Abstract
Traumatic neuropathic pain (TNP) is caused by traumatic damage to the somatosensory system and induces the presentation of allodynia and hyperalgesia. Mitochondrial dysfunction, neuroinflammation, and apoptosis are hallmarks in the pathogenesis of TNP. Recently, mitochondria-based therapy has emerged as a potential therapeutic intervention for diseases related to mitochondrial dysfunction. However, the therapeutic effectiveness of mitochondrial transplantation (MT) on TNP has rarely been investigated. Here, we validated the efficacy of MT in treating TNP. Both in vivo and in vitro TNP models by conducting an L5 spinal nerve ligation in rats and exposing the primary dorsal root ganglion (DRG) neurons to capsaicin, respectively, were applied in this study. The MT was operated by administrating 100 µg of soleus-derived allogeneic mitochondria into the ipsilateral L5 DRG in vivo and the culture medium in vitro. Results showed that the viable transplanted mitochondria migrated into the rats’ spinal cord and sciatic nerve. MT alleviated the nerve ligation-induced mechanical and thermal pain hypersensitivity. The nerve ligation-induced glial activation and the expression of pro-inflammatory cytokines and apoptotic markers in the spinal cord were also repressed by MT. Consistently, exogenous mitochondria reversed the capsaicin-induced reduction of mitochondrial membrane potential and expression of pro-inflammatory cytokines and apoptotic markers in the primary DRG neurons in vitro. Our findings suggest that MT mitigates the spinal nerve ligation-induced apoptosis and neuroinflammation, potentially playing a role in providing neuroprotection against TNP.
Keywords
Introduction
Traumatic neuropathic pain (TNP) is a type of chronic pain that is caused by accidental injury to the somatosensory system and typically affects young adults. 1 It is characterized by spontaneous pain, allodynia, and hyperalgesia and is often resistant to pharmacological treatments. 2 TNP not only significantly impacts patients’ quality of life but also has negative socio-economic consequences. Therefore, developing new therapeutic approaches to address TNP is a crucial issue. Recent research suggests that neuroinflammation, mitochondrial dysfunction, and consequent cell death play a critical role in the development and persistence of TNP.3–6
Traumatic damage to the peripheral somatosensory nerves leads to excessive calcium influx and the infiltration of inflammatory mediators, which causes sensitization of the afferent fibers and amplification of pain signals. This results in local microglia/macrophages and astrocytes activation and the secretion of pro-inflammatory cytokines such as interleukin-6 (IL-6) and tumor necrosis factor (TNF), which recruit circulating immune cells that release large amounts of pro-inflammatory cytokines. 7 TNF activates the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) signaling, 3 which is implicated in the development of hyperalgesia in patients with neuropathic pain.3,6 Additionally, TNF and IL-6, as neuroinflammatory mediators, activate STAT3 and caspase 8, which in turn trigger extrinsic apoptosis pathways.8–10 These unresolving glial reactions and subsequent inflammatory events plays a crucial role in the transition from nerve injury to the development of chronic pain. Hence, managing the trauma-induced nerve injury and the ensuing neuroinflammation in the early stages appears to be crucial in halting the advancement of chronic pain development.
Mitochondria play a crucial role in generating adenosine triphosphate (ATP), which serves as the energy currency of cells. 11 Given the high energy demands of the nervous system, the impaired mitochondrial function can have severe consequences for neurological health. In fact, several neurological disorders, such as stroke, spinal cord injury, Alzheimer’s disease, Parkinson’s disease, and Huntington’s disease, have been linked to mitochondrial dysfunction.12–14 Notably, mitochondrial dysfunction is a common feature of neuropathies 15 and contributes to the development of TNP. 4 Neuroinflammation-induced mitochondrial dysfunction in TNP can lead to significant neuronal loss through intrinsic apoptotic pathways. 16 Therefore, targeting mitochondrial dysfunction may hold promise as a therapeutic approach for treating neurological disorders that involve mitochondrial dysfunction.
Previous research has demonstrated that mitochondria exhibit high mobility and can transfer between cells in the nervous system. For example, neurons can transfer damaged mitochondria to astrocytes for physiological disposal, 17 while astrocytes can transfer functional mitochondria to ischemic neurons to support cell viability and recovery under ischemic stress. 18 The transferability of mitochondria has led to the development of mitochondrial transplantation (MT), which involves the direct transplantation of healthy mitochondria to replace or rescue dysfunctional mitochondria in lesioned sites, resulting in a rapid increase in mitochondrial content. 19 Animal studies have proven the beneficial effects of MT on various injuries, including ischemia/reperfusion-induced myocardial injuries,19,20 brain injury,21,22 and traumatic spinal cord injury.23,24 Clinical studies have also shown the effectiveness of MT in helping pediatric patients with heart failure wean off central extracorporeal membrane oxygenation support. 25 Consequently, MT is a potential strategy for treating diseases associated with mitochondrial dysfunction. However, its therapeutic effectiveness for TNP remains unknown.
Herein, we established both in vivo and in vitro models of neuropathic pain by conducting an L5 spinal nerve ligation (SNL) in rats and exposing the primary dorsal root ganglion (DRG) neurons to capsaicin, respectively, to investigate the therapeutic efficacy of MT in TNP. In vivo, we examined the short-term effects of MT during the acute phase of nerve injury on the SNL-induced pain hypersensitivity, neuroinflammation, and apoptosis in rats. In vitro, the effects of exogenous mitochondrial treatment on capsaicin-induced inflammatory responses and apoptosis in primary DRG neurons were determined.
Materials and methods
Animals
Adult male Sprague-Dawley (SD) rats aged 8 weeks, weighing 250∼300 g, were purchased from BioLASCO (Nangang District, Taipei, Taiwan). The rats were kept in individual ventilated cages with ad libitum access to food and water, and under controlled environmental conditions of temperature (24 ± 1°C) and humidity (55 ± 10%), with an 11-h light/13-h dark cycle (lights turned on at 7 a.m.). All experimental procedures were conducted during the light-on phase.
A total of 35 male SD rats were used, out of which 8 rats served as donors for allogeneic mitochondria and 27 were used in the experiments. Among these 27 rats, 3 were used to determine the distribution of transplanted mitochondria, while the remaining 24 were randomly and equally divided into three groups, namely Sham, SNL/MT−, and SNL/MT+ groups (with eight rats per group). The SNL/MT− rats underwent SNL and received intra-DRG administration of mitochondrial isolating buffer, the vehicle used to resuspend the transplanted mitochondria. The SNL/MT+ rats underwent SNL and received intra-DRG administration of allogeneic mitochondria. The Sham rats underwent the same surgical procedures as the other groups, but without SNL and served as controls. In each group, five rats were subjected to evaluations of pain hypersensitivity and immunoblotting, while the other three rats were used for the immunofluorescence staining. Furthermore, 56 rat pups aged 1–3 days were used for culturing primary DRG neurons.
