Abstract
Background:
CYP24A1 polymorphisms may affect predisposition of cancer, but the results of published studies remain inconclusive. Therefore, the authors conducted this meta-analysis to more robustly assess relationships between CYP24A1 polymorphisms and the predisposition of cancer by pooling the findings of published studies.
Materials and methods:
A comprehensive literature search of PubMed, Embase, Web of Science, Wanfang, and CNKI was endorsed by the authors to identify eligible studies; 17 studies were finally found to be eligible for pooled meta-analysis.
Results:
The pooled meta-analysis results showed that genotypic frequencies of the rs4809960 polymorphism among cancerous patients and controls of Caucasian ethnicity differed significantly, and genotypic frequencies of the rs6022999 polymorphism among cancerous patients and controls of Asian ethnicity also differed significantly. Moreover, we found that genotypic frequencies of the rs2585428 polymorphism among patients with prostate cancer and controls differed significantly, and genotypic frequencies of the rs6068816 polymorphism among patients with prostate cancer/breast cancer and controls also differed significantly.
Conclusions:
This meta-analysis suggests that the rs4809960 polymorphism may affect the predisposition of cancer in Caucasians, and the rs6022999 polymorphism may affect the predisposition of cancer in Asians. Moreover, the rs2585428 polymorphism may affect the predisposition of prostate cancer, while the rs6068816 polymorphism may affect the predisposition of prostate cancer and breast cancer.
Keywords
Introduction
Cancer is the second most common cause of death worldwide.1-2 Although its definite pathogenesis mechanisms are still unclear, it is well established that genetic factors play a role in cancer risk and development. First, compared with those without a family history of the disease, the odds of developing cancer are significantly increased in those with a positive family history in first-degree relatives,3-8 and genetic background is definitely one of the major contributing factors of this family aggregation phenomenon. Second, previous genetic association studies have also detected many susceptibility genes for different types of cancers, such as BRCA1 and BRCA2 for breast cancer, RTEL1 and TP53 for lung cancer, and MYC for gastric cancer.9-12 Nevertheless, genetic contributing factors of cancer are still not fully understood, and thus warrant intensive exploration.
It is thought that the vitamin D metabolic pathway may be involved in the onset and progression of cancer. First, previous epidemiological studies have shown that a higher vitamin D intake is associated with a reduced risk of multiple types of cancers, and serum levels of vitamin D have been found to be inversely associated with disease severity.13-15 Second, many pre-clinical studies have demonstrated that vitamin D has immune-regulatory, anti-proliferative, and anti-angiogenic functions, which may provide favorable protection effects against cancer onset and development.16-17
CYP24A1 serves as the core degradation enzyme of vitamin D, and a higher mRNA expression level of CYP24A1 has been detected in many types of cancers.18-22 Consequently, it is biologically plausible that CYP24A1 polymorphisms may also influence the predisposition of cancer. In the last two decades, investigators all over the world have repeatedly tried to explore relationships between CYP24A1 polymorphisms and the predisposition of cancer, with inconsistent findings. Therefore, the authors conducted this meta-analysis to more robustly analyze relationships between CYP24A1 polymorphisms and the predisposition of cancer by pooling the results of eligible publications.
Materials and methods
The authors strictly followed the PRISMA guidelines when conducting this meta-analysis.
23
We also registered an Open Science Framework account for this meta-analysis (https://osf.io, username:
Literature search and inclusion criteria
A comprehensive literature search of PubMed, Web of Science, Embase, Wanfang and CNKI was conducted by the authors using the following keywords: CYP24A1 and (polymorphism or variant or variation or mutation or SNP or genome-wide association study or genetic association study or genotype or allele) and (cancer or tumor or carcinoma or neoplasm or malignancy). The authors also manually searched the references of retrieved publications to make up for the potential incompleteness of electronic literature searching.
The selection criteria of eligible studies were: (a) case-control or cohort studies; (b) investigated relationships between CYP24A1 polymorphisms and the predisposition of cancer; (c) studies where there was no strict selection criteria regarding how the results were presented, where adjusted or unadjusted results were both acceptable, but raw genotypic frequencies of CYP24A1 polymorphisms in patients with cancer and control subjects had to have been reported; and (d) the full manuscript was retrievable. Articles were excluded if one of the following three criteria was met: (a) not about CYP24A1 polymorphisms and the predisposition of cancer; (b) narrative reviews, systematic reviews, meta-analyses or comments; and (c) case series that only involved participants with cancer. If duplicate publications were retrieved from the literature search, only the study with the largest sample size would be included for pooled meta-analysis.
