Abstract
With the recent progress in personal care robots, interest in wearable exoskeletons has been increasing due to the demand for assistive technologies generally and specifically to meet the concerns in the increasing ageing society. Despite this global trend, research focus has been on load augmentation for soldiers/workers, assisting trauma patients, paraplegics, spinal cord injured persons and for rehabilitation purposes. Barring the military-focused activities, most of the work to date has focused on medical applications. However, there is a need to shift attention towards the growing needs of elderly people, that is, by realizing assistive exoskeletons that can help them to stay independent and maintain a good quality of life. Therefore, the present article covers the rapidly evolving area of wearable exoskeletons in a holistic manner, for both medical and non-medical applications, so that relevant current developments and future issues can be addressed; this includes how the physical assistance/rehabilitation/compensation can be provided to supplement capabilities in a natural manner. Regulatory guidelines, important for realizing new markets for these emerging technologies, are also explored in this work. For these, emerging international safety requirements are presented for non-medical and medical exoskeleton applications, so that the central requirement of close human–robot interactions can be adequately addressed for the intended tasks to be carried out. An example case study on developing and commercializing wearable exoskeletons to help support living activities of healthy elderly persons is presented to highlight the main issues in non-medical mobility exoskeletons. This also paves the way for the potential future trends to use exoskeletons as physical assistant robots, as covered by the recently published safety standard ISO 13482, to help elderly people perform their activities of daily living.
Keywords
Introduction
The term ‘wearable robotics’ came into general existence in the 1960s when research efforts started to focus on developing load augmentation and rehabilitation systems, 1 and interest continues to grow with new innovations reported regularly. Essentially, such exoskeletons (or ‘exos’) are wearable systems, which can help human wearers perform a variety of tasks such as pick-and-place heavy objects, carry heavy loads, reduce the burden in physically demanding tasks and apply rehabilitation treatment to patients who have suffered major trauma such as stokes and so on. Exoskeletons can also be used to assist in carrying out normal daily living tasks such as walking, carrying objects, ascending/descending stairs, perform sit-to-stand transfers (and vice versa) and moving around generally if the physical abilities of a person have degraded. To date, the primary thrust of exoskeleton research has focused on medical applications such as supporting mobility of spinal cord injured (SCI) persons and rehabilitation of major trauma patients as well as some military application to allow soldiers to carry heavy equipment while marching at high speeds in rough terrain. Medical exoskeletons are medical electrical equipment which is used to provide mobility to physically disabled, injured or weak persons, who are unable to walk due to a variety of medical reasons such as SCI, neurological disorders, major trauma like stroke, cerebral palsy and so on. Primarily, these types of exoskeletons are used in controlled environments such as hospitals and rehabilitation centres under the supervision of medical professionals. A recent review paper on lower-limb medical exoskeletons thoroughly discusses the issues related to performance, user interfaces and control strategies keeping in view the needs of patients and operators. 2
Recently, due to ageing societal concerns, new scenarios for providing assistance to elderly people for carrying out daily living activities have also started to receive attention, and there is growing urgency for such assistive technologies to help elderly people remain independent. As humans age, we start to have physical and cognitive issues and when these become severe, we are no longer able to do basic living activities and need care support to help us. An important issue in being independent is personal mobility capabilities. If we can continue to move around, we can stay active and thereby continue to live in our own homes with good quality of life. Physical assistive exoskeletons can help in such situations and, in addition, reduce the burden on healthcare resources. United Nation’s population statistics 3 (see Figure 1) show the ageing population is rising globally and the need for healthcare support will grow alarmingly to unsustainable levels. To reduce the burden on care services, many initiatives have been set up to promote research into developing assistive care technologies such as wearable exoskeletons to meet the perceived needs in the near future. This article presents a comprehensive review of the research conducted in both medical and non-medical exoskeleton technology along with pertinent design and safety regulations which are emerging to create the new markets in these technologies. Survey papers 4 –8 from the last 10 years suggest that non-medical assistive exoskeletons (referred to as physical assistant robots in the recently published ISO safety standard for personal care robot safety 9 ) are hardly explored, and hence attention is also given to them in the current article along with medical and other commercially available exoskeletons. Additionally, it is important for researchers and industry to design and develop wearable exoskeletons that fulfil the internationally published safety regulations. Therefore, the article presents key issues pertaining to regulatory and safety aspects which are emerging with reference to international ISO/IEC standards, so researchers and manufacturers can ensure that the needed solutions are correctly developed and commercialized.

Population of elderly persons, estimated for 2015 and projected for 2050. 3
Figure 1 shows that the proportion of elderly persons is rapidly increasing throughout the world. It can be noted that the projected elderly population in Asia and the percentage in Europe is growing at an alarming rate, and there is an urgent requirement to develop assistive and medical technologies to meet the needs of this significant segment of the society. The article presents the main issues for doing this and is divided into the following sections. The ‘Key issues of medical and non-medical exoskeletons’ section presents an introduction and the key differences between medical and non-medical wearable exoskeletons; the ‘Survey of current exoskeletons: Classification with respect to end-users’ section surveys the existing exoskeletons for medical (paraplegics, rehabilitation applications, amputees and for compensation of lost functions) and non-medical users such as soldiers, workers and healthy people especially elderly persons. The ‘Comparative analysis: 46 exoskeletons compared through eight parameters’ section presents a comparative table of existing exoskeletons for medical and non-medical purposes; the ‘Regulatory issues’ section introduces pertinent regulatory and safety aspects which must be complied with to realize commercially viable wearable exoskeletons; the ‘Example application: Assistive mobility exoskeletons for elderly persons’ section presents an example study for supporting elderly persons via wearable (non-medical) exoskeletons and the ‘Challenges and future trends’ section presents the potential future trends and potential for wearable exoskeletons. The article ends with the ‘Conclusions’ section presenting conclusions which can be drawn from the work.