Allogeneic mitochondrial isolation
Functional extracellular mitochondria were isolated from the soleus, pectoralis major, biceps, gastrocnemius, and abdominal muscles of healthy donor rats. The rats were anesthetized with 4∼5% isoflurane (Panion and BF Biotech Inc., Nangang District, Taipei, Taiwan) and placed in a prone position, after which an incision was made at the midline of their dorsal lower limb, extending from the ankle to the popliteal fossa. The superficial connective tissues and the gastrocnemius muscles were dissected to expose the soleus muscles. Then, the bilateral soleus muscles were excised, immersed in phosphate buffered saline (PBS), cut into tiny pieces, and homogenized with mitochondrial isolation solution (Cat. #: 89801, Thermo Fisher Scientific, Waltham, MA, USA) with a glass tissue grinder. The homogenates were then centrifuged at 700 × g for 10 min at 4°C. The supernatants were collected and subjected to a second centrifugation at 3000 × g for 15 min at 4°C to pull down the mitochondria. The supernatants were discarded, and the pellets were washed with a wash buffer, followed by centrifugation at 12,000 × g for 5 min at 4°C, twice. The washed pellets containing the isolated mitochondria were weighed with microbalance and resuspended in mitochondrial isolating buffer at a concentration of 100 µg/µL until they were ready to be used in both in vivo and in vitro experiments.
Measurements of activity of respiratory chain components within the isolated mitochondria
The activities of complex I, II, and IV within the isolated mitochondria (total 100 µg) were assessed using commercial assay kits (complex I, Cat. #: 700930; complex II, Cat. #: 700930; complex IV, Cat. #: 700990, Cayman Chemical, Ann Arbor, MI, USA), following the manufacturers’ instructions.
Mitochondrial labeling
The mitochondria isolated from the soleus were stained with MitoTracker deep red FM (MTDR, Cat. #: M22425, Thermo Fisher Scientific) at 37°C for 30 min, washed, and suspended in mitochondrial isolating buffer.
Induction of unilateral L5 SNL
The rats were anesthetized by intraperitoneally injecting a mixture of anesthetics, which included Zoletil® 50 (40 mg/kg, Virbac, Carros, France), 9% xylazine (Bayer, Leverkusen, North Rhine-Westphalia, Germany), and 2% atropine sulfate (Cat. #: 1045009, Sigma-Aldrich, St. Louis, MO, USA), and then placed in a prone position. A subcutaneous injection of 1 mL of the antibiotic enrofloxacin transami (5%, Bayer) was administered to each rat. The lower back of each rat was shaved and sterilized with iodine solution. Local anesthesia was achieved by injecting 0.5 mL of 2% xylocaine (Cat. #: L7757, Sigma-Aldrich) 5 min prior to the incision. A 5-cm skin incision was made on the lower back at the level of L4-L6. The right paraspinal muscles were dissected and retracted laterally to expose the L4-L6 mammillary and L4-L5 transverse processes. The L5 spinal nerve was carefully identified, dissected, and traced upward to the L5 DRG. Using a 4-0 silk, the L5 spinal nerve was tightly ligated 1 mm distal to the DRG to induce SNL.
Intra-DRG microinjection
After SNL, ipsilateral intra-DRG microinjections were administered to the rats. A syringe with 30-G needle, containing 1 µL of allogeneic mitochondria solution (100 µg of total protein, resuspended in mitochondrial isolating buffer), was held steadily on a three-dimensional stereotaxic apparatus and connected to a syringe pump (Model #: KDS101, KD Scientific Inc., Holliston, MA, USA) with an injecting rate of 0.2 µL/min. The injection depth was approximately half of the DRG thickness (400 μm). After the injection, which lasted for 5 min, the syringe was withdrawn to prevent liquid spillage from the injection site. The Sham/MT- and SNL/MT- rats received an intra-DRG microinjection of an equal volume of mitochondrial isolating buffer. A 4-0 nonabsorbable silk was used to close the incision wound, and the rats were placed on a heating plate until they regained consciousness.
Mechanical von Frey test
The mechanical stimulation-related pain behavior was evaluated by the von Frey test on the post-SNL day (PSD) 1, 3, 5, and 7, and 1 day prior to SNL (Figure 1(a)). The von Frey tip with an electronic host machine (Model #: BIO-EVF5, BiosebLab, Vitrolles, Bouches-du-Rhône, France) was gently applied onto the rat’s middle plantar surface of the bilateral hind paws and forced upward, extremely slowly. The machine recorded the largest force when the rat withdrew its leg. Each test included 5 repeated measurements, and the average of these 5 repetitions was presented as a single data point, termed the paw withdrawal threshold (PWT). The spatiotemporal distribution of transplanted mitochondria and the effects of MT on SNL-induced mechanical and thermal pain hypersensitivity in rats. (a) The experimental timeline to study the effects of MT on TNP in vivo. (b) Representative micrographs of DAPI (blue) and MTDR (red) signals in the ipsilateral DRG and spinal cord of sham rats and rats receiving SNL with or without MT on PSD7. Scale: 100 µm in the upper and lower rows, 50 µm in the middle row. (c) Quantitative results of the mechanical von Frey test applied on the ipsilateral side. (d) Quantitative results of the mechanical von Frey test applied on the contralateral side. The paw withdrawal threshold (PWT) means the largest force when the rat withdrew its leg. (e) Quantitative results of the thermal Hargreaves test applied on the ipsilateral side. (f) Quantitative results of the thermal Hargreaves test applied on the contralateral side. The paw withdrawal latency (PWL) means the period from the initiation of heating the rat’s paw on the hot plate to the time of the paw withdrawal from the hot plate. N = 5 rats/group. Data were expressed as mean ± standard deviation. **p < .01, ***p < .001, ****p < .0001, repeated measures two-way ANOVA. The details of the statistic results of post-hoc comparisons were described in the main text.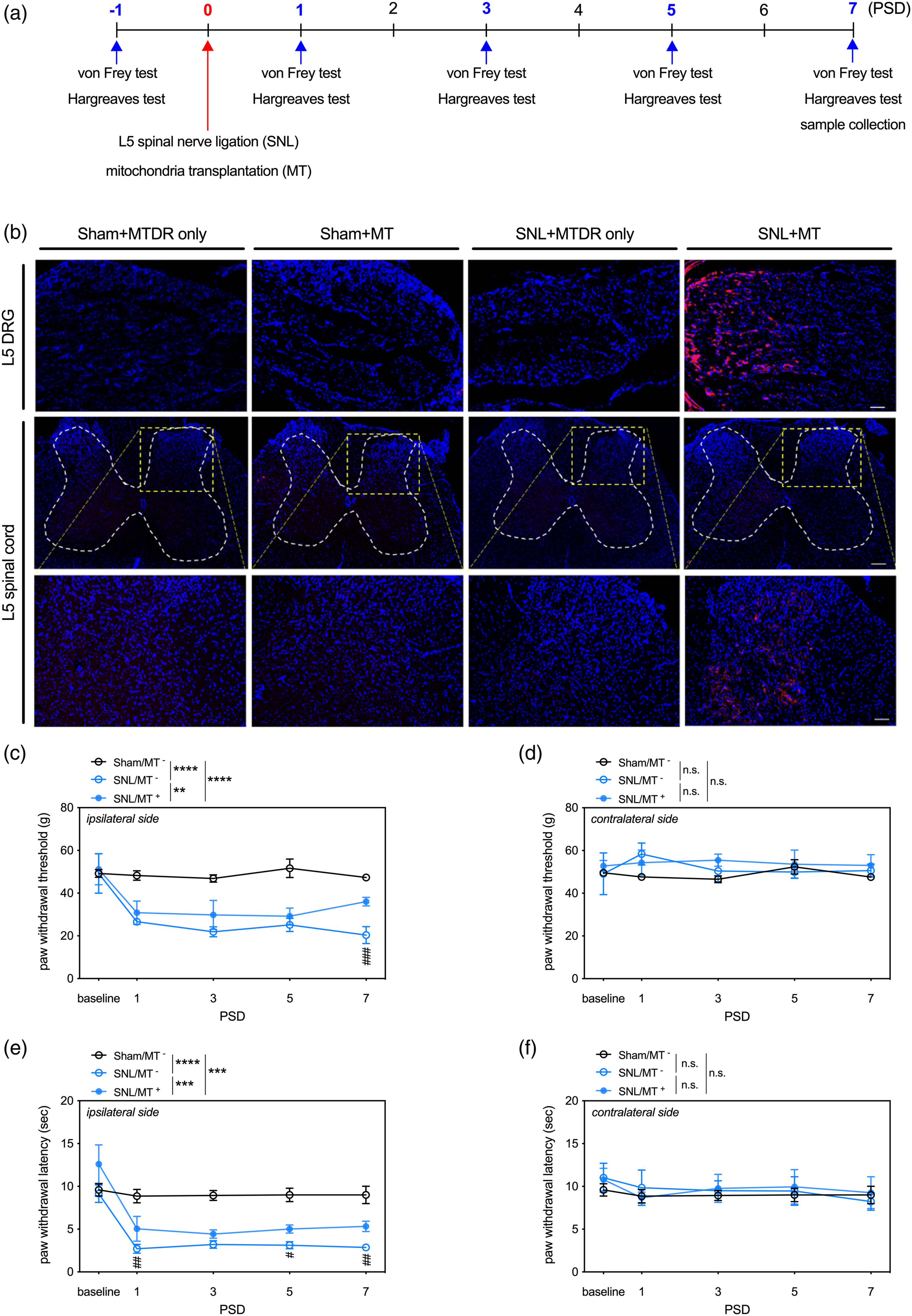
