Abstract
Background:
The optimal induction chemotherapy (IC) regimen for locally advanced nasopharyngeal carcinoma (LA-NPC) remains uncertain.
Objectives:
The study aims to compare the efficacy and safety of nanoparticle albumin-bound paclitaxel plus cisplatin (NAB-TP) versus gemcitabine plus cisplatin (GP) as IC in locoregionally advanced nasopharyngeal carcinoma (LA-NPC).
Design:
A retrospective analysis.
Methods:
This study was conducted in LA-NPC patients treated at Sun Yat-sen University Cancer Center between 2012 and 2024. All patients received IC with either the GP or NAB-TP regimen followed by concurrent chemoradiotherapy (CCRT). Propensity score matching (PSM) was used to balance baseline characteristics between the GP and NAB-TP groups.
Results:
In total, 908 patients with LA-NPC (197 in the NAB-TP group) were enrolled. Before PSM, no statistically significant differences were observed between the NAB-TP and GP group in 5-year overall survival (OS; 98.67% vs 91.67%; p = 0.647), 5-year progression-free survival (PFS; 84.18% vs 66.22%; p = 0.587), 5-year locoregional relapse-free survival (LRFS; 88.13% vs 81.44%; p = 0.106), or 5-year distant metastasis-free survival (DMFS; 98.05% vs 89.88%; p = 0.106). After PSM, no differences were found in 5-year OS (98.60% vs 86.26%; p = 0.536), PFS (84.18% vs 66.22%; p = 0.587), LRFS (88.82% vs 76.14%; p = 0.757), or DMFS (97.95% vs 91.74%; p = 0.105). In the matched cohort, the NAB-TP group showed significantly lower incidences of anemia and thrombocytopenia than the GP group.
Conclusion:
IC with the NAB-TP combined with CCRT showed comparable survival efficacy for LA-NPC patients compared with the GP, with reduced acute toxicity.
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) is a malignant tumor originating from nasopharyngeal epithelial cells, exhibiting distinct geographic distribution patterns. NPC is rare in Western populations, 1 with an annual incidence rate of less than 1 per 100,000 in the United States, whereas it is endemic in South China and Southeast Asia, 2 where the annual incidence reaches approximately 9 per 100,000.3,4 Over two-thirds of newly diagnosed patients present with locoregionally advanced disease. 5 The standard of care involves the addition of induction chemotherapy (IC) to concurrent chemoradiotherapy (CCRT), 6 which serves to eradicate subclinical micrometastases undetectable by imaging and mitigate the risk of poor prognostic factors. In addition, IC administered prior to radiotherapy (RT) reduces the tumor burden, enhances radiosensitivity, and improves treatment tolerance.7,8 Meanwhile, multidisciplinary management and biomarker-guided systemic treatment further reshape the IC decision-making paradigm in NPC.9–11 Gemcitabine-cisplatin (GP) and docetaxel-cisplatin-fluorouracil regimens are currently endorsed by the NCCN guidelines as first-line IC options for NPC, whereas docetaxel-cisplatin (TP) is classified as a 2B evidence-based recommendation. 12 However, the optimal IC regimen remains contentious.
Although conventional paclitaxel demonstrates robust antitumor activity in locoregionally advanced nasopharyngeal carcinoma (LA-NPC), its clinical use is limited by poor solubility, necessitating solvents such as polyoxyethylated castor oil or polysorbate, which elevate infusion-related hypersensitivity reactions and solvent-associated toxicity. 13 Premedication for desensitization and post-treatment complications—including myelosuppression, skin reactions, and nausea/vomiting—further limit its applicability. Nanoparticle albumin-bound paclitaxel (NAB-paclitaxel) was developed to circumvent solvent-related toxicity while enhancing tumor-specific drug delivery and uptake via albumin-mediated tumor penetration. Clinically, NAB-paclitaxel is effective in advanced solid tumors such as breast, lung, and pancreatic cancers.14–16 Compared with docetaxel-based regimens, NAB-paclitaxel-based IC significantly improved disease-specific survival and overall survival (OS) in p16-positive head and neck squamous cell carcinoma patients. 17 Single-arm studies have reported encouraging antitumor efficacy and manageable toxicity for NAB-paclitaxel combined with cisplatin (NAB-TP) as an IC in LA-NPC. 18
Nevertheless, comparative analyses of NAB-TP versus GP regimens in terms of efficacy and adverse events remain unexplored. This retrospective real-world study aims to evaluate the efficacy and toxicity profiles of NAB-TP versus GP regimens as IC for LA-NPC, thereby informing therapeutic decision-making for this patient population.
