Abstract
Neuropathic pain is still a clinical challenge. Inflammatory responses and autophagy in the spinal cord are important mechanisms for the occurrence and maintain of neuropathic pain. PDCD4 is an important molecule that regulates inflammation and autophagy. However, the regulatory role of PDCD4 is unknown in pain modulation. In this study we found that the expression of PDCD4 in the spinal cord of CCI mice was increased. Inhibition of PDCD4 by intrathecal injection of adeno-associated virus alleviated neuropathic pain hypersensitivity and enhanced autophagy in CCI mice, and inhibited the activation of MAPKs, as well as the expression of inflammatory factors. Intrathecal injection of autophagy inhibitor 3-MA reversed PDCD4 inhibition induced pain relief and change of autophagy. Our results indicate that spinal cord inhibition of PDCD4 alleviates pain sensitization in neuropathic pain mice through MAPKs and autophagy, and PDCD4 may be developed into a therapeutic target of neuropathic pain treatment.
Introduction
Neuropathic pain (NP), a chronic pain condition, arises from damage or injurious stimulation to the somatosensory nervous system, and can also be caused directly by diseases.1,2 NP can affect various parts of the nervous system, encompassing a clinical syndrome that includes types of pain such as allodynia, hyperalgesia, and spontaneous pain. 3 Patients with neuropathic pain often experience a range of distinct symptoms, such as burning and electrical sensations, as well as pain triggered by non-painful stimuli like light touch. 4 However, the efficacy of pharmacological treatments is finite, and the toxic side effects associated with these medications impose certain restrictions on their clinical application.5,6 Therefore, it is particularly important to investigate the mechanisms underlying the occurrence and development of neuropathic pain.
Programmed cell death 4 (PDCD4), a tumor suppressor and protein translation inhibitor, exerts its effects by binding to translation initiation factor eukaryotic initiation factor 4F (eIF4A), transcription factors, and other binding factors. PDCD4 inhibits cell growth, promotes apoptosis, and suppresses tumor invasion and metastasis.7,8 As a cancer-inhibiting protein, PDCD4 is involved in the occurrence and development of tumors. 9 Recent years, numerous studies have reported the role of PDCD4 in other diseases such as polycystic ovary syndrome, obesity, diabetes, and coronary artery atherosclerosis.10–13 PDCD4 also plays a crucial regulatory role in inflammation. 14 For instance, in pdcd4 gene knockout mice, the JNK and NF-κB signaling pathways were activated, leading to the elevated expression of TNF-α and IL-6. 15 Additionally, PDCD4 expression was increased in LPS-induced inflammation of central nervous system. 16 Based on the aforementioned evidence, PDCD4 may be an important molecule involved in regulating inflammatory responses of pain modulation. However, there are few reports on the role of PDCD4 in pain regulation. A previous study reported upregulated PDCD4 in the spinal cord of CCI mice, which was regulated by miR-330-3p, but the behind mechanisms remain unclear. 17
Autophagy refers to the process in which abnormal synthesized or misfolded proteins, as well as dysfunctional organelles and other substances within the cell, are enveloped by double-membrane structures and ultimately transported to lysosomes for degradation. 18 Autophagy is crucial for maintaining cellular homeostasis. Increasing evidence suggested that dysregulation of autophagy may be associated with the development of various diseases, in addition to its role in maintaining physiological states.19–21 Studies have found that autophagy in spinal cord is involved in pain regulation, with weakened autophagy in chronic pain and relief of pain with autophagy inducer rapamycin.22,23 Previous research has identified PDCD4 as an important molecule in regulating autophagy. PDCD4 regulates the autophagy process by modulating the translation of autophagy-related proteins and is involved in the regulation of processes such as tumor development and epilepsy.24,25 However, whether PDCD4 participates in the regulation autophagy in chronic pain modulation has not been reported yet. This study aims to delve into the spinal mechanisms by which PDCD4 regulates neuropathic pain, providing a theoretical basis for the treatment of neuropathic pain.
Methods and materials
Animals
One hundred and six C57BL/6 male mice (8 weeks old, weighing 21–25 g) were obtained from Xuzhou Medical University (SYXK 2016-0028). All mice were housed in groups of 4–6 individuals per cage in a standard condition with access to food and water ad libitumin a colony room kept at 19–22°C and 40%–60% humidity, under a 12-h light/dark cycle (light from 08:00 to 20:00). The animals were acclimatized for 7 days before the experiments and were grouphoused with the same cage mates throughout the acclimation and experiment. The study protocol was approved by the Ethics Committee of Xuzhou Medical University and all procedures were performed in accordance with the approved guidelines. All experiments were conducted in compliance with the Animal Research Reporting In Vivo Experiments (ARRIVE) guidelines.
Neuropathic pain model
A neuropathic pain model, known as the CCI model, was established by loosely ligating the sciatic nerve. In brief, Mice were anesthetized by an in traperitoneal injection of Avertin (isoamyl alcohol, 250 mg/kg). An incision was made on the lateral surface of the left thigh (opposite to the site of viral injection) and a section was created through the biceps femoris and gluteus muscles. These muscles were separated through blunt dissection, exposing the sciatic nerve, which was then gently retracted. Three loose ligations were applied to the dorsal third to half of the common sciatic nerve at the upper-thigh level using nonabsorbent 5-0 silk suture (Ethicon).
Intrathecal injection
The drugs injected in this study were prepared as follows. After anesthesia, mice were bent at the waist. Mice were anesthetized by an in traperitoneal injection of Avertin (isoamyl alcohol, 250 mg/kg). The 3-Methyladenine (3-MA) was dissolved in 2% dimethyl sulfoxide in saline (autophagy inhibitor; MCE, HY-19312, 5 µg/d/mouse). Two percent dimethyl sulfide is the concentration of the storage solution. When used for experiments, 3-MA needs to dissolve 2% dimethyl sulfide at a concentration of 5 mM, and add to saline. The final DMSO concentration is 0.6%, and the 3-MA concentration is about 0.5 mg/mL and intrathecally administered 10 µL on postoperative day 7, 8, and 9 during the stage of neuropathic pain according to the previous studies.26,27 pAAV-U6-shRNA (Pdcd4)-CMV-EGFP-WPRE was designed and manufactured by Obio. Ltd. (Shanghai, China). pAAV-U6-shRNA (Pdcd4)-CMV-EGFP-WPRE and Adeno-associated virus (AVV) vector was injected into the cerebrospinal fluid through the L5-L6 lumbar intervertebral space using a 10 μL microsyringe. The accuracy of each injection was assessed by observing a reflexive flick of the tail.