Thermal Hargreaves test
The thermal stimulation-related pain behavior was evaluated by the Hargreaves test with a Hargreaves apparatus (Model #: 7370, Ugo Basile, Gemonio, VA, Italy) on PSD 1, 3, 5, and 7, and 1 day prior to SNL (Figure 1(a)). The heat was generated by an infrared source at a temperature of 55°C, with a cutoff time of 20 s. The infrared heater was directed at the middle plantar surface of the rat’s hind paw. The thermal tolerance interval was defined as the period from the initiation of heating the rat’s paw on the hot plate to the time of paw withdrawal from the hot plate. Each test included 5 repeated measurements, and the average of these 5 repetitions was presented as a single data point, termed the paw withdrawal latency (PWL).
Tissue preparation
On the day of termination of the experiments, the rats were anesthetized with 4–5% of isoflurane (Panion and BF Biotech Inc.) and transcardially perfused with 600 mL of chilled normal saline. Their L5 spinal cord, L4-L6 DRGs, and bilateral sciatic nerves were quickly dissected out. To determine the distribution of the transplanted mitochondria and perform the immunofluorescence staining, the specimens were post-fixed with 4% paraformaldehyde at 4°C for 16 h and dehydrated in PBS solution with 30% sucrose for 3 days. For the immunoblotting, the specimens were immediately immersed in liquid nitrogen and stored at −80°C.
Determination of temporal and spatial distribution of transplanted mitochondria
The fixed and dehydrated specimens were prepared into microsections with a cryostat. The spinal cord specimens were transversely and horizontally cut into 20-μm sections. The DRG specimens were coronally sectioned at a thickness of 8 and 10 μm, respectively. The sections were mounted on the glass slides with media containing DAPI (Cat. #: ab104139, Abcam, Cambridge, UK). The fluorescence of MTDR and DAPI was detected, and the relevant images were captured with a fluorescence optical microscope system (Model: IXplore Standard, Olympus, Shinjuku, Tokyo, Japan) equipped with a digital camera (Model: DP80, Olympus).
Immunoblotting
The frozen spinal cord specimens were homogenized with iced T-PER lysis buffer (Cat. #: 78510, Thermo Fisher Scientific Inc.) containing protease inhibitors (Cat. #: 04693116001, Roche, Basel, Switzerland) and phosphatase inhibitors (PHOSS-RO, Roche). The cultured cells were washed with PBS and homogenized with iced cell lysis buffer (Cat. #: 9803, Cell Signaling Technology, Danvers, MA, USA) containing the same protease inhibitors and phosphatase inhibitors. The homogenates were centrifuged at 14,000 × g for 10 min at 4°C. The supernatants were collected and subjected to the determination of protein concentration using a commercial protein assay kit (Cat. #: 23225, Thermo Fisher Scientific Inc.). In total, 30 µg of the extracted protein from each sample was mixed with the sample buffer (Cat. # S3401, Sigma-Aldrich), denatured by boiling, loaded into each well of a 12% polyacrylamide gel, resolved for 2 h, and transferred onto PVDF membranes (Cat. #: IPVH00010, Merck-Millipore, Burlington, MA, USA) using the wet transfer method.
The membranes were incubated overnight (16 h) at 4°C with appropriate dilutions of primary antibodies, including cleaved caspase-3 (1:2,000, Cat. #: 9661, Cell Signaling Technology), Bcl-2 (1:2,000, Cat. #: 12789-1-AP, Thermo Fisher Scientific Inc.), BAX (1:2,000, Cat. #: 2772, Cell Signaling Technology), TNF (1:2,000, Cat. #: ab6671, Abcam), IL-1β (1:2,000, Cat. #: TA321162, OriGene, Rockville, MD, USA), IL-6 (1:2,000, Cat. #: ab6672, Abcam), NF-κB (1:2,000, Cat. #: 8242, Cell Signaling Technology), and α-tubulin (1:10,000, Cat. #: GTX112141, GeneTex, Irvine, CA, USA). Densitometry was carried out using ImageJ software (v2.0.0-rc-69/1.52p, U.S. National Institutes of Health, Bethesda, MD, USA).
Immunofluorescence staining
The transverse sections of the L5 spinal cord were washed with PBS to remove the embedded compound and sequentially blocked with 3% normal goat serum (Cat. #: S-1000-20, Vector Laboratories, Burlingame, CA, USA) solution prepared in PBS containing 0.1% bovine serum albumin (Cat. #: A7906, Sigma Aldrich) and 0.1% Triton X-100 (Cat. #: X100, Sigma Aldrich) for 20 min. Then, the sections were probed with primary antibodies against glial fibrillary acidic protein (GFAP, 1:400, Cat #: G3893, Sigma Aldrich) and ionized calcium-binding adapter molecule 1 (Iba1, 1:200, Cat. #: GTX632426, GeneTex) for 16 h at 4°C, washed with PBS, and incubated with Alexa Flour 488-conjugated secondary antibodies (Cat. #A28175, Thermo Fisher Scientific Inc.) for 1 h at room temperature. The fluorescence images were acquired using a fluorescence optical microscope system (Model: IXplore Standard, Olympus) equipped with a digital camera (Model: DP80, Olympus). The ratios of the GFAP-immunoreactive (GFAP+) and Iba1-immunoreactive (Iba1+) area to the respective total imaged area of the spinal cord were quantified using ImageJ software (v2.0.0-rc-69/1.52p, U.S. National Institutes of Health) and presented as percentages.
