Abstract
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) is a malignant tumor in the head and neck region that arises from epithelial cells of the nasopharynx and the global incidence is increasing to half a million and causing more than 34.1 million death every year. 1 It diagnosed an estimated 133 354 patients worldwide in 2020. 2 NPC global incidence rates are obvious differences in ethnic and geographical distribution. There are highest in Southeast Asia (especially in southern China), Micronesia/Polynesia, Eastern Asia, and North Africa. Rates are 2 to 3 times higher in men than in women.1,3 Radiotherapy (RT) is the main treatment for NPC. Intensity-modulated radiation therapy (IMRT) can deliver a highly conformed dose to targets while effectively sparing critical normal organs and has the potential to improve local control rate and reduce radiation-related toxicities. 4 Studies showed prolonged locoregional control interval, overall survival (OS), and progression-free survival (PFS) by CCRT,3,5–8 which has become the standard treatment for locoregionally advanced NPC (LA-NPC).
Although CCRT improves the locoregional control (LRC) of LA-NPC, distant metastasis is still the main mode of failure after treatment.9–11 Therefore, in order to ensure the curative effect, strengthened chemotherapy might be needed. Meta-analyses show that chemotherapy has an increasing trend in the comprehensive treatment of LA-NPC and intensified chemotherapy achieved higher survival benefits.6,7 So how to improve the intensity of chemotherapy is a hot topic of research on LA-NPC. There are many reports on induction chemotherapy (IC) and adjuvant chemotherapy (AC) for LA-NPC. Studies have shown that IC plays a significant role in the comprehensive treatment of LA-NPC,12–18 while CCRT plus AC has no more improvement compared with CCRT alone.19–21 The expert guideline panel concluded that cisplatin with docetaxel induction chemotherapy can be offered to patients with stage III-IVA NPC in the absence of any medical contraindications. 10 In addition to IC before RT or AC after RT, a third schema could be considered, that is to strengthen the concurrent chemotherapy. CCRT can enhance the sensitivity of RT and improve LRC 3 and OS of LA-NPC.7,8,20,22,23 Cisplatin-based CCRT has the significantly higher OS and PFS than RT alone in LA-NPC,24–27 and is the standard regimen of concurrent chemotherapy in patients with LA-NPC. However, it has well-known side effects, such as gastrointestinal reactions, nephrotoxicity, ototoxicity, and neurotoxicity.28,29 Due to the high incidence of side effects of cisplatin, some patients cannot tolerate cisplatin-based concurrent chemotherapy, especially for the elderly and patients with renal insufficiency. Docetaxel improves the LRC and OS in patients with squamous cell carcinoma of the head and neck,30–34 with mild gastrointestinal reaction, nephrotoxicity, and ototoxicity than cisplatin, and can be used as an active drug for LA-NPC. If double-drug concurrent chemotherapy can achieve higher survival benefits with acceptable toxicities in patients with LA-NPC, it is worthy of further application. In the present study, it was hypothesized that patients with LA-NPC treated with docetaxel single-drug CCRT might emerge as a new therapeutic approach with similar or higher survival benefits compared with cisplatin single-drug CCRT, especially in patients with contraindications to cisplatin. Therefore, a prospective control study on the feasibility of docetaxel single-drug and docetaxel plus cisplatin double-drug concurrent chemotherapy needs to be carried out. The objective of this trial is to evaluate the feasibility of concurrent helical tomotherapy with docetaxel plus cisplatin, docetaxel, and cisplatin in patients with LA-NPC.
Materials and Methods
Trial Design and Participants
This study was a prospective, single-center, open-label, phase II, randomized trial conducted in the Department of Radiation Oncology, of Chinese People's Liberation Army (PLA) General Hospital, which was approved by the ethics committee of Chinese PLA General Hospital.