Key issues of medical and non-medical exoskeletons
The main features and the key differences of medical and non-medical exoskeletons can be summarized as follows: In medical exoskeletons, the motion trajectories for individual joints cannot be provided by the wearer as the patient cannot make the required movements, whereas healthy persons can normally possess sufficient physical functionality which needs to be ‘topped up’ rather than having to have it ‘fully compensated for or replaced by’ as in medical situations such as providing mobility provision to SCI persons. This means many technical issues such as user interfaces, control strategies, mechanical interfaces and so on need to be designed specifically to cater for the individualistic needs of the patient/wearer. Often medical exoskeletons require specialist medical professionals to deploy and use them so that maximum clinical benefit can be given to the patient and harm prevented. For non-medical exoskeleton applications, the methods for measuring ‘user intention’ are most important so that the actuated mechanisms can be operated to support the user’s plans to make the desired motions in as natural a manner as possible. For maximum effectiveness and acceptability, natural interfaces are needed so that lay users can don, operate and doff the exoskeletons easily, quickly and efficiently. The control and stiffness interfaces need to be generic enough to have mass appeal but be sufficiently adaptive to meet the wide range of variations found in the movement patterns of individuals. The international safety regulatory requirements (published by ISO/IEC) for medical and non-medical exoskeletons are different and must be complied with for successful commercialization. For medical exoskeletons, such regulations are still underdeveloped by the joint working group IEC SC62D and ISO TC299 JWG36 (medical robots for rehabilitation), whereas for non-medical exoskeletons, ISO TC299 WG2 (personal care robot safety) has produced and published the safety requirements for physical assistant robots (restraint and restraint-free types). It is important to be aware of these standards and what is emerging in a regulatory sense, that is, is the wearable robot a medical electrical equipment for patients or a personal care exoskeleton for healthy persons, so that the correct risk assessment and risk reduction can be carried out for non-medical exoskeletons or appropriate risk management carried out for medical exoskeletons? This means identification and application of relevant medical device or machine safety standards. The physical functionality of elderly persons degrades with age, and hence a flexible and adaptable assistive technology needs to be developed so that the level of assistance can automatically adapt to meet the growing need at an individual level rather than having to routinely change the exoskeleton. An important issue to note here is that, although ageing is normal and does not need to be a medical issue, ageing normally involves a variety of medical conditions to arise and this needs to be considered in the design of the exoskeleton. If such medical conditions are relevant to the human motion aspects, then it is likely that the exoskeleton should be classified as medical electrical equipment; on the other hand, if the medical conditions are not related to the motion being supported, the wearable exoskeleton can be a non-medical physical assistant robot and consumer regulations will apply making the product mainstream and allowed to be sold in a variety of outlets including retail stores without the involvement of medical practitioners. This could be a key issue in helping to create new wearable robot markets. Researchers are starting to give attention to these requirements for wearable exoskeletons so that effective solutions can be realized and replace the limited number of assistive products currently available for supporting human motion such as the personal mobility of individuals where wheelchairs, crutches, sticks, mobility scooters and so on have been traditionally used. Simple aids like sticks and crutches are acceptable for low-level or short-term help but can be ineffective when high-level support is needed. Wheelchairs and mobility scooters can provide this for general mobility support in large open plan areas but require major changes to be made to adopt them in confined spaces such as homes. Wheelchairs often must be used as there are no other viable alternatives but people are forced to move to single storey apartments, install chair lifts and ramps, change fittings because they are too high or too low and so on. Such changes can impose significant financial costs on an individual even though the actual cost of the wheelchair or a mobility scooter can be quite low and attractive; in fact, the overall costs for effective adoption can be huge. Replacing these traditional wheel-based mobility solutions by body-fitting exoskeletons becomes attractive because minimal changes need to be made to homes and lifestyles for staying active and independent by allowing for the possibility to continue living in one’s home for as long as possible.
Having presented the overall issues for wearable exoskeletons, we start the detailed discussions by presenting a review of existing exoskeletons based on the main application scenarios so that the different requirements can be highlighted and how these should be explored in the future to meet the global emerging trends.
Survey of current exoskeletons: Classification with respect to end-users
For a comprehensive survey of the exoskeletons developed to date, we will classify them into two main categories, namely medical and non-medical systems, based on the rationale presented above. These are further subdivided according to the application and need as shown in Figure 2. The focus here will be on lower-body exoskeletons in the main and to highlight the mobility issue of the wearer, and only full-body or lower-body exoskeletons are considered. Upper-body exoskeletons, although important in their own right, do not contribute significantly to mobility and hence are omitted here for convenience. Going further into the literature review, it was found that these exoskeletons can be classified into a variety of different categories. However, there are few exceptions due to multifold applications of exoskeletons, which create a dilemma about the explicit category, in which each exoskeleton should be placed. Further, a comparison table is developed through which the various exoskeletons are compared on the basis of their purpose, degrees of freedom (DOF), actuator type, overall weight, battery life and so on. In this table, most of the data are taken from the published literature and patents. In some cases, the data are available on the Internet, videos or commercial websites, so the facts should be taken as suggestive. In the next subsection, each subcategory of Figure 2 is discussed.

Classification of exoskeletons with respect to end users.
Medical exoskeletons
Medical exoskeletons are designed to help the joint/limb motion of a patient in some specific manner where functionality is limited or lost in terms of mobility and strength. Such exoskeletons include ankle exos for drop foot applications or a hip and knee exos for rehabilitation purposes. As stated already, in this article, only lower-limb exoskeleton systems are considered to discuss the technology trends in how the mobility issues are catered for. Upper-limb exoskeletons are discussed in a comprehensive manner by Mann et al. 10 For lower-limb exoskeletons, important medical application subcategories are discussed next.
For paraplegics
Medical exoskeletons for paraplegics are used to assist patients suffering a type of paralysis as shown in Figure 3; paralysis is the inability in the sensory-motor functionality of the lower limbs preventing normal motions such as standing and walking. Several conventional methods have been used including braces and crutches, wheelchairs and orthotic devices. Braces and crutches fail to provide full motion autonomy to the person and hence have limited use. The mode of locomotion through conventional aids such as wheelchairs has its own pros and cons as already stated. Wheelchairs provide effective movement on flat surfaces but cannot be used in tight or unstructured terrains and result in excessive sitting in one posture. Moreover, wheelchairs do not permit eye-level interactions, which creates major social issues of ‘verticality’. Other locomotion options use different orthotic systems like functional electrical stimulation (FES), knee–ankle–foot orthoses (KAFOs) and reciprocal gait orthoses (RGOs). It has been reported in the literature 12 that FES systems were successful for short distance ambulation only but can result in high energy consumption and muscle fatigue. Compared to conventional KAFOs, which are heavy and difficult to wear, the RGOs have been found to offer better ambulation over short distances. 12

Types of paralysis, where the coloured portion is paralysed. (a) Quadriplegia, (b) hemiplegia and (c) paraplegia. 11
As presented in Table 1, there are several medical electrical equipment exoskeletons developed for upright walking of paralysed people. A good example is the ReWalk exo, 14 which has been developed for SCI patients. As a patient-worn backpack device, ReWalk is a self-contained exoskeleton which uses rechargeable batteries to drive the hip and knee joint motors. It uses a tilt sensor to compute the trunk angle and a wristwatch-style controller to activate different motion modes such as stand–sit, sit–stand or walking. ReWalk has been designed to be used by persons weighing less than 100 kg, height in the range of 157–193 cm and having the adequate upper-body strength to use the medical exoskeleton. These constraints can limit its range of end users. Another example for SCI applications is the Indego exoskeleton developed and commercialized by Vanderbilt University. 15 It offers a modular mechanical design which consists of three parts, namely a hip brace, two thigh frames and two shank frames, which can be assembled quickly for putting the exoskeleton on. It has been developed to be used with a conventional set of ankle–foot orthoses and is claimed to be easy to put on (don), take off (doff) and to be adjusted for good fitting single handedly. It has six modes for sitting, standing and walking and the transitions in between, and it costs around $75,000. Other exoskeletons like Ekso, 13 Rex, 16 Atlante 17 and Mindwalker 20 have also been developed for SCI and paraplegic patients. Exoskeletons like ReWalk, Ekso and Rex have been approved by medical regulators such as United States Food and Drug Administration thereby leading to commercialization. This is commercialized as high-value medical products, but the evidence of their real usefulness is still in doubt as there are still a variety of technical and social issues which need to be addressed for making them widely accessible and useful to society. Some of the technical issues include slowness in response by the exoskeletons’ dynamic performances as compared to humans, the weight constraints of the structure, the severe requirement for heavy battery packs when used outdoors, comfort issues for wearability (both short- and long-term) and complexities in sensing and actuation components. The social issues include hesitation towards the use of such assistive technology in everyday life, poor ergonomics of the exoskeletons, the high capital costs and the poor addressing of issues related to the complexities arising in the human-exoskeleton interactions.