Primary DRG neuronal cultures
The DRGs were removed from the postnatal rat pups (1–3 day old), digested with 0.1% collagenase for 1 h, and subjected to 0.25% trypsin digestion for 30 min. The dissociated neurons were seeded at a density of 3 × 105 cells in a poly-L-lysine-coated 6-cm dish, cultured with DMEM-F12 media supplemented with 2% B-27 supplement (Cat. #: 17504044, Thermo Fisher Scientific Inc.), 0.5 mM L-glutamine (Cat. #: 25030081, Thermo Fisher Scientific Inc.), and 10% fetal bovine serum (Cat. #: 10082147, Thermo Fisher Scientific Inc.), and maintained at 37°C in an environment containing 5% CO2. Post seven day-culture, DRG neurons were exposed to 10 µM of capsaicin (Cat. #: 92350, Cayman Chemical) for 24 h. Then 100 µg of exogenous mitochondria was administered to the primary DRG neurons 1 h after the beginning of the capsaicin treatment. Control groups received equivalent volumes of the respective vehicle solutions. After the 24-h capsaicin and/or the 23-h mitochondrial treatments, the cells were used for further experiments.
Mitochondrial membrane potential assay
Mitochondrial membrane potential was measured using the JC-1 Mitochondria Membrane Potential Assay Kit (Cat. #: 10009172, Cayman Chemical). Following the capsaicin and/or mitochondrial treatments, the culture media were replaced to wash out the externally introduced allogenic mitochondria, and the cells were subsequently stained by JC-1 according to the manufacturer’s protocol. The fluorescent intensities at Ex/Em: 535/595 nm and Ex/Em: 485/535 nm were detected with a fluorescent plate reader (Model: SpectraMax iD5, Molecular Devices, San Jose, CA, USA). The ratio of red fluorescence intensity at Ex/Em: 535/595 nm to green fluorescence intensity at Ex/Em: 485/535 nm was presented. For imaging, the primary DRG neurons were seeded in 8-well chamber slides and underwent the same treatments and JC-1 staining. Afterward, the slides were mounted with DAPI-containing media (Cat. #: ab104139, Abcam). The fluorescence images were acquired using a fluorescence optical microscope system (Model: IXplore Standard, Olympus) equipped with a digital camera (Model: DP80, Olympus).
Quantifications of content levels of TNF, IL-1β, and IL-6 in conditioned media
After the 24-h capsaicin and/or the 23-h mitochondrial treatments, the culture media from primary DRG neurons were collected and subjected to subsequent ELISA assays. The content levels of TNF (Cat. #: ab100785, Abcam), IL-1β (Cat. #: ab255730, Abcam), and IL-6 (Cat. #: ab234570, Abcam) in the conditioned media were assessed using commercially available ELISA kits, following the manufacturer’s guidelines.
Terminal deoxynucleotidyl transferase dUTP nick end labeling assay
The primary DRG neurons were seeded in 8-well chamber slides and treated with 10 μM of capsaicin and 100 μg of exogenous mitochondria for 24 h. Then the terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay was performed using ApopTag Red In Situ Apoptosis Detection Kit (Cat. #: S7165, Sigma-Aldrich) following the manufacturer’s protocol. Following this, the slides were mounted with DAPI-containing media (Cat. #: ab104139, Abcam), and the fluorescence images were acquired using a fluorescence optical microscope system (Model: IXplore Standard, Olympus) equipped with a digital camera (Model: DP80, Olympus). The percentage of TUNEL-positive cells was analyzed and reported.
Statistical analysis
All numerical data are expressed as mean ± standard deviation. Statistical analyses were performed using the Prism software (v. 7.0a, GraphPad Software Inc., San Diego, CA, USA). Significance was set at p < .05. The pain behaviors were analyzed using a repeated measures two-way analysis of variance (ANOVA) followed by Sidak’s multiple comparison test with time and treatment as the main factors. A one-way ANOVA followed by Tukey’s multiple comparison test were performed to analyze the effects of SNL and MT on protein expression examined by immunoblotting and immunofluorescence staining. Designs with two independent variables (the in vitro studies) were analyzed by a two-way ANOVA followed by Tukey’s multiple comparison test to identify if the main effects or interactions were significant.
Results
Transplanted mitochondria migrate along the pain-conducting pathway
Initially, we validated the in vitro functionality of allogenic mitochondria isolated from the various rat muscles, including soleus, pectoralis major, biceps, gastrocnemius, and abdominal muscles, by determining the activity of components of respiratory chain within these isolated mitochondria. Our results showed that the mitochondria isolated from all selected muscles exhibited activities of complex I, complex II, and complex IV at both the two-hour and 3-day time points following the isolation procedures (Supplementary Figure 1). Among them, the mitochondria isolated from soleus showed relatively high respiratory chain activity than those isolated from the other muscle tissues (Supplementary Figure 1), hence we adopted soleus muscle as source of allogenic mitochondria in this study.
Next, to explore the viability and spatiotemporal distribution of the allogenic mitochondria, we labeled the transplanted mitochondria with MTDR, a far red-fluorescent probe routinely used to chemically mark the living mitochondria and traced them along the pain-conducting pathway. Our results showed that, on PSD7, the MTDR-labeled allogenic mitochondria were recognized in the ipsilateral L5 DRG (Figure 1(b), upper row), sciatic nerve (Figure 1(b), middle row), and the dorsal horn of spinal cord (Figure 1(b), lower row) of the SNL rats that received MT, but not in those tissues of Sham rats or those injected with MTDR alone (Figure 1(b), leftmost three columns). These results suggest that the transplanted mitochondria remained viable for at least 1 week and preferably migrate into the injured tissues.
MT alleviates SNL-induced pain hypersensitivity
To examine the effects of MT on SNL-induced pain hypersensitivity, we carried out von Frey and Hargreaves tests before (1 day before) and after the SNL (PSD 1, 3, 5, and 7) (Figure 1(a)). Repeated measures of two-way ANOVAs showed that SNL induced mechanical and thermal pain hypersensitivity by reducing the ipsilateral PWT and PWL (SNL/MT− vs Sham/MT−, p < .0001, Figure 1(c) and(e)). MT elevated ipsilateral PWT (SNL/MT+ vs SNL/MT−, p = .0098, Figure 1(c)) and PWL (SNL/MT+ vs SNL/MT−, p = .0004, Figure 1(e)) in the SNL rats. Sidak’s multiple comparisons tests showed that the SNL/MT+ rats had higher ipsilateral PWT than the SNL/MT− rats on PSD 7 (p = .0001 Figure 1(c)) and had higher ipsilateral PWL than the SNL/MT− rats on PSD 1 (p = .0028), 3 (p = .0209), and 7 (p = .0016) (Figure 1(e)). Compared to the Sham/MT− rats, the SNL/MT+ rats displayed lower PWT (p < .0001) and PWL (p = .0004) (Figure 1(c) and (e), respectively). Moreover, the results of the von Frey and Hargreaves tests applied on the intact contralateral paw revealed no differences between the three groups (Figure 1(d) and (f)).
