Abstract
Background
Millions of people suffered from neuropathic pain, which is related to neuroinflammation in the central nervous system. Penehyclidine hydrochloride is a premedication of general anesthesia, which has been confirmed possessing neuroprotective effects against various neurodegenerative or neuroinflammatory diseases. However, it is not clear that whether penehyclidine hydrochloride could suppress neuropathic pain through its anti-neuroinflammatory effects.
Methods
This study investigated the effects of penehyclidine hydrochloride on rat spinal nerve ligation injury-induced neuropathic pain with behavioral, morphological, and molecular biological methods in animals.
Results
The results indicated that penehyclidine hydrochloride could attenuate spinal nerve ligation-induced neuropathic pain without any motor impairment and had no effect on sham-operated animals after repeated intraperitoneal administration. Intraperitoneal penehyclidine hydrochloride could suppress spinal nerve ligation-induced ipsilateral spinal dorsal horn microglial activation with downregulation of OX42 expression. Moreover, intraperitoneal penehyclidine hydrochloride inhibited spinal nerve ligation-induced spinal p-p38 mitogen-activated protein kinase expression, which was specially colocalized with the spinal dorsal horn microglia. Furthermore, intraperitoneal penehyclidine hydrochloride could depress spinal neuroinflammation by suppressing spinal nerve ligation-induced interleukin (IL)-1β over-expression.
Conclusion
These results indicated that the anti-allodynic effects of penehyclidine hydrochloride on spinal nerve ligation-induced neuropathic pain did not rely on motor impairment. Inhibiting spinal microglial p-p38/IL-1β pathway activation might contribute to the anti-allodynic effect of penehyclidine hydrochloride on nerve injury-induced neuropathic pain.
Introduction
Millions of people suffered from neuropathic pain, which is defined as a type of chronic pain induced by peripheral or central nerve injury or degeneration. 1 The pathological process can be mediated through the activation of various cells (neurons, astrocytes, microglia, and Schwann cells), as well as the dissolution of distal axons. 2 It is urgent to developing an effective treatment for this intractable pain. However, the efficacy of current treatments with opioids, nonsteroidal anti-inflammatory drugs, and anticonvulsants are not sufficiently effective in reducing the pain and limited by the extensive side effects observed in patients, underlining the necessity for novel therapeutic targets. Evidence suggested a critical role played by glial cells in the mechanisms underlying the chronicity of pain, both at the site of damage in the periphery and in the dorsal horn of the spinal cord.3,4 Therefore, the involvement of glia should be considered in neuropathic pain treatment.
Thanks to decades of research, it is known that the aberrant excitability might not be a consequence merely of changes in neurons, but rather of multiple alterations in glial cells, such as microglia, the immune cells of the central nervous system (CNS). 5 Glia is activated after peripheral nerve injury and secretes lots of proinflammatory mediators, which played as key regulators during the development of neuropathic pain. This process involves many factors including neuroinflammation, ion channel opening, and ligand–receptor interactions. 6 Microglia are central players in mechanisms underlying neuropathic pain and might be potential targets for chronic pain treatment. 7 A recent study demonstrated that promoting spinal glial “alternative” polarization and restoring the balance between pro- and antinociceptive factors might be a novel therapeutic approach for painful neuropathy. 8
Penehyclidine hydrochloride (PHC) is widely used as a premedication drug of general anesthesia based on its selective anticholinergic effects on both antimuscarinic and antinicotinic activities in China. 9 Recently, PHC has been confirmed possessing neuroprotective effects on different neurodegeneration or neuroinflammation disease. PHC could decrease the frequency and amplitude of mEPSCs to protect hippocampus slice against oxygen glucose deprivation injury by decreasing excitatory amino acids release and increasing the release of inhibitory amino acids. 10 Another study also confirmed that PHC could protect neuronal damage by the regulation of N-methyl-D-aspartate (NMDA) receptor activity after cerebral ischemia. 11 Furthermore, PHC exhibited anti-inflammatory and protective effects by inhibiting toll-like receptor (TLR) activation. TLR is widely recognized as a pattern recognition receptor for triggering the innate immunity. 12 In addition, PHC showed improved neurological deficits and blood–brain barrier integrity and reduced infarction volume, brain water content, and apoptosis after cerebral ischemia/reperfusion (I/R) injury by suppressing p38 mitogen-activated protein kinase (MAPK) signaling pathway activity. 13 Interestingly, PHC significantly depressed the expression of pro-inflammatory factors in lipopolysaccharide (LPS)-activated microglia and effectively inhibited the translocation of nuclear factor (NF)-kappaB from the cytoplasm to the nucleus. The activities of NF-kappaB and p38 MAPK in LPS-treated microglia were significantly lowered after pretreatment of PHC. 14 It is concluded that PHC could inhibit neuroinflammation by blocking p38 MAPK and NF-kappaB pathways in microglia. This may offer a novel therapeutic option for PHC to confine microglial overactivation in neuroinflammatory diseases, including nerve injury-induced neuropathic pain.