Comparison of lower-limb exoskeletons developed throughout the world.

ZMP: zero moment point.
Rehabilitation applications
Rehabilitation is needed for persons with gait disorders, which can result from a variety of medical conditions such as lesions in the central nervous system, cerebrovascular accidents, cerebral palsy and so on. These gait disorders force the persons to depend on wheelchairs for mobility as the only viable way of performing stable travelling movements to carry out normal daily life activities. In contrast, Copilusi et al. 56 –58 presented the design and simulation of a lower-body exoskeleton to fulfil the main human locomotion tasks. Subsequently, lower-limb rehabilitation exoskeletons have been presented as a possible solution 59 for these people as the medical exoskeletons can provide therapy and retrain the patient to walk again. Medical gait training exoskeletons also come into this category and can be treadmill based and stationary in nature. Treadmill-type gait training exoskeletons do not fully come into the domain of wearable exoskeletons for moving around to support daily activities, but they apply forces to individual human joints in the same way to retrain a patient to relearn a lost motion function. Chen et al. 60 comprehensively explained the use of gait training devices and lower-limb exoskeletons for such rehabilitation applications with/without body weight support (BWS) systems, where six types of exoskeletons as shown in Figure 4 have been presented.
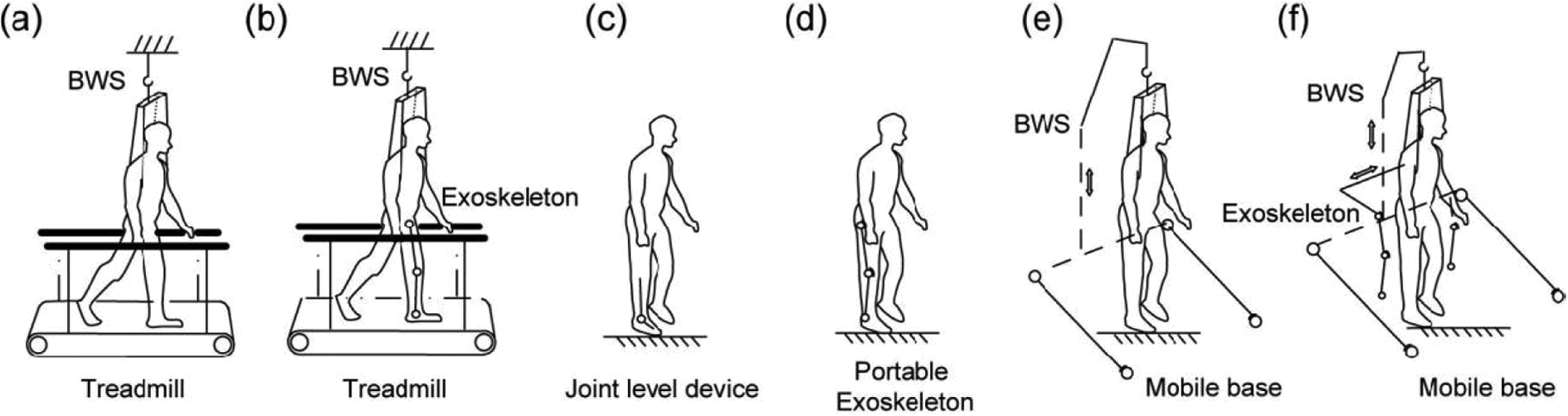
(a) Treadmill-based BWS system, (b) treadmill-based exoskeleton, (c) joint level device, (d) portable exoskeleton, (e) mobile robotic trainer, (f) exoskeleton with mobile platform. 60 BWS: body weight support.
An exoskeleton named ‘Lokomat’ developed by Hocoma (Switzerland) is a treadmill-based BWS device which includes audio-visual biofeedback, using a screen in front of the user; it is powered at the hip and the knees. Other rehabilitation exoskeletons like Lokomat, 61 Lokohelp 62 and so on fall into this category and are briefly discussed in Table 1. Such treadmill-based exoskeletons need two essential improvements, namely improving their effectiveness in treating patients and reducing the overall capital cost, which is high thus limiting widespread clinical use currently.
For amputees
Amputees are persons who have parts of one or more limbs removed due to causes such as accident, trauma or some medical condition. For restoring the mobility of amputees, various devices are used, for example, walkers, braces, orthotic devices, artificial limbs and so on. These medical devices attempt to restore the mobility of the person as best as possible but are normally limited in their functionality. For improved restoration of personal mobility in such cases, better fitting and functioning robotic prosthetics and orthotics need to be used which not only support the person in providing the missing limb but also how it should be actuated and controlled during various human motions. Such robotic prosthetics fall into the category of medical wearable devices and hence exoskeletons. 63 Research in this area is gaining pace as society is accepting the technology in preference over conventional methods wherever possible. Specific products are manufactured according to the needs of the user and type of amputation. Some examples are shown in Figure 5, for example, for a trans-femoral amputee (above the knee), the device comprises a knee prosthetic, a socket, an ankle–foot prosthetic and a link between them. Ottobock 65 is a world leader in such robotic prostheses and orthoses and its ‘C-Leg’ product is popular with trans-femoral amputees; the device weighs around 1.2 kg and is made of the carbon frame. It has an on-board microprocessor for mapping the actuator feedback and is claimed to be quite comfortable compared to other similar products. Other notable devices include the prosthetic foot made by Ossur 66 and so on.

Types of amputation: (a) amputation at the hip and pelvis region, (b) transfemoral amputation, (c) amputation at the knee (knee disarticulation), (d) transtibial amputation and (e) amputation at the foot. 64
Hydraulic actuators have been used to power knee prostheses which gave high resistive torques and it is claimed that users feel more security and stability in the various motion stance phases during walking. 63 However, with the rising introduction of more efficient electronics, such hydraulic components are being phased out and the term ‘intelligent prostheses’ has been growing where some form of ‘smart’ strategy is used to control energy generation and application at the supported joint. Intelligent prostheses and orthosis have changed the outlook of the industry, but some pending issues remain; these include weight and size incompatibility and overall efficiency of the electromyographic sensors used. The next era is expected to comprise ‘cybernetic devices’ which will incorporate more invasive and integrated robotics technology into human bodies to assist the amputees. Bio-inspired robotics is also part of this trend. The control strategies’ reproduction of gait patterns utilizes solutions from nature and advances made in science and engineering. In the future, it is envisaged that such cybernetic systems will be common and widely used by amputees to regain their mobility and be able to move around like healthy persons without even getting noticed.
Compensation of lost functions
This category includes multipurpose power-assist exoskeletons to replace or restore lost functions and also falls under the medical sector. Examples are wide and varied and can include the ‘nurse power assist’ 30 designed for medical staff to help lift patients in hospitals and care institutions thereby reducing the likelihood of injury or fatigue. In this device, pneumatic rotary actuators are used and installed on the back of the nurse (wearer) to take the weight of the patient. Another device, namely the Pheonix, 32 manufactured by SuitX (Berkeley, CA, USA) for people with mobility disorders also falls under this category. This adjustable device is commercially available; for actuation, it uses DC motors and has a battery life of 4 h. Other exoskeletons like the Honda-leg (Honda Inc., Japan), Keeogo (B-TEMIA Inc., Canada) and so on are aimed also to compensate for some lost function in patients. It should be noted that such compensation exoskeletons face major challenges of weight and cost reduction to make them popular and be widely used. Further, there are issues in developing flexible human-like joints and efficient control to accurately replicate the lost functions.