MT represses inflammation and glial activation in the spinal cord of rats with SNL
To explore possible mechanisms underlying the beneficial effects of MT on SNL-induced pain hypersensitivity, we evaluated the inflammatory profiles in the spinal cord among all groups. Immunoblotting results showed that SNL upregulated the expression of proinflammatory mediators, including TNF (p = .0100), IL-1β (p = .0061), IL-6 (p = .0199), and NF-κB (p = .0244) in the spinal cord (SNL/MT− vs Sham/MT−, Figure 2(a)–(e)). MT repressed the SNL-induced upregulation of TNF (p = .0036), IL-1β (p = .0003), and IL-6 (p = .0017), but not of NF-κB (p = .1064) in the spinal cord (SNL/MT+ vs SNL/MT−, Figure 2(a)–(e)). Moreover, we analyzed the formation of gliosis in the spinal cord by examining the local expression of GFAP and Iba1, the markers for astrocytes and microglia, respectively. Immunofluorescence staining showed that SNL increased the expression of GFAP (p = .0001) and Iba1 (p = .0062) in the dorsal horn of the spinal cord (SNL/MT− vs Sham/MT−, Figure 2(f)–(i)). MT repressed the SNL-induced upregulations of GFAP (p = .0041) and Iba1 (p = .0361) (SNL/MT+ vs SNL/MT−, Figure 2(f)–(i)). Interestingly, we also found that SNL induced an increase in the fraction of Iba1-immunoreactive area in the ventral horn of spinal cord (SNL/MT− vs Sham/MT−, Figure 2(j) and (k)), which was revered by MT (SNL/MT+ vs SNL/MT−, Figure 2(j) and (k)). The effects of MT on expression of pro-inflammatory cytokines and degree of glial activation in the ipsilateral spinal cord of rats. (a) Representative micrographs of immunoblots of TNF, IL-1β, IL-6, and NF-κB. (b)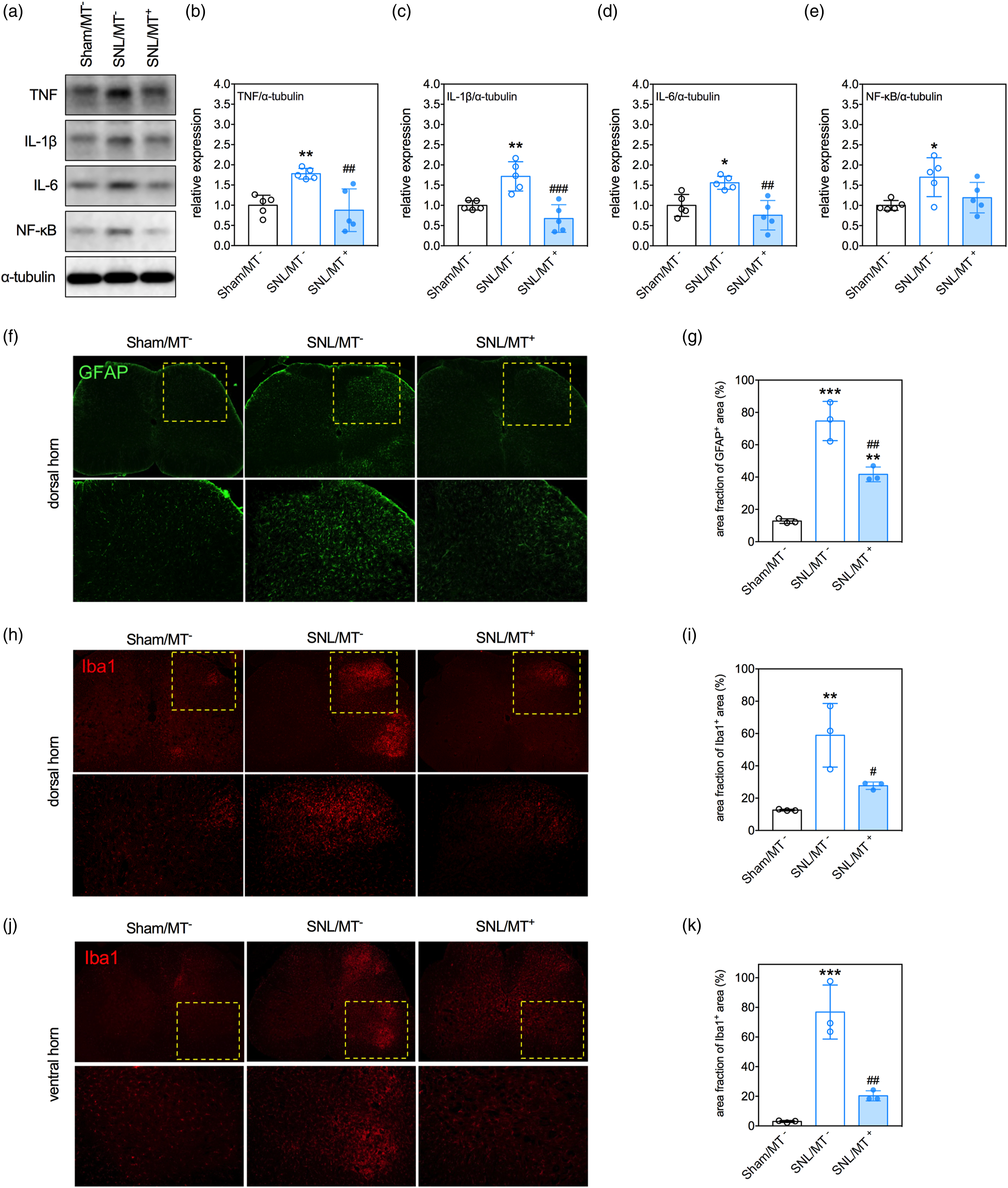
MT ameliorates SNL-induced apoptosis in the spinal cord
The effects of SNL and MT on apoptosis in the spinal cord of rats were determined by examining the levels of cleaved caspase-3, anti-apoptotic Bcl-2, and pro-apoptotic BAX, the molecules involved in the apoptotic pathway, with immunoblotting. Figure 3 showed the levels of cleaved caspase-3 with an increasing trend in the spinal cords of SNL-treated rats although it lost significance. Simultaneously, SNL significantly elevated the levels of the pro-apoptotic protein, BAX, in the spinal cord (p = .0005, SNL/MT− vs Sham/MT−) while the Bcl-2 levels were not affected. Excitingly, the SNL/MT+ rats had lower levels of cleaved caspase-3 (p = .0074) and BAX (p < .0001), and a higher expression ratio of Bcl-2 to BAX (p = .0146), compared to the SNL/MT− rats. The effects of MT on the expression of apoptosis mediators in the