Eligibility criteria included the following: age between 18 and 70 years; histologic confirmation of keratinizing squamous cell carcinoma, nonkeratinizing differentiated carcinoma, and nonkeratinizing undifferentiated carcinoma that was according to the 4th edition of the World Health Organization (WHO) classification of head and neck tumors; newly diagnosed stage III to IVA disease that was according to the American Joint Committee on Cancer (AJCC)–Union for International Cancer Control (UICC) 8th edition stage-classification system; a Karnofsky performance-status (KPS) score of at least 70; previously untreated, and adequate hematologic, hepatic, and renal function. Exclusion criteria included treatment with palliative intent, a history of previous RT, chemotherapy, or surgery (except diagnostic procedures) to the primary tumor or nodes, previous malignancy, pregnancy or lactation, or any severe coexisting disease. All patients had complete history and experienced physical examination, hematologic and biochemical analyses, nasopharyngeal and skull base magnetic resonance imaging (MRI), endoscopic evaluation, chest computed tomography (CT), neck and abdomen ultrasound, and bone scanning. Positron emission tomography (PET) was optional. Written informed consent was obtained from all the patients before enrollment. Patients could withdraw consent at any time after enrollment and could discontinue the trial if disease progression or severe coexisting conditions occurred during treatment. The reporting of this study conforms to the CONSORT statements. 35
Randomization and Masking
The randomization procedure was carried out by Dr Lei Du. SPSS software was used to generate a list of 150 random numbers. Randomization was stratified after the patients were diagnosed as NPC with a stage of III or IVA. Eligible patients were randomly assigned (1:1:1) to receive two cycles of IC with docetaxel plus cisplatin followed by helical tomotherapy concurrently with docetaxel plus cisplatin (docetaxel plus cisplatin CCRT group, DP group), docetaxel (docetaxel CCRT group, D group), and cisplatin (cisplatin CCRT group, P group). The treatment group assignment was not masked.
Chemotherapy
For IC, docetaxel plus cisplatin was administered as 70 mg/m2 docetaxel intravenously on day 1 and 80 mg/m2 cisplatin intravenously on days 1 to 2, 3 cycles were administered at intervals of 3 weeks. For CCRT, in the DP group, 70 mg/m2 docetaxel and 80 mg/m2 cisplatin were administered intravenously every 3 weeks on days 1 to 2, 22 to 23, and 43 to 44 concurrently with RT; in the D group, 70 mg/m2 docetaxel was administered intravenously every 3 weeks on days 1, 22, and 43 concurrently with RT; in the P group, 80 mg/m2 cisplatin was administered intravenously every 3 weeks on days 1, 22, and 43 concurrently with RT. During IC and CCRT phases, dose modifications of chemotherapy were dependent on the hematological and nonhematological toxicities from the previous cycle. Dose modifications of docetaxel were planned for grade 3 or higher neutropenia, leukopenia, anemia, thrombocytopenia, pulmonary toxicity, and impaired liver function. The docetaxel dose was reduced by one level (14 mg/m2) for the first episode of the above toxicities and by two-level (28 mg/m2) for the second episode. Dose modifications of cisplatin were planned for grade 3 or higher gastrointestinal reactions, nephrotoxicity, ototoxicity, and neurotoxicity. The cisplatin dose was reduced by one level (16 mg/m2) for the first episode of the above toxicities and by two-level (32 mg/m2) for the second episode. Patients with delayed recovery from nonhematologic toxicities for more than 3 weeks was allowed to delay chemotherapy. The criteria of stop chemotherapy included the following: patients with delayed recovery from any grade 3 or higher acute toxicities except alopecia for more than 3 weeks; patients had grade 3 or higher nerve toxicities. To prevent chemotherapy-induced nausea and vomiting, we allowed the use of 5-HT3-receptor antagonists (ondansetrone or granisetron), dexamethasone, NK-1-receptor antagonist aprepitant, and metoclopramide. We allowed prophylactic granulocyte colony-stimulating factor (G-CSF) in patients with grade 3 or higher neutropenia during the preceding cycle.
Radiotherapy
All eligible patients were irradiated with helical tomotherapy as described in detail in our previous studies. 36 Plain and enhanced CT images with 3-mm slice thickness were taken for treatment planning and then transmitted to the Pinnacle3 8.0 workstation and fused. The pre-treatment enhanced CT, MRI, or PET/CT images were used as a guide for target contours. According to ICRU 50, 62, and 83 reports, the gross target volume of local tumor (GTVnx) and regional lymph nodes (GTVnd) were, respectively, defined as the visible tumor and involved nodes. The pGTVnx was obtained by expanding the corresponding GTVnx with a margin of 3 mm while limited by the brainstem, spinal cord, optic chiasma, and optic nerve. The pGTVnd was the GTVnd with an expansion of 3 mm. Clinical target volume 1 (CTV1) covered nasopharynx, high-risk local structures (ie, skull base, clivus, parapharyngeal space, retropharyngeal lymph nodes, sphenoid sinus, pterygopalatine fossa, posterior part of the nasal cavity and maxillary sinus, and oropharynx), as well as positive lymph nodes and nodes at level IB (when nodes at level IIA were involved), level II and superior part of VA. Clinical target volume 2 (CTV2) included lymph nodes at level Ш, IV, VB, and inferior part of VA as a prophylactic irradiated volume. Planning target volume1 (PTV1) and 2 (PTV2) were generated with a 3 mm margin of CTV1 and CTV2 at least 3 mm from the skin. The prescription doses were 67.5 Gy (2.25 Gy per fraction) to pGTVnx and pGTVnd, 60 Gy (2 Gy per fraction) to PTV1, and 54 Gy (1.8 Gy per fraction) to PTV2 in 30 fractions. CCRT started within 21 to 28 days after the first day of the second cycle of IC. The correction has been made in the revised version. CCRT started within 21 to 28 days after the first day of the second cycle of IC.
