Abstract
Purpose
To evaluate the prognostic effect of pretreatment serum superoxide dismutase (SOD) activity in locoregionally advanced nasopharyngeal carcinoma.
Methods
A total of 498 patients diagnosed with stage III–IVA nasopharyngeal carcinoma between January 2013 and December 2016 were involved in this study. The X-tile program was used to determine the cut-off value of pretreatment serum SOD activity based on disease-free survival. Kaplan–Meier methods and Cox proportional hazards models were used to evaluate the impact of serum SOD levels on survival outcomes. The receiver operating characteristic (ROC) curve analysis was used to compare the prognostic value of clinical stage, pretreatment serum SOD level, and the combination of them regarding disease-free survival.
Results
Based on the X-tile plot, the optimal cutoff value of pretreatment serum SOD activity for disease-free survival was 146.0U/mL. As a dichotomous variable, SOD was significantly higher in non-keratinizing differentiated disease (P = 0.027) and early T stage (P = 0.011). Compared with the lower subset, higher SOD activity predicted an inferior 3-year rates of overall survival (84.6 vs. 94.7%, P < 0.001), distant metastasis-free survival (78.3 vs. 92.8%, P < 0.001) and disease-free survival (78.2 vs. 92.8%, P < 0.001). Multivariate analysis verified that the SOD activity was an independent prognostic indicator to predict distant metastasis, disease progression, and death. The area under the ROC curve (AUC) of the combination was superior to that of clinical stage or SOD alone for disease-free survival (both P < 0.01).
Conclusion
Serological SOD activity before treatment is an important prognostic indicator for patients with stage III–IV non-metastatic nasopharyngeal carcinoma undergoing chemoradiation therapy.
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) is mainly distributed in southern China and countries in Southeast Asia with around 133,000 new cases and 80,000 disease-related deaths occurred in 2020 worldwide.1,2 Over 70% of NPC patients are diagnosed with locoregionally advanced disease at presentation. 3 With the utilization of intensity-modulated radiotherapy (IMRT) and platinum-based chemotherapy, the survival outcomes of locoregionally advanced NPC (LA-NPC) patients have greatly improved.4,5 However, locoregional recurrence and distant metastasis are still the main causes of treatment failure. 3 Currently, the tumor node metastasis (TNM) staging system remains the gold standard for risky group classification. Nevertheless, there are profound individual differences in clinical outcomes between NPC patients presenting with an identical stage of disease. 6 Thus, the identification of clinical prognostic indicators to complement the TNM staging system may allow for an individualized approach tailored to the management of NPC patients.
Reactive oxygen species (ROS), which consist of superoxide and hydroxyl free radicals and hydrogen peroxide (H2O2), are produced by inflammation, hyperoxia, and reperfusion after ischemia, as well as by radiotherapy and radical-producing anticancer drugs, such as cisplatin (DDP).7–9 Superoxide dismutase (SOD) is the main enzyme that forms the first line of defense against oxygen-derived free radicals and catalyzes the removal of superoxide free radicals. 10 The regulatory functions of SOD in metabolism, growth, and the oxidative stress response are increasingly recognized as critical for carcinogenesis, tumor progression, and sensitivity to anticancer therapy. 11
There have been lots of studies regarding SOD in cancerous tissues of humans. A number of studies have reported an increased level of SOD activity in tumor tissue at multiple sites.12–14 Conversely, in other reports, the tumor tissue SOD activity was down-regulated in carcinomas of liver, 15 kidney, 16 cervix, 17 and colon. 18 The results of the SOD status in various organs therefore remain controversial, probably due to differences in the cancerous tissues studied. Until now, only a few studies have been launched to illustrate the role of SOD in serum and its possible relationship with the prognosis of malignancies.
On the basis of this premise, we performed a retrospective clinical analysis containing 498 LA-NPC patients to explore the correlation between pretreatment SOD activity in serum and the characteristics and prognosis of patients.
Methods
Patients
The protocol was approved by the Institutional Review Board of the Affiliated Cancer Hospital & Institute of Guangzhou Medical University, and informed consent was obtained from all patients. This study also complied with the Declaration of Helsinki. The NPC records of patients hospitalized in the Affiliated Cancer Hospital & Institute of Guangzhou Medical University between January 2013 and December 2016 were reviewed.
Data from patients who met the following criteria were selected for further analysis: (a) biopsy-proven non-keratinizing nasopharyngeal carcinoma based on the current World Health Organization (WHO) pathologic classification; (b) stage III–IVA disease based on the 8th edition of the American Joint Committee on Cancer (AJCC) staging system; (c) Eastern Cooperative Oncology Group grade rank of 0–2; (d) without severe complications; (e) availability of complete clinical information; (f) no evidence of distant metastasis. The exclusion criteria were: (a) patients with history of chemotherapy or radiotherapy before our research; (b) patients with serious complications or other primary malignancies when diagnosed as NPC; (c) pregnancy or lactation.
Baseline evaluation
The routine pretreatment evaluation for NPC patients consisted of a comprehensive medical history, physical examination, hematological and biochemical testing, nasopharynx biopsy, magnetic resonance imaging of the nasopharynx and the whole neck, abdominal ultrasonography, chest radiography, emission computed tomography bone imaging, and/or 18F-fluorodeoxyglucose positron emission tomography-computed tomograph if applicable. Serological SOD activities were determined within 4 weeks before the start of treatment applying a Siemens ADVIA 2400 chemistry analyzer (Siemens Healthcare, Germany). Normal serum activities of SOD were defined to 129.0–216.0U/mL. In the measurement of SOD, the coefficient of variance was <5.0%.