ipsilateral spinal cord of rats. (a) Representative micrographs of immunoblots of cleaved caspase 3, Bcl-1, and BAX. (b)–(d) Quantitative results of the relative expression of caspase 3, Bcl-1, and BAX. (e) Quantitative results of the expression ratio of Bcl-2 to BAX. Data were expressed as mean ± standard deviation. ***p < .001, versus Sham/MT−, Tukey’s multiple comparisons. #p < .05, ##p < .01, ####p < .0001, versus SNL/MT−, Tukey’s multiple comparisons. N = 5 rats/group.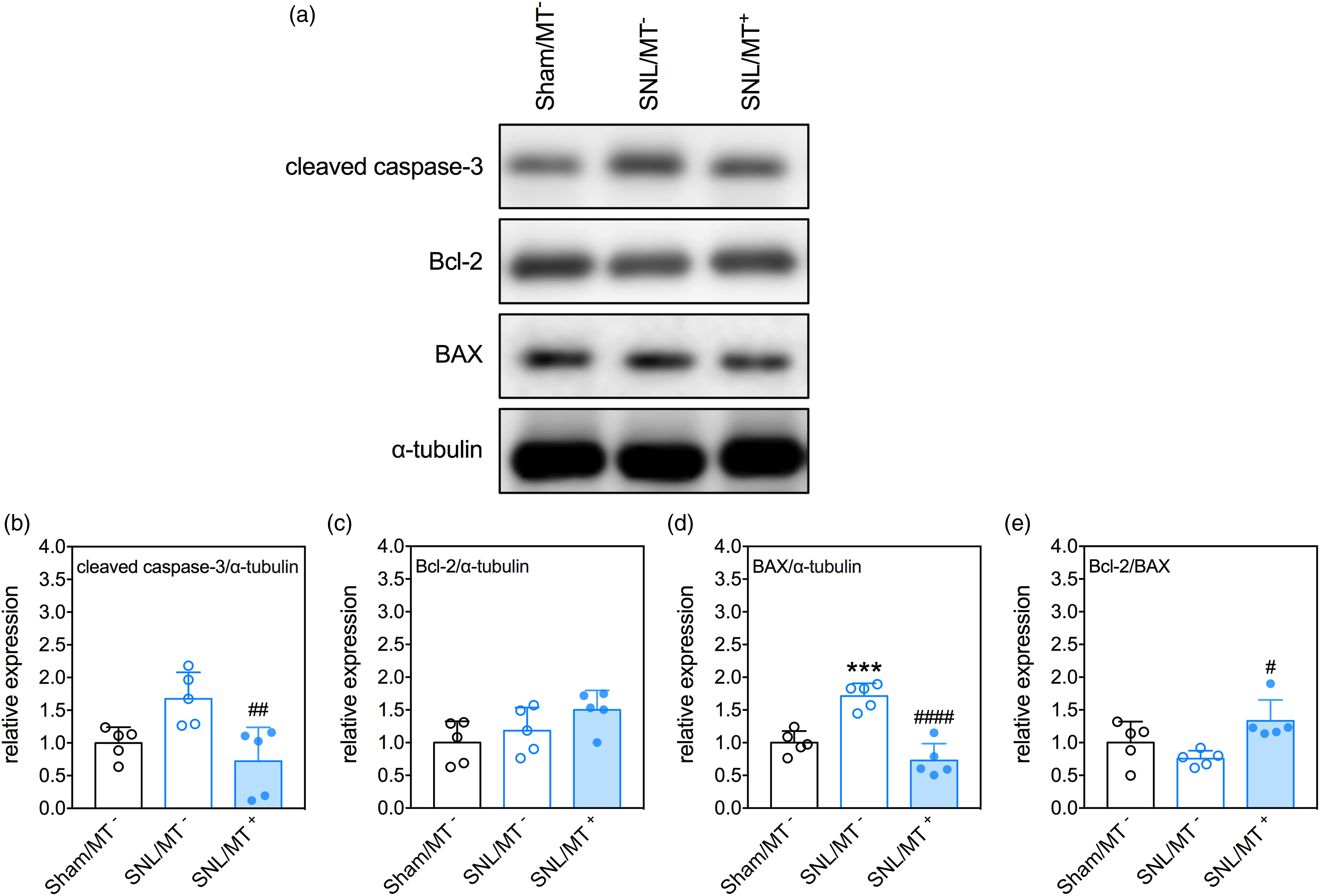
Exogenous mitochondria counteract the capsaicin-induced reduction of mitochondrial membrane potential in primary DRG neurons
Neurons isolated from DRG in culture display similar characteristics to those of their in vivo counterparts.
26
Capsaicin, a substance extracted from hot pepper, is known to induce responses similar to the events of pain stimulation in the neuronal cells, including the release of substance P and apoptosis.27,28 Accordingly, we studied the effects of exogenous mitochondria on capsaicin-induced responses in the primary DRG neurons. First, we determined the differences in mitochondrial membrane potentials of the cells, which were detected by JC-1 staining among the groups. Figure 4 showed that a 24-h treatment with 10 μM of capsaicin reduced the mitochondrial membrane potentials in the primary DRG neurons (p < .0001, capsaicin+/mitochondria− vs capsaicin−/mitochondria−). Administering 100 μg of mitochondria partially reversed the capsaicin-induced loss of mitochondrial membrane potentials in the primary DRG neurons (p = .0032, capsaicin+/mitochondria+ vs capsaicin+/mitochondria−). The effect of treatments with capsaicin and exogenous mitochondria on mitochondrial membrane potential in the primary DRG neurons. (a) Representative images of JC-1 staining and DAPI on primary DRG neurons treated with capsaicin and exogenous mitochondria. JC-1 demonstrates a potential-dependent accumulation within the mitochondria, leading to the formation of J aggregates emitting red fluorescence; upon depolarization, it remains as monomer showing green fluorescence. As a result, mitochondrial depolarization is indicated by a decrease in the red/green fluorescence intensity ratio. Scale: 200 µm. (b) Quantitative results of the red-to-green fluorescence intensity ratio of JC-1 staining. Data were expressed as mean ± standard deviation. *p < .05, ****p < .0001, versus capsaicin−/mitochondria−, Tukey’s multiple comparisons. ##p < .01, versus capsaicin+/mitochondria−, Tukey’s multiple comparisons. N = 5 biological replications.