Data extraction and quality assessment
The authors extracted the following data from eligible studies: (a) the last name of the first author; (b) the publication year; (c) the country and ethnicity of the study population; (d) the number of patients and controls; (e) the genotypic frequencies of CYP24A1 polymorphisms in patients and controls as well as the P values of Hardy-Weinberg equilibrium (HWE) test (the threshold of HWE deviation was set at 0.05; if P > 0.05, then we considered that the genotypic distribution of the investigated polymorphism was in agreement with HWE); (f) the age and gender distributions of the study subjects; (g) the source of controls in eligible studies; and (h) the genotyping method used in the eligible studies.
The authors used the Newcastle-Ottawa scale (NOS) to assess the quality of eligible studies. 24 The score range of NOS was from 0 to 9, and the methodology quality of a study was considered to be good if it cannot achieve a score higher than 7.
Data extraction and quality assessment of the eligible studies were performed separately by two authors. In case of any discrepancy between the two authors, it was agreed that a thorough discussion would be undertaken until a consensus was reached. The authors would write to the corresponding authors of eligible studies for additional information if we failed to extract all necessary data from the manuscripts of included studies.
Statistical analyses
All statistical analyses in this meta-analysis were performed with Review Manager Relationships between CYP24A1 polymorphisms and the predisposition of cancer were estimated by using odds ratio (OR) and its 95% confidence interval (CI). The statistically significant P value was set at 0.05. I 2 statistics were used to determine