Materials and methods
Patient cohort
The Ethics Committee of Sun Yat-sen University Cancer Center (SYSUCC) approved this study (Approval No. SL-B2024-836-01) and waived the requirement for written informed consent due to its retrospective nature. From January 2012 to December 2024, 2203 consecutive previously untreated patients with newly diagnosed non-metastatic NPC were identified at our study institute. The eligibility criteria were as follows: (a) histologically confirmed NPC; (b) Stage III–IVA disease according to the 8th edition of the International Union Against Cancer/American Joint Committee on Cancer Staging system; (c) administration of IC followed by CCRT; (d) IC regimens: NAB-TP (260 mg/m2 albumin-bound paclitaxel on Day 1 and 80 mg/m2 cisplatin on Day 1, intravenously every 3 weeks) or gemcitabine plus cisplatin (GP: 1 g/m² gemcitabine on Days 1 and 8, 80 mg/m2 cisplatin on Day 1, intravenously every 3 weeks), followed by cisplatin-based CCRT (cisplatin 100 mg/m2 every 3 weeks) with RT. The exclusion criteria were as follows: (a) incomplete clinical data; (b) concurrent malignancies; and (c) failure to complete the planned dose of RT. The study design is illustrated in Figure 1. Ultimately, 908 eligible patients were included. TNM staging examinations include magnetic resonance imaging (MRI) of the nasopharynx and neck, fiberoptic nasopharyngoscopy, 18F-fluorodeoxyglucose positron emission tomography (PET), and computed tomography (PET-CT) or whole-body bone scan (ECT) or CT of the chest and abdomen. The reporting of this study conforms to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement (Supplemental File 1).
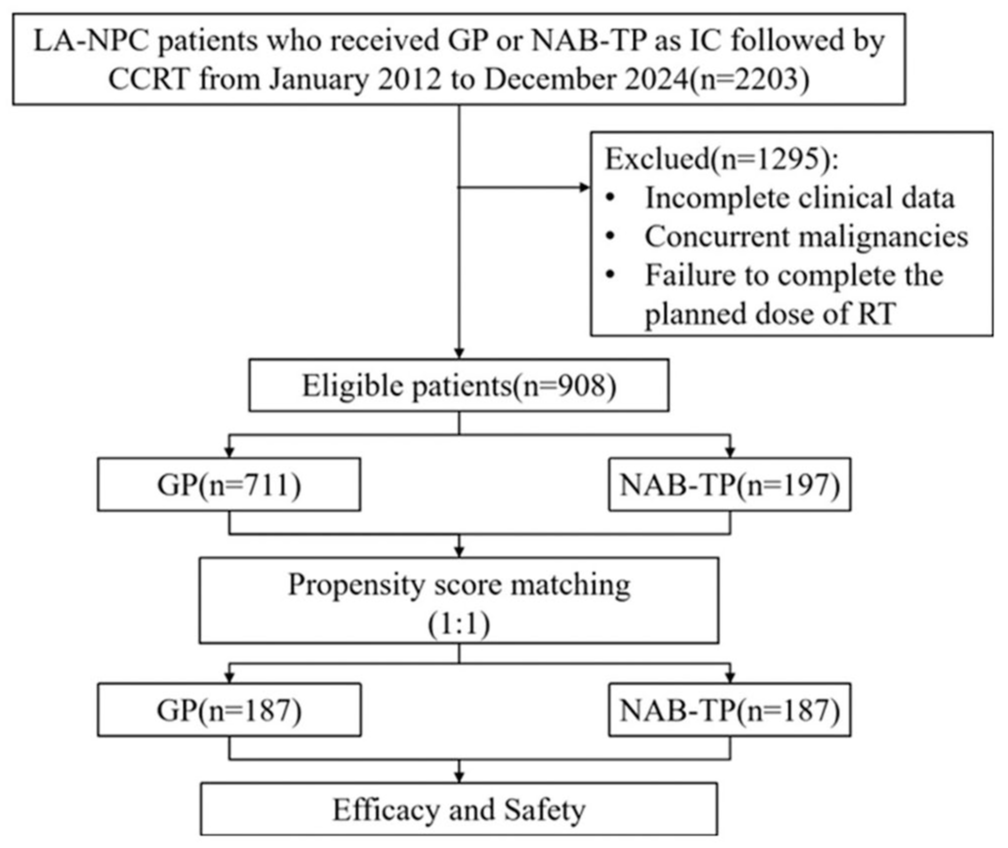
Flowchart of participant enrollment.
Follow-up and endpoints
After completing radical therapy, patients underwent follow-up examinations every 3 months for the first 2 years, every 6 months for the subsequent 3 years, and annually thereafter. During follow-up visits, physical examinations, including visual inspection of the nasopharynx via indirect nasopharyngoscopy, were performed at each appointment. All patients received regular annual examinations comprising chest X-rays, abdominal ultrasound, and MRI of the nasopharynx and neck. Additional investigations, such as CT, ECT, or biopsies, were conducted when clinically indicated. Local recurrence was diagnosed based on MRI and biopsy results, with documentation of tumor recurrence/metastasis locations and timing. The primary endpoints were OS and progression-free survival (PFS). OS was defined as the time