Non-medical exoskeletons
Non-medical exoskeletons are physical assistant robots falling under the category of personal care robots which are designed for various applications to assist healthy persons in carrying out some activity; examples include strength augmentation for workers to perform physically demanding tasks, for soldiers to carry heavy equipment and march at fast speed over rough terrain and for supplementation scenarios where healthy people need help to carry out normal daily chores. The latter includes applications to support the elderly in activities of daily living to allow them to stay active and independent with a good quality of life. Such general-purpose exoskeletons have also been developed recently which are used for multiple applications, such as exercising, sports and recreation, and also for space scientists. Non-medical exoskeletons can be further divided into four categories according to the need of the users and these are discussed next.
For soldiers
Soldiers are fit/strong young persons who may be required to perform combat missions when they reach their destination. To assist in such cases, military exoskeletons have been developed to amplify the capabilities of the soldier so that he/she feels only 5%–10% of the actual load carried so that long distances can be travelled with reduced metabolic costs. Berkley Lower Extremity Exoskeleton 36 is one prominent system developed at the University of California–Berkeley under a DARPA-funded project to develop a versatile transport platform for mission-critical equipment. It comprises a portable power unit and uses linear hydraulic actuators to allow soldiers to carry around 100 kg without fatigue. Other similar military exoskeletons include Sarcos XOS2 (Raytheon, USA), 37 HULC (Berkeley Robotics and Human Engineering Laboratory, CA, USA), 41 Exo-climber (Berkeley Robotics and Human Engineering Laboratory, CA, USA) 39 and so on which have been designed to augment capabilities of soldiers in wartime and in emergency operations. The main challenge in developing load augmentation exoskeletons for soldiers is how to reduce the overall weight yet maintain high levels of augmentation for long durations so that soldiers will stay fresh for combat. These devices are likely to be practically useful only if they reduce the metabolic cost significantly while augmenting the load carrying capacity of soldiers. However, the high torque actuators require more power and bigger batteries for the long operational times; it is clear longer-lasting lightweight batteries are needed to realize solutions able to contribute in this sector. Preventing misalignment of the human joints during actuation over the long wear times is another open research challenge for the future. Figure 6(a) and (b) shows soldiers using exoskeletons during various operations in the field.

For workers
Workers in industrial environments often have to perform physically demanding tasks which can lead to stress and adverse effects on health. Tasks can include lifting heavy parts for fitting on an assembly line or handling heavy equipment in pick and place operations. Exoskeletons have been designed to help workers in performing specialized motions by defining reachable spaces for the tasks to be carried out as shown in Figure 6(c) and (d). In this way, the worker is supported specifically for 3D joint movements needed to be carried out so that health is not compromised due to the nature of the physically demanding human motions in the work. In this respect, passive exoskeletons have also been realized, for example, ROBOMATE. 83 Certainly, one can think why other types of robotized/ automation solutions are not used if the nature of work is not within human limits. However, it is essential that human workers should not be replaced totally, and well-balanced approaches for developing collaborative human–robot solutions should be sought, by optimizing the overall effectiveness of the final design. In this regard, exoskeletons for physically supporting human workers are an important research topic for the future.
For healthy persons (including elderly person applications)
This non-medical area of exoskeletons covers a wide area of users but one unique sector likely to grow in importance in the coming years is the support that wearable robots can provide to elderly persons. Healthy elderly persons have no serious disability or disease as such, which demands the intervention of medical personnel (except that they are old and their physical ability could be reduced). Perhaps they are not as mobile as when they were younger or perhaps they get tired more quickly by doing activities which require significant physical effort. This sector is often referred to as active and assisted living (AAL) applications in Europe, 71 or the silver/grey society in Japan and so on. Despite the global concerns, only a few multipurpose exoskeletons have been developed which can be used by elderly persons and no commercial exoskeleton is available to date. PhaseX, a spin-out company created by University of Gävle in Sweden, aims to focus on commercializing assistive exoskeleton technology developed within the AAL Call 4 project EXO-LEGS. For this, simplified lower-body mobility exoskeletons able to support some key human motions was developed, which is discussed in detail in ‘Example Application’ section. Such wearable exoskeletons have the potential to meet the mobility requirements of the elderly for performing daily living activities. The intention must be to develop user-centric solutions, so affordable mass-market consumer products are realized rather than focusing on high-value low-volume exoskeletons.
For general purpose
General-purpose exoskeletons include all those wearable devices which are designed with no specific target application. These are multi-application devices which can cater to different types of users, for example, for exercise, sports, recreation, astronauts and so on. Due to the general nature, these exoskeletons are heavy and large in size as they do not target any particular user. NASA’s X1 54 is an example of such a device which was developed by NASA and IHMC with many application areas including assisting motions of astronauts in space/extraterrestrial surfaces, walking of paraplegics, for exercising and other applications and so on. General-purpose exoskeletons are useful devices but cannot target mass markets, as they lack a dedicated design approach to optimize the overall solution. Moreover, exclusive devices for specific application areas are becoming available as already explained.
Comparative analysis: 46 exoskeletons compared through eight parameters
Having presented an approach to classify wearable exoskeletons into medical and non-medical applications, we are able to compare the exoskeletons which have been produced by researchers and companies. Table 1 presents a comparison as well as provides insight into the key features of the main wearable exoskeletons realized that includes mechanics (joint mechanisms, number of DOF, material, etc.), types of actuators used, control strategy, sensors deployed, battery life and so on. It is important to note that Table 1 intends to render comparative details of medical and non-medical exoskeletons in order to provide the readers a comprehensive idea of recent trends in the field.
Regulatory issues
In emerging domains such as wearable exoskeletons, it is important to not just focus on developing technology and prototypes for testing in research labs, but have a clear vision for how the advances can make real impacts in society. In this regard, how wearable exoskeleton markets can be created should be focused upon, so stakeholders are aware of the issues needing attention. In this section, an overview is given to international regulations and where the new wearable exoskeletons discussed in this article fit. We start with a brief introduction to international technical regulation.
Governments have the responsibility to ensure that products sold in their territories are acceptable and meet the market requirements. For this purpose, governments adopt appropriate laws to ensure that the objectives are met and product safety is the primary goal so that harm cannot be caused to citizens by unsafe products being sold. When commercialization involves technical products, it is important to have in place a technical regulatory framework, where an appropriate regional authority makes the legislation that can apply to all levels of government (national, provincial, state, municipal, etc.) and covers product services and actions so that provisions (mandatory or not) can be put into place over and above the free market system.
Figure 7 shows a typical technical regulatory framework so that technology is adopted appropriately in products, and the safety of its citizens guaranteed. The key elements within such a technical regulatory system are the following:
Regulator: Must be established by law and must be a legal persona ‘juristic person’ in the country. The powers of the regulator must be made known (as well as the limitations), and these must not have conflicts of interest with standards or conformity assessment bodies
Standards/regulations: Standards are voluntary, but can be referred to or incorporated in regulations (which are the mandatory laws/rules in a country). There are a variety of standards resulting from international, regional, national and even private initiatives and activities. They are produced and adapted for specific purposes, but international standards produced by International Organization for Standardization (ISO; www.iso.org) and International Electro-technical Commission (IEC; www.iec.ch) have the strongest presence as they consist of international networks of national standard bodies which represent the majority of stakeholders in each country, from all regions of the world, working in partnership with international organizations such as the United Nations, its specialized agencies and the World Trade Organization (WTO). The fundamental basis of developing international standards is that they are globally acceptable, is that they must involve a public inquiry phase, allowing all stakeholders to be involved, and they must be based on consensus. Once such international standards (as well as other relevant information) are available, it is the regulators who set the legal regulations based on information gathered by a public enquiry, and these regulations are backed by the government.