Enzyme-linked immunosorbent assay (ELISA)
The concentrations of IL-1β, TNF-α, IL-6 levels in L4-6 lumbar enlargement spinal cord tissues measured by ELISA Kits (KE10007 for IL-6 and KE10002 for TNF-α, Proteintech; HA722166 for IL-1β, Huabio). Spinal cord tissues were homogenized with lysis buffer including the protease inhibitor. ELISAs were performed and the standard curve was depicted according to the manufacturer’s protocol.
qRT-PCR
L4-6 spinal cord tissues were collected, the total RNA was extracted by TRIzol (TABAKA) according to the manufacturer’s instructions. Hifair III 1sr Strand cDNA Synthesis SuperMix for qPCR (Yeasen) was used to synthesize cDNA according to the manufacturer’s instructions. qPCR was performed on a TOptical Real-Time qPCR Thermal Cycler (Analytik Jena) using the SYBR Green method.
Pain behavioral quantification
Thermal hyperalgesia was assessed by measuring the paw withdrawal latency (PWL) in response to a radiant heat s stimulation. A Plantar Analgesia Meter (IITC Life Science Inc., CA, USA) was used as a radiant source. Briefly, mice were placed individually in perspex observation chambers on an elevated glass plaform, and a radiant heat source was applied to the glabrous surface of the paw through the glass plate. The nociceptive endpoints included the characteristic lifting or licking of the hind paw. The heat was maintained constant intensity, which produced a stable PWL of 12–14 s in normal animals. A 20-s cutoff was used to prevent tissue damage. After acclimation to the test chambers, both hind paws were tested independently with 3-min intervals between trials.
Paw withdrawal threshold was measured in response to von Frey filament stimulation (Aesthesio, Danmic Global, San Jose, CA, USA) to determine the presence of mechanical allodynia. Briefly, following three consecutive days of acclimatization, the mouse was kept on a wire net floor in a plexiglass chamber and allowed to familiarize for 10–15 min before the experiment is initiated. The mice’s mid-plantar surface of the hind paw was applied with a series of filaments (0.4, 0.6, 1.4, 2, 4, 6, 8, 10, and 15 g) with sustaining pressure to elicit a paw withdrawal reflex or to bend the filament for 5 s within 5 s. Application of each of the filaments was done five times, followed by the calculation of the 50% threshold (g) employing the formula: maximum bending force value [(maximum bending force value minimum bending force value) (positive rate of the maximum bending force − positive rate of the minimum bending force)] (positive rate of the maximum bending force 50%).
Immunofluorescence
Under deep anesthesia, the L4-5 spinal segment of mice was dissected and post-fixed in 4% PFA for 3 h, and subsequently transferred into Tris-buffered saline (TBS) containing 30% sucrose and conserved at 4°C until use. The spinal sections were transversely cut 25 μm thick in a cryostat and stored in TBS. The sections were first blocked with 5% donkey serum in 0.3% Triton X-100 for 1 h at room temperature and then incubated overnight at 4°C with the following primary antibodies: rabbit anti-PDCD4 polyclonal antibody (1:500, 9535S, CST, USA), c-Fos rabbit mAb (1:500, #2250, CST, USA), rabbit anti-p-ERK polyclonal antibody (1:100, 28733-1-AP, Huabio), rabbit anti-p-JNK polyclonal antibody (1:400, 80024-1-RR, Huabio), rabbit anti-p-p38MAPK polyclonal antibody (1:200, 28796-1-AP, Huabio). The sections were rinsed three times for 10 min in TBS and then incubated for 1 h at room temperature with corresponding secondary antibodies (conjugated to Alexa Fluor 488 or 594, Invitrogen, Carlsbad, CA, USA) overnight at 4°C. For double immunostaining, the stained sections were incubated secondly with the same immunostaining procedure described above. The immunofluorescent images were captured with a confocal scanning laser microscope (FluoView FV1000, Olympus Co., Tokyo, Japan). For obtaining the semiquantitative determination of c-Fos and p-CREB fluorescence, 20 fields spreading the whole section of dorsal horn in each group were scanned and analyzed at the same exposure time to generate the data of green fluorescence intensity.
Western blot
The dorsal horn of the L4-6 spinal cords were quickly removed from mice under deep anesthesia, then the tissue samples were dissected and stored in liquid nitrogen. Then equivalent amounts of protein (80 μg) were separated using 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto a polyvinylidene fluoride (PVDF) membrane. The membranes were blocked with 5% non-fat milk or BSA at room temperature for 2 h followed by overnight incubation with the following primary antibodies at 4°C: rabbit polyclonal anti-PDCD4 antibody (1:500, 9535S, CST, USA), Beclin1 (1:200, HA721216, Huabio, China), p62 (1:500, HA72117, Huabio, China), and glyceraldehyde-3-phosphate dehydrogenase (1:10000, GAPDH, G9545, Sigma, USA), respectively. After washing with TBST three times, the blots were incubated with HRP conjugated secondary antibody and detected in SuperLumia ECL solution (MedChemExpress, Monmouth Junction, NJ, USA). Band intensity was quantified using Quantity One Analysis Software (Version 4.6.5, Bio-Rad Laboratories, Hercules, CA, USA). The target protein was expressed as the fold change of the control group after normalization against GAPDH.
Statistical analyses
GraphPad Prism 9 (GraphPad Software Inc., CA, USA) was used to conduct all statistical analyses. Alterations of detected protein expression were tested using one-way ANOVA with repeated measures, followed by the Dunnett multiple comparison test, and changes of behavioral response to radiant heat stimuli over time among groups were tested using two-way ANOVA with repeated measures, followed by Bonferroni post hoc test. All data are expressed as means ± SEM. Statistical differences were considered significant if p < 0.05.