from administration to the date of death or last follow-up. PFS was defined as the time from administration to disease progression or death, whichever occurred first. The secondary endpoints included distant metastasis-free survival (DMFS), locoregional relapse-free survival (LRFS), and safety evaluations. Adverse events were recorded using the National Cancer Institute Common Terminology Criteria for Adverse Events version 5.0 (CTCAE v5.0). Tumor response was assessed according to the Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST v1.1) and immune-related RECIST criteria.
Statistical analysis
Statistical analyses were performed using R software (version 4.3.0). Categorical variables were compared using Chi-square tests, corrected Chi-square tests, or Fisher’s exact tests, whereas continuous variables were analyzed with the Mann–Whitney U test. Survival outcomes were calculated via the Kaplan–Meier method, with differences assessed by log-rank tests. Variables impacting survival were evaluated through univariate and multivariate Cox regression models. Propensity score matching (PSM) was employed to ensure patient comparability based on balanced baseline characteristics. The match ratio used was 1:1. All p values were two-tailed, and p < 0.05 was considered statistically significant for all analyses.
Results
Patient characteristics
A total of 908 patients were enrolled, with a median age of 45 years (range: 15–72), comprising 671 males (73.9%) and 237 females (26.1%). Among them, 197 patients (21.7%) received IC with NAB-TP, whereas 711 (78.3%) received IC with GP. Before PSM, the NAB-TP group had a significantly higher proportion of patients with stage N2 disease compared to the GP group (54.82% vs 41.49%), whereas the GP group had a greater percentage of patients with stage N1 disease (36.85% vs 24.87%), indicating statistically significant differences in N stage distribution (p = 0.002). In addition, the proportion of patients with a family history of NPC was significantly higher in the NAB-TP group (13.20% vs 7.45%, p = 0.011), whereas the GP group had more patients with baseline lactate dehydrogenase (LDH) > 250 U/L (8.44% vs 3.05%, p = 0.010). After 1:1 PSM, 187 patients received NAB-TP followed by CCRT, and 187 received GP followed by CCRT, achieving balanced distributions in N stage, family history of NPC, and baseline LDH levels; all reported parameters were balanced between groups, with no statistically significant differences detected. The baseline characteristics of the study cohort are summarized in Table 1.