Compliance: Conformity assessment to the regulations should uphold international principles and use accredited service providers. The use of mutual recognition agreements is recommended, as are third-party independent organizations which respect WTO rules in a transparent manner.
Sanctions: For the regulatory system to work well, the regulator’s powers are normally well defined in detail in terms of entry of premises, samples to be tested, what can be confiscated and disposed off, what fines can be imposed, suspension and prohibitions of trading and so on.

A typical model of a technical regulatory system.
Hence, the role of robot standardization is to support such technical regulation by ensuring robot products are safe, reliable and of sufficient quality to work as intended, through clear and unambiguous provisions agreed by all the relevant stakeholders to facilitate trade and communication. By having international standards, it is possible for the companies to sell their products anywhere they want because they contain all the information needed about differences between individual countries they want to export to. By using an international standard, a company loses less time (and uses fewer resources) and can produce products that operate in a predictable way anywhere in the world. For example, in Europe there are several EC Directives adopted as law and two such directives relevant to the exoskeletons being discussed here are the following:
Directive 2006/42/EC: machinery directive.
72
This is written to promote the design of machinery that is as safe as possible according to the latest technological developments. This directive applies to machines generally defined as devices with at least one moving part, containing actuators, control and power circuits. Exceptions to the machinery directive exist and are normally covered by other regulations, as well as machines in which the main risk is of electrical origin (in which case only the low-voltage directive would apply). Most robots to date have been classified as machines and hence the robot safety standards need to comply with this directive.
Directive 2001/104/EC: medical device directive.
73
The key issues in the definition of a medical device are the following: any instrument, apparatus, appliance, software, material or other article, whether used alone or in combination, together with any accessories, including the software intended by its manufacturer to be used specifically for diagnostic and/or therapeutic purposes and necessary for its proper application, intended by the manufacturer to be used for human beings for the purpose of (1) diagnosis, prevention, monitoring, treatment or alleviation of disease, (2) diagnosis, monitoring, treatment, alleviation of or compensation for an injury or handicap, (3) investigation, replacement or modification of the anatomy or of a physiological process, and which does not achieve its principal intended action in or on the human body by pharmacological, immunological or metabolic means, but which may be assisted in its function by such means. These issues are important and hence any robot designed to meet a medical need must be regulated as a medical device (under this medical directive) rather than as a machine (under the machine directive).
When focusing on the ageing society problem, exoskeletons complying with both machinery and medical device regulations are needed as discussed in this article. The former exoskeletons are likely to be mostly needed for healthy elderly persons (customers) who require support to continue living in their own homes for as long as possible, whereas the medical exoskeletons are for patients with specific medical issues and where medical consultation is needed and the appropriate medical aids need to be prescribed by medical professionals.
Before presenting summary details of the machinery and medical issues for robots, it is worth noting that, prior to 2005, the main robot standardization work was carried out by ISO for only industrial robots complying with safety requirements for machines. These industrial robots were (and still are) powerful and dangerous machines and hence the requirements (published in ISO 10218-1, -2 74 ) relied upon keeping humans and robots separated by the use of real or virtual work cells with physical barriers to automatically apply safety interlocks to turn power off to the robot if a human enters the robot’s operational workspace or to control robot speed and power. With the emergence of service robots (which includes exoskeleton robots), it was not possible to develop the needed robots since they demand close human–robot interaction for providing the ‘service’, and new safety requirements have been published for personal care robots in ISO 13482. The service robot safety standardization efforts have also initiated consideration of robots as medical devices (known formally as medical electrical equipment) and requirements for basic safety and essential performance are being produced by a joint working group under IEC SC62D and ISO TC299. Brief summaries of the standardization work in the machinery and medical application sectors are presented in the next subsections as knowledge of this is essential to appreciate what is needed for wearable exoskeletons to be commercialized in these sectors.
Standardization of assistive exoskeletons as machines
It is important to have an appreciation of traditional robot safety from its industrial manufacturing roots, where robots are normally seen as powerful machines and the design has been based on keeping the industrial robots and humans separated by real or virtual cages to prevent harm to the humans as already stated. The industrial robot safety requirements are published in ISO 10218-1, 2. Since the late 1990s, as service robots have evolved, it has become clear that this ‘separation’ philosophy cannot deliver the needed new robots, and close human–robot collaboration is essential for service robots. And of course, for exoskeletons, the wearable robots are designed to be physically attached to a human during normal intended use. For this to be possible, new international robot safety requirements were needed, and the important work to identify the new safety requirements and design guidelines was started in 2006 for personal care robots and has recently resulted in the publication of ISO 13482 in 2014. Personal care robots typically perform tasks to improve the quality of life of intended users, irrespective of age or capability.
In the course of developing ISO 13482, a number of key steps have been taken by the international robot standardization community. Some of these are the following: The definition of the term ‘robot’ has been updated to programmed actuated mechanism with a degree of autonomy, moving within its environment and to perform intended tasks. A new term ‘autonomy’ has been defined as the ability to perform the intended tasks based on current state and sensing, without human intervention. New types of robots have been defined as follows: ▪ Service robot: Robot that performs useful tasks for humans or equipment excluding industrial automation applications. ▪ Personal care robot: Service robot that performs actions contributing directly towards improvement in the quality of life of humans, excluding medical applications. ▪ Physical assistant robot: Personal care robot that physically assists a user to perform required tasks by providing supplementation or augmentation of personal capabilities. ▪ Medical robot: Robot intended to be used as medical electrical equipment or as a medical electrical system.
Hence, wearable exoskeletons as discussed here are physical assistant robots as defined in the new terminology, and ISO 13482 has defined two types of physical assistant robots, which are as follows:
Restraint-type physical assistant robot: Physical assistant robot that is fastened to a human during use.
Restraint-free-type physical assistant robot: Physical assistant robot that is not fastened to a human during use.
The restraint-type assistant robot allows free holding/releasing of the robot by the human in order to control or stop the physical assistance. ISO 13482 presents the needed safety requirements using the well-accepted risk assessment and risk reduction principles presented in the type A standard (ISO 12100
75
) via the three-step method, namely: Try to achieve the safety requirements by means of inherently safe design measures. If inherently safe designs are not possible, then try to achieve the requirements by means of safeguarding or complementary protective measures. If neither of these solutions is possible, then provide information for use to the operator (warnings and instructions) to assist the operator in achieving acceptable safety.
These steps ensure the risk is reduced to an acceptable level and, if this is not possible, the machine (exoskeleton) should not be used. The generic methodology has been used to identify hazards which can arise for personal care robot scenarios under possible single fault conditions. The list of hazards presented is extensive and the most relevant hazards for wearable exoskeletons are likely to be associated with the following: (1) uncontrolled release of stored energy; (2) power failure or shutdown; (3) hazards due to shape of exoskeleton; (4) hazardous vibrations; (5) hazards due to stress, posture and usage; (6) hazards due to exoskeleton motion; (7) mechanical instability generally and while carrying loads; (8) instability in case of collision; (9) instability while donning/doffing exoskeleton and (10) hazardous physical contacts (with exoskeleton and moving parts). For each hazard identified, steps that shall be adopted are presented on the three-step method and ways to provide acceptable protective measures provided.