Results
Expression and distribution of PDCD4 in the spinal cord of CCI mice
We first examined the timecourse of pain behaviors in CCI mice, behaviors tests showed that after CCI, mechanical and thermal allodynia were appeared in CCI mice. Then we examined expression of PDCD4 in the spinal cord of CCI mice at different time points after surgery. Western blot results revealed a significant increase in PDCD4 expression at 7, 14, and 21 days post-CCI (Figure 1(c)). To determine the cellular localization of PDCD4 in the spinal cord of CCI mice, we performed immunofluorescence co-staining of PDCD4 with neuronal marker NeuN, astrocyte marker GFAP, and microglia marker IBA1. The immunofluorescence results demonstrated PDCD4 was colocalized mostly with neurons (NeuN), and a minority with microglia (IBA1), but not astrocytes (GFAP) in the spinal cord dorsal horn (Figure 1(d)).
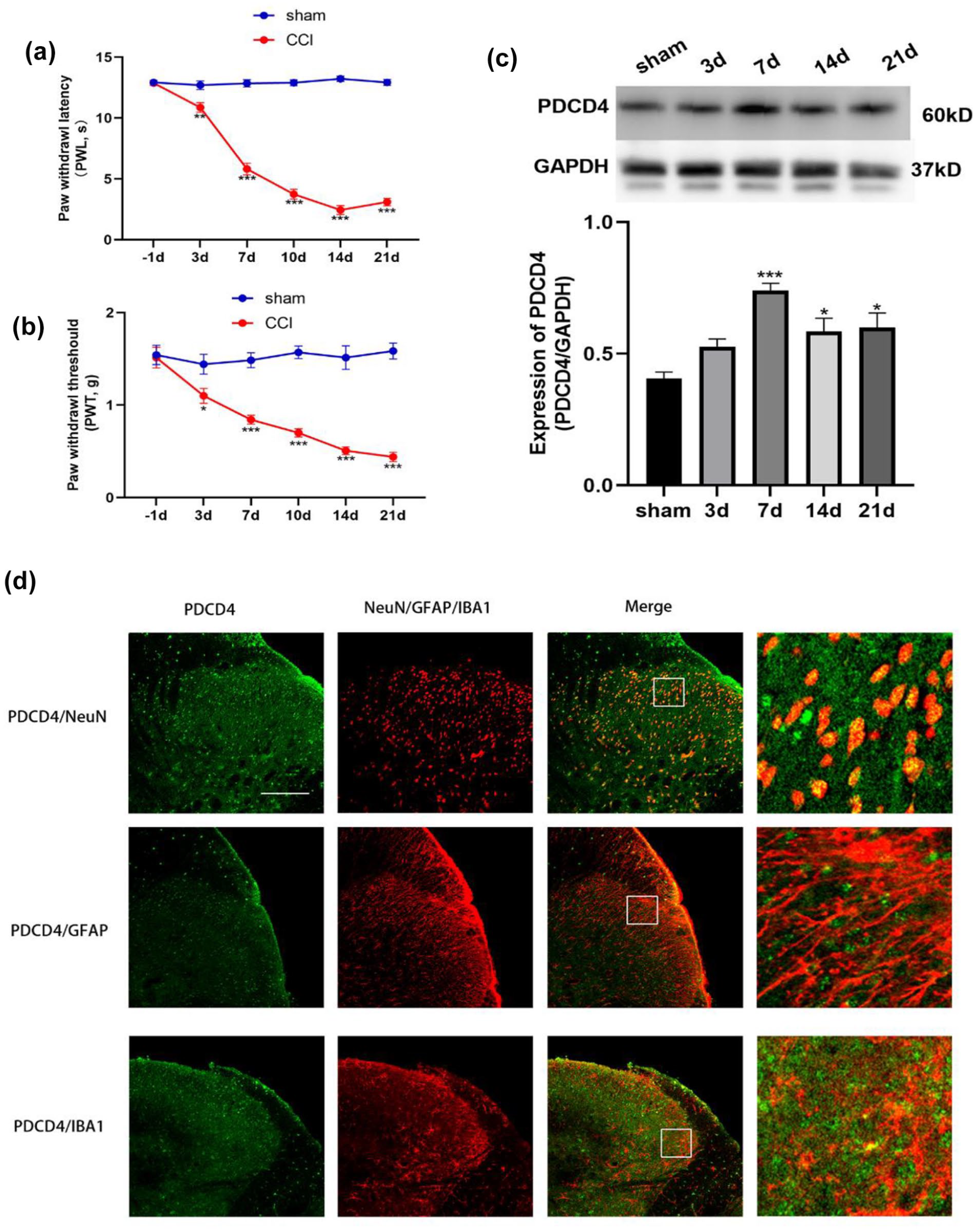
Expression and distribution of PDCD4 in spinal cord of CCI mice. (a–b) Behavioral tests of CCI and sham mice, PWL and PWT were lower in CCI mice after operation. (*p < 0.05, **p < 0.01, ***p < 0.001 compared with the sham group, n = 8 per group). (c) WB showed time-course of PDCD4 in the spinal cord of CCI mice. (***p < 0.001; *p < 0.05 compared with the sham group, n = 6 per group). (d) Distribution of PDCD4 in the spinal cord dorsal horn following CCI. PDCD4 was colocalized mostly with neurons (NeuN), and a minority with microglia (IBA1), but not astrocytes (GFAP) in the spinal cord dorsal horn. Tissues were collected at day 14 after CCI (n = 3 per group, scale bar = 100 μm).
