bowel syndrome (IBS), report recent exposure to significant stress.1–3 Previous studies in patients with IBS have also shown that visceral stimulation leads to altered activity in emotional-arousal and stress-related circuits including the anterior cingulate cortex (ACC), insula, primary somatosensory cortex, medial thalamus, and hippocampus; thereby supporting changes in supraspinal processing contributing to functional pain.4–8
Stress is a common trigger for IBS symptoms, and several hypotheses have been put forth to explain the neurobiological mechanisms contributing to the pathophysiology of this syndrome including disruptions in gut-brain axis signaling.9,10 Previous research has demonstrated that psychological factors such as stress and anxiety can increase the severity of GI-related symptoms and that these factors serve as good predictors for the development of this syndrome, as well as other chronic pain conditions such as temporomandibular disorder (TMD).1–3,11,12 Indeed, IBS patients report that stress exacerbates their abdominal pain and discomfort,4,13 and stress-induced alterations in colonic motility and heightened visceral sensitivity in these patients have been documented. Moreover, females with IBS report greater stress or anxiety compared to males with IBS or healthy controls, and the incidence of comorbid conditions including mood and stress-related psychiatric disorders are high in this patient population.14,15 This condition is also more prevalent among females, suggesting that sex-related hormonal influences on visceral pain sensitivity may, in part, underlie the pathophysiology of IBS.10,16,17
The brain mechanisms underlying stress-induced visceral hyperalgesia are unknown. In the current study, we focus on the ACC, insula, and amygdala, as putative sites involved in mediating altered gut-brain signaling and changes in visceral sensitivity often described in the IBS literature. Cingulate and insular activations are commonly reported in neuroimaging studies performed in humans with various chronic pain conditions 18 and in rodent models. 19 Patients with IBS have abnormal insula activation to visceral stimulation, 7 and hypersensitive IBS patients have increased insula activity compared to non-hypersensitive IBS patients. 20 The insula is considered to be important in aspects of pain affect, intensity coding, and descending modulation (e.g., via connections with periaqueductal gray, PAG, via the amygdala), 21 as well as interoceptive awareness and the integration of viscerosensory and motor responses.22,23 In rats, stress induced by a forced-swim paradigm has been shown to increase inflammatory hyperalgesia and cFos expression in the insula cortex. 24 Bilateral lesioning of this structure inhibits the development of visceral hypersensitivity to chronic partial restraint, 25 suggesting the insula is necessary for stress-induced hyperalgesia. The ACC is involved in many aspects of pain processing, modulation, and affective components of chronic pain.26–28 The amygdala also plays an important role in the pain response, and inflammation-mediated hyperalgesia has been shown to be dependent upon activation of the right amygdala. 29 In rodent neuroimaging studies, the amygdala is typically activated by noxious visceral stimulation30–32 and activation of the amygdala, either with the stress hormone corticosterone or optogenetically, increases anxiogenic and visceral pain-like behaviors.32,33
In the current study, we aimed to use functional MRI (fMRI) to examine brain activation associated with colorectal distension (CRD) and the role of gonadal hormones in a model of visceral hypersensitivity in rats. Rodent neuroimaging has been useful in identifying changes associated with the development of chronic pain models.34–36 Here, we examined visceral hypersensitivity-related activity of the whole brain with a particular focus on the ACC, insula, and amygdala. We hypothesized that noxious visceral stimulation would be associated with activation of the aforementioned brain areas, previously reported to be activated during CRD in both humans and animals. We further hypothesized that estrogen-dependent visceral hypersensitivity would enhance activation of these regions and also recruit other brain areas involved in affect and reward processing.