Baseline characteristics of the patients in the entire cohort and matched cohorts.

CRP, C-reactive protein; EBV, Epstein–Barr virus; GP, gemcitabine plus cisplatin; LDH, lactate dehydrogenase; NAB-TP, nanoparticle albumin-bound paclitaxel plus cisplatin; NPC, nasopharyngeal carcinoma; PSM, propensity score matching; SMD, standardized mean difference.
Survival outcomes
The median follow-up duration was 27 months (interquartile range: 15–37 months). The 5-year OS, PFS, LRFS, and DMFS rates for the entire included population were 92.66% (95% confidence interval (CI): 88.47%–97.05%), 76.17% (95% CI: 69.45%–83.53%), 83.08% (95% CI: 77.50%–89.07%), and 91.95% (95% CI: 86.32%–97.96%), respectively. Before PSM, the 5-year OS rates were 98.67% (95% CI: 96.82%–100.00%) in the NAB-TP group and 91.67% (95% CI: 86.54%–97.10%) in the GP group (p = 0.647; Figure 2(a)). For the NAB-TP group versus the GP group, the 5-year PFS rates were 84.18% (95% CI: 72.41%–97.87%) versus 66.22% (95% CI: 46.60%–94.10%; p = 0.587; Figure 2(b)), the 5-year LRFS rates were 88.13% (95% CI: 78.77%–98.59%) versus 81.44% (95% CI: 74.65%–88.85%; p = 0.106; Figure 2(c)), and the 5-year DMFS rates were 98.05% (95% CI: 95.85%–100.00%) versus 89.88% (95% CI: 81.82%–98.73%; p = 0.106; Figure 2(d)), indicating no significant survival differences between the NAB-TP and GP groups. After PSM, the 5-year OS, PFS, LRFS, and DMFS rates for the entire included population were 91.13% (95% CI: 83.34%–99.65%), 76.23% (95% CI: 65.27%–89.03%), 82.82% (95% CI: 73.58%–93.23%), and 94.20% (95% CI: 90.82%–97.71%), respectively. The NAB-TP versus GP groups showed 5-year OS rates of 98.60% (95% CI: 96.66%–100.00%) versus 86.26% (95% CI: 73.12%–100.00%; p = 0.536; Figure 2(e)), 5-year PFS rates of 84.18% (95% CI: 72.41%–97.87%) versus 66.22% (95% CI: 46.60%–94.10%; p = 0.587; Figure 2(f)), 5-year LRFS rates of 88.82% (95% CI: 79.20%–99.61%) versus 76.14% (95% CI: 59.84%–96.88%; p = 0.757; Figure 2(g)), and 5-year DMFS rates of 97.95% (95% CI: 95.65%–100.00%) versus 91.74% (95% CI: 86.68%–97.09%; p = 0.105; Figure 2(h)), confirming that there were no significant survival differences between the matched groups (Table 2).