Radiation therapy
In the present study, all patients underwent RT with 6-MV photons by application of an IMRT technique. Detailed information with respect to the planning and delivery of IMRT has been previously described.19,20
The primary tumor of the nasopharynx, defined as the clinically and/or radiologically observed nasopharyngeal primary lesion, was treated with 66–72Gy in 28–33 fractions. The metastatic lymph node, defined as the enlarged lymph node detected on clinical examinations and radiographs, was treated with 64–70Gy. Radiotherapy was delivered using simultaneous-integrated boost IMRT. All patients were treated following a routine schedule with five daily fractions per week for 6–7 weeks.
Chemotherapy
Neoadjuvant or adjuvant chemotherapy mainly contain platinum-based regimens including 5-fluorouracil (1000 mg/m2, 120 h of continuous intravenous infusion) with cisplatin/nedaplatin (80 mg/m2, first day of chemotherapy course) (PF), or docetaxel (75 mg/m2, first day of chemotherapy course) with cisplatin/nedaplatin (75 mg/m2, first day of chemotherapy course) (TP), or docetaxel (60 mg/m2, first day of chemotherapy course) plus cisplatin/nedaplatin (60 mg/m2, first day of chemotherapy course) with 5-fluorouracil (600 mg/m2, 120 h of continuous intravenous infusion) (TPF) every 3 weeks for two or three cycles. Concomitant chemotherapy consisted of two to three cycles of cisplatin/nedaplatin (80 mg/m2, first day of chemotherapy course) every 3 weeks.
Clinical outcome assessment and follow-up
In the course of treatment, patients were assessed at least once per week. After completion of treatment, patients were then followed up once every 3 months in the first 3 years, once every 6 months for the following 2 years, and once every year thereafter until death. The procedures of evaluation were similar with those carried out before treatment. The primary endpoint was disease-free survival (DFS) rates at 3 years. The secondary endpoints included locoregional relapse-free survival (LRRFS), distant metastasis-free survival (DMFS), as well as overall survival (OS). Patient follow-up was calculated from the first day of treatment to either the day of death or the last day of examination.
Statistical analysis
All statistical analyses employed the Statistical Package for the Social Sciences software package (SPSS 25.0, SPSS, Inc., Chicago, IL) and the X-tile version 3.6.1 (Yale School of Medicine, New Haven, CT, USA). 21 The X-tile program was utilized to identify the optimal cutoff values for the activity of SOD based on their associations with disease-free survival (DFS) of patients in the entire cohort. Independent sample t tests were applied to compare the SOD as a continuous variable. Chi-square tests were used to determine the significance of differences for patients grouped by SOD as a dichotomous variable. Survival rates were calculated using Kaplan–Meier estimates and compared to Log-rank test. Multivariate prognostic analyses were performed by Cox proportional hazards regression model to test independence, significance, and hazard discrimination. The area under curve (AUC) of receiver operating characteristic (ROC) was utilized to calculate the validity of prediction for clinical stage, pre-treatment serum SOD level, and the combination of these two factors (the maximal AUC>0.50 was considered as medically significant). The prognostic values of them were compared by AUC values using Medcalc version 19.0.4 (Ostend, Belgium).22,23 Statistical significance was taken as P value < 0.05 (two tailed).
Results
Patient characteristics and treatment
The clinicopathological characteristics of 498 patients are summarized in Table 1. The median age at diagnosis was 47 years (interquartile range: 40–54 years). The patients consisted of 358 males (71.9%) and 140 females (28.1%). According to the WHO criteria, 95.6% of patients had non-keratinizing undifferentiated carcinoma and 4.4% non-keratinizing differentiated carcinoma. Based on the new AJCC staging system, 276 of the cases (55.4%) were stage III and 222 (44.6%) stage IVA.
