Abstract
Objective:
This study aimed to investigate the correlation of A-kinase interacting protein 1 (AKIP1) with chemokine (C-X-C motif) ligand 1 (CXCL1) and CXCL2, as well as their associations with clinical characteristics and prognosis in prostate cancer patients.
Methods:
A total of 248 eligible prostate cancer patients who underwent surgery were consecutively recruited, and tumor tissues were collected during the surgery. AKIP1, CXCL1, and CXCL2 expression in tumor tissues were assessed by immunohistochemistry. Disease-free survival and overall survival were recorded, and the median follow-up time was 27 months.
Results:
The proportion of patients with AKIP1, CXCL1, and CXCL2 high expression was 56.5%, 63.7%, and 56.9%, respectively. Additionally, AKIP1 expression positively correlated with CXCL1 expression (P<0.001) and CXCL2 expression (P<0.001), and CXCL1 expression was positively associated with CXCL2 expression (P<0.001). Furthermore, AKIP1 expression positively correlated with pathological T stage (P<0.001) and pathological N stage (P=0.003). CXCL1 expression was positively associated with pathological T stage (P<0.001) and pathological N stage (P<0.001) as well. However, the CXCL2 expression only positively correlated with pathological T stage (P=0.002). Also, AKIP1 high expression correlated with worse disease-free survival (P=0.049) and OS (P=0.013), and CXCL1 high expression was associated with unfavorable disease-free survival (P=0.023) but not overall survival (P=0.052). CXCL2 expression was not correlated with disease-free survival (P=0.083) or overall survival (P=0.065). Multivariate Cox’s regression disclosed that AKIP1 high expression independently predicted worse overall survival (P=0.009).
Conclusion:
AKIP1 positively associates with CXCL1/2 and is a potential biomarker for disease monitoring as well as prognosis in prostate cancer.
Introduction
Prostate cancer is the second most common malignancy in males in over a half of the countries worldwide, and its mortality is still rising in many Asian countries despite of the development of screening technology using the prostate-specific antigen (PSA) test. 1 Also, currently, there is a controversy in the application of PSA screening among males because it requires an invasive procedure for the suspected individuals.2-4 Although favorable prognosis is achievable in the localized prostate cancer patients, the survival profile of non-localized patients is terrible. They are often tortured by pain, physical inconvenience, high relapse rate, and multiple side effects caused by chemotherapy or hormone therapy. 5 Therefore, it is very necessary to explore biomarkers promoting the early diagnosis and intervention of prostate cancer, which could subsequently improve patients’ prognosis.
A-kinase interacting protein 1 (AKIP1)—a protein initially found in breast cancer cells and prostate cancer cells—is shown to be implicated in the promotion of tumor progression.6-8 Chemokine (C-X-C motif) ligand 1 (CXCL1) and CXCL2 are two chemokines involved in the regulation of tumorigenesis of various cancers, such as breast cancer, colon cancer, and more importantly, prostate cancer.9-11 Notably, a previous report illustrates that AKIP1 enhances angiogenesis and tumor growth, and promotes cancer cell proliferation by regulating CXCL1, CXCL2, and CXCL8. 12 Accordingly, we hypothesized that AKIP1, CXCL1, and CXCL2 might possess clinical value in the management of prostate cancer, while it lacks related study. Thus, we aimed to investigate the correlation of AKIP1 with CXCL1 and CXCL2, as well as their associations with clinical characteristics and prognosis in prostate cancer patients.
Methods
Patients
A total of 248 eligible prostate cancer patients were consecutively recruited in our hospital from January 2015 to December 2018. The inclusion criteria of this study were: (a) diagnosed as prostate cancer by histopathology; (b) clinical status suitable for surgery and about to receive prostatectomy; and (c) age ⩾18 years old. The exclusion criteria were: (a) relapsed and secondary prostate cancer; (b) complicated with or history of malignancies; and (c) history of prostate relative surgeries. The Ethics Committee of our hospital approved the study, and all patients provided the written informed consents.