The impact of intrathecal adenovirus-mediated PDCD4 inhibition on behavioral outcomes in CCI mice
Previous investigations have revealed an upregulation of PDCD4 expression in the spinal cord of CCI mice. In this part, we employed an innovative tool, AAV-shPDCD4, to suppress the expression of PDCD4. The construction of AAV-encoded shPDCD4-EGFP or control EGFP was shown in Figure 2(a). Immunofluorescence staining results showed the expression of EGFP in the spinal cord dorsal horn (Figure 2(b)). Then, the expression of PDCD4 following AAV-shPDCD4-EGFP and AAV-EGFP delivery has been examined in the spinal cord (Figure 2(c)). Results showed that PDCD4 protein was significantly suppressed by AAV-encoded shPDCD4 EGFP in the spinal cord compared with the AAV-control EGFP group. There was no significant difference in pain behaviors among the groups before or after shPDCD4-EGFP or control injection. Timecourse of pain behaviors showed that PDCD4 inhibition had a significant inhibitory effect on the PWL (Figure 3(b)) and PWT (Figure 3(c)) evaluation revealed that mechanical allodynia and thermal allodynia were attenuated in AAV- shPDCD4-EGFP treated mice at all time points after CCI compared with AAV-control-treated mice. However, there was no significant change in pain behaviors in the AAV-control group during the whole experiment. These results revealed that inhibition of PDCD4 in the spinal cord is responsible for pain relief induced by CCI.
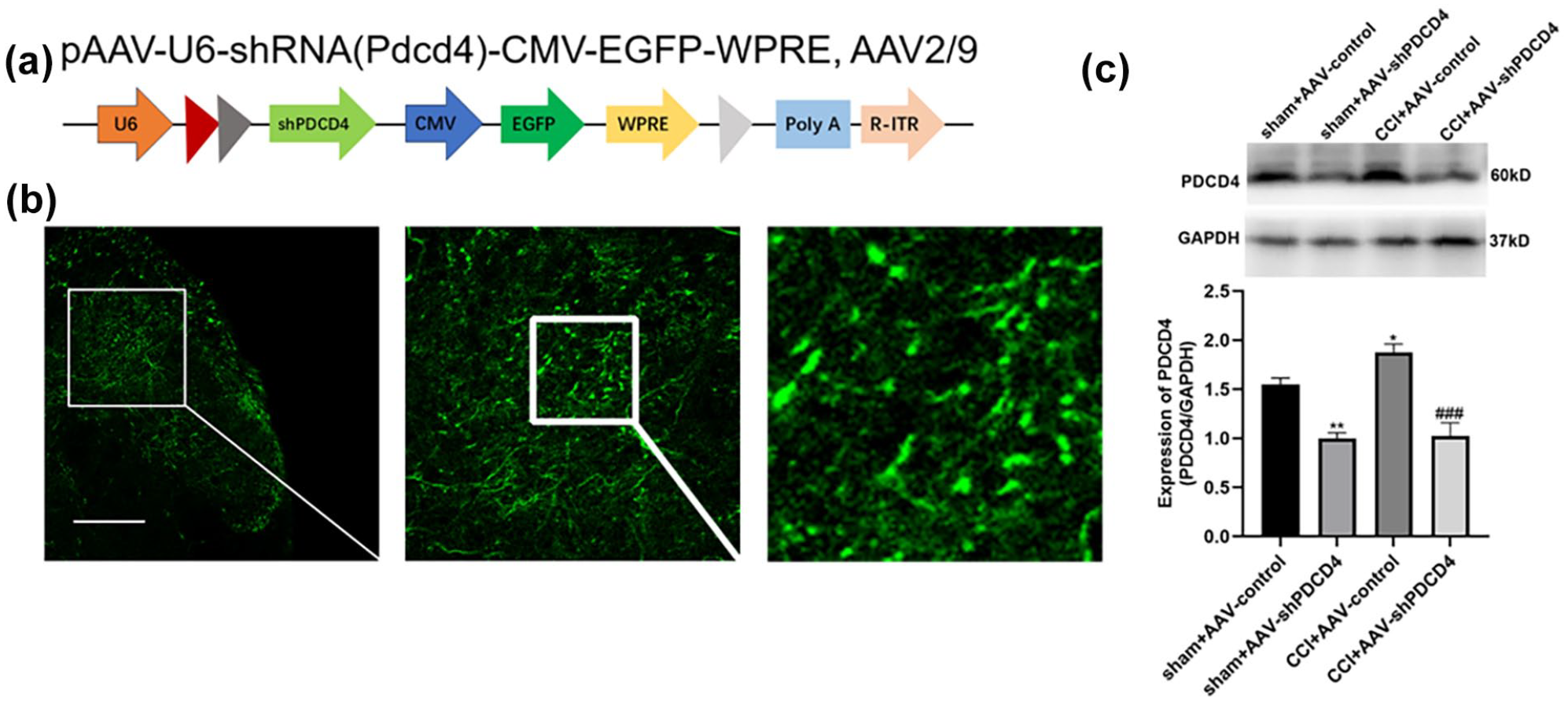
Effects of intrathecal injection of AAV encoded shPDCD4 (AAV-shPDCD4) on the expression of PDCD4 protein. (a) Schematics of AAV constructs shPDCD4 or control. CMV, cytomegalovirus promoter; EGFP, enhanced GFP; WPRE, woodchuck hepatitis virus posttranscriptional regulatory element; ITR, inverted terminal repeats. (b) EGFP immunoreactivity was detected in the superficial laminae of the dorsal horn at 4 weeks after AAV-shPDCD4 (5.0 × 1012 v.g/mL) injection (scale bar = 100 μm). (c) Representative bands for the expression of PDCD4 in the spinal cord of mice at 4 weeks after intrathecal injection of AAV-shPDCD4 or AAV-control, CCI models were performed 2 weeks after AAV injection (**p < 0.01 compared with the indicated group, n = 4 per group).

Effects of PDCD4 inhibition on existing pain hyperalgesia after CCI. (a) Schematic timeline for drug administration, behavioral detection, and tissue extraction. (b–c) Analgesic effects of i.t. AAV-shPDCD4 on thermal hyperalgesia (PWL) and mechanical allodynia (PWT) in ipsilateral to CCI. (***p < 0.001, **p < 0.01 vs sham+AAV-control group. #p < 0.05, ###p < 0.001 vs CCI+AAV-control, n = 8 per group).