Association of pretreatment serum SOD level with clinicopathological factors in patients with locoregionally advanced nasopharyngeal carcinoma.
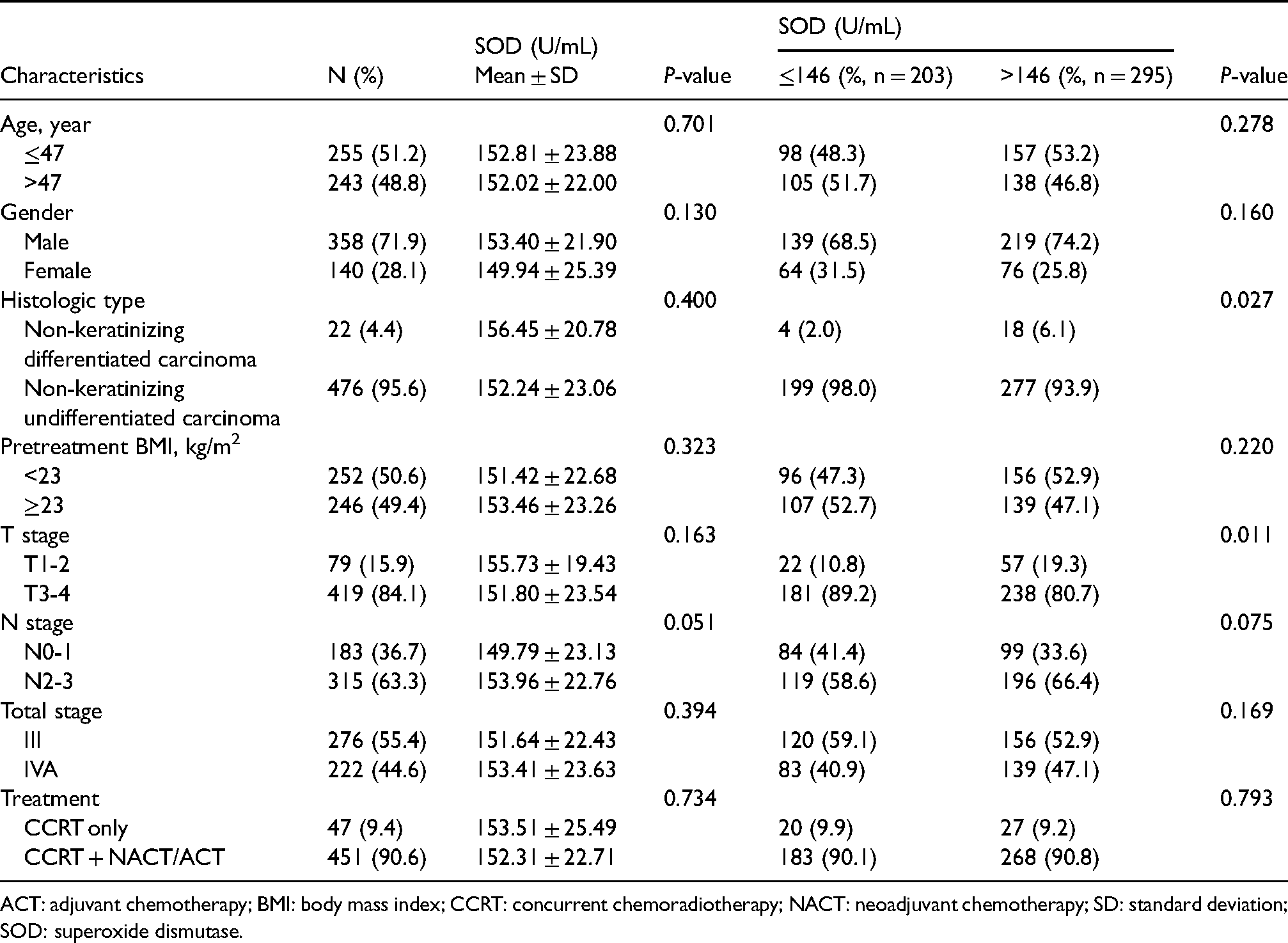
ACT: adjuvant chemotherapy; BMI: body mass index; CCRT: concurrent chemoradiotherapy; NACT: neoadjuvant chemotherapy; SD: standard deviation; SOD: superoxide dismutase.
A total of 47 patients (9.4%) received concurrent chemoradiotherapy (CCRT) only and 451 (90.6%) CCRT combined with neoadjuvant chemotherapy (369/498, 74.1%) or adjuvant chemotherapy (82/498, 16.5%). For CCRT, which was only administered to 47 patients, 31 (66.0%) received two cycles and 16 (34.0%) received three cycles of cisplatin/nedaplatin. A total of 77.0% (284/369) of patients received two cycles of neoadjuvant platinum-based chemotherapy and the remaining patients (85/369, 23.0%) could complete three cycles of neoadjuvant platinum-based chemotherapy. For the adjuvant chemotherapy in 82 patients, 43 (52.4%) had two cycles and 39 (47.6%) had three cycles of PF, TP, or TPF chemotherapy. All patients completed the full course of radiation therapy.
Treatment response
Twelve weeks after the end of radiotherapy, 465 patients (93.4%) had achieved complete regression considering the primary tumor and neck together, 18 patients (3.6%) achieved a partial response, 15 patients (3.0%) were non-assessable, and none had a stable or progressive disease. Among the patients with partial response, only 2 received surgical intervention for residual nodal disease.
Determination of the optimal cutoff value of SOD
X-tile software was used to calculate the predictive value of SOD for DFS in patients with LA-NPC. When applying an SOD of 146.0U/mL as the cut point, the maximum of × 2 log-rank values of 27.96 (P < 0.001) was achieved for DFS (Supplementary Figure 1). Therefore, 146.0U/mL was chosen as the optimal cutoff value of SOD for DFS in LA-NPC. We then classified all patients into low and high subgroups by the predetermined cutoff point.
The relationship between SOD and clinicopathological factors
At the time of diagnosis, 87.3% (435/498) of patients with LA-NPC had serum levels of SOD within the normal limits (129.0–216.0U/mL), 57 patients (11.4%) lower than the normal limits (<129.0U/mL), and 6 patients (1.2%) higher than the normal limits (>216.0U/mL). The mean and median serum levels of SOD for the entire cohort were 152.4 and 151.0U/mL, respectively, and the values ranged from 97.0 to 306.0U/mL. As a continuous variable, SOD activity was independent of patient's age, gender, histologic type, body mass index, T stage, N stage, total stage, and treatment strategy. As a dichotomous variable, SOD activity was significantly higher in non-keratinizing differentiated disease (P = 0.027) and early T stage (P = 0.011, Table 1).
Survival outcome
The median follow-up time for all patients was 37 months (range, 3–76 months), and the median times to failure were 16 months (3–57 months) and 20 months (8–56 months) for distant metastasis and locoregional recurrence, respectively. The rates of 3-year survival for the entire cohort were: OS, 88.9%; LRRFS, 94.7%; DMFS, 84.3%; and DFS, 84.2%.
Univariate and multivariate analysis
In the univariate analysis, high serum SOD activity was significantly related to lower 3-year OS, DMFS, and DFS. The 3-year rates of OS, LRRFS, DMFS, and DFS for high versus low serum SOD groups were 84.6 versus 94.7% (P < 0.001), 93.5 versus 96.2% (P = 0.513), 78.3 versus 92.8% (P < 0.001), and 78.2 versus 92.8% (P < 0.001), respectively (Table 2 and Supplementary Figure 2). Additionally, patients with advanced N stage and total stage had inferior 3-year OS, DMFS, and DFS, and female patients tended to have better 3-year DMFS and DFS. In the multivariate analysis, a high serum SOD activity was proved to be a statistically significant independent predictor of the OS, DMFS, and DFS. Besides, age and total stage were also independent prognostic factors (Table 3).