Data collection
After enrollment, demographics and clinicopathological features (such as age and PSA level) of patients were recorded. After resection, the surgical margin status (e.g. the pathological T stage, the pathological N stage, and the Gleason score) was also documented. In addition, the assessment criteria for pathological T stage Pt2 were as follows: Pt2 was the tumor locating within the prostate, Pt2a was the tumor occupying less than 1/2 in simple leaf of the prostate, Pt2b was the tumor occupying more than 1/2 in simple leaf of the prostate but limited in this leaf, and Pt2c was the tumor occupying two leaves of the prostate.
Immunohistochemistry (IHC) staining
The tumor tissues (N=248) and paired adjacent non-tumor tissues (N=74) resected from the surgery were fixed using formalin and embedded using paraffin. The expression of AKIP1, CXCL1, and CXCL2 in the tumor tissues was assessed by IHC. Briefly, the tumor tissues were cut into 4 μm sections, then the sections were deparaffinized with xylene and rehydrated with graded ethanol. After antigen retrieval, the non-specific binding and peroxidase activity of the sections were blocked using 0.3% H2O2 and 10% normal goat serum (Sigma-Aldrich, USA), respectively. Subsequently, the sections were incubated with the primary antibody (Rabbit AKIP1 Polyclonal Antibody (1:30, Invitrogen, USA), Rabbit CXCL1 Polyclonal Antibody (1:100, Invitrogen, USA) or CXCL2 Recombinant Rabbit Monoclonal Antibody (1:20, Invitrogen, USA)) overnight at 4℃. The following day, the sections were incubated with horseradish peroxidase-conjugated Goat anti-Rabbit IgG (H+L) secondary antibody (1:10000, Invitrogen, USA) for 60 min at 37℃. Finally, the sections were stained using diaminobenzidine (DAB) (Sigma-Aldrich, USA), counterstained using hematoxylin (Sigma-Aldrich, USA), and sealed using neutral resin (Sango Biotech, China).
IHC assessment
The staining result of IHC was assessed by a semiquantitative scoring method, which included a staining intensity score and a staining density score. 13 The staining intensity was scored as 0 (no staining), 1 (week staining), 2 (moderate staining), and 3 (intense staining); the staining density was scored as 0 (negative), 1 (⩽25%), 2 (26–50%), 3 (51–75%), and 4 (⩾76%). The total IHC score ranged from 0 to 12. The sections with a total IHC score >3 were defined as high expression, and the sections with a total IHC score ⩽3 were defined as low expression. 13
Follow-up
After surgery, appropriate treatments were given to the patients by their attending physicians. All patients were regularly followed up to December 31, 2018, and the median follow-up duration was 27.0 months. During follow-up, the relapse or death event was recorded for calculating disease-free survival (DFS) and overall survival (OS). DFS was defined as the duration from resection to relapse or death, and OS was defined as the duration from resection to death. The patients who were lost to follow-up were censored on the date of the last visit for the DFS calculation or they were censored on the date of last known to be alive for the OS calculation.
Statistical analysis
Statistical analysis was performed using SPSS 24.0 (IBM, USA), and figures were made using GraphPad Prism 7.00 (GraphPad Software, USA). Correlations among AKIP1, CXCL1, and CXCL2 expressions were determined by the Chi-square test. The correlation of AKIP1, CXCL1, and CXCL2 expressions with clinicopathological features was determined by the Chi-square test or Spearman’s rank correlation test. DFS and OS were displayed by Kaplan–Meier curves, and comparisons of DFS and OS between two groups were determined by the log-rank test. Factors correlated with DFS and OS were analyzed by univariate and forward stepwise multivariate Cox’s proportional hazard regression model. P value <0.05 was considered as significant.
Results
The prostate cancer patients’ characteristics
The 248 prostate cancer patients included in our study had an average age of 62.5±9.3 years (Supplementary Table 1). As for other characteristics, the number of patients with pathological T stage of pT2a, pT2b, pT2c, pT3a, pT3b, and pT4 was 76 (30.7%), 39 (15.7%), 34 (13.7%), 50 (20.2%), 44 (17.7%), and 5 (2.0%), respectively. Also, there were 181 (73.0%) patients with pathological N stage of pN0 and 67 (27.0%) patients with pathological N stage of pN1. The number of patients with Gleason score ⩽6, =7 and ⩾8 was 54 (21.8%), 138 (55.6%), and 56 (22.6%), respectively. In addition, there were 67 (27.0%), 126 (50.8%), and 55 (22.2%) patients who had a PSA level of ⩽10 ng/mL, 10–20 ng/mL and ⩾20 ng/mL, respectively. In addition, 202 (81.5%) patients had a negative surgical margin, and 46 (18.5%) patients had a positive surgical margin.