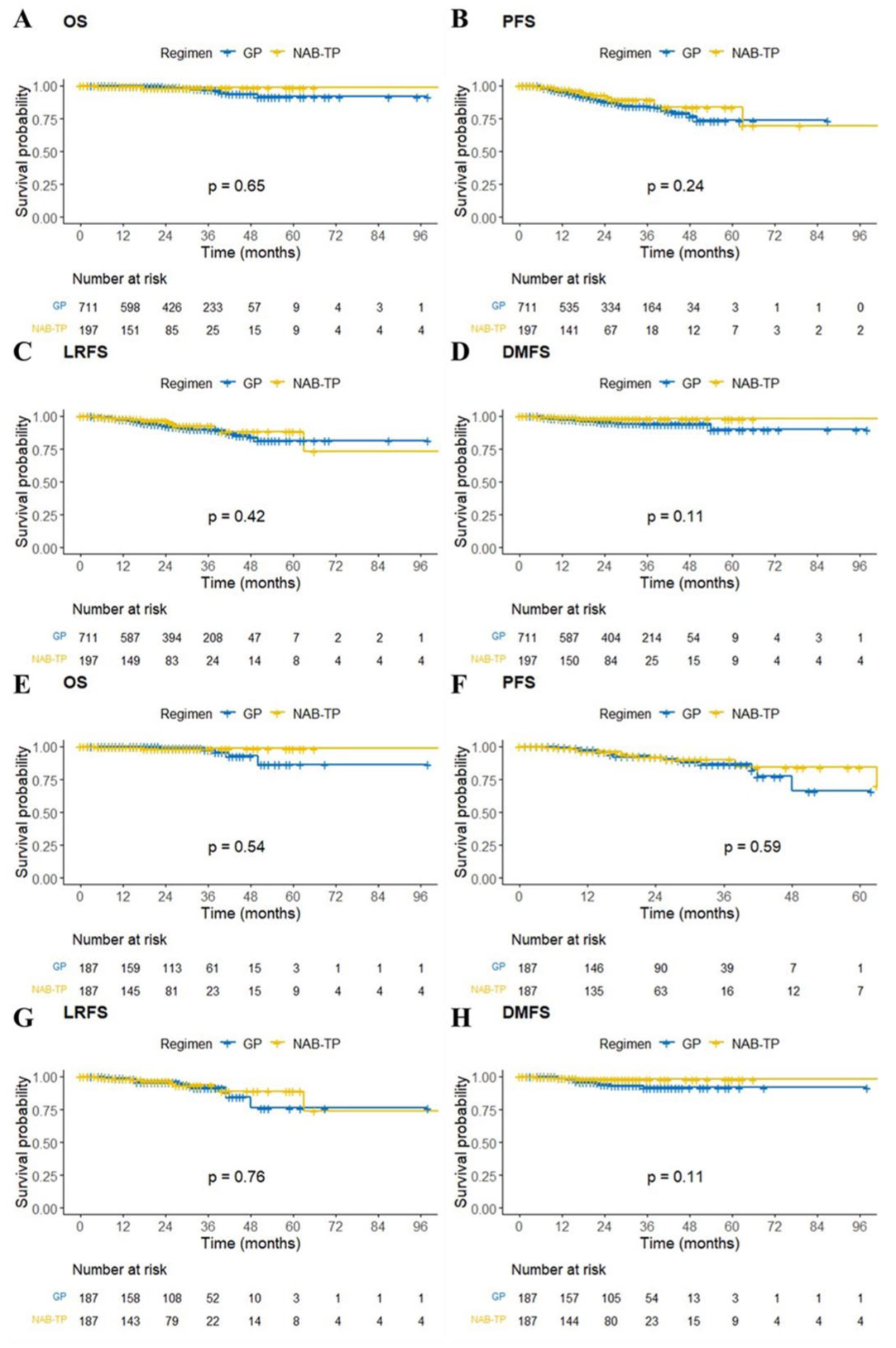
Kaplan–Meier curves of OS (a, e), PFS (b, f), LRFS (c, g), and DMFS (d, h) for LA-NPC patients receiving GP versus NAB-TP before (a–d) and after (e–h) PSM analysis.
Survival outcomes of the patients in the entire cohort, and matched before and after cohorts.

CI, confidence interval; DMFS, distant metastasis-free survival; GP, gemcitabine plus cisplatin; LRFS, locoregional relapse-free survival; NAB-TP, nanoparticle albumin-bound paclitaxel plus cisplatin; OS, overall survival; PFS, progression-free survival; PSM, propensity score matching.