Univariate analysis of prognostic factors with locoregionally advanced nasopharyngeal carcinoma.
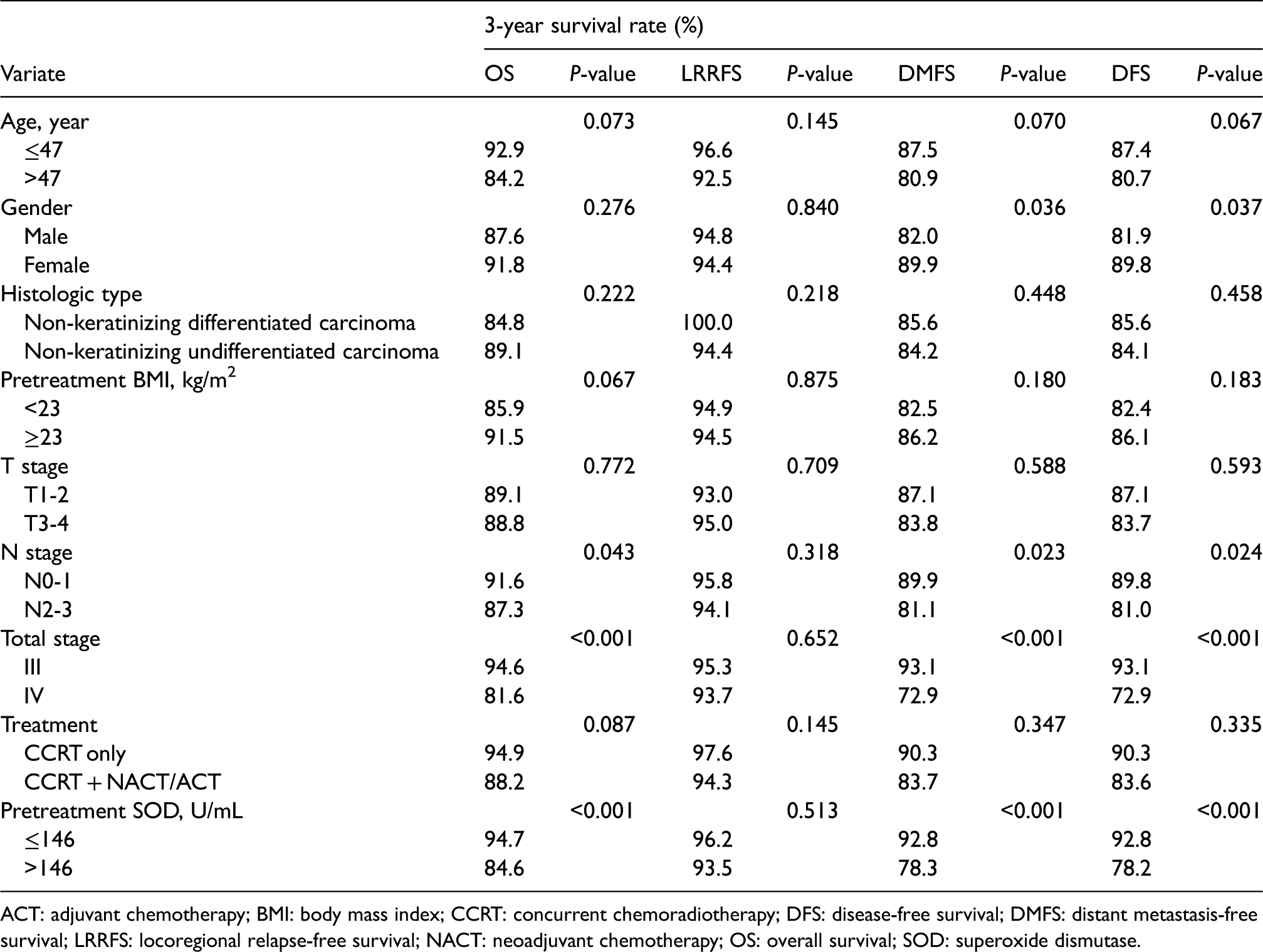
ACT: adjuvant chemotherapy; BMI: body mass index; CCRT: concurrent chemoradiotherapy; DFS: disease-free survival; DMFS: distant metastasis-free survival; LRRFS: locoregional relapse-free survival; NACT: neoadjuvant chemotherapy; OS: overall survival; SOD: superoxide dismutase.
Multivariate analysis of prognostic factors with locoregionally advanced nasopharyngeal carcinoma.