Methods
Animals
Female Sprague Dawley rats (200–225 g) were purchased from Harlan (Frederick, MD). Rats were housed in ventilated plastic cages with soft bedding and maintained on a 12/12 h light/dark cycle (lights on at 07:00). Rats were fed standard rat chow and had access to tap water ad libitum. All procedures were approved by the Institutional Animal Care and Use Committee at the University of Maryland and performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Surgical procedures
Rats were ovariectomized prior to the baseline scan for fMRI (n = 10) or visceromotor response (VMR) testing (n = 7) and singly housed following surgery for the duration of the study. Rats were anesthetized with isoflurane delivered by a precision vaporizer (5% initially by nose cone, then reduced to 2%) and ovariectomized by a dorsolateral approach using standard surgical procedures under aseptic conditions. For rats undergoing VMR testing, electromyogram (EMG) electrodes made from Teflon-coated 32 gauge stainless steel wire (Cooner Wire Company, Chatsworth, CA) were stitched into the ventrolateral abdominal wall. The electrode leads were tunneled subcutaneously and exteriorized at the back of the neck. Rats were given at least 10 days for recovery.
Hormone treatment
17β-estradiol (E2; 50 µg in 100 µl safflower oil) or vehicle (safflower oil) was injected subcutaneously (s.c.) every four days (see Figure 1). This mimics the fluctuation of estrogen during the estrous cycle in intact rats.
33
Scans and VMR recordings were made 24 hours (h) following E2 or vehicle injection.
Schematic illustrating the study timeline. Rats were ovariectomized (OVx) and electrode implantation for EMG recordings was performed 15 days prior to the baseline (pre-stress) fMRI scan or visceromotor response (VMR) testing. Estradiol (E2; 50 µg in 100 µl safflower oil) or vehicle (oil; safflower) was injected s.c. every four days. Stress was produced by a three-day forced-swim (FS) paradigm. Three scans were performed for each rat: baseline (pre-stress), 2 days post-FS, and 18 days post-FS. In separate groups of E2 and oil-treated rats, VMR testing was performed at these same study time points (baseline, 2 days post-FS; 18 days post-FS).
Stress induction
Stress was produced by a three-day forced-swim paradigm starting one day following the E2/vehicle injection. This stress paradigm induced thermal hyperalgesia, facilitated inflammation induced hyperalgesia and induced chronic visceral hypersensitivity when paired with orofacial inflammation.24,37,38 Rats were placed in a rectangular Plexiglas container (30 cm × 30 cm × 50 cm) that contained 20 cm of water maintained at 25℃–26℃. Half the water was changed between rats, and all fecal pellets were removed. At the start of the experiment, rats were placed in the center of the tank and released. The swim duration was 10 min on the first day and 20 min on the following two days. At the end of the swim, rats were patted dry with paper towels and placed in a cage with warm air blowing in part of the cage. Rats were kept there for 15 to 20 min and then returned to their home cage.
CRD and the visceromotor response
CRD was performed as previously described.39–41 Rats were fasted for 18 to 24 h to allow balloon placement. Water was available ad libitum. Briefly, under isoflurane sedation, a 5 to 6 cm balloon attached to tygon tubing was inserted into the colorectum. The distal end of the balloon was at least 1 cm proximal to the external anal sphincter and the tubing was taped to the tail. For the VMR recordings, rats were loosely restrained in plastic rodent restrainers and given 30 min to recover from sedation. The EMG signals were recorded with a CED 1401 and analyzed using Spike 2 software (Cambridge Electronic Design, Cambridge, UK). CRD was produced by computer-controlled inflation of the distention balloon (three trials, eight distentions per trial, two each at 20, 40, 60, 80 mmHg, 20 s duration, 3 min interstimulus interval). The EMG was analyzed by rectifying the signal and subtracting background activity of the same duration before the distention stimulus. The mean response was calculated for each pressure for the last two trials and the four responses were summed to obtain an overall VMR value.
Experimental design
Figure 1 illustrates the experimental design and study time line. Three fMRI scans or VMR recordings were performed for each rat: baseline (prior to stress), 2 days post-stress, and 18 days post-stress. fMRI scans were performed during CRD with 80 mmHg, a pressure which has been shown to reliably produce a VMR response indicative of noxious stimulation. 42 We performed statistical analysis for the group-by-time interactions of whole-brain activity related to CRD.