Expressions of AKIP1, CXCL1, and CXCL2 in tumor tissue
The examples of AKIP1 high/low expression, CXCL1 high/low expression and CXCL2 high/low expression in tumor tissues by IHC staining are presented in Figure 1(a). There were 108 (43.5%) patients who presented with AKIP1 low expression, and the other 140 (56.5%) patients presented with AKIP1 high expression (Figure 1(b)). With regard to CXCL1 and CXCL2, the number of patients with CXCL1 low expression and CXCL1 high expression was 90 (36.3%) and 158 (63.7%), respectively; the number of patients who had CXCL2 low expression as well as patients who had CXCL2 high expression was 107 (43.1%) and 141 (56.9%), respectively. Additionally, the positive control and negative control of AKIP1 expression in tumor tissue were presented in Supplementary Figure 1(a). We also found that the AKIP1 expression was increased in tumor tissue (N=248) compared with adjacent non-tumor tissue (N=74) (P = 0.001) (Supplementary Figure 1(b)).
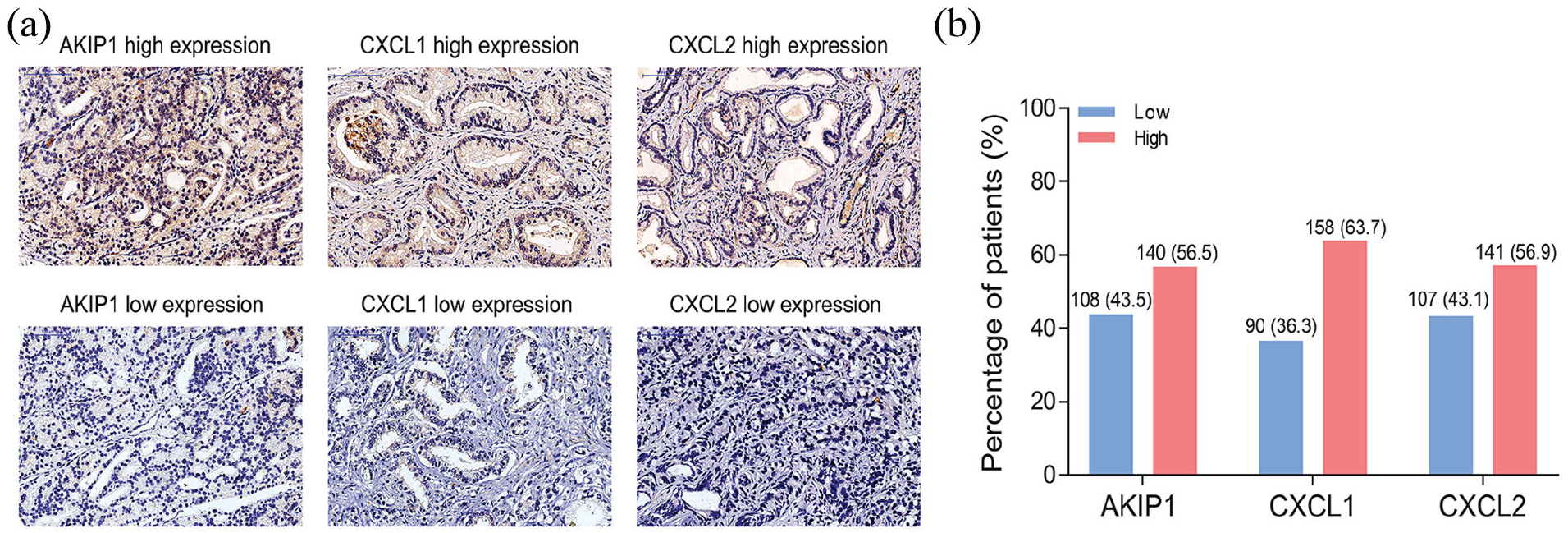
Comparison of AKIP1/CXCL1/CXCL2 high/low proportions in tumor tissue. The illustrations of AKIP1 high/low expression, CXCL1 high/low expression and CXCL2 high/low expression in tumor tissue assessed by IHC staining (a), and the percentage of patients with AKIP1/CXCL1/CXCL2 high or low expression (b).