In addition, ISO 13482 has introduced low and general risk types of personal care robots. For exoskeletons, the low-risk type is defined as one ‘where low-powered physical assistance is provided and the user can overpower the exoskeleton if needed in a single fault condition’. In this way, design issues can be simplified, yet the mandatory safety requirements maintained by the wearer being able to resist hazardous situations likely to arise under fault situations. Work is continuing within ISO TC299/ WG2 (personal care robot safety) to develop two complementary technical reports to support the new safety standard ISO 13482. These work projects are ISO TR 23482-1 safety-related test methods for ISO 13482. The technical report provides recommended test methods to verify compliance with the safety criteria of personal care robots. The tests describe the principle of each test, the apparatus that may be used, the test procedure that should be followed and the key data that should be presented in the test report. Important tests included for wearable robots assess physical hazards characteristics posed (interactive forces and displacement/velocity generated at contact surfaces between exoskeleton and human skin, endurance characteristics, safety-related control functions, etc.). Publication is expected in 2019. ISO TR 23482-2 application guide to ISO 13482. The technical report is being produced for robot manufacturers with limited experience of risk assessment and risk reduction to facilitate personal care robots to be designed and assessed for safety compliance with the new ISO 13482 standard. Furthermore, clarification is provided on terms and the new safety requirements introduced for allowing close human–robot interaction and human–robot contact. Publication is expected in 2019.
In commercialization and opening up novel new robot markets, the ISO 13482 safety standard and the two technical reports could be valuable so that experience can be gained within the holistic robotics community and priority areas identified for focusing further research advances, for example, developing better sensors and actuators and more ergonomic controllers.
Standardization of exoskeletons as medical electrical equipment
When exoskeletons are discussed, especially for elderly persons, it is clear that medical applications (including aids for the disabled) need to be addressed because ageing often involves medical complications and hence medical consultation is needed. The standardization work has been started jointly by ISO and IEC, and the work is continuing with the intention to publish IEC 80601-2-78 (medical electrical equipment – Part 2-78: particular requirements for basic safety and essential performance of medical robots for rehabilitation, assessment, compensation or alleviation) in late 2018. Cleary, some exoskeleton robots will be classified and regulated as medical devices rather than as machines, and this poses an interesting boundary question being pondered by the international community because it will lead to different regulatory requirements. The issue is how the boundary between medical and non-medical wearable exoskeletons can be defined. For example, exoskeletons can be used for rehabilitation of injured persons (which is clearly a medical application) as well as physically helping a healthy person to carry a heavy load (clearly a non-medical application), but there are applications that are not so clear, namely an assistive exoskeleton for helping the mobility of healthy elderly persons. In non-medical applications, we refer to the user as a ‘normal person’ (consumer), whereas in medical applications, the term ‘patient’ is defined as ‘living being (person or animal) undergoing a medical, surgical or dental procedure’ and used to carry out the risk management process. Further discussions are still needed to reach consensus on how the boundary will be defined and when a ‘consumer’ for non-medical exoskeletons becomes a ‘patient’ for medical exoskeletons.
The key regulatory process to follow for designing medical devices is the application of risk management presented in ISO 14971. 76 Here it is not that risk should be reduced to ‘an acceptable level’ under a single fault condition but it is important to focus on managing the risk by performing a risk–benefit analysis for the patient. This is because it is sometimes necessary to cause ‘harm’ to a patient in the short term to treat them, for example, for medical robots to perform surgical cuts to carry out the needed surgery. The requirements for a medical electrical equipment cover basic safety and essential performance. Hence, medical equipment needs to perform the risk–benefit analysis for the patient to ensure basic safety which is along the lines of the machine regulations as well as ensures the essential clinical functionality is maintained under single fault conditions. This needs to be stated by the manufacturer when the risk management file is prepared. The particular standard IEC 80601-2-78 being developed is formulating the requirements for medical wearable exoskeletons related to the patient’s movement functions following any impairment.
Example application: Assistive mobility exoskeletons for elderly persons
The article has covered holistic issues in developing and commercializing lower-limb exoskeletons and how the sector is evolving. In this section, we go into specific details which should be addressed by focusing on developing assistive exoskeletons for AAL applications. The work described here was carried under the AAL Call 4 project EXO-LEGS aimed at developing mobility exoskeletons for elderly persons (www.exo-legs.org, AAL project 2012-03255). The project brought together end users, companies and research organizations at the EU level to specify the indoor and outdoor mobility needs of elderly persons to enable them to continue living independently with good quality of life in their own homes for as long as possible. The aim was to ensure a variety of exoskeleton prototypes could be designed, developed, tested and validated in research labs before being validated by elderly users in Sweden, Germany, Spain, Switzerland and the United Kingdom. In order that such a project has its work properly grounded to the needs of actual end users, the project work plan needs to be centred around end users.
As part of the EXO-LEGS project an end user’s group was created to comprise the following:
Primary AAL end users: Elderly persons who will use AAL wearable exoskeletons.
Secondary AAL end users: Friends and relatives of primary end users or AAL care organizations.
Tertiary AA end users: Organizations contributing in organizing and supporting the AAL sector.
In total of 118 end users participated in the research with ethical approval from Swedish Ethical Board in Uppsala. The end users were engaged with via specially designed questionnaires to seek their views on various aspects of the work to be carried out within the project and the findings were in fact implemented in the EXO-LEGS project. We provide summary reports on the key areas. Further information can be found on AAL website 71 and a previous paper by one of the co-authors. 77 .
Specification of mobility functionalities for AAL exoskeletons
The main mobility tasks needed to be carried out for normal daily living were presented to end users for ranking in order of priority where they felt that assistance via an exoskeleton would be needed. The ranked list of mobility functionalities produced is as follows: stable standing on open space; sit-to-stand transfers and vice versa; straight walking on flat ground; walking and turning on flat ground; bending down (to pick item from floor); crouching with support (pushing rollator); walking up/downstairs; stepping over objects; crouching without support (e.g. to avoid low ceiling); walking on ramps; speed walking on flat ground; walking on uneven ground; walking on slippery ground; exercising; using leg to open/close door.
This ranked list was used to develop different category exoskeletons (basic, standard and deluxe) which could be designed to meet different markets in a modular way to allow easy upgrading to enhance functionality. Using a modular approach the mobility functionalities 1, 2, 3 and 6 are felt to be essential and hence included in the simplest exoskeleton (basic model); functionalities 4, 5, 7, 8 and 9 are felt to be more challenging technically and hence included in the next higher grade of exoskeleton (standard model) and functionalities 10–14 are included in the most complex exoskeleton needed (deluxe model) as the motion tasks are both very challenging and also not essential for most elderly persons in their daily living activities. The EXO-LEGS project focused on designing, building and testing the basic exoskeleton since this included both the simplest technologies and the most urgently needed mobility functionalities allowing research effort to be usefully targeted. The realized basic EXO-LEGS exoskeleton is number 39 in Table 1. In addition, the EXO-LEGS basic model has been designed to provide 30% support in the sagittal plane to the wearer at the hip, knee and ankle joints in both legs making it a low-power exoskeleton as defined within the new safety standard for personal care robots (ISO 13482). Its regulatory requirements are therefore minimal as it can be overcome by the user and it cannot cause harm to the wearer under single fault conditions when risk assessment is carried out for it.