fMRI data acquisition
Data were acquired using a Bruker BioSpec 70/30USR Avance III 7-Tesla scanner (Bruker Biospin MRI GmbH, Germany). The system was equipped with a BGA12S gradient system and interfaced to a Bruker Paravision 5.1 console. A Bruker 40 mm circular polarized volume coil was used for acquisition. Isoflurane concentrations were kept below 1.5% and maintained throughout the scan session. During each scan session, respiration and heart rate were continuously monitored using a small animal monitoring and gating system (SA Instruments, Inc., Stony Brook, NY, USA). The body temperature was maintained at 36–37℃, using a circulating warm water heater. A high resolution T1-weighted image was obtained (RARE, TR = 2000 ms, TE = 14 ms, 256 × 256, in plane resolution = 100 µm, 24 axial slices, 1 mm slice thickness) for anatomical reference. Functional scans were acquired using a spin-echo echo-planar imaging (EPI) sequence (TR = 1500 ms, TE = 35.096 ms, 75 × 75, in plane resolution = 0.35 × 0.35 × 1 mm, 24 axial slices; 620 volumes). fMRI scans were performed during CRD (80 mmHg for 15 s with a 60 s interstimulus interval, 15 trials, ∼15 min, TR = 1.5 s, resolution = 0.35 × 0.35 × 1 mm). Distention was produced by rapidly depressing a syringe plunger connected to the balloon. Pressure was monitored in parallel by a pressure gauge.Three scans were performed for each rat in each group: baseline (no visceral hypersensitivity in either group), 2 days post-stress (E2-dependent visceral hypersensitivity in E2 group only), and 18 days post-stress (no visceral hypersensitivity in either group). During scanning, CRD was performed by inserting a 5- to 6-cm balloon attached to Tygon tubing inserted into the descending colon and rectum (see previous section for details of the CRD procedure).
fMRI preprocessing and analysis
Data were preprocessed using SPM8 (http://www.fil.ion.ucl.ac.uk/spm/) and MATLAB (R2012b, The MathWorks, Natick, MA). Functional images were slice time corrected and realigned to the mean image. A study-specific template was created by co-registering and averaging the 30 (n = 5 rats × 2 groups × 3 time points) high-resolution T1-weighted images. Subsequently, all functional scans were co-registered and normalized to this template using previously described methods. 35 Functional images were interpolated to a voxel size of 0.5 mm isotropic during normalization, then smoothed with a 1 mm full-width at half maximum (FWHM) Gaussian kernel.
At the single-subject level, functional images were high-pass filtered (0.0078 Hz), and the time series was submitted to a general linear model. In this model, the onsets, offsets, and durations of the inflation and deflation periods were specified as regressors and convolved with the canonical hemodynamic response function. Additionally, the standard six movement parameters associated with head movement were modeled as regressors of no interest. Due to the high degree of overlap between the task and head motion, 12 supplementary regressors of no interest were created to account for the possible delayed effects of motion on the blood oxygenation level dependent (BOLD) signal. Specifically, two additional sets of six motion parameters were created by “lagging” the original motion parameters by one or two TRs, respectively. 43
Second level analyses included a one sample t test to assess overall activation patterns related to CRD and a flexible factorial model to assess the interaction effects of group and time on CRD-related activity. The one sample t test included the averaged (across time points) activation maps for each rat. The flexible factorial model included time and group main effects and their interaction. The contrast tested the group-by-time interaction effect. Because of the small sample size and exploratory nature of the study, second level maps were voxel-wise thresholded at p < 0.05 and cluster corrected for multiple comparisons at p < 0.05. In addition, given our a priori hypotheses regarding insula, ACC, and amygdala activation, we report results in these regions at uncorrected thresholds.
Results
Stress-induced visceral hypersensitivity
Behavior testing included four rats in the E2 group and three rats in the vehicle group. Visceral sensitivity was recorded prior (baseline; pre-stress) to and 2 and 18 days following the forced-swim stress paradigm. For the VMR tested animals, there were significant treatment effects (two-way repeated measures ANOVA, p < 0.05) two days following forced-swim in the E2-treated rats, which returned to baseline by 18 days, but there was no stress-induced visceral hypersensitivity in the vehicle-treated (oil) rats (Figure 2).