The associations among AKIP1, CXCL1, and CXCL2 in tumor tissue
The AKIP1 expression was positively correlated with CXCL1 expression (P < 0.001) and CXCL2 expression (P < 0.001) in tumor tissue (Supplementary Table 2). CXCL1 expression was also positively associated with CXCL2 expression (P < 0.001).
The associations of AKIP1, CXCL1, and CXCL2 in tumor tissue with patients’ characteristics
The AKIP1 expression in tumor tissue was positively correlated with pathological T stage (P < 0.001) and pathological N stage (P = 0.003), but was not correlated with the Gleason score (P = 0.304) (Table 1). In terms of CXCL1 and CXCL2, the CXCL1 expression was positively associated with both pathological T stage (P < 0.001) and pathological N stage (P < 0.001). The CXCL2 expression was only positively correlated with pathological T stage (P = 0.002). In addition, CXCL1 expression (P = 0.353) and CXCL2 expression (P = 0.077) were not correlated with the Gleason score.
Correlation of AKIP1, CXCL1, and CXCL2 expressions with clinicopathological features.
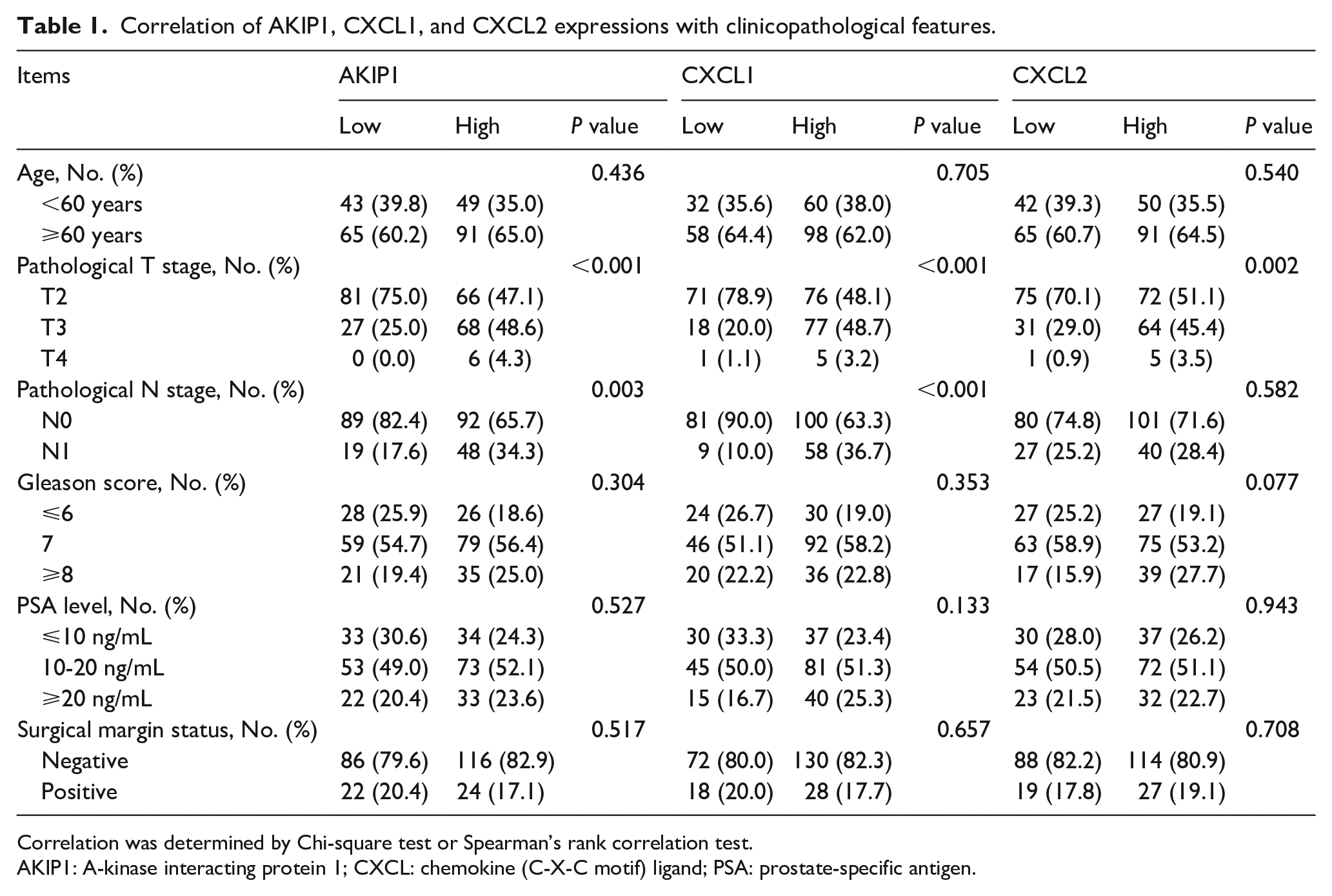
Correlation was determined by Chi-square test or Spearman’s rank correlation test.
AKIP1: A-kinase interacting protein 1; CXCL: chemokine (C-X-C motif) ligand; PSA: prostate-specific antigen.
The associations of AKIP1, CXCL1, and CXCL2 in tumor tissue with patients’ survival profiles