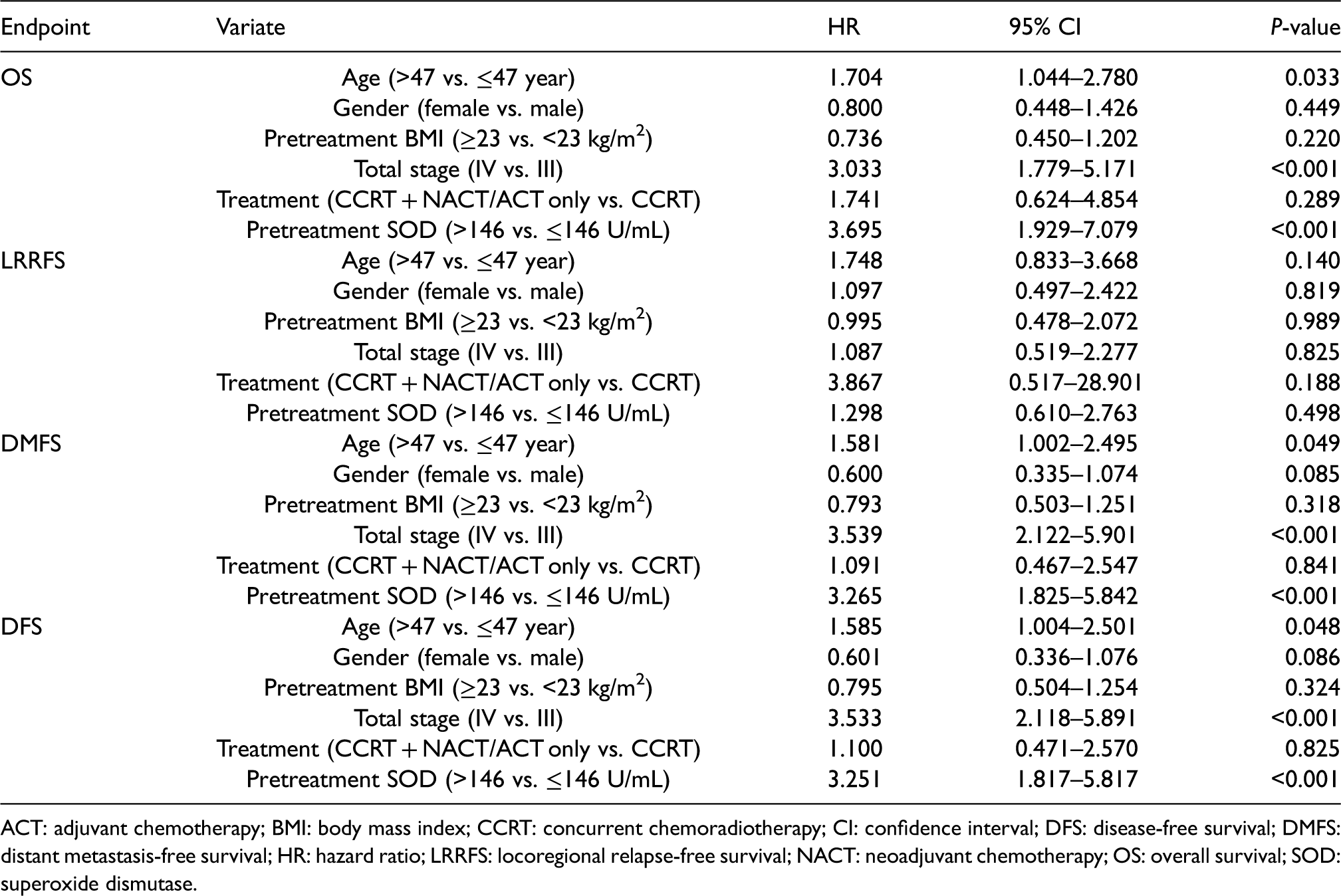
ACT: adjuvant chemotherapy; BMI: body mass index; CCRT: concurrent chemoradiotherapy; CI: confidence interval; DFS: disease-free survival; DMFS: distant metastasis-free survival; HR: hazard ratio; LRRFS: locoregional relapse-free survival; NACT: neoadjuvant chemotherapy; OS: overall survival; SOD: superoxide dismutase.
Combination of the clinical classification and baseline serum SOD level
Both univariate and multivariate analysis revealed that clinical classification and pretreatment serum SOD status were prognostic predictors for survival. Therefore, patients were divided into four subgroups according to these two factors: group 1 (stage III and SOD≤146U/mL), group 2 (stage III and SOD>146U/mL), group 3 (stage IV and SOD≤146U/mL), and group 4 (stage IV and SOD>146U/mL). The 3-year rates of OS, LRRFS, DMFS, and DFS for the four subgroups were 98.7% versus 91.1% versus 88.8% versus 77.0% (Figure 1(a)), 95.5% versus 95.1% versus 97.3% versus 91.2% (Figure 1(b)), 97.3% versus 89.8% versus 86.1% versus 64.9% (Figure 1(c)), and 97.3% versus 89.7% versus 86.0% versus 64.9% (Figure 1(d)), respectively. The OS, DMFS, and DFS for patients with stage III disease + lower SOD and stage IV disease + lower SOD were significantly better than those with stage III disease + higher SOD and stage IV disease + higher SOD, respectively (P < 0.05 for all groups). However, patients with stage III disease + higher SOD had similar OS, LRRFS, DMFS, and DFS as those with stage IV disease + lower SOD (Figure 1(a) to (d)). Additionally, ROC curves were generated to compare the prognostic value of clinical stage, SOD and the combination of these two factors. As shown in Figure 2, the AUC for clinical stage, pre-treatment serum SOD level and the combination of them with regard to DFS were 0.664, 0.632, and 0.723, respectively; thus, the prognostic ability of the combination was significantly superior to clinical stage or SOD alone (both P < 0.01).