The magnitude of the visceromotor response (VMR) in estradiol (E2) and vehicle-treated (oil) rats. (a) Stimulus-response curves in E2 (red, n = 4) and oil (blue, n = 3) treated rats. The mean curve at baseline (open square), 2 days post-stress (closed square) and 18 days post-stress (closed circle) are shown. (b) The area under the curve (AUC) after data were normalized to the baseline response to 80 mmHg. At 2 days post-stress, there was visceral hypersensitivity in the E2 (red), but not oil-treated (blue) rats. The visceral sensitivity returned to baseline in the E2 rats by 18 days post-stress. *p < 0.05 versus baseline; #p < 0.05 versus oil.
Neuroimaging results
Neuroimaging was performed on a separate group of rats and included five rats in the E2 and five rats in the vehicle group. Two scans were not usable because of excess motion and were excluded from the analysis. Excluded scans consisted of EPI images from time point 2 for a rat in the E2 group and time point 3 for a rat in the vehicle group. Therefore, a total of 28 out of the 30 scans were included in the analysis.
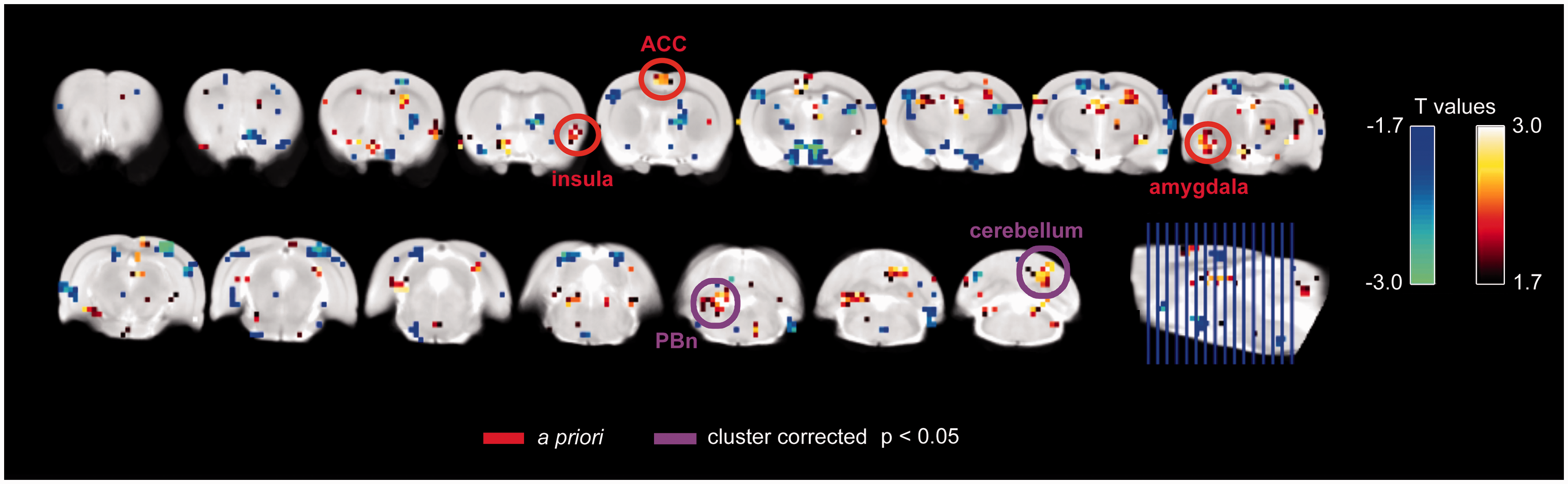
Across time, CRD induced functional activation in the ACC and insula. An example of ACC and bilateral insula activation with distension is shown in Figure 3 for a single rat. Group maps revealed activations in a priori regions, which included the ACC, right insula, and left amygdala, although these regions did not reach cluster corrected significance (Figure 4). Two regions that did meet correction criteria included the right cerebellum and left rostral brainstem. Activations and deactivations at lower cluster corrected thresholds are reported in Supplemental Figure 1.
Example of colorectal distention-related fMRI activation in a single rat. Activation map from a single rat (left panel), highlighting distention-related activation in the bilateral insula and right cingulate cortex. The plot (right panel) shows the hemodynamic response function (HRF) convolved stimulus (red) and the blood oxygenation level dependent (BOLD) signal (blue) for the right insula. Map is thresholded at t > 3. Colorectal distention-related activation across E2 (n = 5) or vehicle (n = 5) groups of rats. All 10 rats (E2 + vehicle groups combined) were included in the analysis, with data averaged within each rat over the three time points. Shown at p < 0.05 with significant clusters and a priori regions indicated. Abbreviations: ACC = anterior cingulate cortex; PBn = parabrachial nucleus.