The impact of intrathecal AAV-shPDCD4 injection on MAPK pathway activation
Studies have demonstrated that PDCD4 is a crucial regulator of inflammatory responses, and MAPKs play a pivotal role in regulating neuroinflammation. 28 Previous researches have indicated that PDCD4 modulated the MAPK pathway in processes such as tumor development and fulminant hepatic failure.29,30 In this study, we examined the effects of PDCD4 inhibition on MAPK pathway activation. Immunofluorescence results indicated that intrathecal AAV-shPDCD4 injection suppressed the expression of p-ERK, p-JNK, and p-p38MAPK in the spinal cord after CCI (Figure 4(a)). WB showed p-ERK, p-JNK, and p-p38MAPK protein was suppressed by AAV-shPDCD4 injection (Figure 4(b)–(e)). These findings suggested that PDCD4 may regulate neuropathic pain through the modulation of the MAPK pathway activation.
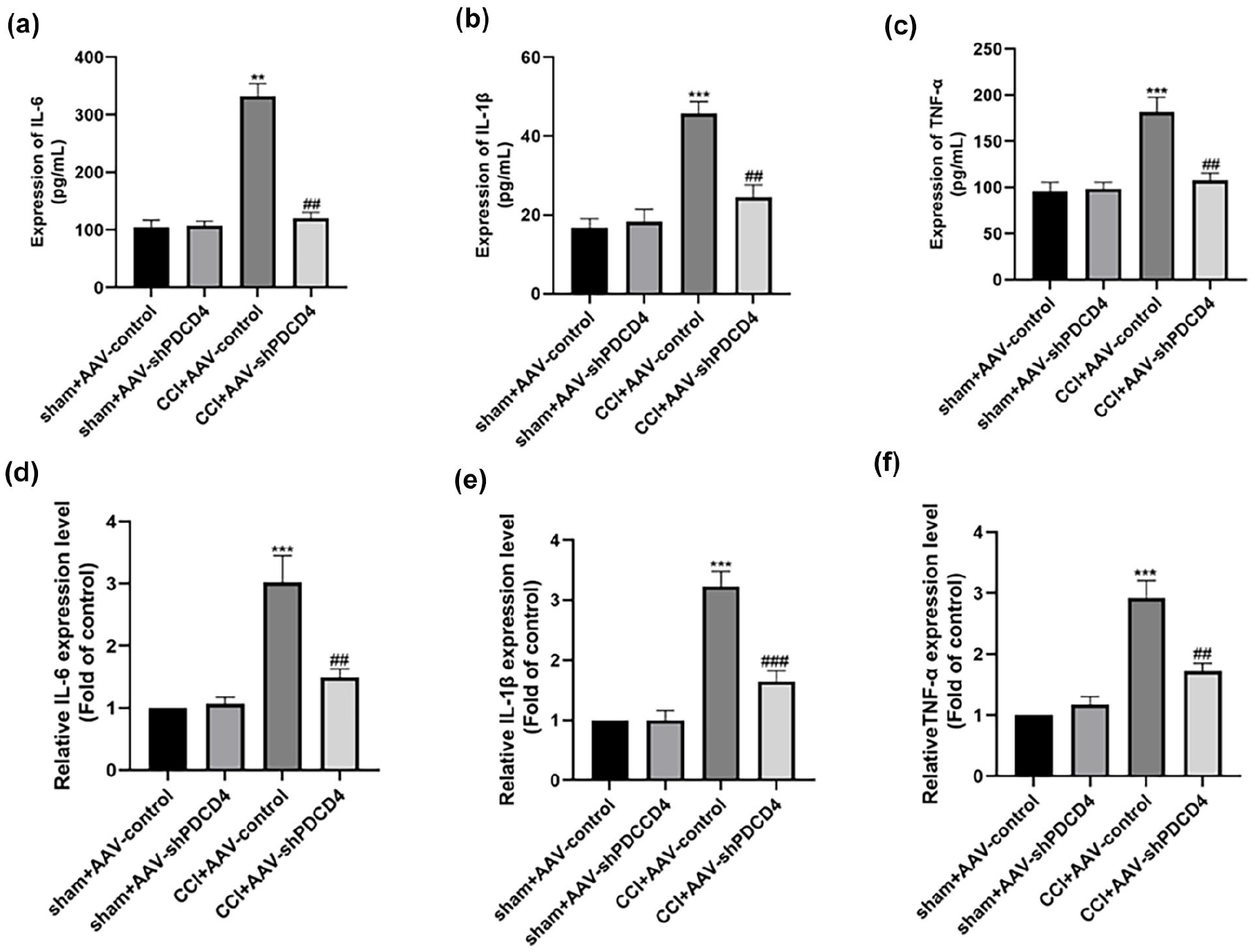
The effects of PDCD4 inhibition proinflammatory factors. (a–c) ELISA data showed the expression of proinflammatory cytokines in the L4-6 spinal cord were downregulated by intrathecal injection of AAV-shPDCD4 after CCI (**p < 0.01, ***p < 0.001 compared with the sham group, ##p < 0.01 compared with CCI+AAV-control group, n = 4 per group). (d–f) qRT-PCR results of proinflammatory cytokines in the L4-6 spinal cord were downregulated by intrathecal injection of AAV-shPDCD4 after CCI (***p < 0.001 compared with the sham group, ##p < 0.01, ###p < 0.001 compared with CCI+AAV-control group, n = 4 per group).
The impact of intrathecal AAV-shPDCD4 injection on neuroinflammation
PDCD4 plays an important role in regulating neuroinflammation, studies have shown that PDCD4 was upregulated in the cerebral cortex of LPS induced neuroinflammation, and inhibition of PDCD4 inhibits microglial activation through the MAPK pathway. 16 We tested the expression of proinflammatory factors, ELISA results further demonstrated a reduction in the expression of proinflammatory factors IL-6, IL-1β, and TNF-α following AAV-mediated PDCD4 inhibition (Figure 6(a)–(f)). These findings suggest that PDCD4 may regulate neuropathic pain by modulating neuroinflammation.