In view of patients’ survival profiles, the DFS was worse in patients who had AKIP1 high expression compared to patients who had AKIP1 low expression in tumor tissue (P = 0.049) (Figure 2(a)). The DFS was shorter in patients presenting with CXCL1 high expression compared to patients presenting with CXCL1 low expression (P = 0.023) (Figure 2(b)). However, no difference of DFS was discovered between patients with CXCL2 high expression and patients with CXCL2 low expression (P = 0.083) (Figure 2(c)). OS was less prolonged in patients with AKIP1 high expression compared to patients with AKIP1 low expression (P = 0.013) (Figure 2(d)). Nonetheless, the OS did not vary between patients with CXCL1 high expression and patients with CXCL1 low expression (P = 0.052) (Figure 2(e)), nor between patients presented with CXCL2 high expression and patients with CXCL2 low expression (P = 0.065) (Figure 2(f)).
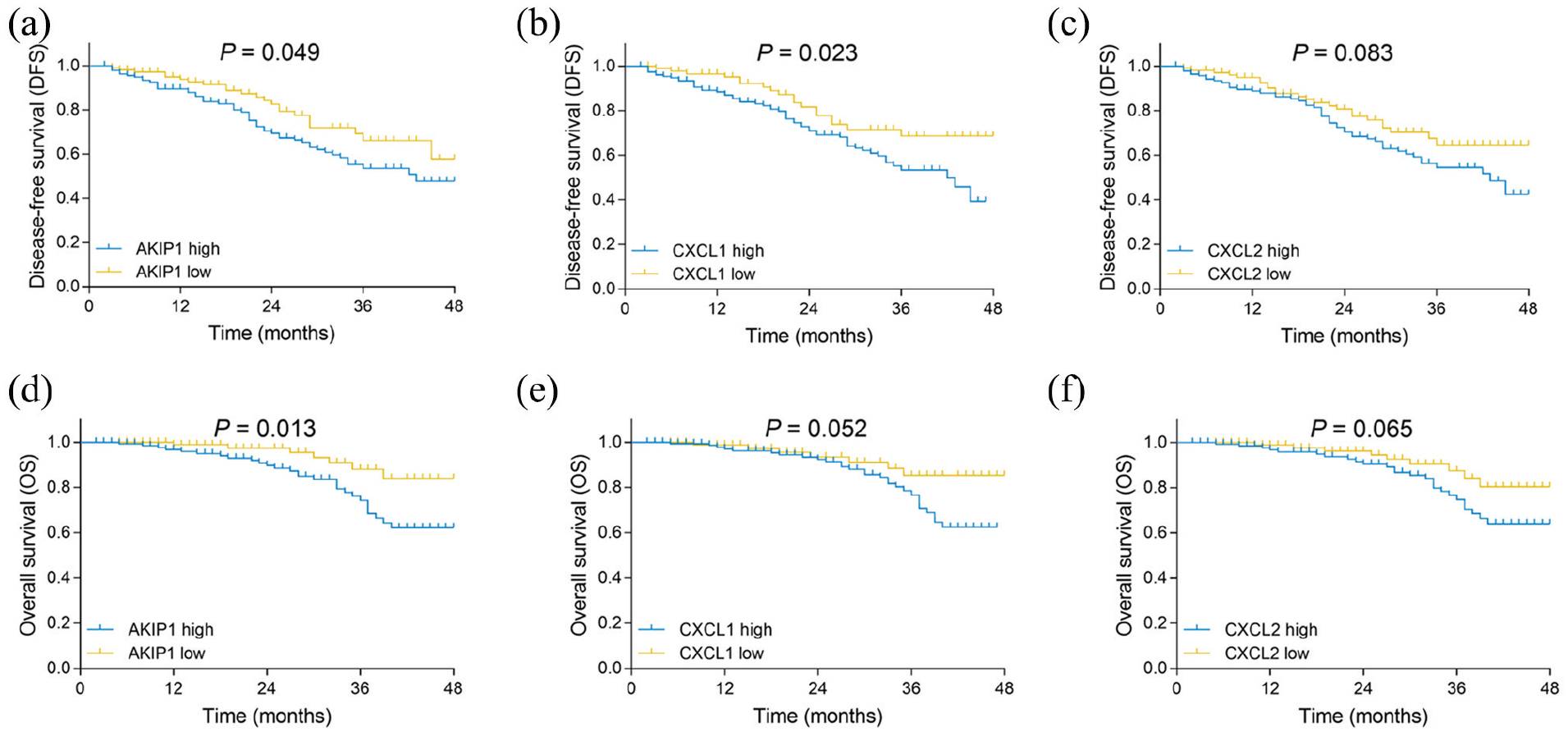
Comparison of DFS between patients with different (high/low) AKIP1, CXCL1, and CXCL2 expressions. The comparison of DFS between patients with AKIP1 high expression and low expression (a), the comparison of DFS between patients who had CXCL1 high expression and low expression (b), and the comparison of DFS between patients with CXCL2 high and low expression (c). The comparison of OS between patients with AKIP1 high and low expression (d), the comparison of OS between patients with CXCL1 high and low expression (e), and the comparison of OS between patients with CXCL2 high and low expression (f).