whether significant heterogeneities existed among included studies. The DerSimonian-Laird method would be used to pool the results if I 2 was larger than 50%. Otherwise, the Mantel-Haenszel method would be used to pool the results. Subgroup analyses by ethnic groups and types of cancers were further performed to obtain the ethnic-specific effects of CYP24A1 polymorphisms on the predisposition of cancer and to elevate the source of heterogeneity. Stabilities of pooled results were examined by omitting one study each time and pooling the results of the other studies. Publication biases were tested with funnel plots and Egger’s tests.
Results
Characteristics of included studies
There were 176 articles retrieved by the authors using our literature searching strategy. The authors assessed 28 articles for eligibility after excluding unrelated and repeated publications. Six reviews were further excluded, and another 5 studies without all necessary genotypic data were also excluded. A total of 17 studies met the selection criteria and were finally included for pooled meta-analysis (Figure 1). The eligible studies were published between 2007 and 2020. All eligible studies were population-based case-control studies, and all genotypic analyses were performed using blood samples. Extracted data of eligible studies are summarized in Table 1 and Supplementary Table 1.
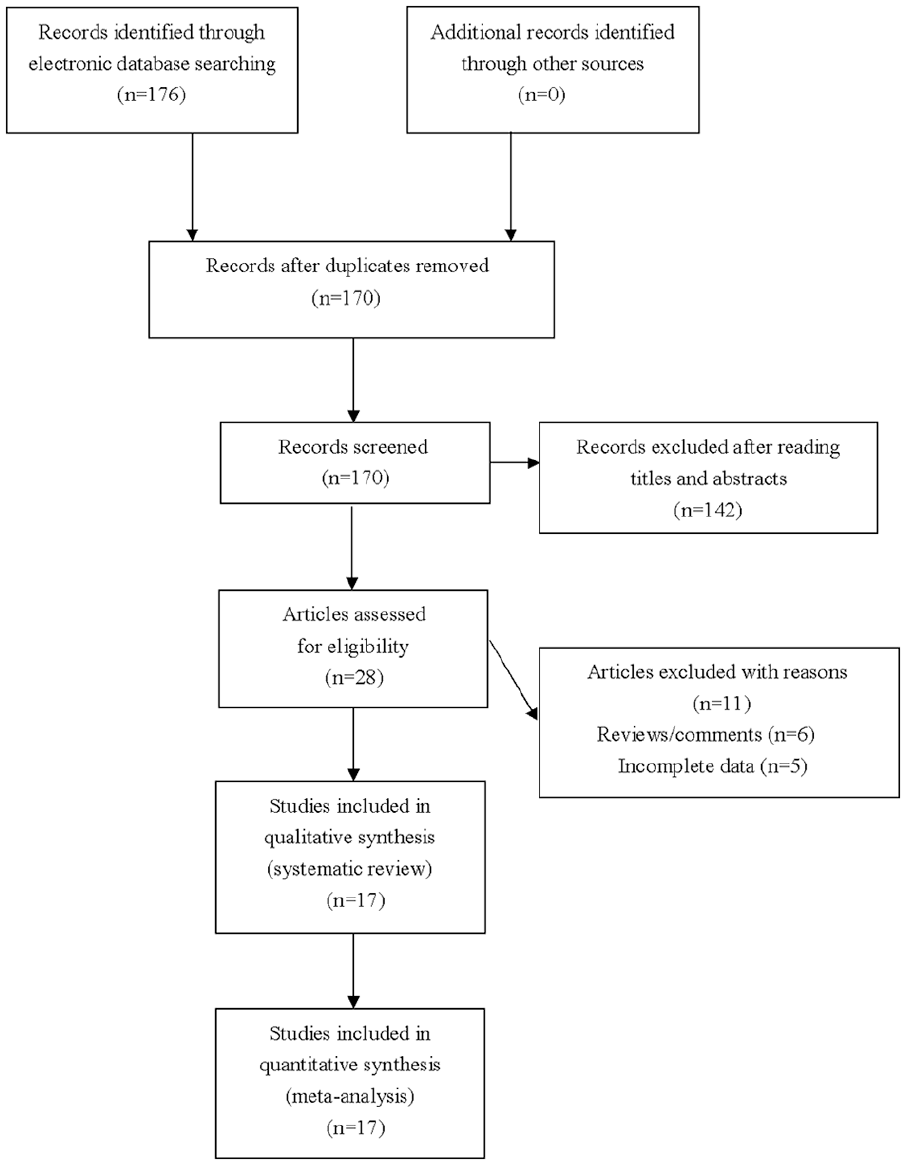
Flowchart of the study selection for the meta-analysis of CYP24A1 polymorphisms and cancer.
The characteristics of studies included in the current meta-analysis.
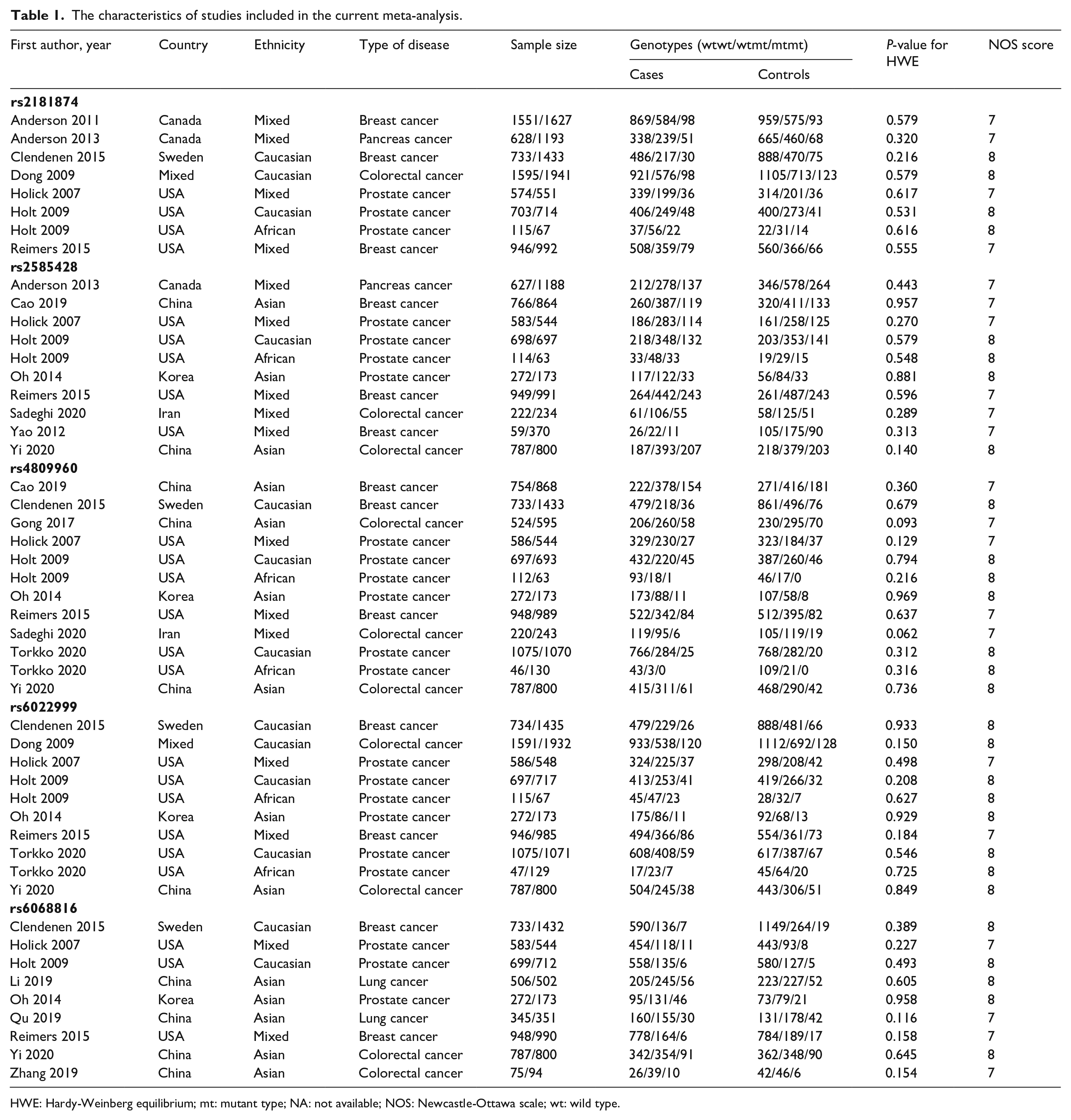
HWE: Hardy-Weinberg equilibrium; mt: mutant type; NA: not available; NOS: Newcastle-Ottawa scale; wt: wild type.
Meta-analyses of CYP24A1 polymorphisms and cancer