The influence of intrathecal AAV-shPDCD4 injection on spinal cord autophagy in CCI mice
Enhanced autophagy has been shown to alleviate neuropathic pain in mice. 31 PDCD4 is an important molecule regulating autophagy, so whether autophagy is involved in the regulation of PDCD4 in pain modulation? Our findings thus far suggested that PDCD4 regulated neuropathic pain in CCI mice. To explore the underlying mechanisms, here we investigated the relationship between PDCD4 and autophagy in neuropathic pain regulation. As shown in Figure 5, western blot results indicated that intrathecal AAV-shPDCD4 injection significantly enhanced the expression of Beclin1 and LC3B, while reducing the expression of p62 (Figure 6(a)–(c)). These results suggested that PDCD4 may participate in neuropathic pain regulation through the modulation of autophagy.

The effects of PDCD4 inhibition on MAPK activation. (a): Immunofluorescence showed the expression of p-ERK, p-JNK, and p-p38MAPK were inhibited by intrathecal injection of AAV-shPDCD4 in the L4-6 spinal cord (n = 4 per group, scale bar = 100 μm). (b–e): WB showing the expression of p-ERK, p-JNK, and p-p38MAPK were inhibited by intrathecal injection of AAV-shPDCD4 in the L4-6 spinal cord (*p < 0.05, **p < 0.01, compared with CCI+AAV-control; ###p < 0.001 compared with sham+AAV-control, n = 4 per group).
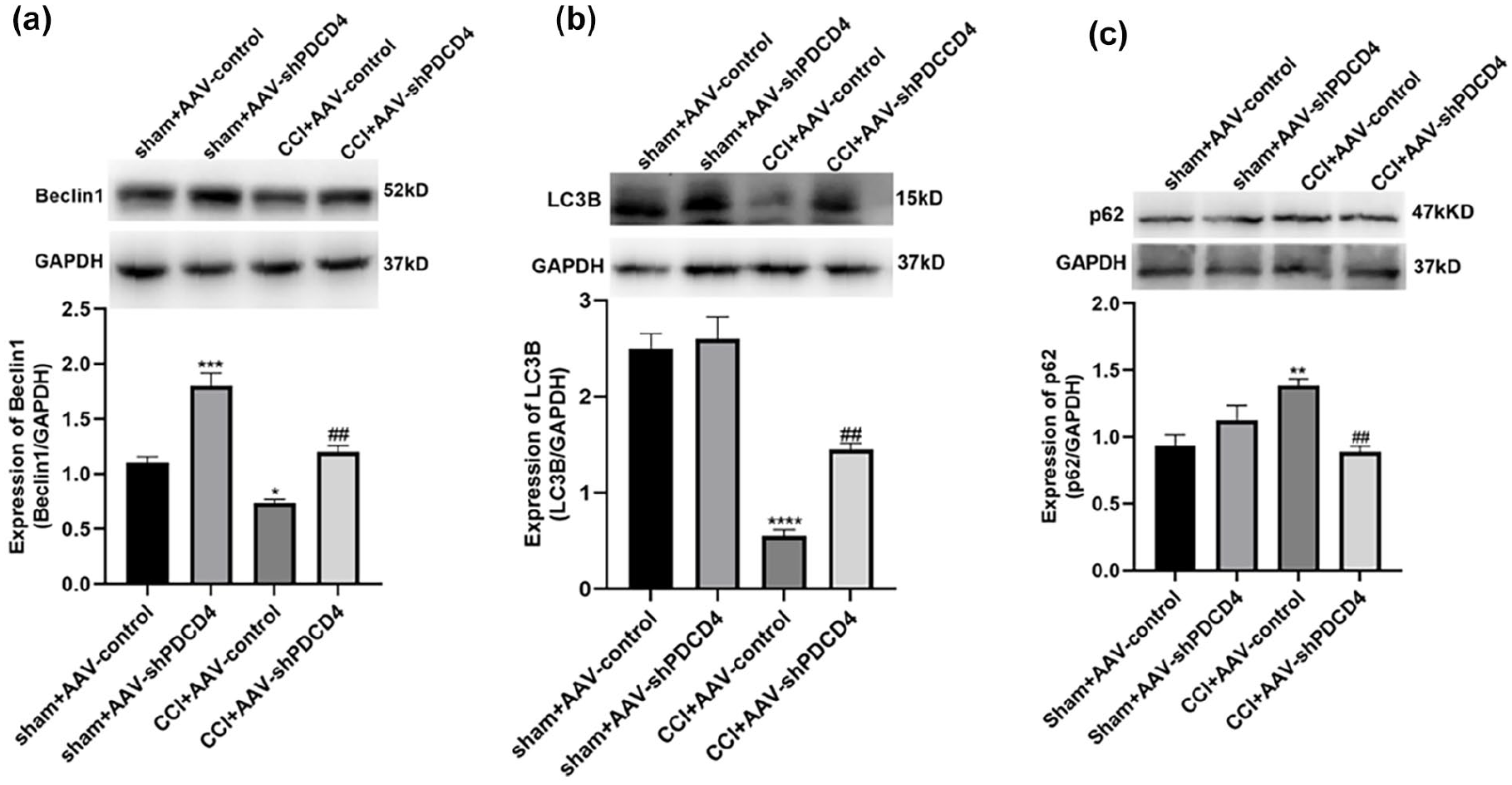
The effects of PDCD4 inhibition on autophagy. (a–c) WB results showed expression of Beclin1, LC3B and p62 were reversed after AAV-shPDCD4 intrathecal injection in CCI mice. (*p < 0.05, **p< 0.01, ***p < 0.001, ***p< 0.0001 compared with CCI+AAV-control; ##p < 0.01 compared with sham+AAV-control, n = 4 per group).
Autophagy activation was required for PDCD4 inhibition induced analgesia
Above studies have confirmed that inhibiting spinal cord PDCD4 alleviated pain hypersensitivity in CCI mice, possibly through activation of autophagy. To further clarify the regulatory relationship between PDCD4 and autophagy, we confirmed it through the following experiments. Two weeks following direct injection of AAV-shPDCD4, the mice were established neuropathic pain model by CCI. To assess the contribution of autophagy in the spinal cord of CCI mice, an autophagy inhibitor 3-MA (i.t., 10 μL, 5 μg/d/mouse) or vehicle (10 μL) was given within the sheath once daily from day 7 to day 9 after CCI (Figure 7(a)). As shown in Figure 7(b) (F = 7.300)-C (F = 7.309), treatment with 3-MA markedly reversed the analgesic effect of PDCD4 inhibition, the change of Becline1, LC3B p62 and induced by PDCD4 inhibition in CCI mice were also reversed by 3-MA (Figure 7(d)–(f)). Moreover, the reduction of inflammatory cytokines was reversed by 3-MA as well (Figure 7(g)–(l)). These results going further suggested PDCD4 may regulated neuropathic pain via autophagy.