Guidance on the important design criteria
The EXO-LEGS end users were asked their views on identifying general design concepts that were preferred. These included how the exoskeleton should be attached to the body, how it should look and be adjusted to fit the human for good comfort, how it should be worn and battery life. The top-ranked features were identified to be as follows: good, useful functionality; easy/natural to use; comfortable, good fit; easy to put on/take off (don/doff); wear without help; lightweight.
How the assistive exoskeletons should be made available to potential end users
Another vital survey carried out with the EXO-LEGS end users was their views on how the EXO-LEGS technologies should be commercialized and what prices can be accepted by different markets. The main findings are as follows:
Nearly 100% believe there should be exoskeleton centres set up where users can go to test them 95.2% believe healthcare bodies should add assistive exoskeletons to approved lists; 96.8% are prepared to contribute personally to acquiring an assistive exoskeleton if insurance schemes are available; 95.2% think renting/leasing options should exist for accessing exoskeletons; 93.7% prefer to have certified safe exoskeletons even if they are more expensive; 96.8% prefer to use an exoskeleton over other mobility aids; 84.1% like the idea that exoskeletons should be made available in care homes; 85.7% agree that a price range of €2000–3000 would be widely acceptable;
Following the information provided by the end users, detailed technical work was carried out to study various human motion tasks and component design and testing to allow a basic EXO-LEGS exoskeleton to be designed and built. A CAD drawing and an image of the realized basic exoskeleton are shown in Figure 8.
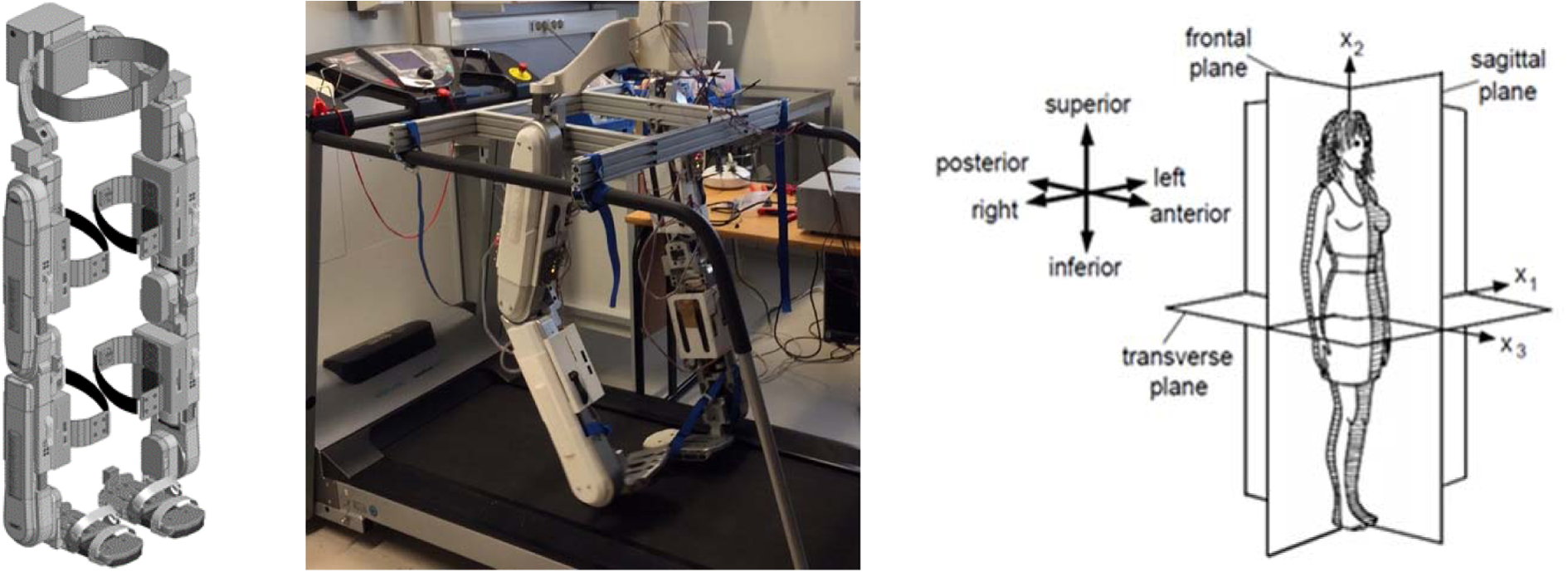
Basic EXO-LEGS model exoskeleton designed for sagittal plane support of key daily living mobility tasks.
The basic exoskeleton was tested by primary end users in Sweden after ethical approval obtained for the following tests: walking on treadmill; sit-to-stand transfers.
Figure 9 shows some images of the end user testing carried out in Sweden.

End user testing of the basic exoskeleton for straight walking and sit-to-stand transfers.
Overall the results of the testing were that the design of the basic EXO-LEGS model was acceptable to the end users and they could feel the assistance being provided in the two motion tests and such assistive exoskeletons are seen as commercially viable products in the foreseeable future for elderly persons to maintain an independent life. However, improvements are felt to be needed in terms of reducing weight, improving comfort and making it much easier for putting on and taking off. These are generally understood as the main goals for developing wearable exoskeletons to assist elderly persons to perform daily living tasks.
In another study, 78 the gait analysis tests on eight different users in age band 19–27 years, 3D accelerations of upper and lower body were measured for different walking speeds and the data were collected and transferred using wireless and Bluetooth network, respectively. Motion characterization normally includes stiffness, bending and torsional information, through location, directions and speeds of accelerometers and gyroscopes. Another work 79 proposed an effective way of measuring the dynamic behaviour of human torso using inertial and magnetic sensing tools. It has been discussed that incorporation of a correct amount of stiffness in the humanoid joints and links has utmost importance in the design of robots and exoskeletons. The work 56 presented useful analytical and experimental techniques to estimate correct amount of stiffness. A compact unifying procedure is proposed for the calculation of numerical and experimental stiffness matrices of the multibody robotic system. 56 In all, motion parameters are analysed for better repeatability, accuracy and payload capacity of exoskeletons.
Challenges and future trends
From the last two decades, the research into wearable exoskeletons has risen steeply; this can be estimated from the number of exoskeletons reported in the survey section of this article and the number of publications emerging in the scientific literature. The prominent reason of acceptance of this technology in modern society is that lifestyles are fast and people want to remain active and independent with good quality of life; this independence may relate to being a patient, a worker, a soldier, a nurse, an elderly person and so on. This leads researchers to focus effort on designing and developing wearable exoskeletons which not only give mobility or strength to the wearer but also makes them more independent by allowing them to continue their desired activities. Exoskeleton technology is widely seen as a key enabling technology for providing individualized personal mobility solutions to millions of people immobilized by injury, trauma, disabilities or ageing. However, the technology is in its early phase and there are still many factors that need to be addressed to achieve ‘fully effective exoskeletons at an affordable cost to the masses’. The EXO-LEGS case study presented in section ‘Example application: Assistive mobility exoskeletons for elderly persons’ clearly makes this conclusion.