Assessment of Outcomes
Clinical efficacy was assessed 16 weeks after CCRT, according to the Response Evaluation Criteria in Solid Tumors (RECIST), version 1.1. 37 Toxic effects were assessed weekly during IC and CCRT and at subsequent predefined intervals. Acute toxicities were graded according to the established criteria of the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) and Radiation Therapy Oncology Group and the European Organization for Research and Treatment of Cancer (RTOG/EORTC). 38 Patients were followed up every 3 months in the first year, every 4 months in the second and third years, every 6 months in the fourth and fifth years, and then 12 months until progression or death. All the end points were assessed or confirmed by the physician in charge.
Endpoints
The primary end point was the incidence of grade 3 or 4 acute toxicity, which was investigated weekly and peak toxicities were recorded from the first day of IC to 30 days after CCRT. Secondary end points included clinical efficacy, OS, PFS, locoregional failure-free survival (LRFFS), and distant failure-free survival (DFFS). The changes in tumor size were measured via electronic nasopharyngoscopy and MRI of the nasopharyngeal and neck areas at 16 weeks after CCRT. The clinical efficacy was evaluated as complete remission (CR), partial remission (PR), stable disease (SD), and progressive disease (PD). Objective response rate (ORR) = (CR + PR)/the total × 100%, and disease control rate (DCR) = (CR + PR + SD)/the total × 100%. OS was calculated from the date of randomization to death; moreover, PFS was calculated from the date of randomization to the date of locoregional recurrence, distant metastasis, or death from any cause, whichever occurred first; LRFFS and DFFS were calculated from the date of randomization to the date of first locoregional and distant failure, respectively.
Statistical Analyses
This trial aimed to examine the acute toxicity of 3 concurrent chemotherapy regimens. Wen et al 39 reported that the incidence of grade 3 to 4 nausea/vomiting in S-1 concurrent chemoradiotherapy and cisplatin concurrent chemoradiotherapy was 9.1% and 40.0%, respectively. According to the guidelines for the prevention and treatment of vomiting caused by antitumor therapies, 40 docetaxel and tegafur (the main component of S-1) are chemotherapeutic drugs with a low risk of emesis (the incidence of vomiting is 10%-30%). Xie et al 41 reported that the incidence of grade 3 to 4 leukopenia in cisplatin concurrent chemoradiotherapy and docetaxel plus cisplatin concurrent chemoradiotherapy was 12.0% and 60.7%, respectively. Therefore, this trial assumed that the incidence of grade 3 or above acute toxicity was 40% in the docetaxel group, 52% in the cisplatin group, and 70% in the docetaxel plus cisplatin group. We estimated that the trial would have 80% power with a two-sided significance level of .05. Approximately 135 patients were required to undergo randomization (45 patients per group). We further assumed that 10% of the patients would be lost to follow-up or would prematurely discontinue the trial. This yielded a final sample size of 150 (50 patients per group for 3 groups).
Analysis was based on intention to treat. The classification variables were described by the number of cases and percentage, and the comparison between the 2 groups and the 3 groups was analyzed by chi-square test or Fisher exact test and Bonferroni correction. Continuous variables were described by mean ± standard deviation or median and quartile according to their normal distribution or skew distribution. Independent sample t-test, paired t-test, or rank-sum test were used to compare the 2 groups, and one-way ANOVA or Kruskal Wallis h-test were used to compare the 3 groups. A chi-square test was used to analyze the differences between groups. Survival rates were assessed using the Kaplan-Meier method. The Log-rank test and the Cox proportional hazards model were used to identify prognostic factors independently associated with survival and to estimate hazard ratios (HRs). Analyses were conducted with the use of SPSS software, version 26.0 (Armonk, NY: IBM Corp). A two-tailed P-value of less than .05 was considered as statistically significant.
Results
Patient Characteristics
From June 2017 to November 2019, 125 newly diagnosed LA-NPC patients were enrolled in this study. All patients were treated with two cycles of IC followed by CCRT.