Altogether, this study will detect the effect of PHC on spinal nerve ligation (SNL)-induced neuropathic pain. Besides, the MAPK and pro-inflammatory factors related to spinal microglial activation will be discussed to disclose the underlying mechanisms.
Material and methods
Animals
Male Sprague–Dawley (SD) rats (180–200 g) with food and water available were raised in plastic cages and kept on a 12:12 h light/dark cycle under conditions of 22°C–25°C ambient temperature. All procedures received approval from the Animal Use and Care Committee for Research and Education of the Fourth Military Medical University (Xi’an, China) and the ethical guidelines to investigate experimental pain in conscious animals. 15 Efforts were made to minimize animal suffering and the number of animals used.
Drug administration
PHC (1 mg/ml, No: 121107, Chengdu List Pharmaceutical Company, China) was diluted with preservative-free 0.9% NaCl before injection (2 mg/kg, intraperitoneally). Normal saline (0.9%) was used as the negative control. Animals were randomly divided into four groups for administration: Sham-Saline Group, SNL-Saline Group, SNL-PHC Group and Sham-PHC group. The PHC and normal saline were injected 1 h before SNL and once again on the postoperative day 1 (POD1). The dosage of PHC applied systemically in this study was based on previous reports13,14,16 and pilot experiment, which has no obvious side effects such as dizziness, urine retention, delirium, and rise in body temperature when given at a large dosage. 17
SNL animal model
To produce the SNL animal model, the left L6 transverse process of SD rat was removed to expose the L4 and L5 spinal nerves with pentobarbital anesthesia [45 mg/kg, intraperitoneally]. The L5 spinal nerve was isolated and ligated carefully with 6–0 silk thread tightly. 18 The surgical procedure for sham groups was identical to that of the SNL groups, except for SNL.
Behavioral tests
Rats were acclimatized to the experimental environment for three days before baseline assessment. Then, animals were put in inverted plastic boxes (30 × 30 × 50 cm3) with an elevated mesh floor 30 min before the threshold testing for habituation. A model 2390–5 IITC Electronic von-Frey Anesthesiometer (Woodland Hills, CA, USA; blunt polypropylene tip, diameter 0.5 mm) was used to detect mechanical allodynia threshold. A paw-flick reflex response was recorded as threshold by applying an increasing force (measured in gram) using a plastic filament focused on the middle of the plantar surface of the ipsilateral hindpaw. The force applied was initially below detection threshold, then increased from 1 to 50 g in 0.1 g steps over 20 s, then held for another 10 s at 50 g. The rate of the increase in force was 2.5 g/s. The cutoff value was 50 g. This was determinate as the mean of three measurements at 1 min intervals.
Immunofluorescence
Animals were perfused through the ascending aorta with 100 ml 0.9% saline followed by 500 ml 0.1 M phosphate buffer (PB, pH 7.3) that contained 4% paraformaldehyde and 2% picric acid after deep anesthesia (Pentobarbital, 60 mg/kg, intraperitoneally).The L5 spinal segment was then removed and postfixed in the same fixative solution for 2 to 4 h and cryoprotected for 24 h at 4°C in 0.1 M PB containing 30% sucrose. Transverse frozen spinal sections (30 µm thick) were collected serially by a cryostat (Leica CM1800; Heidelberg, Germany) within three dishes. The sections from one dish selected randomly were rinsed in 0.01 M phosphate-buffered saline (PBS, pH 7.3) three times (10 min each), blocked with 2% goat serum in 0.01 M PBS that contained 0.3% Triton X-100 for 1 h at room temperature, and then applied for immunofluorescence (IF).
The sections were incubated with primary antibodies: mouse anti-OX42 (1:500; Abcam, Cambridge, UK) and rabbit anti-phosphorylated p38 (p-p38) antibody (1:500; Cell Signaling Technology, Beverly, MA, USA) overnight at 4°C. Other primary antibodies were as follows: mouse anti-NeuN (1:3000; Chemicon, Temecula, CA, USA) and mouse anti-glial fibrillary acidic protein (GFAP) (1:5000; Chemicon). Sections were incubated with a mixture of two primary antibodies followed by a mixture of the two respective secondary antibodies (Alexa 488 donkey anti-rabbit IgG and Alexa 594 donkey anti-mouse IgG, 1:500; Invitrogen, Carlsbad, CA, USA) for double IF. Images were acquired with a confocal laser microscope (FV1000; Olympus, Tokyo, Japan, 1 µm thickness of optical section), and digital images were captured with Fluoview 1000 (Olympus).