Challenges
The following address the key challenges where advances are needed: The key component is the actuator which gives the ability of motion. Many types of actuators are used in exoskeletons depending on the application, for example, heavy hydraulic ones used in military applications for providing large levels of augmentation whereas light/compact actuators are preferred in medical applications, so patients can carry small loads. Compact actuators are costly and cheap ones are bulky and less energy efficient. Better actuators which are lighter, more compact, safer, and more affordable are the main open research issues. Whether the current actuators provide sufficient core details to allow focusing on what is needed for supporting the motion of human joints is also worth exploring. For example, current electrical motors tend to be focused on providing very tight positioning control capabilities whereas for supporting human motion applications, such level of precision is not the most important issue. The performance of actuators depends on the control strategy adopted to provide the required power at individual joints for the specific motion being performed. Various control strategies have their own pros and cons. For example, EMG control is not totally reliable due to errors in sensing the muscle movement and mismatch with wearer’s intentions. Fixed trajectory control methods face problems of synchronization and they can disturb natural gait cycles so that the human motions appear to be robot-like. Fully efficient and robust control strategies for specific human motions for mobility functionalities are still missing. The area must be explored and a range of sensory interfaces deploying multiple sensors and diversity explored to realize the advances needed. Long-term power supplies to provide the needed energy for the actuators is a major issue in making the exoskeleton fully autonomous for long period wear to be possible. As electric actuators are widely used, batteries used for powering them need to be improved. Most batteries have the problem of weight, limited lifespans, a limited number of charge/discharge cycles and so on. This poses challenges for researchers to develop lighter, denser and longer-lasting battery technologies. Safety is the major concern of the governmental and regulatory organizations before allowing new products to be commercially available to the public. In this regard, only a few exoskeletons like the ones from ReWalk, Ekso Bionics and Cyberdyne have been certified to comply with the newly emerging international safety requirements in medical and non-medical applications. This is a major concern as it affects the widespread commercialization and restricts the opening up of new exoskeletons markets globally. Consumers and patients need to be able to have confidence in the new technologies to ensure global impacts and compliance with international safety standards and use of widely recognized certification processes is essential for this. Other factors like more lightweight materials with the needed strength for making the exoskeleton structures, better sensors to detect a variety of human motion intention and further miniaturization and integration of the electronics are needed to reduce the overall size and weight.
Future trends
Future trends of lower-limb exoskeletons will primarily depend on consumer responses to how the needed mass markets will emerge; these are likely to depend on factors like affordability, effectiveness, comfort and safety certification. Some key issues in this regard are summarized next to highlight the urgently needed future trends in developing the area of lower-limb exoskeletons: To make future exoskeletons more convenient and comfortable for the wearers, the exoskeletons should be as flexible, adaptable and adjustable to the human wearer to allow better synchronization with the natural human movements while providing the needed assistance. A larger sized and stiffly weighted exoskeleton structure will make the human look robot-like which will create resistance from social use scenarios. Therefore, further work is needed to develop exoskeletons, which can be more easily embedded in normal clothing. In this regard, the area of soft exoskeletons could be interesting. Future actuators for exoskeletons must be lightweight, low cost, noise-free, compact yet have sufficient precision and reliability. Also, they should be able to provide high needed torques at the required speeds without the use of expensive harmonic drives. Series elastic actuators could be developed keeping in mind the torque, speed and weight requirements. It is need of the hour to develop custom-made motors and mechanical transmission systems
80
instead of using the general-purpose motors and drives (e.g. Maxon motors, harmonic drives, PAMs, etc.). The widely used electrical motor – transmission combination is not so smooth and compact and, hence, a notable amount of effort is lost between the actuator-exoskeleton interactions. Construction materials should have low density, high strength and toughness such as non-metals, flexible textile fabrics, carbon fibre and so on. The advanced technological developments in material engineering and nano-technology field can reduce the size and weight of mechanical structures of exoskeletons drastically. The overall comfort and better fit of the exoskeletons need to be worked on as this will not only allow for long duration wearability but will also add a sense of satisfaction, comfort and independence. General-purpose or multipurpose devices should be avoided at this stage as they are likely to be not very effective. However, specific application or specific task functionality exoskeletons should be developed by adopting concepts such as modularity where components can be reused and functionalities enhanced in a straightforward manner. New powering technologies apart from conventional batteries should be explored which are not only autonomous but also more cost-effective and environment friendly. This can be realized by taking power from human efforts and using alternative power sources. It is important to note that the battery technology has not progressed so fast as compared to the tremendous developments in portable electronic systems. Therefore, even though lightweight electronic controllers and sensors are developed but the batteries needed to run these systems are still heavier and have limited power durations. Thus, a periodic battery charging is a frequent necessity, which is a serious challenge that is needed to be addressed urgently. Research on energy harvesting from human motion is also being carried out. However, the amount of electrical energy which is extracted so far from human walking or running activity is not so sufficient; for example, a few milliwatts harvesting systems have been developed and these are unable to power exoskeletons yet. However, such methods can be explored to utilize human motion harvested energy to power sensors and other low-power electronics of the exoskeleton. The efficiency of energy harvesting from human motion activities can be further enhanced by improving the design of the harvester, energy extracting and storage circuitry, tuning the harvester to the frequency of human motion, developing highly efficient energy conversion materials and so on.
81,82
More focus is required on studying the details of the various human motions have to be performed and make the use of exoskeletons more comfortable and trouble free for the wearers. Only a few exoskeletons have been realized that are able to provide full stabilization, without supporting crutches/sticks since this increases the weight of the exoskeleton significantly. This aspect needs to be worked on, to reduce weight without compromising human stability. Better mechanisms for torque variation and motion reversal at joints to enable quick motions without fuss are needed. The level of assistance for lower-limb exoskeleton for elderly should be configurable so that the wearer can decide the level of assistance needed from the worn exoskeleton. The higher prices of exoskeletons is major challenge restricting popularity among society. The general price range of lower-body mobility exoskeleton systems is between US $30,000 and US $130,000, which is beyond the affordable limit of most people who need them. More efforts are needed to reduce these prices by developing cheaper components so that the technology become more accessible. As most of the end users are vulnerable, fragile elderly people, human safety is of paramount importance. International safety organizations have devised pertinent rules and regulations, but further research is needed to improve the safety testing aspects to assess compliance.
Conclusions
The article has presented a review of lower-body wearable exoskeletons classified on the basis of end applications for medical and non-medical purposes. The current exoskeletons which have emerged have been compared to give insight into their design and functionality from mechanical and electrical perspectives including key issues such as actuators, control strategy, sensors, powering methods, mechanisms and materials. A comprehensive literature survey was done for this, highlighting the less explored research areas and suggesting suitable trending applications where exoskeletons should be employed. The other aim of the classification and comparison of wearable exoskeletons is to bring out issues where improvements are needed in the existing wearable exoskeletons and to allow the research and development focus to be made in relevant technology areas for advancing lower-limb exoskeletons. Further, the need for safety standardization and regulation in medical and non-medical wearable devices was introduced and its impact on the overall process to deliver the urgently needed products to the marketplace in medical and non-medical applications. As a whole, it is hoped that this article will help researchers and academicians to have a comprehensive state-of-the-art presentation on the lower-limb exoskeletons and will enable the community to review and realign research efforts to maximize impact on the rapidly evolving area of personal care robots by keeping in mind the issues of safety and standardization.
Footnotes
Acknowledgements
Part of the work presented here was carried out under the AAL Call 4 project EXO-LEGS within Europe with funding from Sweden, Germany, Spain, Switzerland and United Kingdom. The help and support from the EXO-LEGS partners for the work carried out is gratefully acknowledged.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