This study was based on 150 patients randomized in a ratio of 1:1:1 (50 patients per group). SPSS software was used to generate a list of 150 random numbers. Patients were enrolled in the study and assigned to the treatment groups in chronological order according to the computer-generated list. However, the investigator found that the incidence of acute toxicities in the DP group was serious and the chemotherapy plans were delayed. The preliminary analysis following the advice from clinicians was conducted when the recruited patients were up to 25 cases or more. A total of 77 patients were enrolled and randomized into 3 groups: DP group (n = 25), D group (n = 27), and P group (n = 25). In the course of CCRT, 88.0% (22/25) of patients in the DP group, 63.0% (17/27) in the D group, and 52.0% (13/25) in the P group experienced grade 3 or 4 toxicities (P = .020). The incidence of grade 3 or 4 acute toxicities in the DP group was significantly higher than that in the D group (P = .037) and the P group (P = .012). The clinical ethics committee decided that patient recruitment into the DP group was terminated according to ethical principles. We believed that the prematurely terminated recruitment in the DP group would not affect the accuracy of experimental results. Eligible patients continued to be enrolled in the D and P groups in chronological order according to the computer-generated list after the preliminary analysis.
Finally, 125 patients with LA-NPC were assigned to DP (n = 25), D (n = 50), and P (n = 50) groups. All patients completed safety assessment, and there was no case of loss of follow-up. In all, 125/125 (100%) patients were treated with 2 cycles of IC with docetaxel plus cisplatin, and 124/125 (99.2%) completed protocol-defined RT. Only one patient in the D group terminated RT early due to grade 3 pharyngo-esophageal toxicity. During CCRT, 66.4% (83/125) patients completed 3 cycles, 24.0% (30/125) completed 2 cycles, and 9.6% (12/125) completed one cycle of concurrent chemotherapy according to the research plan; 42/125 (33.6%) patients discontinued chemotherapy ahead of schedule, of which 34 patients had grade 3 or 4 acute toxicities, and 8 patients withdrew their consent (Figure 1). The characteristics of the patients at baseline were well balanced among the 3 groups (Table 1).

Enrollment, randomization, and follow-up.
Characteristics of Patients.
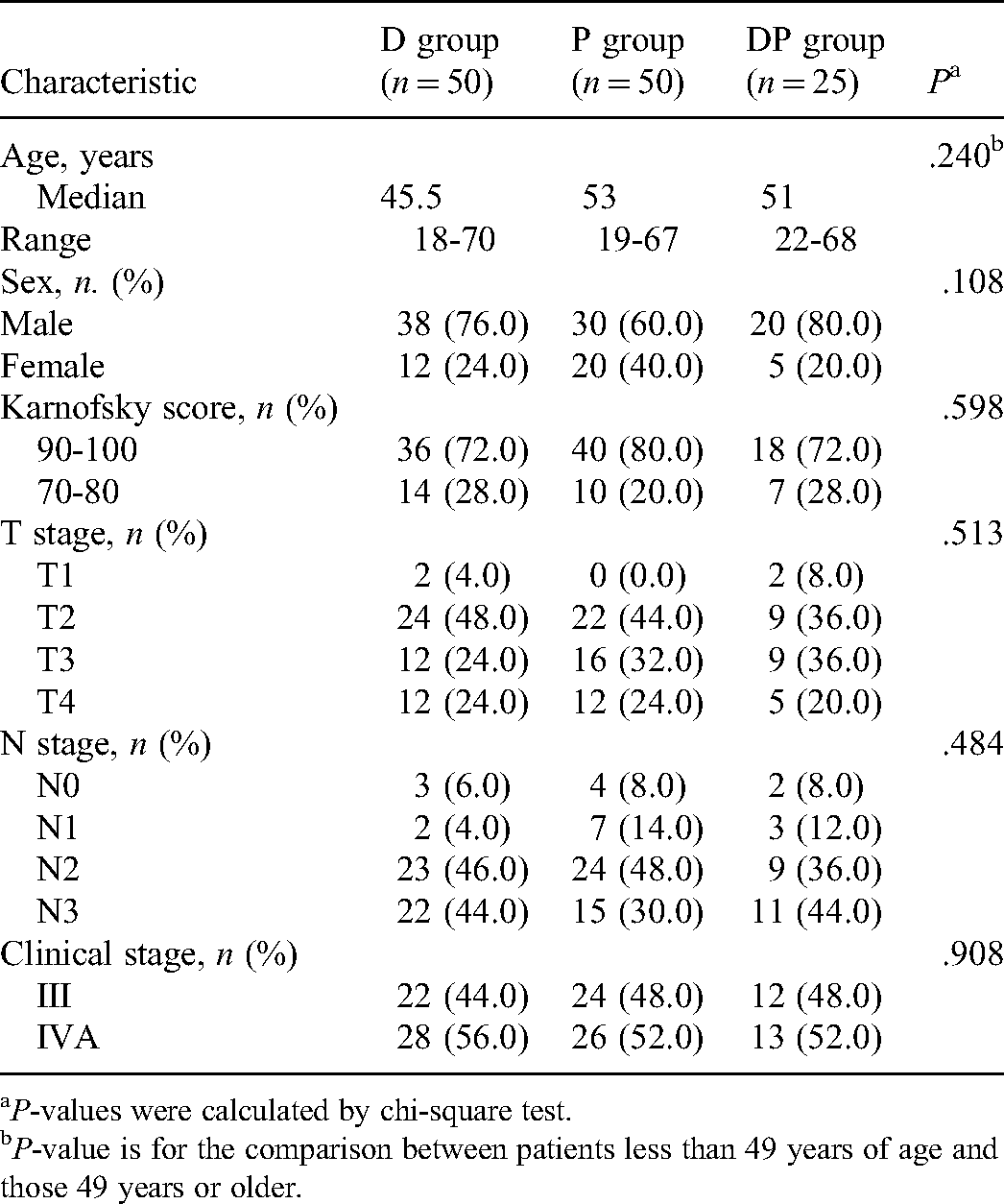
P-values were calculated by chi-square test.
P-value is for the comparison between patients less than 49 years of age and those 49 years or older.
Acute Toxicities