Seven studies with 6845 cases and 8518 controls were eligible for estimation of the relationship between the rs2181874 polymorphism and the predisposition of cancer; 9 studies with 5077 cases and 5924 controls were eligible for estimation of the relationship between the rs2585428 polymorphism and the predisposition of cancer; 10 studies with 6754 cases and 7601 controls were eligible for estimation of the relationship between the rs4809960 polymorphism and the predisposition of cancer; 8 studies with 6850 cases and 7857 controls were eligible for estimation of the relationship between the rs6022999 polymorphism and the predisposition of cancer; and 9 studies with 4948 cases and 5598 controls were eligible for estimation of the relationship between the rs6068816 polymorphism and the predisposition of cancer. CYP24A1 rs4809960 polymorphism was found to be significantly associated with predisposition of cancer in Caucasians (over-dominant comparison: OR=0.86, P=0.01), and rs6022999 polymorphism was found to be significantly associated with predisposition of cancer in Asians (dominant comparison: OR=1.47, P<0.0001; over-dominant comparison: OR=0.73, P=0.0007; allele comparison: OR=1.37, P<0.0001). In addition, we found that rs2585428 polymorphism was significantly associated with predisposition of prostate cancer (allele comparison: OR=1.12, P=0.02), whereas rs6068816 polymorphism was significantly associated with predisposition of both breast cancer (recessive comparison: OR=0.52, P=0.04) and prostate cancer (dominant comparison: OR=0.83, P=0.04; allele comparison: OR=0.84, P=0.02). We did not observe any other positive associations in our pooled analyses (see Table 2 and Supplementary Figure 1).
Pooled meta-analysis results of the current study.