The predictive values of AKIP1, CXCL1, and CXCL2 in tumor tissue for patients’ survival profiles
A univariate Cox’s regression analysis revealed that AKIP1 high expression (P = 0.043) and CXCL1 high expression (P = 0.022) in tumor tissue predicted worse DFS (Table 2). Higher pathological T stage (P = 0.002), higher pathological N stage (P = 0.006), Gleason score⩾8 (P = 0.031), and positive surgical margin status (P = 0.008) were also predictors for worse DFS. Additionally, in the multivariate Cox’s regression analysis using the forward stepwise method, higher pathological T stage (P = 0.001) and positive surgical margin status (P = 0.005) were independent predictors for unfavorable DFS. For OS, AKIP1 high expression (P = 0.013), CXCL1 high expression (P = 0.048) could predict shorter OS, and the other predictors for shorter OS were higher pathological T stage (P = 0.008), higher pathological N stage (P = 0.002), Gleason score=7 (P = 0.040), Gleason score⩾8 (P = 0.012), and positive surgical margin status (P = 0.003) (Table 3). Then, a forward stepwise multivariate Cox’s regression analysis disclosed that AKIP1 high expression (P = 0.009) was an independent predictive factor for worse OS. The other independent predictive factors for less prolonged OS included a Gleason score ⩾8 (P = 0.015) and positive surgical margin status (P = 0.010).
Analysis of factors correlated with DFS.