During IC, 17/125 (13.6%) patients had grade 3 or 4 acute toxicities. Neutropenia was the most common toxicity (15/125, 12.0%). During CCRT, 22/25 (88.0%) patients in DP group, 36/50 (72.0%) in D group, and 28/50 (56.0%) in P group had grade 3 or 4 acute toxicities. Overall, 86/125 (68.8%) patients had grade 3 or 4 acute toxicities. The proportion of patients with grade 3 or 4 acute toxicities was significantly different among the 3 groups (P = .015), while no significant difference was detected between the D and P groups (72.0% vs 56.0%, P = .096) (Table 2). Mucositis was the most common toxicity (50/125, 40.0%), followed by leukopenia (37/125, 29.6%), neutropenia (33/125, 26.4%), and pharyngo-esophagitis (15/125, 12.0%). The incidence of grade 3 or 4 leukopenia and neutropenia was higher in the D group (48.0% and 42.0%) and the DP group (36.0% and 32.0%) than that in the P group (8.0% and 8.0%) (P < .001). The incidence of grade 3 or 4 vomitings was higher in the DP group (28.0%) than that in the P group (8.0%, P = .035), and no one in the D group (P < .001).
Grade 3 or 4 Acute Toxicity of the 3 Groups.

P-values were calculated using the chi-square test (or Fisher's exact test).
P-value for the 3 groups.
P-value for D and P groups.
Short-Term Outcomes and Survival Analysis
Forty-eight (38.4%) (18 patients in D group, 22 patients in P group, and 8 patients in DP group) of the 125 patients had achieved CR, 69 (55.2%) (29 in D group, 25 in P group, and 15 in DP group) achieved PR, and 7 (5.6%) (3 in D group, 2 in P group, and 2 in DP group) achieved SD, considering the primary tumor and regional nodes together. Only one patient had PD (bone metastasis) in the P group. The whole effective rate was 93.6% (117/125), with an ORR of 94.0% (47/50), 94.0% (47/50), and 92.0% (23/25) in the D group, P group, and DP group; respectively, without significant difference among the 3 groups (P = 1) (Table 3).
Clinical Outcomes of the 3 Groups.

Abbreviations: CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease; ORR, objective response rate; DCR, disease control rate.
P-values were calculated using the chi-square test (or Fisher's exact test).
P-value for the 3 groups.
P-value for D and P groups.
The final date of data collection was January 31, 2022, with a median follow-up of 38 (range, 10-53) months. In all, 25/125 (20.0%) patients experienced treatment failure or died during the follow-up, including 6 local recurrences, 3 regional recurrences, and 10 distant metastases. Fourteen patients (14/14, 100%) died of NPC, and among them, 4 experienced hemorrhages and 10 died from systemic failure after salvage treatment (Table 4). The 3-year OS was 87.0% (44/50), 87.6% (44/50), and 96.0% (24/25) in the D group, P group, and DP group, respectively, without significant difference among the 3 groups (P = .800). The 3-year PFS was 79.7%, 76.9%, and 92.0%, LRRFS was 91.8%, 92.7%, and 96.0%, DFFS was 90.0%, 89.0%, and 100%, respectively, without significant difference among the 3 groups either (P = .532,.824, and.265; respectively, Figure 2).