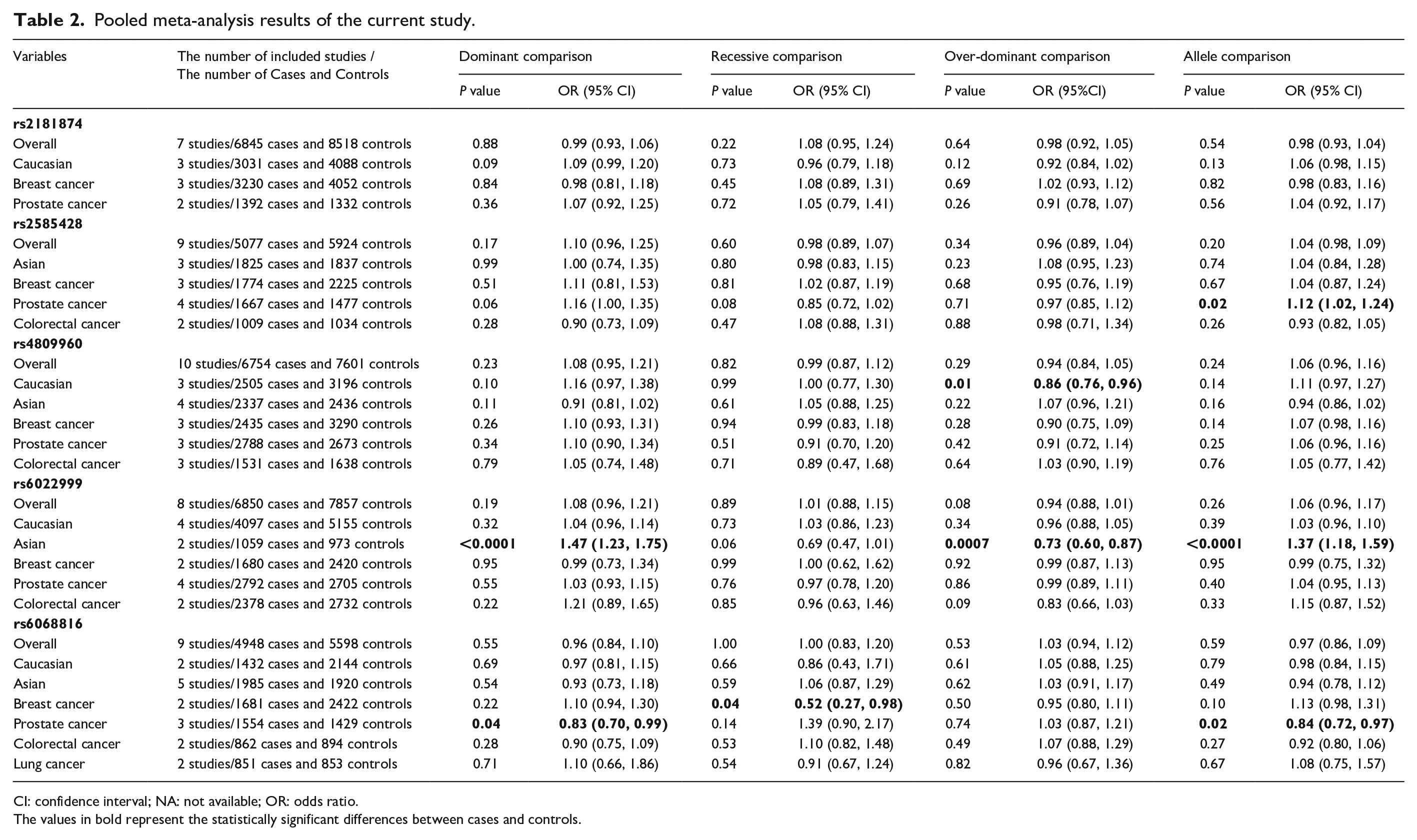
CI: confidence interval; NA: not available; OR: odds ratio.
The values in bold represent the statistically significant differences between cases and controls.
Sensitivity analyses
By omitting one study each time and pooling the results of the other studies, the authors examined stabilities of pooled meta-analysis results in the overall population. The trends of associations were not significantly altered in the sensitivity analyses. No positive associations were observed in the overall population; similarly, in the sensitivity analyses, no positive associations were detected, which indicated that our pooled meta-analysis results were statistically stable. (See datasets uploaded to the Open Science Framework.)