Exogenous mitochondria repress capsaicin-induced expression of pro-inflammatory cytokines in primary DRG neurons
We analyzed the alteration of production of proinflammatory cytokines in the primary DRG neurons treated with capsaicin and/or mitochondria. The ELISA results showed that a 24-h treatment with 10 μM of capsaicin increased the secretions of TNF (p < .0001) and IL-6 (p = .0001), while not affecting IL-1β, in the primary DRG neurons (capsaicin+/mitochondria− vs capsaicin−/mitochondria−, Figure 5). Moreover, the administration of 100 μg of exogenous mitochondria attenuated capsaicin-induced TNF (p < .0001) and IL-6 (p = .0024) production in primary DRG neurons (capsaicin+/mitochondria+ vs capsaicin+/mitochondria−, Figure 5(a) and (c)). The effect of treatments with capsaicin and exogenous mitochondria on the production of pro-inflammatory cytokines in the primary DRG neurons. (a) Quantitative results of levels of TNF in the conditioned media. (b) Quantitative results of levels of IL-1β in the conditioned media. (c) Quantitative results of levels of IL-6 in the conditioned media. Data were expressed as mean ± standard deviation. ***p < .001, ****p < .0001, versus capsaicin−/mitochondria−, Tukey’s multiple comparisons. ###p < .001, ####p < .0001, versus capsaicin+/mitochondria−, Tukey’s multiple comparisons. N = 5 biological replications.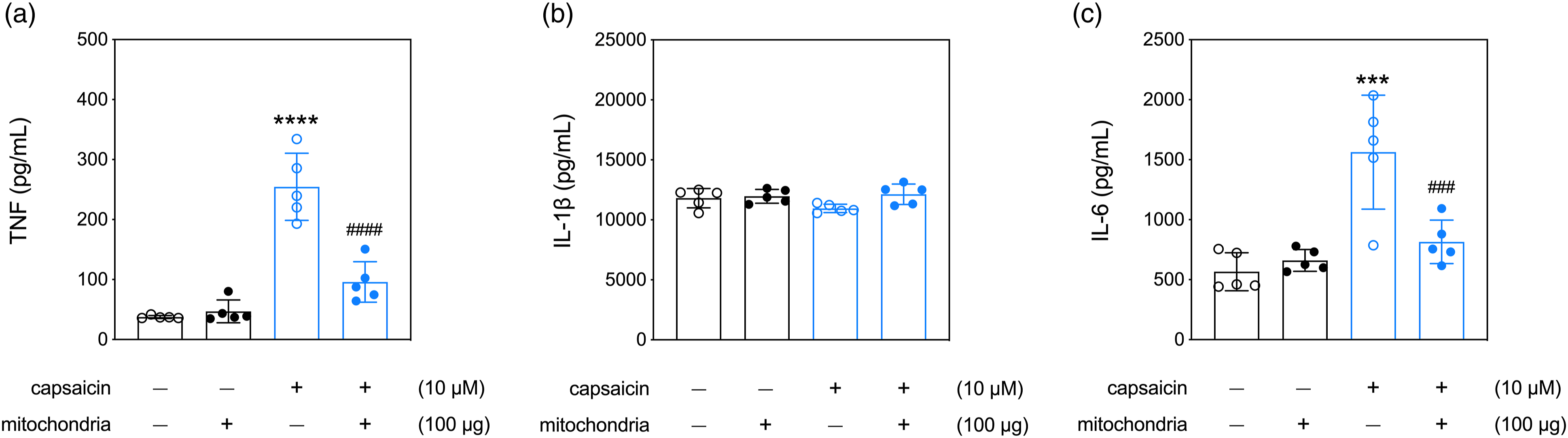
Exogenous mitochondria reverse capsaicin-induced apoptosis in primary DRG neurons
We determined the effects of capsaicin and mitochondria on apoptosis in the primary DRG neurons. First, we used immunoblotting to examine the expression of cleaved caspase-3, Bcl-2, and BAX in the primary DRG neurons. Quantitative results revealed that 10 μM of capsaicin upregulated the levels of cleaved caspase 3 (p = .0473) and BAX (p = .0101) without affecting the expression of Bcl-2 (capsaicin+/mitochondria− vs capsaicin−/mitochondria−, Figure 6(a)–(d)). Exogenous mitochondria at a dosage of 100 μg reduced the expression level of cleaved caspase 3 (p = .0029, capsaicin−/mitochondria+ vs capsaicin−/mitochondria−, Figure 6(a) and (b)). Moreover, exogenous mitochondria reversed the capsaicin-induced increases of levels of cleaved caspase 3 (p = .0002) and BAX (p = .0003) (capsaicin+/mitochondria+ vs capsaicin+/mitochondria−, Figure 6(a), (b) and (d)). Second, the TUNEL assay was performed to visualize the population of apoptotic cells in the primary DRG neuronal cultures. The results showed that treatment of exogenous mitochondria suppressed the capsaicin-induced increase of apoptotic cell density (Figure 6(e) and(f)). The effect of treatments with capsaicin and exogenous mitochondria on apoptosis in the primary DRG neurons. (a) Representative micrographs of immunoblots of cleaved caspase 3, Bcl-1, and BAX. (b)–(d) Quantitative results of the relative expression of cleaved caspase 3, Bcl-1, and BAX. (e) Representative images of the TUNEL and DAPI stainings. Scale: 200 µm. (f) Quantitative results of TUNEL staining. Data were expressed as mean ± standard deviation. *p < .05, ****p < .0001, versus capsaicin−/mitochondria−, Tukey’s multiple comparisons. ###p < .001, ####p < .0001, versus capsaicin+/mitochondria−, Tukey’s multiple comparisons. N = 5 biological replications.
Discussion
In this study, we determined the therapeutic effects of MT on TNP in vivo and in vitro. We found that the functional transplanted mitochondria migrated to the injured tissues and effectively alleviated pain hypersensitivity in the SNL rats. MT also suppressed neuroinflammation and apoptosis in the spinal cord of rats with SNL. Similarly, mitochondrial treatments reversed the capsaicin-induced reduction of mitochondrial membrane potentials, upregulation of pro-inflammatory cytokines, and apoptosis in vitro. Our findings suggest that MT ameliorates apoptosis and neuroinflammation resulting from SNL, potentially contributing to neuroprotection against TNP. These findings offer foundational support for the prospective translation of MT as a treatment for TNP.
MTDR is a mitochondrial potential-dependent dye that labels viable mitochondria.29,30 In this study, the MTDR-labeled allogenic mitochondria were recognized not only in the L5 DRG of rats, but also in the ipsilateral L5 spinal cord and sciatic nerve, highlighting the migrating ability and viability of the transplanted mitochondria. Moreover, by JC-1 staining, we also found that the exogenous mitochondrial could enhance the bioenergetics of mitochondria within the DRG neurons. It is worth noting that we washed out the extracellularly administered allogenic mitochondria by replacing the culture media before the JC-1 staining procedures to minimize any confounding effects on the analysis of JC-1 signals. To our surprise, we did not detect any MTDR fluorescent signals within the cells. This unexpected result was consistently observed in primary DRG neurons treated with capsaicin at intervals of 1, 6, and 24 h from the beginning of MT (Supplementary Figure 2), indicating that the introduced allogenic mitochondria were not internalized by the primary DRG neurons. These findings are consistent with the outcomes observed in our own and other in vivo studies. In the context of experimental myocardial injury model, it was observed that a very limited number of exogenous transplanted mitochondria was incorporated into the host cells. 20 Similarly, in our previous study investigating the therapeutic effects of MT on traumatic spinal cord injury, we found that when viable allogenic mitochondria were administered regionally, they primarily distributed into the surrounding area, with most of these exogenous organelles being located interstitially rather than being taken up by the recipient cells. 23 Given that, the mechanisms underlying how MT attenuates neuroinflammation and apoptosis following traumatic nerve injury necessitate further investigations.