The full interaction model revealed increased (Group × Time) activation in the right amygdala for E2 relative to vehicle-treated rats (E2 > vehicle), and significant clusters in the right ventral striatum-pallidum (extending to the nucleus accumbens), left superior and inferior colliculi/V1M/V2L (primary and secondary visual cortices, medial and lateral areas, respectively), and cerebellum. The Group × Time analysis for the reverse contrast (vehicle > E2) yielded significant clusters in the midline mediodorsal thalamus/hypothalamus, right hippocampal formation (postsubiculum), and right deep mesencephalic nuclei (DpMe). These results are shown in Figure 5. Activations and deactivations at lower cluster corrected thresholds are reported in Supplemental Figure 2. Because this was an exploratory study, we present activation maps for each group at each time point, as well as cluster corrected results at p < 0.1 and p < 0.2 in Supplemental Figure 3. The relative activations in E2 versus vehicle group, despite the sparsity of activation in the E2 group, could reflect a difference between groups that is slightly positive in E2 rats and slightly negative in vehicle rats, or strongly negative in vehicle rats.
Results from the full interaction model (group-by-time). Flexible factorial model full interaction [increasing activation over time in E2 rats (n = 5, n = 4 at 2 days time point) relative to decreasing activation over time in vehicle rats (n = 5, n = 4 at 18 days time point)]. Shown at p < 0.05 with significant clusters and a priori regions indicated. Abbreviations: VP = ventral palladus; CPu = putamen; MD = mediodorsal thalamus; DpMe = deep mesencephalic nuclei.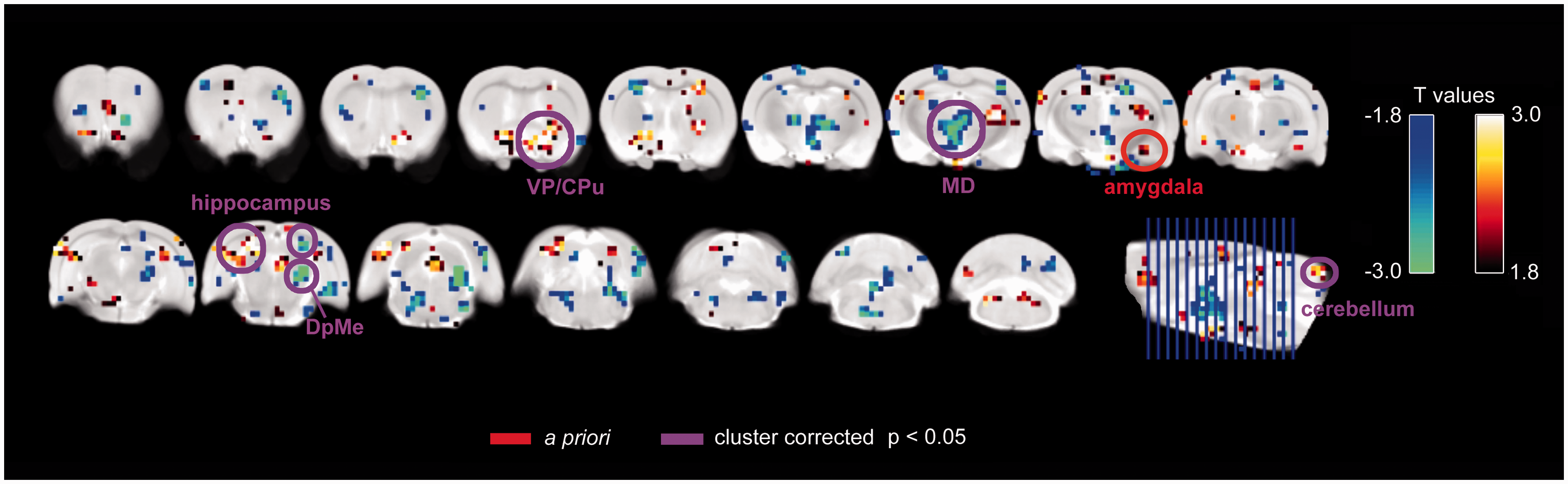
Discussion
The main objective of the current study was to examine the effects of stress and gonadal hormones in a model of estrogen-dependent visceral hypersensitivity in female rats using fMRI. We hypothesized that ACC, insula, and amygdala would have important roles in visceral hypersensitivity and would be modulated by our stress induction model in estrogen-treated rats. Across all rats, there was activation of the ACC, right insula, and left amygdala based on our a priori criteria. While insula and ACC were strongly activated in some rats during CRD, this activation pattern was not consistent (uncorrected ACC, insula, and amygdala clusters are evident at the whole group level, indicating a trend toward activation).