Publication biases
The authors examined potential publication biases in this meta-analysis with funnel plots and Egger’s tests. Funnel plots were found to be overall symmetrical (see Supplementary Figure 2). We further calculated the P values of the Egger’s tests to quantitatively analyze publication biases, and found that these P values were all greater than 0.05 (for rs2181874 polymorphism: dominant comparison, P = 0.226; recessive comparison, P = 0.493; over-dominant comparison, P = 0.371; allele comparison, P = 0.168; for rs2585428 polymorphism: dominant comparison, P = 0.362; recessive comparison, P = 0.173; over-dominant comparison, P = 0.454; allele comparison, P = 0.278; for rs4809960 polymorphism: dominant comparison, P = 0.333; recessive comparison, P = 0.419; over-dominant comparison, P = 0.112; allele comparison, P = 0.574; for rs6022999 polymorphism: dominant comparison, P = 0.438; recessive comparison, P = 0.627; over-dominant comparison, P = 0.218; allele comparison, P = 0.535; for rs6068816 polymorphism: dominant comparison, P = 0.274; recessive comparison, P = 0.316; over-dominant comparison, P = 0.588; allele comparison, P = 0.392). These results jointly indicated that our pooled analyses results were unlikely to be severely deteriorated by publication biases.
Evaluation of heterogeneity
The findings of eligible studies were generally found to be homogenous, and no severe heterogeneities (I2 > 75%) were detected in this meta-analysis. For dominant and allele comparisons of rs4809960, rs6022999, and rs6068816 polymorphisms, obvious heterogeneities (I2 > 50%) were observed. In further subgroup analyses by ethnic groups, we noticed that degrees of heterogeneity for the abovementioned comparisons were all significantly reduced, which suggests that for these comparisons, the ethnic origin of study subjects may serve as a major source of heterogeneity. (See Supplementary Figure 1 and datasets uploaded to the Open Science Framework.)
Discussion
This meta-analysis, comprehensively summarized associations between CYP24A1 polymorphisms and predisposition of cancer. The pooled meta-analysis results demonstrated that the rs4809960 polymorphism was significantly associated with the predisposition of cancer in Caucasians, and the rs6022999 polymorphism was significantly associated with the predisposition of cancer in Asians. We also found that the rs2585428 polymorphism was significantly associated with the predisposition of prostate cancer, whereas the rs6068816 polymorphism was significantly associated with the predisposition of both breast cancer and prostate cancer. In sensitivity analyses, the trends of associations were not significantly altered, suggesting that our pooled meta-analysis results are quite stable.
Several points should be considered when interpreting our findings. First, it is plausible that the investigated CYP24A1 polymorphisms may (a) alter the enzymatic function of CYP24A1 or the mRNA expression level of CYP24A1; (b) impact the metabolism of vitamin D; and (c) influence the predisposition of cancer. Nevertheless, the functionalities of the investigated polymorphisms remain unclear, and thus warrant further exploration. Second, we found that the trends of associations for different types of cancers were somehow inconsistent, which suggest that the effects of CYP24A1 polymorphisms on different types of cancers may vary significantly. However, considering that only a few studies were found to be eligible for pooled analyses, maybe the sample sizes of our pooled meta-analysis—especially some subgroup analyses—were still inadequate to reveal the real associations of CYP24A1 polymorphisms with cancer. Therefore, future studies with larger sample sizes need to confirm our findings. Third, we aimed to analyze all CYP24A1 polymorphisms at the beginning. However, we did not find a sufficient number of eligible studies to support meta-analyses of other polymorphisms, so we could only focus on five polymorphisms in this pooled meta-analysis. Fourth, it is biologically plausible that subjects with genetic predisposition factors may be more liable to develop cancer earlier in their lives. Therefore, age distribution may also influence the genetic associations between CYP24A1 polymorphisms and the predisposition of cancer. The age of the study subjects in the eligible studies are summarized in Supplementary Table 1. Where the studies reported the age of the subjects, we noticed that the median ages generally matched, so the observations in this meta-analysis were unlikely to be seriously influenced by age distribution. Fifth, it is worth noting that a recent meta-analysis by Zhu et al. 25 also tried to investigate associations between CYP24A1 polymorphisms and the predisposition of cancer. Nevertheless, this previous meta-analysis only covered relevant genetic association studies published before 2015. Since our literature search revealed that many related studies were published after 2015, we believed that an updated meta-analysis was warranted. In contrast to this previous meta-analysis, we found that the rs6068816 polymorphism was significantly associated with the predisposition of both breast cancer and prostate cancer, whereas the rs6022999 polymorphism was significantly associated with the predisposition of cancer in Asians, but the positive association observed between the rs4809960 polymorphism and the predisposition of breast cancer by Zhu et al. 25 was no longer found to be statistically significant in our pooled analyses. Considering that the current meta-analysis was based on more eligible studies and more study participants, our pooled analyses should be considered as a valuable supplement to pre-existing literature.