Western blot
Animals were sacrificed rapidly, and the L5 dorsal horns were removed and frozen on dry ice promptly. The spinal dorsal horn was dissected using the open book method. 19 The selected region was homogenized with a hand-held pestle in sodium docyl sulfate (SDS) sample buffer (10 ml/mg tissue), which contained a cocktail of proteinase and phosphatase inhibitors. The electrophoresis samples were heated at 100°C for 5 min and loaded onto 10% SDS-polyacrylamide gels with standard Laemmli solutions (Bio-Rad Laboratories, CA, USA). The proteins were electroblotted onto polyvinylidenedifluoride membranes (Immobilon-P, Millipore, Billerica, MA, USA). The membranes were placed for 1 h in a blocking solution (containing Tris-buffered saline with 0.02% Tween (TBS-T) and 5% non-fat dry milk) and then incubated for two nights at 4°C temperature under gentle agitation with primary antibodies: rabbit anti-p-p38 antibody (1:1000; in 5% bovine serum albumin (BSA), Cell Signaling Technology), rabbit anti-p38 antibody (1:2000, in 5% BSA; Cell Signaling Technology), rabbit anti-interleukin (IL)-1β antibody (1:1000, NB600-633, Novus Biologicals, Littleton, USA), or mouse anti-β-actin (1:1000; Sigma, St Louis, MO, USA), respectively. Bound primary antibodies were detected with the anti-rabbit or anti-mouse horseradish peroxidase-conjugated secondary antibody (1:10,000; Amersham Pharmacia Biotech Inc., Piscataway, NJ, USA). After each step, the immunoblots were rinsed with TBS-T. All reactions were detected by the enhanced chemiluminescence detection method (Amersham). The densities of protein blots were analyzed by using Labworks Software (Ultra-Violet Products, UK). The densities of immunoreactive bands were quantified with background subtraction. A square of the same size was drawn around each band to measure the density, and the background near that band was subtracted. The density of specific p-p38 bands was measured and normalized against total p38 level. The density of IL-1β bands was measured and normalized against β-actin level.
Rotarod test
Rotarod tests were performed in order to assess whether the PHC used in the present experiment could influence motor function and behavioral tests or not. Animals with no previous exposures to rotarod test were put on the UgoBasile 7650 Rotarod accelerator treadmill (UgoBasile, Varese, Italy) set at the minimal speed for training sessions of 1 to 2 min at intervals of 30 to 60 min. After training, animals were placed on to the rotarod at a constant speed of 25 r/min. As the animal took a grip of the drum, the accelerator mode was selected on the treadmill, that is, the rotation rate of the drum was increased linearly at 20 r/min. Thereafter, the time was measured from the start of the acceleration period until animal fell off the drum. The cut-off time was 30 s. The time that the animal remained on the rotarod was recorded and expressed as a percentage of that animal’s own mean control performance. 20
Quantification and statistical analysis
Data were collected by researchers blinded to experiments and were presented as mean ± standard deviation.
Immunofluorescent data were collected according to the previous report. 21 Five nonadjacent sections (30 µm) from the L5 segments were selected randomly from each animal for the quantification of OX42 immunopositive cell profiles in the spinal cord with six rats in each group. Image (450 × 338µm 2 ) was captured under 20× objective on the medial two thirds of the superficial dorsal horn (laminas I–III). 22 All of the positively stained cells in the area were evaluated by a computer-assisted image analysis program (MetaMorph 6.1), which set the low and high thresholds for the immunofluorescent intensity that was determined to be a signal. The same configuration was used to measure cell areas in all experimental groups. The measured areas were transferred to Excel automatically for the following statistical analysis. MetaMorph 6.1 is calibrated to provide standardization of area measurements. A standardized field area was sampled arbitrarily from regions within randomly selected dorsal horn sections. 22 Data from IF were expressed as a fold change against that of Sham-Saline group. Analysis of variance (ANOVA) followed by the least significant difference test was used for statistical analysis.
Western blot (WB) data were analyzed using one-way ANOVA, followed by the least significant difference test. Behavioral test data were tested by repeated measures ANOVAs followed by Fisher’s protected least significant difference post hoc comparisons, where appropriate. SPSS® version 16.0 software (SPSS Inc., Chicago, IL, USA) was used for statistical analyses, and p < 0.05 was considered statistically significant.