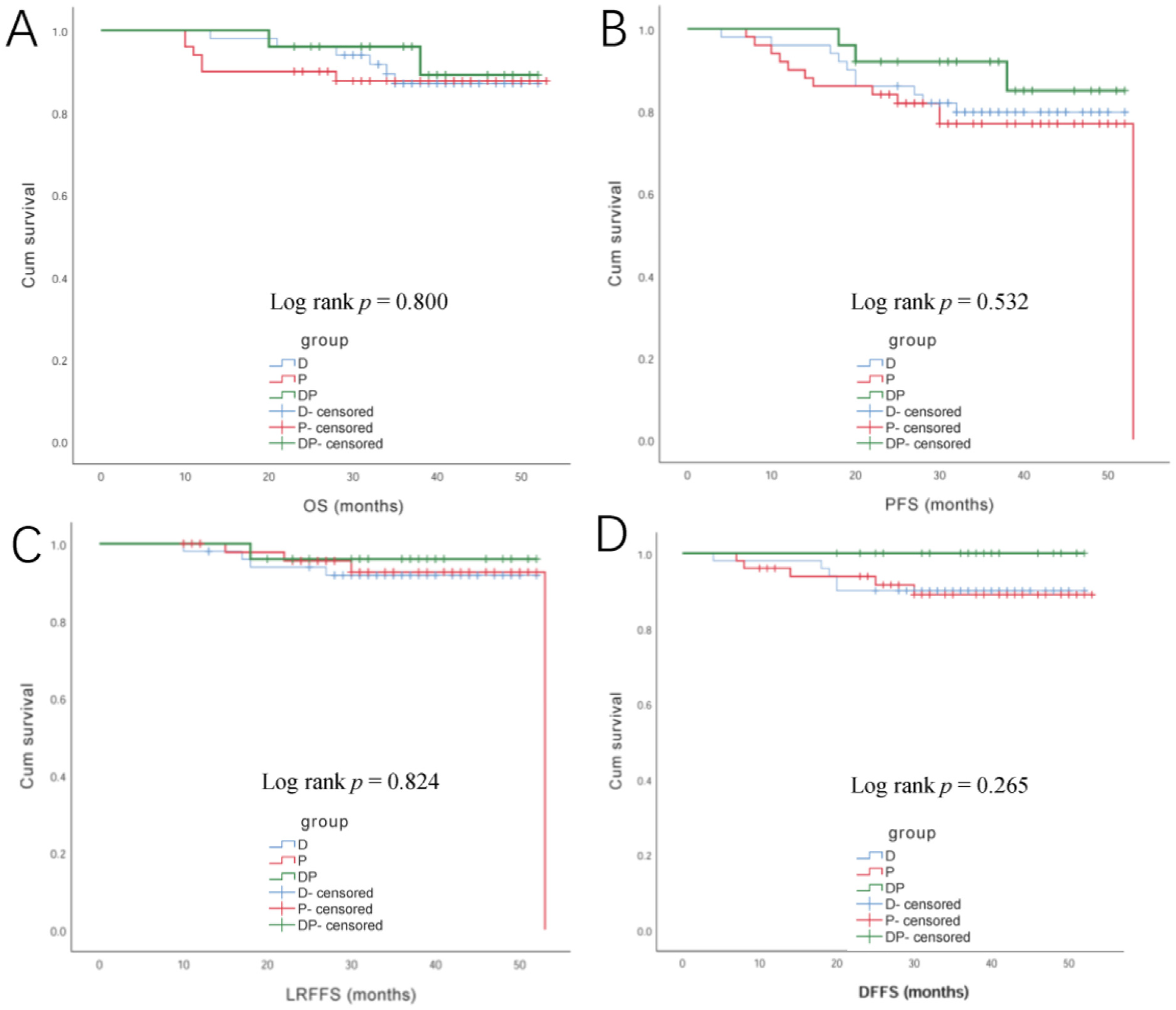
Kaplan-Meier curves for OS (A), PFS (B), LRFFS (C), and DFFS (D) of patients in the D group, P group, and DP group.
Patterns of Treatment Failure.

P-values were calculated using the chi-square test (or Fisher's exact test).