Several major limitations of our pooled meta-analysis should also be acknowledged. First, our meta-analyses results were only based on unadjusted pooling of previous publications. Without access to raw data of eligible studies, we can only estimate associations based on re-calculations of raw genotypic frequencies. However, we admit that lack of further adjustment for baseline characteristics such as age, sex, alcohol intake, and smoking status may certainly influence the authenticity of our findings. 26 Additionally, cancer characteristics—such as stage, grade, histology features, tumor size, metastasis status and the presence of well-established pathogenic mutations—may also influence the effects of CYP24A1 polymorphisms on the predisposition of cancer. Nevertheless, due to the fact that the detailed genotypic data of CYP24A1 polymorphisms according to these cancer characteristics were not available from eligible studies, it is impossible for us to perform analyses accordingly. Second, environmental factors may also affect relationships between CYP24A1 polymorphisms and the predisposition of cancer. However, the majority of authors only investigated genetic associations in their works, so genetic–environmental interactions could not be explored in our meta-analysis. 27 Third, we did not enroll grey literature for analysis because these literature are always incomplete and it is hard to assess the quality of their methodology. Hence, even though the funnel plots were found to be symmetrical overall, we acknowledge that publication biases still may affect the reliability of our pooled results. 28 Fourth, it is notable that some of the eligible studies had relatively small sample sizes. Therefore, even though the methodologies of these eligible studies were acceptable according to the NOS scale, these studies still may be influenced by potential selection biases, which may impact the reliability of our pooled analyses results. 29
Conclusions
This meta-analysis demonstrates that the rs4809960 polymorphism may affect the predisposition of cancer in Caucasians, and the rs6022999 polymorphism may affect the predisposition of cancer in Asians. Moreover, the rs2585428 polymorphism may affect the predisposition of prostate cancer, while the rs6068816 polymorphism may affect the predisposition of both prostate and breast cancer. Further studies with larger sample sizes are still needed to confirm our findings. Additionally, scholars should try to explore the underlying molecular mechanisms of associations between the abovementioned CYP24A1 polymorphisms and the predisposition of cancer in the future.
Supplemental Material
Supplementary_Figure_1-Forest_plots – Supplemental material for Genetic associations between CYP24A1 polymorphisms and predisposition of cancer: A meta-analysis
Supplemental material, Supplementary_Figure_1-Forest_plots for Genetic associations between CYP24A1 polymorphisms and predisposition of cancer: A meta-analysis by Guoqiang Zhang and Maohe Jin in The International Journal of Biological Markers
Supplemental Material
Supplementary_Figure_2-Funnel_plots – Supplemental material for Genetic associations between CYP24A1 polymorphisms and predisposition of cancer: A meta-analysis
Supplemental material, Supplementary_Figure_2-Funnel_plots for Genetic associations between CYP24A1 polymorphisms and predisposition of cancer: A meta-analysis by Guoqiang Zhang and Maohe Jin in The International Journal of Biological Markers
Supplemental Material
Supplementary_Table_1_5 – Supplemental material for Genetic associations between CYP24A1 polymorphisms and predisposition of cancer: A meta-analysis
Supplemental material, Supplementary_Table_1_5 for Genetic associations between CYP24A1 polymorphisms and predisposition of cancer: A meta-analysis by Guoqiang Zhang and Maohe Jin in The International Journal of Biological Markers
Footnotes
Acknowledgements
None.
Authors’ contributions
Guoqiang Zhang and Maohe Jin designed this meta-analysis, searched the literature, analyzed data, and wrote the manuscript. Both authors approved the final manuscript as submitted.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Ethics
This article does not contain any studies with human participants or animals performed by any of the authors, thus ethical approval and informed consent are not required.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