Experimental protocols
The baseline of behavioral test and rotarod test was performed prior to PHC or normal saline administration on the SNL or Sham surgical day. SNL or Sham operation was conducted after PHC or normal saline administration on the POD0. PHC or normal saline was applied once a day on POD0 to POD1. Then, the behavioral test and the rotarod test were carried out on POD3. Finally, all rats were sacrificed for IF or WB experiments after the behavioral test on POD3 (Table 1).
The experimental protocols.

PHC: penehyclidine hydrochloride; POD: postoperative day; SNL: spinal nerve ligation; IF: immunofluorescence; WB: Western blot.
Results
The effect of intraperitoneal PHC on rotarod test
Nociceptive response may be easily affected by motor dysfunctions. Therefore, a group of SNL-free animals were assessed with the rotarod test to evaluate the effect of intraperitoneal PHC (2 mg/kg) on motor functions. The results indicated that repeated intraperitoneal PHC did not influence motor function compared with their own baseline and with the normal saline group (Figure 1). This result makes sure that the effects of PHC on SNL-induced neuropathic pain did not rely on motor impairment.

Effects of intraperitoneal PHC on motor performance with rotarod test. PHC or normal saline was given intraperitoneally to experiment-free rats, and the rotarod test was performed at POD1 and POD3 after the baseline response obtained at POD0. The score of each group was normalized as a percentage of the baseline value. There was no statistical difference obtained after PHC intraperitoneal injection compared with that of baseline in rotarod test. PHC: penehyclidine hydrochloride; POD: postoperative day.
Intraperitoneal PHC could attenuate SNL-induced neuropathic pain
After the baseline of mechanical allodynia conducted, PHC was injected intraperitoneally 1 h before SNL and once again on POD1. The effect of PHC on SNL-induced neuropathic pain was observed at POD3.
SNL induced significant mechanical allodynia on POD3 in the SNL-Saline group (p < 0.05, compared with that of Sham-Saline) (Figure 2). Intraperitoneal PHC suppressed SNL-induced mechanical allodynia apparently on POD3 after repeated administration (p < 0.05, compared with that of SNL-Saline). However, PHC had no effect on Sham-operated animals after intraperitoneal repeated administration. This result indicated that intraperitoneal PHC could exert anti-allodynia effect on SNL-induced neuropathic pain.

Effect of intraperitoneal PHC on neuropathic mechanical allodynia after SNL. SNL induced obvious mechanical allodynia on POD3 shown by the pain behavioral tests. PHC injection exerted an effective and reliable anti-allodynia effect on mechanical allodynia after SNL. #p < 0.05, compared with that of SNL-Saline. *p < 0.05, compared with that of Sham-Saline. PHC: penehyclidine hydrochloride; POD: postoperative day; SNL: spinal nerve ligation.
Intraperitoneal PHC could suppress SNL-induced ipsilateral spinal dorsal horn microglial activation
SNL apparently induced ipsilateral spinal dorsal horn microglial activation, indicated by the high expression of OX42 on POD3 (Figure 3, SNL-Saline group). Meanwhile, the results showed that spinal microglia were not sparked in the Sham-Saline group, and PHC had no effects on the expression of spinal microglial OX42 in the Sham-PHC group. However, intraperitoneal PHC could markedly suppress SNL-induced spinal dorsal horn OX42 expression (Figure 3, SNL-PHC group), which indicated that PHC could inhibit SNL-induced microglial activation during the development of neuropathic pain.