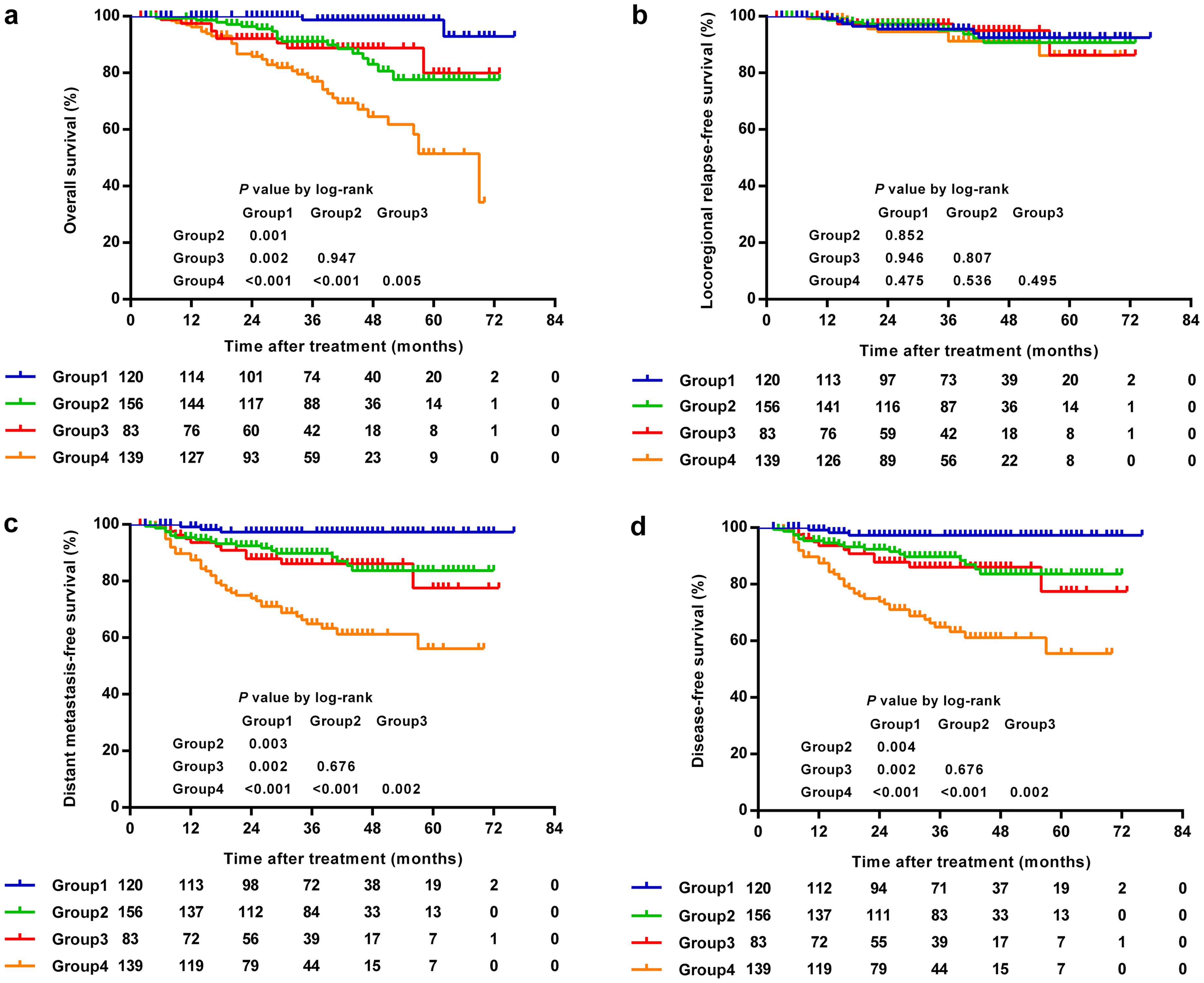
Kaplan–Meier curves according to clinical stage and SOD level. The OS (a), DMFS (c) and DFS (d) for patients with stage III disease + lower SOD and stage IV disease + lower SOD were significantly better than those with stage III disease + higher SOD and stage IV disease + higher SOD, respectively (P < 0.05 for all groups). Patients with stage III disease + higher SOD had similar OS (a), LRRFS (b), DMFS (c) and DFS (d) as those with stage IV disease + lower SOD. DFS: disease-free survival; DMFS: distant metastasis-free survival; LRRFS: locoregional relapse-free survival; OS: overall survival; SOD: superoxide dismutase.
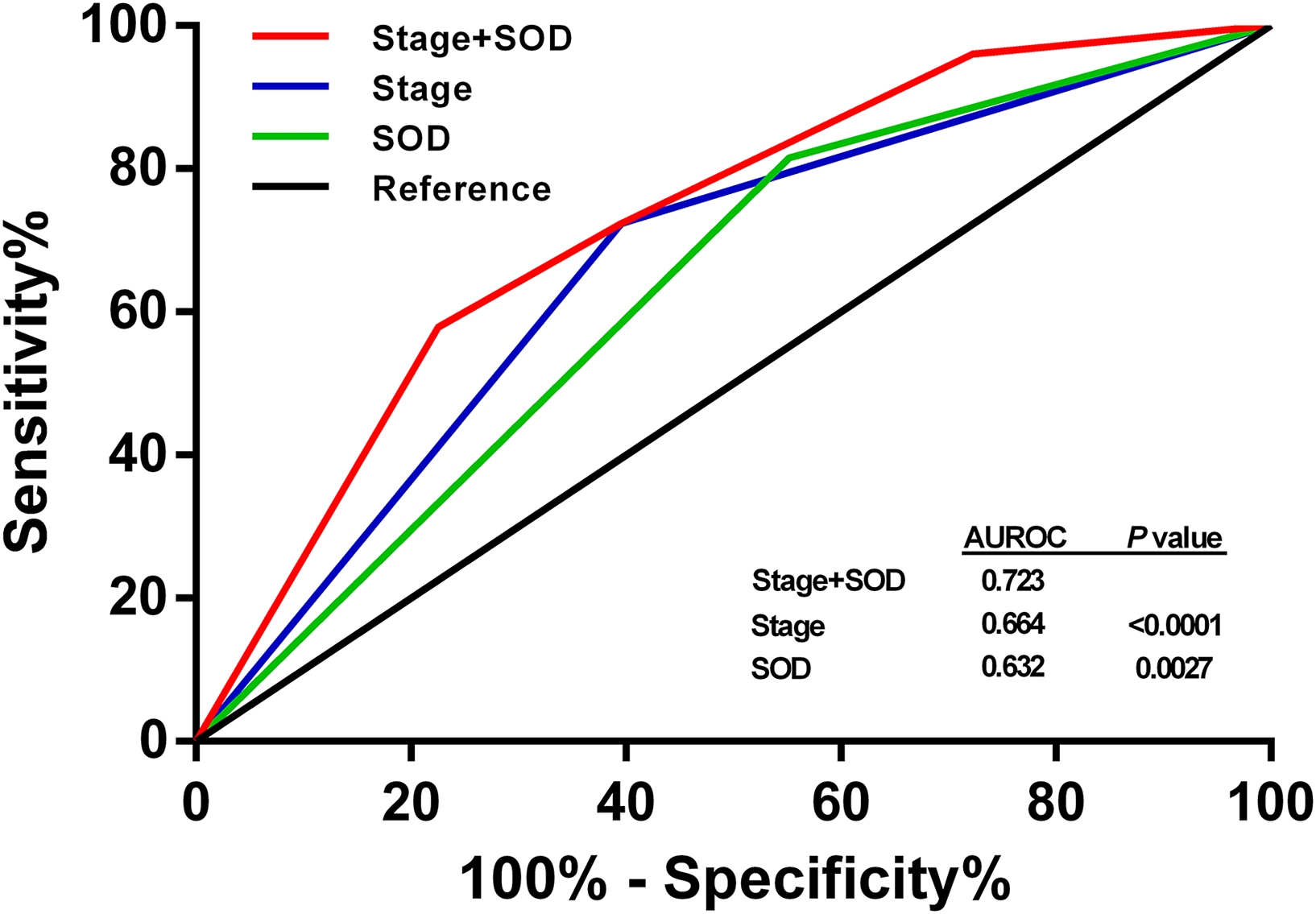
Receiver operating characteristic (ROC) curve analysis. The prognostic ability of the combination was significantly superior to clinical stage or SOD alone with regard to DFS (both P < 0.01). DFS: disease-free survival; SOD: superoxide dismutase.
Discussion
Previous studies have compared the level of serum SOD between cancer patients and healthy donors; however, the findings remain controversial. For example, the serum elevation of SOD was reported as a tumor marker in malignant melanoma, 24 gastric cancer, 25 and Hodgkin's disease. 26 On the other hand, several studies emphasized the serum decrease of SOD in leukemia 26 and NPC.27,28 Our study further demonstrated that for LA-NPC patients, the serum SOD was significantly decreased in advanced T stages (T3–4) than in early stages (T1–2).
The progression of primary tumor in NPC patients may be explained by the lack of balance between ROS and antioxidant defenses. Reactive oxygen metabolites, including hydroxyl radical, superoxide anion, and hydrogen peroxide, are involved in the initiation and progression of cancer.29–31 During multistep carcinogenesis, exposure to carcinogens could lead to DNA damage mediated by ROS. 32 Antioxidant enzymes may protect normal cells from the toxic effects of excessive concentrations of ROS generated in the process of cellular metabolism. As one of the most critical antioxidant enzymes involved in the primary cellular defense, SOD may contribute to the protection against carcinogenesis or progression of malignancies. In the present study, for NPC patients with lower level of serum SOD, oxidative damage can arise from the deficiency of antioxidant defense, leading to more aggressive behavior and more invasive features of primary tumor.