As predicted, E2-dependent hypersensitivity was associated with an enhanced response of the right amygdala and ventral striatum-pallidum, and decreased activity in the medial thalamus, mesencephalic nuclei, and cerebellum. The activation of the amygdala during visceral stimulation is consistent with the literature,30–32,44 and in particular, this enhancement in activity of the right amygdala in stressed E2 rats fits with a lateralized response to the right amygdala to pain-like behaviors in mice. 29 Furthermore, our findings of ventral striatum-pallidum activation is in accord with a growing literature implicating the ventral striatum and nucleus accumbens in chronic pain states.45,46 Decreased activation of the medial thalamus, hippocampus, brainstem regions could reflect a dysregulation in descending modulatory circuits. 47 The cerebellum might be an important structure in chronic pain conditions because of its sensorimotor functions and role in aversive learning. 48
Some limitations to this study should be noted. First, to have confidence in these findings, the current results will need to be replicated with more control groups—including intact males and females with larger sample sizes. For example, the effects of E2 alone on the CRD-related VMR (Figure 2, baseline time point) and fMRI response (Supplemental Figure 3) are apparent in our data and could affect nociceptive processing in multiple ways. Second, the data generally had more motion than in our previous experiments35,36 because of the nature of the stimulus, which results in passive motion of the animal. While we did extensive cleanup of the data to account for motion, there were likely still residual motion-related artifacts. Future work incorporating additional control groups to isolate the main effects and interactions of sex hormones, stress, and chronic pain will strengthen the present findings. Additionally, ongoing work will examine the effects of interventions in reducing stress-dependent pain.
In summary, in this pilot study, we compared brain activity related to visceral stimulation in ovariectomized rats with or without estrogen replacement both before and after stress. This is the first fMRI study to show that estrogen-dependent stress-induced hyperalgesia is associated with altered brain activity in regions that would suggest increased affective response and decreased descending modulation. Future work including more control groups and sensitive behavioral tests are needed.
Conclusions
The major aim of the current study was to use a longitudinal fMRI paradigm to examine brain activation associated with CRD and the role of gonadal hormones in a rat model of visceral hypersensitivity. We showed that stress-induced activation of emotional-arousal and stress-related circuits are modulated, in part, by estrogen. Although preliminary, these findings show that E2-treated rats, compared to controls, showed enhanced BOLD signal responses in the right amygdala and ventral striatum, as well as decreased responses in the medial thalamus and brainstem regions, indicating a potential estrogen-dependent upregulation in stress-related emotional-arousal circuitry during noxious visceral stimulation. Only a few previous studies have examined brain activity associated with CRD with either fMRI 49 or autoradiography to measure cerebral blood flow.50–52 Thus, the current study contributes novel findings to our limited understanding of this topic. Understanding the brain circuitry involved in IBS using a rodent fMRI model could allow us to define targets for pharmacological and non-pharmacological therapies in humans.
Footnotes
Author contributions
CSH, JMK, and DAS ran the MRI studies; JMK performed all the animal procedures and surgeries; DAS, CSH, AJF, and JTS analyzed data and prepared figures; RJT and DAS designed the study; DAS, CSH, and RJT wrote the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported in part by seed funding from the Organized Research Center on Persistent Pain at the University of Maryland, Baltimore. Grant 2014/20983-1, São Paulo Research Foundation (FAPESP) to JTS.