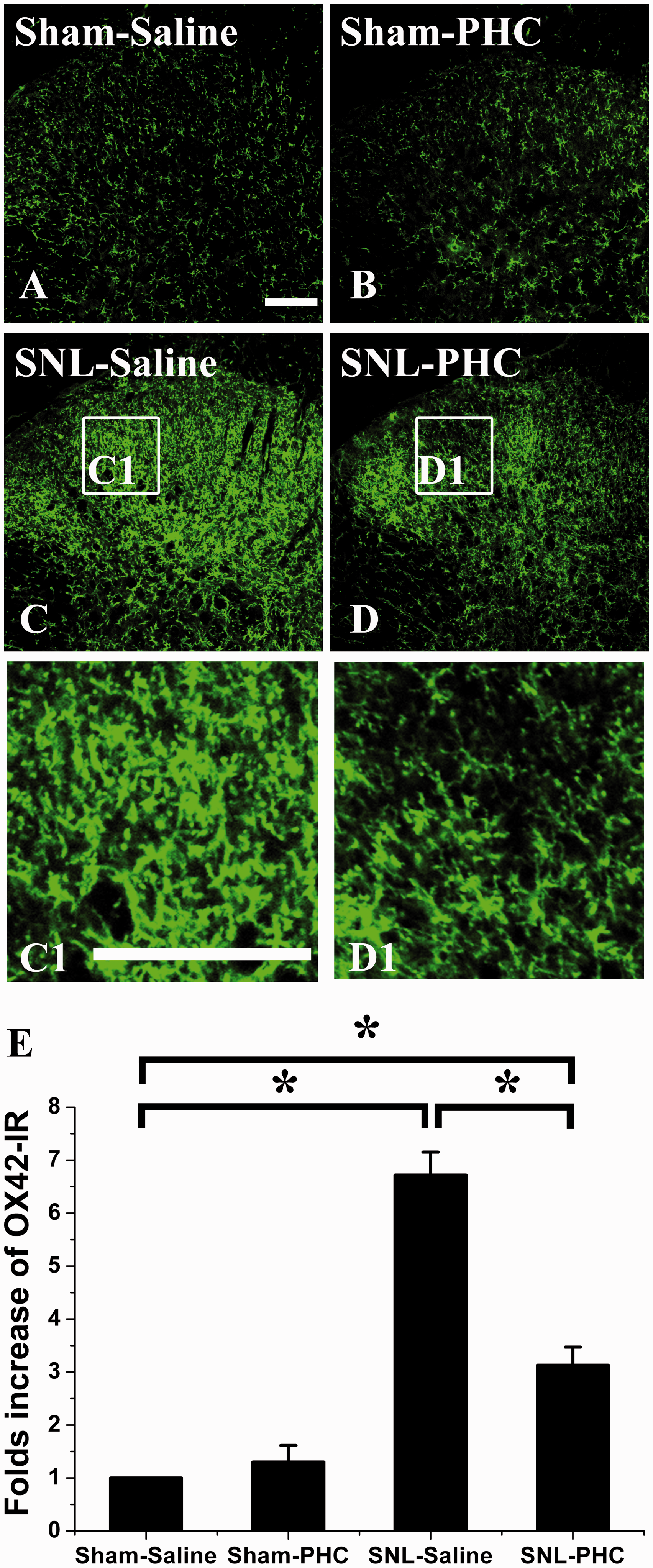
Effect of PHC injection on spinal microglia activation after SNL. SNL induced remarkable spinal microglia activation indicated by OX42 up-expression in the ipsilateral dorsal horn after SNL (SNL-Saline, (c)) compared with that of Sham-Saline (a). Intraperitoneal PHC did not affect the expression of microglial OX42 expression in Sham-PHC group (b). Intraperitoneal administration of PHC depressed SNL-induced microglial OX42 up-expression in SNL-PHC group (d) compared with that of Sham-Saline. C1 and D1 showed the migration and proliferation of microglia after SNL treated with saline or PHC. (e) Statistical analysis of the spinal microglia activation after different treatments with the density of OX42 expression. Asterisk (*) indicates statistically significant difference with p < 0.05 between groups. Scale bar = 100 µm. PHC: penehyclidine hydrochloride; SNL: spinal nerve ligation.
These results suggested that intraperitoneal PHC could depress SNL-induced spinal microglia activation, which might contribute to the anti-allodynic effect of PHC.
Intraperitoneal PHC depressed SNL-induced spinal microglial MAPK/p-p38 activation
Spinal dorsal horn MAPK/p-p38 was highly expressed in the ipsilateral spinal dorsal horn (Figure 4(b)) as same as microglia (Figure 4(a)) three days after SNL. The double immunofluorescent staining with antibodies against MAPK/p-p38 and OX42 indicated that all MAPK/p-p38 colocalized with OX42-positive microglia (Figure 4(c) and (d)). These results suggested that spinal dorsal horn MAPK/p-p38 was a special signal pathway in spinal dorsal microglia after SNL.

Intraperitoneal PHC depressed SNL-induced ipsilateral spinal dorsal horn microglial MAPK/p-p38 expression. SNL induced a remarkable MAPK/p-p38 up-expression in the ipsilateral spinal dorsal horn (b) as well as microglial OX42. Double immunofluorescent staining between OX42 and MAPK/p-p38 ((c) and (d)) demonstrated that all MAPK/p-p38 positive cells were microglia with OX42 expression. High-magnification image (d) was captured from (c) showing the colocalization of OX42 and MAPK/p-p38. Intraperitoneal PHC injection has no obvious effect on spinal dorsal horn MAPK/p-p38 expression of Sham-PHC group compared with that of Sham-Saline group ((e) and (f)). MAPK/p-p38 was up-expression remarkable in the SNL-Saline group ((e) and (f)). PHC injection suppressed MAPK/p-p38 up-expression apparently in SNL-PHC group compared with that of SNL-Saline group ((e) and (f)). Asterisk (*) indicates statistically significant difference with p < 0.05 between groups. Scale bars: 50 µm in A, B and C; 10 µm in D. PHC: penehyclidine hydrochloride; SNL: spinal nerve ligation.