According to the WHO standards, NPCs are grouped into keratinizing squamous cell carcinoma and non-keratinizing carcinoma. The latter group can be further divided into non-keratinizing differentiated carcinoma and non-keratinizing undifferentiated carcinoma. 33 The keratinizing subtype constitutes less than 20% of cases worldwide, and is relatively rare in endemic areas such as southern China; the non-keratinizing subtype accounts for most cases in endemic areas (>95%) and is predominantly connected with Epstein–Barr virus (EBV) infection.34,35 We excluded patients with keratinizing subtype since their etiology and outcomes were different from the non-keratinizing counterparts. 36 We found that in LA-NPC patients, SOD activity was significantly higher in non-keratinizing differentiated disease. This could be related to the effects of SOD on tumor cell differentiation, pending further supportive evidence in a larger number of patients.
Intriguingly, elevated serum SOD concentration was related to inferior survival outcomes. This result is consistent with other reports, which found that the expression of manganese superoxide dismutase (MnSOD) was negatively associated with OS in patients with NPC. 37 Therefore, although not yet tested, it is reasonable to speculate that the increased SOD in the serum of NPC patients was likely to be the seepage of SOD enzymes from the tumor tissue into the peripheral blood. Furthermore, multivariate analysis using the Cox proportional hazards model verified that pretreatment serum SOD was an independent prognostic factor in LA-NPC patients. Patients with higher SOD levels had 3.43 times the risk of distant metastasis, 3.41 times the risk of disease progression and 4.05 times the risk of death of those with lower SOD levels. Moreover, higher serum SOD remained to be an independent unfavorable prognostic factor in both stage III and IV patients.