Univariate and multivariate Cox analyses
Cox proportional hazards regression models were subsequently employed to identify various prognostic factors. Before PSM, univariate analysis revealed that age, N stage, clinical stage, and LDH level were significant potential prognostic factors for OS (Figure 3(a)). Age, N stage, clinical stage, smoking history, and EBV-DNA status were significant potential prognostic factors for PFS (Supplemental Figure 1(A)). Age and history of smoking were significant potential prognostic factors for LRFS (Supplemental Figure 2(A)). Sex, T stage, N stage, clinical stage, EBV-DNA, and LDH were significant potential prognostic factors for DMFS (Supplemental Figure 3(A)). After PSM, multivariate analysis revealed that N stage was a potential prognostic factor for OS (Figure 3(b)). Age, N stage, clinical stage, and family history of NPC were potential prognostic factors for PFS (Supplemental Figure 1(B)). Age, N stage, and family history of NPC were potential prognostic factors for LRFS (Supplemental Figure 2(B)). Age, N stage, clinical stage, and family history of NPC were potential prognostic factors for DMFS (Supplemental Figure 3(B)). Further multivariate analysis showed that clinical stage and LDH were independent prognostic factors for OS before PSM (Figure 3(c)). T stage and N stage were independent prognostic factors for DMFS before PSM (Supplemental Figure 3(C)). After PSM, family history of NPC was a common independent prognostic factor for PFS/LRFS/DMFS, age was a common independent prognostic factor for PFS/DMFS, and N stage and clinical stage were independent prognostic factors for LRFS and DMFS, respectively (Supplemental Figures 1(D), 2(D), and 3(D)).

Forest plots describing the univariate (a, b) and multivariate (c) associations of clinical and biochemical indicators with OS before (a, c) and after (b) PSM analysis.
Toxicity
Before PSM, retrospective assessment of patient toxicity revealed that, compared with the GP group, the NAB-TP group presented a significantly lower incidence of any-grade anemia (88.8% vs 94.5%, p = 0.005) and any-grade thrombocytopenia (9.1% vs 44.9%, p < 0.001). In addition, for grade 3–4 adverse reactions, the NAB-TP group presented reduced rates of anemia (3.0% vs 8.2%, p = 0.013) and thrombocytopenia (2.5% vs 8.6%, p = 0.004). No statistically significant differences were observed between the two groups in terms of the incidence of leukopenia, neutropenia, hepatotoxicity, or nephrotoxicity. After PSM, compared with the GP group, the NAB-TP group presented a significantly lower incidence of any-grade thrombocytopenia (9.6% vs 39.6%, p < 0.001). However, there was no significant difference in any-grade anemia (89.3% vs 94.7%, p = 0.057) between the two groups. In addition, for grade 3–4 adverse reactions, the NAB-TP group presented reduced rates of anemia (3.2% vs 8.0%, p = 0.043) and thrombocytopenia (2.7% vs 8.6%, p = 0.013). No statistically significant differences were observed between the two groups in terms of the incidence of leukopenia, neutropenia, hepatotoxicity, or nephrotoxicity (Table 3).
Maximum toxicity grade in patients treated with GP or NAB-TP after matching.

GP, gemcitabine plus cisplatin; NAB-TP, nanoparticle albumin-bound paclitaxel plus cisplatin; PSM, propensity score matching.
Discussion
To the best of our knowledge, this is the first study to compare the efficacy and safety of NAB-TP versus GP as IC followed by CCRT in LA-NPC, reporting data from the largest cohort to date, including 908 patients. Our data demonstrated that among LA-NPC patients, those receiving IC with NAB-TP followed by CCRT achieved comparable efficacy to those treated with the conventional GP regimen, and this finding was further confirmed by multivariate analysis. To control for potential confounders, PSM analysis was performed, which further confirmed the consistency of these results. Regarding treatment-related adverse events, the NAB-TP group exhibited a significantly lower incidence of anemia and thrombocytopenia. Therefore, NAB-TP may be considered a viable IC alternative for patients with LA-NPC.