SNL induced ipsilateral spinal dorsal horn microglial MAPK/p-p38 high expression in SNL-Saline group compared with that of Sham-Saline group (Figure 4(e) and (f)). Intraperitoneal PHC obviously suppressed spinal microglial MAPK/p-p38 expression in the SNL-PHC group compared with that of SNL-Saline group (Figure 4(e) and (f)). However, intraperitoneal PHC did not affect the spinal microglial MAPK/p-p38 expression in the Sham-PHC group compared with that of Sham-Saline group (Figure 4(e) and (f)). These results indicated that intraperitoneal PHC depressed SNL-induced spinal microglial MAPK/p-p38 pathway activation, which might involve in the effect of PHC on nerve injury-induced neuropathic pain.
SNL-induced ipsilateral spinal dorsal horn pro- and mature-form of IL-1β over-expression was suppressed by intraperitoneal PHC
Ipsilateral spinal dorsal horn pro- and mature-form of IL-1β were over-expressed in SNL-Saline group compared with that of Sham-Saline group (Figure 5). Intraperitoneal PHC markedly suppressed SNL-induced ipsilateral spinal dorsal horn pro- and mature-form of IL-1β over-expression but did not influence the pro- and mature-form of IL-1β expression in the Sham-PHC group (Figure 5). These results indicated that intraperitoneal PHC inhibited SNL-induced ipsilateral spinal dorsal horn IL-1β (both pro- and mature-form) over-expression, which might contribute to the anti-allodynic effect of PHC on nerve injury-induced neuropathic pain.

Effect of intraperitoneal PHC injection on SNL-induced IL-1β expression. SNL could induce ipsilateral spinal dorsal horn pro- and mature-form of IL-1β up-expression in SNL-Saline group compared with that of Sham-Saline group significantly. Intraperitoneal PHC depressed SNL-induced ipsilateral spinal dorsal horn pro- and mature-form of IL-1β up-expression while had no effect on sham-operated animals. *p < 0.05, compared with that of Sham-Saline group. #p < 0.05, compared with that of SNL-Saline group. PHC: penehyclidine hydrochloride; SNL: spinal nerve ligation.
Discussions
This study revealed that PHC could attenuate SNL-induced neuropathic pain without any motor impairment and had no effect on Sham-operated animals after repeated intraperitoneal administration. PHC could suppress SNL-induced ipsilateral spinal dorsal horn microglial activation. Moreover, intraperitoneal PHC inhibited SNL-induced spinal MAPK/p-p38 activation, which was specially colocalized with the spinal dorsal microglia. Furthermore, intraperitoneal PHC could depress spinal neuroinflammation by suppressing SNL-induced IL-1β over-expression. These results indicated that the anti-allodynic effects of PHC on SNL-induced neuropathic pain did not rely on motor impairment. Inhibiting spinal microglial MAPK/p-p38/IL-1β pathway activation might contribute to the anti-allodynic effect of PHC on nerve injury-induced neuropathic pain.
Neuropathic pain is characterized by allodynia, hyperalgesia and spontaneous pain, and comorbidities such as sleep deprivation, depression, and anxiety. 23 It is a chronic and long-lasting condition especially after the lesion of sensory nerve system. The activation of microglial cells in response to nerve injury has been implicated in the development of neuropathic pain. 23 Not only activation but also proliferation of microglia could be observed in the spinal cord after nerve injury. It has been reported that 5-bromo-2′-deoxyuridine (BrdU)-positive cells on the ipsilateral dorsal horn of the spinal cord were significantly increased at seven days after nerve ligation and were highly colocalized with another microglia marker, ionized calcium-binding adaptor molecule 1 (Iba1), but neither with GFAP nor NeuN, in the spinal dorsal horn of nerve-ligated mice. 24 Another study suggested that there was a close temporal correlation between microglial proliferation in the spinal cord dorsal horn and the abnormal pain responses, indicating a contribution of the new microglia to the genesis of the neuropathic pain symptoms. 25 However, no study has tested the effect of PHC on microglial proliferation or migration, which remains to be an interesting potential target for PHC.