The paradoxical finding that higher SOD levels were seen predominantly in patients with early T stage but associated with worse survival outcomes may be explained by the different functions of SOD in the process of tumor progression and anti-cancer treatment. The relationship between SOD and cancer treatments is of great interest, because radiation and some chemotherapeutic drugs induce anti-tumor effects through provoking an enormous number of noxious ROS beyond the capacity of the endogenous antioxidant system. 38 There is an augmentation in the production of ROS when tumor cells are irradiated. Due to the cell being approximately 80% water, the majority of the radiation energy deposited in the cells is originally absorbed in water; thus, the reactive radicals are produced quickly. These unstable free radicals rapidly react with other molecules, leading to the formation of more free radicals in a chain reaction that takes place generally in mitochondria and within the endoplasmic reticulum. The peroxidation of lipids, modification of proteins, as well as fragmentation of DNA, are induced by high concentrations of reactive radicals, consequently damaging and destroying cells. 39 Regarding anticancer drugs, platinum-based chemotherapy is at present recommended as an inherent component of standard therapy for patients with LA-NPC. Previous studies have demonstrated that the activation of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase (NOX) mediated by cisplatin can trigger oxygen (O2) to superoxide radical (O2•−) and its downstream H2O2. H2O2 was able to be catalyzed by Fe2+/Fe3+ to the noxious hydroxyl radicals (•OH) through the effect of Fenton's reaction. These radicals are ROS that brings about oxidative injury to lipids, proteins, and DNA. 40
In the process of radiotherapy and chemotherapy, instead of playing a role as a protector of normal cells from ROS-mediated carcinogenesis, SOD usually confers cancer cell resistance to ROS-induced cytotoxicity. For instance, in colon cancers, highly expressed MnSOD confers resistance to radiation, hyperthermia, and doxorubicin; and suppression of the endogenous MnSOD expression level can overcome the resistance of these cancers. 41 In Xiao's study, 34 differential proteins in the radioresistant NPC cell line CNE2-IR were identified by proteomic approach, and the study further verified that the expression abnormality of MnSOD might result in NPC radioresistance and be a potential biomarker for the prediction of NPC response to radiation therapy. 42 Zhao and colleagues have also shown that MnSOD expression participates in radioresistance of NPC, and silencing of MnSOD can enhance the radiosensitivity of NPC cells to ionizing radiation (IR) injury.39,43 Therefore, among patients with LA-NPC, the determination of serum SOD activity is useful in identifying those who will be resistant to chemoradiation therapy. Furthermore, the combination of clinical stage and the pre-treatment serum SOD level had better prognostic value than stage or SOD alone by comparing the AUC values of ROC curves.
Antioxidants, such as β-Carotene, N-Acetyl Cysteine, glutathione, played an anti-apoptotic role against radiation through reducing ROS production and inactivating the mitogen-activated protein kinase (MAPK) pathway in NPC cells. 44 In contrast, temozolomide-perillyl alcohol was shown to exhibit its cytotoxicity via ROS accumulation, which might result from mitochondrial membrane potential collapse and lead to activated MAPKs signaling, DNA damage, and cell cycle arrest, and thus suppress tumor proliferation. 45 Therefore, the use of supplemental antioxidants during radiotherapy should be avoided because of the possibility of tumor protection and reduced treatment efficacy; conversely, ROS-increasing agents should be investigated further as a potentially effective therapy for LA-NPC patients with a high level of pretreatment SOD.
There are some limitations associated with this study. First, this retrospective study was conducted on a single-center sample with a relatively short follow-up time. Thus, prospectively collected data from different institutions with longer follow-up are essential to validate our results. Second, EBV DNA information was not included in this cohort because the detection of EBV DNA has only been available since 2018 in our institution. The comparison of prognostic abilities between serum SOD and EBV DNA concentration is warranted in the future. Another caveat is that tissue SOD activity was not tested in the current study cohort. In gastric cancer, the activity of serum SOD is not necessarily considered to reflect the activity of tissue SOD. 25 Therefore, the study on the relationship between them was urgently needed for patients with NPC. Finally, data on the dynamic serum SOD activity at different time points was not presented in this study. The dynamic serum SOD activity as a result of therapy might be of vital clinical importance, especially for the development of personalized treatment strategies; thus, we will continue to explore the predictive and prognostic roles of serial changes in serum SOD level.
Conclusion
Our research indicated that high pretreatment serum SOD activity was an independent poor prognostic indicator for stage III–IV nonmetastatic NPC. Measurement of pretreatment serum SOD activity may contribute to improving the prognostic value of the TNM staging system.
Supplemental Material
sj-docx-1-jbm-10.1177_17246008221075042 - Supplemental material for Prognostic impact of pretreatment serum superoxide dismutase activity in patients with locoregionally advanced nasopharyngeal carcinoma
Supplemental material, sj-docx-1-jbm-10.1177_17246008221075042 for Prognostic impact of pretreatment serum superoxide dismutase activity in patients with locoregionally advanced nasopharyngeal carcinoma by Wenze Qiu, Jiali Jiang, Zejiang Zhan, Laiji Huang, Jin Deng, Jiacai Ye, Guo Li, Kai Liao, Huanhuan Zhang, Yan Ding, Yawei Yuan and Ronghui Zheng in The International Journal of Biological Markers
Supplemental Material
sj-docx-2-jbm-10.1177_17246008221075042 - Supplemental material for Prognostic impact of pretreatment serum superoxide dismutase activity in patients with locoregionally advanced nasopharyngeal carcinoma
Supplemental material, sj-docx-2-jbm-10.1177_17246008221075042 for Prognostic impact of pretreatment serum superoxide dismutase activity in patients with locoregionally advanced nasopharyngeal carcinoma by Wenze Qiu, Jiali Jiang, Zejiang Zhan, Laiji Huang, Jin Deng, Jiacai Ye, Guo Li, Kai Liao, Huanhuan Zhang, Yan Ding, Yawei Yuan and Ronghui Zheng in The International Journal of Biological Markers
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author contributions
Wenze Qiu, Jiali Jiang, and Zejiang Zhan contributed equally to this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (No. 82002858), Guangzhou Key Medical Discipline Construction Project and Key Clinical Technology of Guangzhou (2019ZD17).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