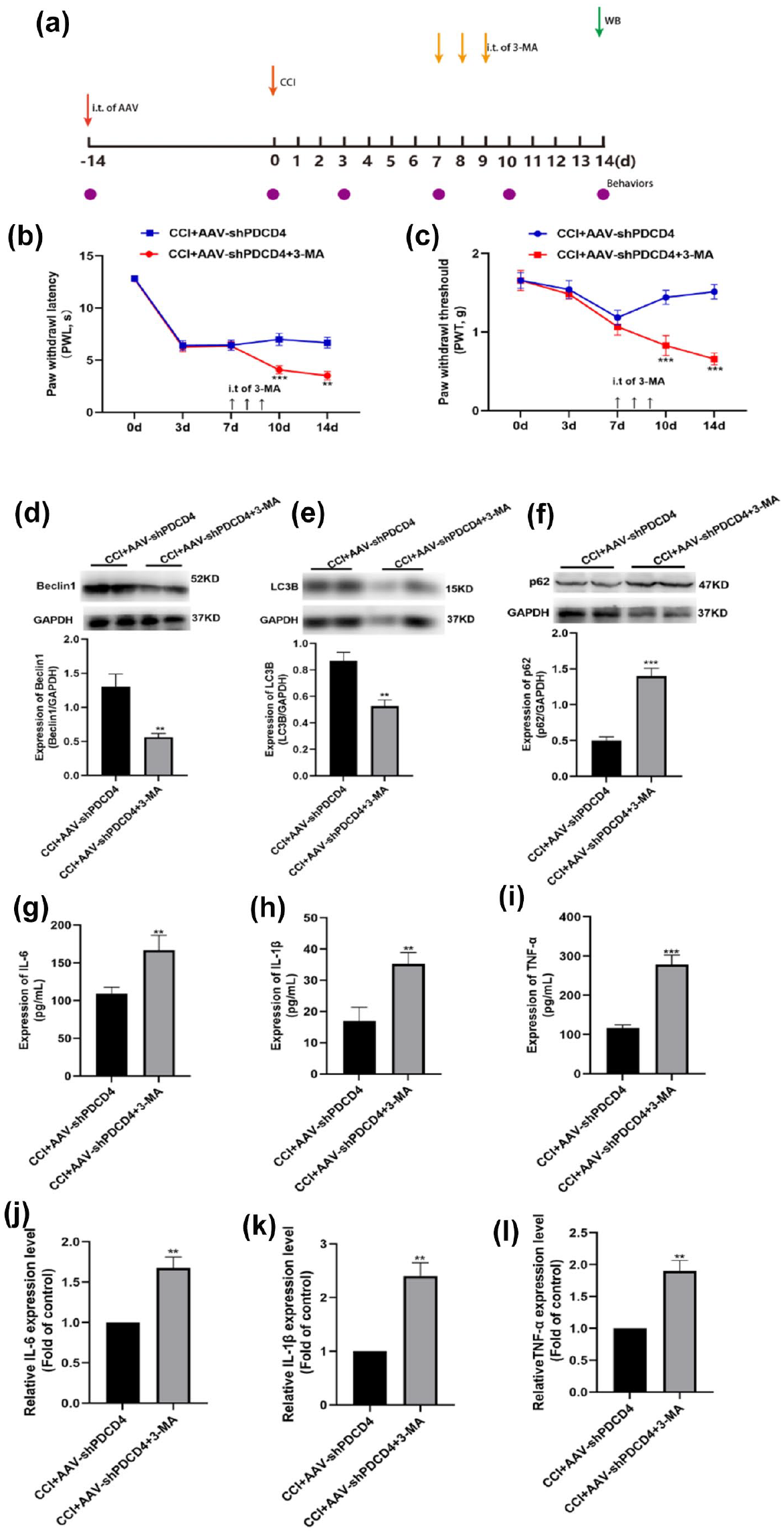
Effect of the autophagy inhibitor 3-MA on pain-related behaviors, autophagy and inflammatory cytokines following AAV-shPDCD4 injection. (a) A schematic timeline for drug administration, behavioral detection, and tissue extraction. (b–c) Intrathecal injection of autophagy inhibitor 3-MA markedly reversed the analgesic effect of PDCD4 inhibition (**p < 0.01, ***p < 0.001 compared with the CCI+AAV-shPDCD4 group, n = 8 per group). (d–f) Intrathecal injection of autophagy inhibitor 3-MA suppressed the autophagy activation induced by PDCD4 inhibition (**p < 0.01, ***p < 0.001 compared with the CCI+AAV-shPDCD4 group, n = 4 per group). (g–i) ELISA data showed intrathecal injection of autophagy inhibitor 3-MA markedly reversed the suppression of pro-inflammatory cytokines (IL-1β, IL-6, and TNF-α) by PDCD4 inhibition. (**p < 0.01, ***p < 0.001 compared with the CCI+AAV-shPDCD4 group, n = 4 per group). (j–l) qRT-PCR results showed intrathecal injection of autophagy inhibitor 3-MA markedly reversed the suppression of pro-inflammatory cytokines (IL-1β, IL-6, and TNF-α) by PDCD4 inhibition. (**p < 0.01, ***p < 0.001 compared with the CCI+AAV-shPDCD4 group, n = 4 per group).
Discussion
Neuropathic pain remains a challenge in clinical practice, and its underlying mechanisms are not yet fully understood. In this study, we explored the role of spinal dorsal horn PDCD4 in regulating neuropathic pain. (1) CCI induces upregulation of PDCD4 expression in spinal dorsal horn; (2) Inhibiting PDCD alleviates pain hyperalgesia in CCI mice; (3) PDCD4 may regulates neuropathic pain through autophagy; (4) PDCD4 may regulate neuropathic pain by regulating the MAPK and neuroinflammation.