Microglia are key factor for many neurodegenerative diseases after neuronal damage occurred, which played an important role in the inflammatory reaction as the first line of defense.26–28 Microglia are the main effectors in the inflammatory process of the CNS. 28 Microglia, the resident macrophages of the CNS, are the first responders after insults to the CNS and comprise a major link between the inflammation and neurodegeneration. 29 Microglia will be activated by pathogen invasion or cell debris and rapidly release some inflammatory cytokines to mediate inflammatory reaction to defense the host. Although the precise mechanism of microglial cells in the development of nerve injury-induced neuropathic pain has not been completely clarified, research into these different activating factors and cytokines is providing further insight into the role of microglial cells in the development and maintenance of neuropathic pain. 23
Activated microglia were found surrounding lesions of various neurodegenerative diseases, which were involved in the removal of necrotic neurons. However, microglial overactivation may promote the development of neurodegenerative disease. 28 Microglia can release some cytotoxic chemokines and cytokines, such as IL-6, IL-1β and tumor necrosis factor (TNF)-α that could gather to the lesion to result in further damage to neuron and cause the painful symptoms. It is becoming increasingly apparent that an intricate network of cytokines and cellular signaling mechanisms underpin the complex relationship between microglia and various cell types, such as neuron, astrocytes, oligodendrocytes, mast cells, and T-cells. 23 Therefore, inhibiting the activity of microglia appropriately may be an effective way for the treatment of neurodegenerative diseases including neuropathic pain.
PHC is used as an anesthetic assistant during the induction of anesthesia widely in China but also displays some anti-inflammatory effects. 14 Previous study incubated microglia with LPS in the presence or absence of PHC, SB203580 (p38 MAPK inhibitor), and pyrrolidine dithiocarbamate (NF-kappaB inhibitor) to investigate whether PHC could exhibit anti-inflammatory effects on activated microglia in the CNS. 14 It was found that PHC could apparently depress nitric oxide (NO), prostaglandin E2, IL-1β, and TNF-α expression. In addition, PHC effectively suppressed the phosphorylation of p38 MAPK and the translocation of NF-kappaB from the cytoplasm to the nucleus. LPS-induced microglial NF-kappaB and p38 MAPK activities were significantly inhibited by PHC pretreatment. It is concluded that PHC could inhibit LPS-induced release of inflammatory mediators from microglia. This effect may result from blocking p38 MAPK and NF-kappaB pathways in activated microglia. These results may offer a novel therapeutic manner to control microglial activation when treating neurodegenerative diseases. 14
Recently, various reports have suggested that PHC, a new cholinergic antagonist, exhibits a variety of biological actions including anti-inflammation, anti-oxidation, cardioprotection, and neuroprotection.30,31 PHC has protective effects on LPS-induced acute kidney injury, including abatement of renal tissue inflammation and functional improvement, with downregulation of proinflammatory factors TNF-α and IL-1β. 30 This study demonstrated that SNL could induce ipsilateral spinal dorsal horn IL-1β over-expression. Moreover, PHC obviously depressed SNL-induced ipsilateral spinal dorsal horn IL-1β over-expression but had no effect on that of the sham animals. This result indicated that the anti-allodynic effect of PHC on nerve injury-induced neuropathic pain might rely on suppressing SNL-induced inflammation in the spinal dorsal horn.
The mechanisms of anti-inflammatory properties of PHC have been evaluated by some new reports. It was reported that acute lung injury (ALI) led to significant reductions in heart rate and mean arterial blood pressure and was associated with significant increases in proinflammatory mediators, p-p38 MAPK, and TLR4 expression. Intraperitoneal PHC inhibited the development of ALI effectively. Besides, PHC attenuated TLR4 expression, reduced the activation of p-p38 MAPK, and downregulated the expression of proinflammatory mediators, such as IL-1β, IL-6, and so on. 16 This study demonstrated that PHC might exert an anti-inflammatory effect by inhibiting the TLR4/p-p38 MAPK/IL-1β signaling pathway. This study also showed that PHC could inhibit spinal microglial p-p38/IL-1β pathway activation, which might contribute to the anti-allodynic effect of PHC on nerve injury-induced neuropathic pain. Moreover, another study concluded that the microglial TLR4 signaling pathway promotes hyperalgesia induced by acute inflammation by stimulating the production of proinflammatory mediators. 32 Taken these together, the microglial TLR4/p-p38 MAPK/IL-1β signaling pathway may contribute to the effect of PHC on SNL-induced neuropathic pain.