P-value for the 3 groups;
P-value for D and P groups.
Systemic failure: patients died from systemic failure after primary or salvage treatment.
Univariate analysis showed that age was an independent factor for 3-year PFS (P = .039) and OS (P = .003). Multivariate analysis was performed with Cox proportional hazard model. Age (< 49 vs ≥ 49 years) was the main factor affecting the 3-year PFS and OS (P = .046 and .016, respectively).
Discussion
According to a historical overview, NPC is a highly radiosensitive and chemosensitive tumor type. Due to its anatomic location, in close vicinity to critical structures, RT is the mainstay of treatment for the nonmetastatic disease. Chemotherapy is generally considered except for patients with stage I disease.10,42 Numerous studies reported the superiority of CCRT for tumor control and survival in patients with LA-NPC.6,7,22,23,43–45 The landmark MAC-NPC meta-analysis confirmed that the addition of chemotherapy to RT significantly improved OS in nonmetastatic stage II-IV NPC (HR 0.79; P < .001). 7 IC has received OS benefit when added to CCRT in several trials.12,16,46 In a phase II study, patients with stage III-IVB NPC (according to the 1997 classification from the Union for International Cancer Control) were randomly allocated to receive CCRT with or without 2 cycles of induction docetaxel and cisplatin. 46 The 3-y OS was significantly superior in the induction arm (HR 0.24; P = .012). At present, the NCCN guidelines recommend IC followed by CCRT as level 2A evidence, and CCRT alone as level 2B evidence for stage II-IV NPC. 3 IC + CCRT has emerged as the core of treatment for LA-NPC according to the CSCO and ASCO guidelines. 47 Distant metastasis is the main failure mode of NPC after CCRT,9,11,48 and is the main cause of tumor-related death of NPC.11,43,49 Strengthening the comprehensive treatment for patients with NPC is an important way to improve the curative effect and reduce the long-term mortality, among which chemotherapy is the key link to improve the curative effect.19,22,50–52 Therefore, the treatment of LA-NPC needs strong enough chemotherapy to ensure the curative effect. Three meta-analyses showed that the combination of chemotherapy with RT improved survival and that CCRT was more effective than IC or AC.8,44,53 The same as in addition to increasing IC or AC, it is meaningful to try double-drugs-based CCRT on the basis of single-drug-based CCRT. However, the addition of concurrent chemotherapy seems to increase the risk of severe late toxicities in patients with NPC, especially for high-dose cisplatin regimen which causes severe ototoxicity. 54 Therefore, a concurrent chemotherapy regimen should be carefully considered for patients with LA-NPC. We carried out this randomized phase II trial study in order to compare the toxicities and survival outcomes of 3 current chemotherapy regimens including docetaxel (D), cisplatin (P), and docetaxel plus cisplatin (DP). We noticed that the incidence of grade 3 or 4 acute toxicities in DP group was 88.0%, which was significantly higher than that of the D group (63.0%) and the P group (52.0%) (P = .015). However, there was no significant difference in the incidence of grade 3 or 4 acute toxicity between the D and P groups (P = .096).
It is pointed out that the acute toxic effects of multidrug-based CCRT are more serious than that of single-drug-based CCRT. According to the study of Komatsu et al, 55 which consisted of 2 cycles of concurrent chemotherapy with paclitaxel, cisplatin, and 5-FU during definitive RT, once every 3 weeks. Because of serious toxic and side effects, only 17 cases (17/24, 70.8%) completed the scheduled CCRT. The second course of chemotherapy was discontinued in 7 cases. The most common causes of chemotherapy discontinuation were myelosuppression (4/7, 57.1%) and renal dysfunction (2/7, 28.6%). At present, three-drug-based CCRT is rarely used in LA-NPC. Nakahara et al 56 reported the result of weekly low-dose docetaxel (5-10 mg/m2) and cisplatin (20 mg/m2) CCRT (up to 6 cycles) for patients with LA-NPC. Twenty-eight patients (28/31, 90.0%) completed CCRT as planned. Grade 3 or 4 mucositides occurred in 16 (51.6%) patients. Grade 3 or higher acute hematologic toxicity (except lymphopenia) was uncommon. Grade 3 or higher leukopenia only occurred in 2 (6.5%) patients. In our study, the completion rate of concurrent chemotherapy in the DP group was lower than that in the above study, 17 patients (17/25, 68.0%) completed 2 cycles of concurrent chemotherapy as planned. Grade 3 or 4 mucositides of CCRT occurred in 15 (60.0%) patients, which was similar to previous reports. However, grade 3 or 4 leukopenia and neutropenia of concurrent chemotherapy with docetaxel (70 mg/m2) and cisplatin (80 mg/m2) every 3 weeks occurred in 9 (36.0%) and 8 (32.0%) patients, higher than those of previous reports. And grade 3 or 4 acute toxicity in the DP group was significantly more serious than that of the P group (88.0% vs 52.0%, P = .006), without significant improvement of 3-year OS (96.0% vs 87.6%, P = .543).