Over the past two decades, the use of CCRT has reduced local recurrences in LA-NPC patients, yet distant metastasis remains a primary concern. Currently, IC followed by CCRT has become the standard of care for LA-NPC. Chemotherapy and intensity-modulated radiotherapy reduce local recurrence, whereas CCRT provides the highest benefit for OS and PFS. The addition of IC exerts the most significant effect on the control of distant metastasis. Compared with CCRT alone, incorporating IC has demonstrated survival benefits in multiple studies.8,19–23 Recent guidelines recommend over five distinct induction regimens for stage III–IVA NPC patients, though the optimal induction strategy remains unclear. Identifying the most effective IC regimen—balancing improved outcomes, minimized toxicity, and enhanced patient compliance—is a critical unresolved issue. While optimization studies for induction regimens exist,24–27 no head-to-head comparisons between GP and NAB-TP induction regimens are available for NPC. Recently, an individual patient data meta-analysis of 8214 patients suggested that taxane-based regimens yield superior OS benefits compared to non-taxane IC. 28 In other cancers, comparisons of these regimens have been explored. In first-line non-small cell lung cancer patients, an open-label phase II trial revealed that weekly NAB-TP exhibited comparable efficacy and tolerability to the GP regimen. 29 In our retrospective study, the NAB-TP induction group demonstrated a trend toward superior OS and DMFS compared with the GP group, albeit without statistical significance.
The GP regimen has been established as the standard treatment for LA-NPC, with its pivotal role recognized in international guidelines. Ma Jun’s phase III study demonstrated that, compared with CCRT alone, IC with the GP regimen followed by CCRT significantly improved the 3-year recurrence-free survival rate to 85.3% and OS to 94.6%. 22 However, we observed a significantly higher overall incidence of acute adverse events in patients receiving the GP regimen followed by CCRT than in those receiving CCRT alone. Notably, the GP group exhibited significantly higher rates of severe neutropenia, thrombocytopenia, anemia, nausea, and vomiting. Furthermore, since gemcitabine requires administration on Days 1 and 8 of each cycle, the GP regimen demands increased treatment adherence. As reported in the induction cisplatin-gemcitabine chemotherapy study, only 39% of patients completed all three cycles of concurrent chemotherapy. 22 A previous study also indicated that prolonged intervals between IC and RT might exacerbate disease progression, raising concerns about an increased risk of local recurrence. 30 The optimal IC regimen for LA-NPC is still being explored. A phase II clinical trial demonstrated that IC with TP followed by CCRT significantly improved 3-year OS compared with CCRT alone (94.1% vs 67.7%; hazard ratio = 0.24; 95% confidence interval (CI): 0.078–0.73; p = 0.012). 19 However, solvent-related hypersensitivity reactions and dose-limiting toxicities such as neuropathy during paclitaxel or docetaxel infusion limit its clinical utility. In contrast, NAB-paclitaxel employs nanotechnology to conjugate hydrophobic paclitaxel with human serum albumin carriers, offering advantages over solvent-based paclitaxel, including shorter infusion times for higher drug doses and elimination of premedication for hypersensitivity prevention. Preclinical xenograft models have shown that NAB-paclitaxel achieves higher mean maximum circulating and intratumoral free paclitaxel concentrations than sb-paclitaxel does. 31 A phase II study by Lv et al. reported 3-year PFS and cancer-specific survival rates of 86.1% (95% CI: 69.8%–99.8%) and 91.7% (95% CI: 68.9%–100.0%), respectively, for LA-NPC patients treated with NAB-TP induction followed by CCRT, indicating promising antitumor activity and manageable toxicity. 18 Consistent with the results of previous studies, 22 our retrospective matched cohort analysis revealed comparable 3-year OS (98.6% vs 97.5%) and PFS (90.2% vs 85.0%) rates between the NAB-TP and GP induction groups, further validating the satisfactory survival benefits of both regimens.