Previous study investigated the effects of PHC on cerebral I/R injury and evaluate whether p38 MAPK pathway is involved in the protective effects of PHC. 13 It is reported that PHC improved neurological deficits and reduced infarction volume compared with that of the I/R group. Besides, PHC significantly inhibited the expression of IL-1β and the phosphorylation level of p38 MAPK. 13 Another study was to investigate the inhibitory effect of PHC on LPS-induced NO and inducible nitric oxide synthase (iNOS) production. 33 LPS treatment significantly induced p38 MAPK activation, iNOS expression, and NO production, which could be attenuated by PHC pretreatment. Furthermore, LPS-induced NO production and iNOS expression were suppressed by p38 MAPK inhibitor SB203580 pretreatment. PHC attenuates NO production and iNOS expression by suppressing the activation of p38 MAPK pathway, thereby implicating a mechanism by which PHC may exert its protective effects against LPS-induced injury. This study demonstrated that SNL induced ipsilateral spinal dorsal horn MAPK/p-p38 over-expression. Intraperitoneal PHC could suppress MAPK/p-p38 expression markedly, which might contribute to the anti-allodynia effect of PHC on nerve injury-induced neuropathic pain. In addition, it could also be noticed that activated microglia were still observed even after PHC treatment, especially in layers II–III the dorsal horn. In this situation, activities of interneurons that project to lamina I were not reduced. It has been reported that microglial-released molecules would influence interneurons activity. 34 A recent study also showed a dynamic change of Gamma-Amino Butyric Acid (GABA)(B)R1a protein expression after SNL, and the SNL-induced down-regulation of GABA(B)R1a expression in the spinal dorsal horn could be prevented by intrathecal administration of a p38/MAPK inhibitor SB203580. 35
Studies indicated that increased presynaptic NMDA receptor (NMDAR) activity could enhance glutamate release to spinal dorsal horn neurons from the primary afferent terminals, which was critical for the synaptic plasticity associated with neuropathic pain induced by peripheral nerve injury. Targeting NMDAR could decrease afferent nociception to the dorsal horn neurons, which might be a new strategy for treating neuropathic pain.36,37 Besides, there were a variety of NMDAR antagonist agents used for the treatment of neuropathic pain in clinic. Recent study reviewed 8 NMDAR antagonists within 58 randomized controlled trials, which showed analgesic effects on neuropathic pain. 38 Furthermore, previous study investigated the effect of PHC on NMDAR expression in hippocampus after global cerebral I/R. The results showed that the expression of NMDAR was significantly decreased after PHC treatment, which indicated that PHC might has protective effect in hippocampus on global cerebral I/R by inhibiting NMDAR expression. 39 Another study also confirmed that PHC could protect neuronal damage by regulation NMDAR activity after cerebral ischemia. 11 NMDAR was considered to express exclusively in neurons for a long time. However, recent studies have found functional NMDAR in glia. 40 Glial NMDAR indicated the complex nature of intercellular signaling mechanisms in the CNS. Therefore, in this study, the spinal NMDAR may be involved in the effect of PHC on peripheral nerve injury induced neuropathic pain.
However, there are some limitations in the present research. First, systemic injection of PHC may not be exact for the detection of the role of spinal microglia, because the involvement of brain microglia and other region in the generation of neuropathic pain cannot be excluded. Therefore, the intrathecal injection could be the best way to analyze the effect and mechanisms of analgesic drugs at the spinal level. However, systemic injection could mimic the clinical use of drugs. It is suggested that systemic PHC could affect the microglia in the spinal cord. Therefore, this study thus first tests the effect of PHC through the systemic injection. In addition, the author is conducting the experiment of intrathecal PHC model to further analyze the effect of PHC on microglial activity.
This study was conducted to disclose the effect of PHC on the early phase of SNL-induced neuropathic pain. Since microglial activation was observed in the early phase of nerve injury-induced neuropathic pain. Thus, in this study, PHC was administrated on the POD0 and POD1 once a day. Therefore, the behavioral and morphological results were observed on POD3. However, further study should be done to disclose the effects of PHC in the later phase of neuropathic pain.
In conclusion, this study demonstrated that PHC could suppress SNL-induced ipsilateral spinal dorsal horn microglial MAPK/p-p38/IL-1β pathway activation, which might contribute to the anti-allodynic effect of PHC on nerve injury-induced neuropathic pain.
Footnotes
Acknowledgments
The authors thank for the critical scientific opinions and help in English by Dr. Jerome Staal from the Menzies Institute for Medical Research and University of Tasmania.
Authors’ Contributions
ZSH and DN performed the experiments. YCY and WW contributed to the experiment of revised manuscript. MXP designed and wrote the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants from the National Natural Science Foundation of China (30901400 and 81270016), the Natural Science Foundation of Shaan-Xi Province (2018JM7101), the Clinical Research Award of the First Affiliated Hospital of Xi'an JiaoTong University (XJTU1AF-CRF-2018–018), the Fundamental Research Funds for the Central Universities (xjj2017128/0816–1191329727), and the Foundation of Chinese Medical Association (220160900005, CSDE012017120009).