This study indicates that DP concurrent chemotherapy appears to increase the incidence of grade 3 or 4 acute toxicities over D and P single-drug regimen, without significant improvement of 3-year OS, PFS, LRFFS, and DFFS. Single-agent regimen with docetaxel and cisplatin had a similar incidence of grade 3 or 4 acute toxicities and 3-year OS, PFS, LRFFS, and DFFS. The 3-year OS and PFS of the DP group were up to 96.0% and 92.0%, which were higher than those of the other 2 single-drug groups (87.0% and 79.7% in the D group, and 87.6% and 76.9% in the P group, respectively). Although no statistically significant difference was detected among the 3 groups, prolonged follow-up is necessary to assess the long-term outcome.
The incidence of serious clinical events (such as hospitalization, death, etc) and treatment delay caused by leukopenia and/or neutropenia is lower than in the past. Therefore, docetaxel single-drug CCRT may be a feasible treatment for patients with LA-NPC contraindicated by cisplatin. However, cumulative evidence on docetaxel single-drug CCRT for LA-NPC remains inadequate for changing the standard treatment, and much larger randomized studies are needed to investigate the validity of docetaxel single-drug CCRT.
This study found that grade 3 or 4 oral mucositis (50/125, 40.0%), pharyngo-esophagitis (15/125, 12.0%), leukopenia (37/125, 29.6%), and neutropenia (33/125, 26.4%) were still common acute toxicities, in the course of CCRT, as our team found in previous studies. Oral mucositis and pharyngo-esophagitis appeared around the 10th fraction of RT. The most severe oral mucositis and pharyngo-esophagitis occurred during the 20th to 25th fraction and then gradually relieved. 36 Our results contrast with those from similar studies conducted by Zhang et al 57 and Chen et al 23 The most serious acute toxicity was mucositis during CCRT. We need to pay attention to the prevention and treatment of oral mucositis. At present, there are few clinical treatment guidelines and effective drugs in this field. Therefore, it is necessary to further study the prevention and treatment of oral mucositis.
In this study, the incidence of grade 3 or 4 acute toxicities in the D group and P group had no difference (72.0% vs 56.0%, P = .096), and the 3-year OS was the same (87.0% vs 87.6%, P = .750). The incidence of grade 3 or 4 leukopenia and neutropenia in the D group was 48.0% and 42.0%, respectively. No grade 3 or higher vomiting occurred. In the P group, the incidence of grade 3 or 4 leukopenia and neutropenia was only 8.0% and 8.0%, respectively, but grade 3 or higher vomiting occurred in 4 patients (8.0%). It appears that the types of severe toxicity were different in the 2 groups. During chemotherapy, the clinical application of G-CSF in patients with LA-NPC can actively prevent and improve leukopenia and/or neutropenia.
In the present study, several factors have to be considered. First of all, due to the high incidence of severe acute toxicities in the DP group, the patient recruitment was terminated in the middle of the study, which may have partly influenced the results. Second, we only analyzed the 3-year OS, PFS, LRFFS, and DFFS, longer follow-up would be needed. Finally, the conclusion reached in this study needs to be further verified through a large-sample multi-center randomized study, so as to provide more potent bases for the options of regimens in treating patients with LA-NPC.
Conclusions
Docetaxel plus cisplatin CCRT had a higher incidence of acute toxicities in patients with LA-NPC, with similar 3-year OS, PFS, LRRFS, and DFFS compared with docetaxel and cisplatin single-drug CCRT. Single-drug docetaxel used concurrently with helical tomotherapy had similar acute toxicity and short-term clinical outcome compared with cisplatin CCRT in patients with LA-NPC. More prospective randomized controlled clinical trials are needed to confirm this conclusion.
Footnotes
Abbreviations
Authors’ Note
Yanrong Luo, Boning Cai, and Bo Li contributed equally to this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The trial was approved and consented by the research ethics committee of the Chinese PLA General Hospital (S2017-030-02), and written informed consent was obtained for all patients.
Trial Registration
This trial was registered at ClinicalTrials.gov with a registration code of NCT03177174.