Before PSM, univariate and multivariate Cox analyses revealed that clinical stage and N stage were independent prognostic factors for OS and DMFS, respectively. After PSM, the clinical stage and N stage remained independent prognostic factors for DMFS and LRFS, respectively. In the univariate analysis of PFS, both the clinical stage and N stage were significantly different before and after PSM. However, multivariate analysis indicated that clinical stage was not an independent predictor of PFS. This observation may be attributable to the intrinsic correlation between disease stage and T/N classification. In multivariate modeling, the dominant effects of T/N stage likely diminished the prognostic impact of clinical stage on PFS. Nonetheless, the TNM staging system—grounded in anatomical principles—remains a critical biomarker for outcome prediction. Notably, we observed that neither induction regimen significantly affected patient prognosis, further confirming the comparable therapeutic efficacy of the two regimens.
Regarding treatment-related toxicity, acute toxicity during IC is primarily hematological, with these events being uncomplicated and readily manageable. Notably, in the matched cohort, the NAB-TP induction group demonstrated significantly lower incidences of anemia (grade 3–4) and thrombocytopenia (any-grade and grade 3–4) than the GP regimen. No statistically significant differences were observed in the rates of leukopenia, neutropenia, hepatotoxicity, or nephrotoxicity between the two groups. These findings suggest that the NAB-TP induction regimen achieves comparable survival benefits to the GP regimen while exhibiting a more favorable short-term toxicity profile. In clinical practice, the NAB-TP regimen offers greater convenience owing to its simplified dosing schedule; the Day 1 and Day 8 dosing requirements of gemcitabine in the GP regimen often lead to poor patient compliance or treatment delays due to unresolved myelosuppression, further highlighting the practical advantages of NAB-TP over GP as an IC.
This study has several limitations. First, as a retrospective analysis was conducted at a single center in an endemic region where undifferentiated non-keratinizing carcinoma predominates, validation in non-endemic areas is necessary to enhance broader applicability. Second, longer follow-up durations are required to confirm the findings presented. Third, as a real-world study, inherent heterogeneity and unavoidable selection bias exist. Finally, although adjusted Cox proportional hazards models and PSM mitigated certain biases, the potential influence of other unmeasured confounders remains unclear. Well-designed, comprehensive, multicenter, prospective, randomized controlled trials are warranted to conduct head-to-head comparisons of the efficacy and safety between NAB-TP and GP regimens in LA-NPC patients.
Conclusion
Our study demonstrated that NAB-TP, as an induction regimen before CCRT, exhibited comparable survival efficacy for LA-NPC patients compared with the GP regimen, with reduced acute toxicity. These findings provide a basis for prospective randomized trials and inform clinical decision-making regarding IC in patients with LA-NPC.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251400977 – Supplemental material for NAB-paclitaxel and cisplatin versus gemcitabine and cisplatin as induction chemotherapy in locoregionally advanced nasopharyngeal carcinoma: a retrospective, propensity score-matched analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359251400977 for NAB-paclitaxel and cisplatin versus gemcitabine and cisplatin as induction chemotherapy in locoregionally advanced nasopharyngeal carcinoma: a retrospective, propensity score-matched analysis by Qin Wang, Zimeng Wang, Qi Yang, Xiaotong Duan, Lan Peng, Rui You, Youping Liu, Tianliang Xia, Mingyuan Chen and Peiyu Huang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251400977 – Supplemental material for NAB-paclitaxel and cisplatin versus gemcitabine and cisplatin as induction chemotherapy in locoregionally advanced nasopharyngeal carcinoma: a retrospective, propensity score-matched analysis
Supplemental material, sj-docx-2-tam-10.1177_17588359251400977 for NAB-paclitaxel and cisplatin versus gemcitabine and cisplatin as induction chemotherapy in locoregionally advanced nasopharyngeal carcinoma: a retrospective, propensity score-matched analysis by Qin Wang, Zimeng Wang, Qi Yang, Xiaotong Duan, Lan Peng, Rui You, Youping Liu, Tianliang Xia, Mingyuan Chen and Peiyu Huang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