Additionally, our results regarding the spatiotemporal distribution of transplanted mitochondria provide the first evidence that transplanted mitochondria are capable of moving along the neural structures propagating pain sensation in a TNP model. However, in a rat model of traumatic spinal cord injury, the transplanted mitochondria are restricted around the injection site. 31 This discrepancy may have resulted from the nature of the disease models used. Upon traumatic spinal cord injury, the subsequent glial scar formation may limit the migration of the transplanted mitochondria, whereas glial scars are not evident in the SNL model. The manner in which exogenous mitochondria migrate remains to be elucidated. Since mitochondria function as a cellular energy source, it has previously been assumed that transplanted mitochondria increase ATP concentration to support the injured cells in the vicinity. However, this hypothesis was rejected by the finding that the administration of mitochondrial components or ATP/ADP did not reproduce the therapeutic effects of MT on ischemic heart injury. 20 The current findings provide novel evidence that transplanted mitochondria could repress the inflammatory microenvironment.
Nerve injury evokes severe neuroinflammatory responses and subsequently induces the release of various pro-inflammatory cytokines. 32 Elevated pro-inflammatory cytokine levels are positively correlated with the severity of neuropathic pain.33,34 Among them, TNF, IL-1β, and IL-6 play critical roles in the pathogenesis of neuropathic pain.35,36 TNF is upregulated after nerve injury, elicits excess inflammatory effects by activating NF-κB signaling, triggers the extrinsic apoptotic pathway by binding to membrane-bound TNF receptors, and consequently induces neuropathic pain.3,37,38 The IL-1β and IL-6 knockout mice show milder pain behaviors after nerve injury than the wild-type ones.39,40 Hence, several studies investigating the therapeutic effects of anti-inflammatory approaches on neuropathic pain have been conducted. For example, inhibiting TNF signaling was found to induce pronounced pain-relieving effects in nerve injury models.6,41 Herein, we also demonstrated that SNL upregulated the expression of TNF, IL-1β, and IL-6 in the spinal cord of rats. Importantly, we found that MT suppressed the spinal expression of TNF, IL-1β, and IL-6 in the SNL rats. These findings are in agreement with those of a previous study showing that injecting viable mitochondria into the ischemic-reperfusion injured heart reduces the local expression of TNF and IL-6.2 24 In the primary DRG neuronal cultures, we also demonstrated that treating exogenous mitochondria counteracted the capsaicin-induced upregulation of TNF and IL-6. Moreover, our histologic results showed that SNL induced microgliosis in both the dorsal and ventral horns of the spinal cord, which was attenuated by MT. Microgliosis in the spinal dorsal horn is known as a major pathomechanism underlying the development of central sensitization of neuropathic pain.42,43 Damaged proprioceptive sensory afferents and axotomized motoneurons have the capacity to trigger microgliosis in the spinal ventral horn, specifically in laminae 8 and 9, along their central projections.44–46 Collectively, our findings suggest that the heightened presence of pro-inflammatory mediators and the increased reactivity of glial cells within the spinal cord of rats subjected to SNL were effectively suppressed by MT treatment. This effect of MT may contribute to the pain-relieving benefits associated with MT treatment. Furthermore, MT may be better than current pharmacological reagents targeting a single cytokine or pro-inflammatory mediator in controlling inflammation in neuropathic pain. However, it is noteworthy that since apoptosis and inflammation affect each other, the suppression of neuroinflammation by MT may occur through the amelioration of apoptosis rather than through direct mediation of the inflammatory responses, and vice versa.
Nerve injury-induced neuroinflammation leads to mitochondrial dysfunctions and subsequent neural apoptosis.16,47 We found that SNL upregulated the apoptotic markers in the ipsilateral spinal cord of rats, which were reversed by MT. In the primary DRG neurons, we also found that exogenous mitochondria reduced the capsaicin-induced increases in the expression of apoptotic markers and the density of apoptotic cells. In line with our findings, it has been reported that MT in the ischemia myocardium 24 and injured spinal cord 31 exerts anti-apoptotic effects. Since the extrinsic apoptosis pathway is mainly triggered by TNF signaling,37,38 MT may palliate SNL-induced apoptosis by inhibiting the TNF pathway. Notably, the activation of intrinsic apoptotic pathway is highly related to mitochondrial dysfunction. 48 Thus, transplantation of viable mitochondria has the potential to correct mitochondrial dysfunction and then attenuate intrinsic apoptosis.
Although we demonstrated that MT alleviated SNL-induced pain hypersensitivity by repressing neuroinflammation and apoptosis, it still exists a few limitations in our study. First, the mitochondria were directly injected into the L5 DRG, which could cause damage to the DRG. Since this pilot study aimed to delineate the therapeutic effects of the transplantation of allogenic mitochondria on TNP, direct injection into the ganglion could guarantee that the transplanted mitochondria had access to neural tissues. Our results have shown positive therapeutic effects. Therefore, alternative delivery routes with less invasion for MT, e.g., intravenous and distal sciatic nerve injection, should be considered for further translational applications. Second, a single injection of allogenic mitochondria performed in the study showed the therapeutic effects on SNL-induced TNP. Since neuroinflammation in neuropathic pain can persist for a long time, a single shot of MT may not be sufficient to suppress the lasting inflammatory events. A study comparing the therapeutic effects of single and repetitive administrations of allogenic mitochondria on TNP should be conducted in the future.
In conclusion, we demonstrated the therapeutic effects of the transplantation of allogeneic mitochondria on SNL-induced TNP in rats. Transplanted mitochondria migrate along the neural structures propagating pain sensation and are still active. These exogenous mitochondria suppress inflammation and apoptosis in the TNP models in vitro and in vivo. We provide basic pre-clinical evidence for the further translational application of MT in treating TNP.
Supplemental Material
Supplemental Material - Mitochondrial transplantation attenuates traumatic neuropathic pain, neuroinflammation, and apoptosis in rats with nerve root ligation
Supplemental Material for Mitochondrial transplantation attenuates traumatic neuropathic pain, neuroinflammation, and apoptosis in rats with nerve root ligation by Chi-Chen Huang, Hsin-Yi Chiu, Po-Hsuan Lee, Shih-Yuan Fang, Ming-Wei Lin, Hui-Fang Chen and Jung-Shun Lee in Molecular Pain
Footnotes
Acknowledgements
We are grateful to thank Dr. Yen-Chin Liu for providing the instruments for behavior tests.
Author contributions
MWL, SYF, and JSL designed the study. HYC, CCH, PHL, and HFC performed the animal experiments and measured the behavior function. HYC, CCH and CYH performed tissue collection and analysis. PHL; SYF, and JSL contributed to the statistical analysis and interpretation of data. HYC, CCH, and JSL contributed to drafting the manuscript. All authors read and approved the final version of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Cheng Kung University Hospital, Taiwan (grant number: NCKUH-11006016) and Ministry of Science and Technology of Taiwan (grant number: MOST- 110-2314-B-006 -075 -MY2).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