In our study, we find an elevation of PDCD4 expression in the spinal cord of CCI mice, which started on postoperative day 7 and persisted until day 21, consistent with previous research. 17 Further investigation reveals the expression of PDCD4 is in neurons and microglia in the spinal dorsal horn. Notably, inhibition of PDCD4 using AAV-shPDCD4 significantly attenuates pain hypersensitivity in CCI mice, highlighting the significance of PDCD4 as a key molecule in regulating neuropathic pain.
Autophagy serves as an immune defense mechanism by which the body combats external stimuli. It is a lysosome-mediated process that involves the degradation of cellular organelles, proteins, and RNA. 18 This cellular response mechanism acts as a resistance mechanism against external survival pressures and plays a crucial role in maintaining cellular substance and energy balance. 32 Morphologically, it is characterized by the appearance of autophagosomes in the cytoplasm, which subsequently fuse with lysosomes for degradation. When autophagy is induced, LC3, the most important and reliable index for autophagy induction in mammals, can be transferred from cytoplasm to autophagosome membrane and transformed from LC3I to LC3II. P62 could interact with LC3II, and the accumulation of p62 indicates autophagy inactivation. Beclin-1, one autophagy biomarker promotes autophagosome maturation. Dysregulation of cellular autophagy has been implicated in the occurrence and progression of neuropathic pain. 31
Activation of cellular autophagy has been shown to effectively alleviate neuropathic pain. 27 Activation with autophagy agonist rapamycin can alleviate pain through autophagy. 33 These studies indicate autophagy plays an important role in pain regulation. PDCD4, an important molecule involved in autophagy regulation, plays an important role in various physiological and pathological processes. 7 In this study, it was observed that the expression of LC3B, Beclin-1 were decreased and p62 was increased in the spinal cord of CCI mice. However, inhibiting PDCD4 expression through AAV delivery increases the expression of Beclin-1and LC3B, suppressed the expression of p62, indicating that PDCD4 regulates neuropathic pain in mice through autophagy. Pre-injection of autophagy inhibitor 3-MA reversed the pain relief and autophagy regulation effects of AAV-shPDCD4 in CCI mice. These results suggested that PDCD4 regulates neuropathic pain in mice through autophagy. It is interesting that AAV-shPDCD4 increased the expression of Beclin1 in sham group mice, and we speculate that it may be due to the inhibition of PDCD4, which led to an increase in autophagy levels in sham operated mice.
Emerging lines of evidence indicated central sensitization through neuronal and non-neuronal immune cells centrally plays a central role in the initiation and maintenance of chronic pain processing.34–36 Spinal neuroinflammation is a crucial mechanism underlying chronic pain. In chronic pain models, activation of spinal glial cells leads to the release of various inflammatory mediators, which bind to receptors on neurons/glial cells, initiating intracellular signaling and amplifying pain signals, ultimately resulting in central sensitization.37–39 Previous studies have suggested a possible link between nociceptive behavior and glial activation after CCI. 40 Moreover, glial activation could secrete glial mediators, including pro-inflammatory cytokines, which could enhance neuronal hyperactivity to further facilitate pain in the spinal cord of CCI mice. 41 Recent studies have found that PDCD4 plays a regulatory role in LPS-induced neuroinflammation. 42 In this study, it was found that PDCD4 is expressed in spinal neurons and microglia of CCI mice. Inhibiting PDCD4 through AAV delivery reduces the expression of inflammatory cytokines suggesting that PDCD4 regulates neuroinflammation in CCI mice.
The MAPK pathway is a crucial signaling pathway involved in the regulation of chronic pain and neuroinflammation. 43 Spinal MAPK pathway was activated in chronic pain procession. 44 Previous studies have shown that PDCD4 regulates MAPK pathway, which are implicated in the pathogenesis of tumors and other diseases.45,46 In this study, p-ERK, p-JNK, and p-p38MAPK are upregulated in spinal cord of CCI mice, inhibiting PDCD4 suppressed the expression of the phosphorylation and activation of all three substreams of the MAPK signaling including ERK, JNK, and p38MAPK, indicating that PDCD4 is involved in the regulation of neuropathic pain in mice through the modulation of the MAPKs. In particular, it is generally believed that chemokines and proinflammatory cytokines released from activated glial cells may together form a chemokine-cytokine networks to regulate the secretion of themselves in a positive feedback manner, which facilitates the development of central sensitization at the spinal level, and leads to persistent pain.47,48 Furthermore, the MAPK pathways could be major downstream targets for PDCD4/autophagy-mediated neuroinflammation and pain sensitization. 16 Interestingly, in our study, PDCD4 was mainly expressed in neurons, but JNK was mainly expressed in astrocytes and p38MAPK was mainly expressed in microglia during pain state. We all know that in pain states, the cross-talk between neurons and glial cells, as well as between glial cells, affects each other. Therefore, we speculate that PDCD4 may indirectly affect the MAPKs pathway of astrocytes and microglia through these cross-talks.
In conclusion, we demonstrated for the first time that the significant role of tumor suppressor PDCD4 in the regulation of neuropathic pain mice through autophagy and neuroinflammation. PDCD4 may emerge as an important therapeutic target for the treatment of neuropathic pain in the future.
Footnotes
Acknowledgements
Funding from the National Natural Science Foundation of China is gratefully acknowledged.
Author contributions
Ting Zhang designed and performed the experiments, Le Qi and Kai Sun acquisition, analysis, or interpretation of data for the work. Xiang Huan and Hao Zhang provided animals drafting the work. Liwei Wang coordinated the study, interpreted the data, and provide approval for publication of the content. All authors read and approved the final manuscript.
Data availability
The data used in the study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by National Natural Science Foundation of China (82001169) to Ting Zhang.
Ethics approval
Ethics approval and consent to participate. All procedures regarding the care and use of animals were approved by the ethics committee of Xuzhou Medical University of China. All methods were performed in accordance with approved guidelines.
Consent for publication
Not applicable.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