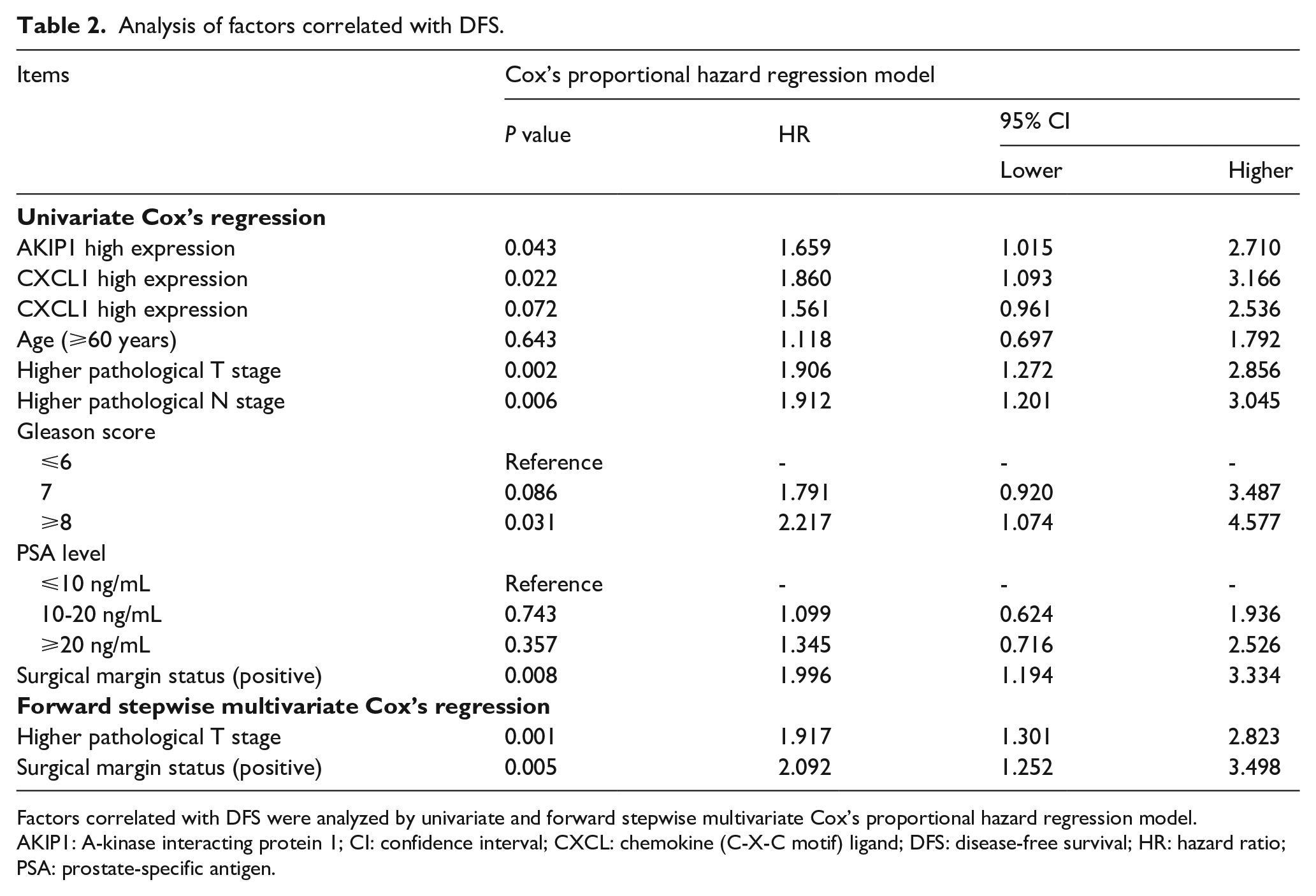
Factors correlated with DFS were analyzed by univariate and forward stepwise multivariate Cox’s proportional hazard regression model.
AKIP1: A-kinase interacting protein 1; CI: confidence interval; CXCL: chemokine (C-X-C motif) ligand; DFS: disease-free survival; HR: hazard ratio; PSA: prostate-specific antigen.
Analysis of factors correlated with OS.
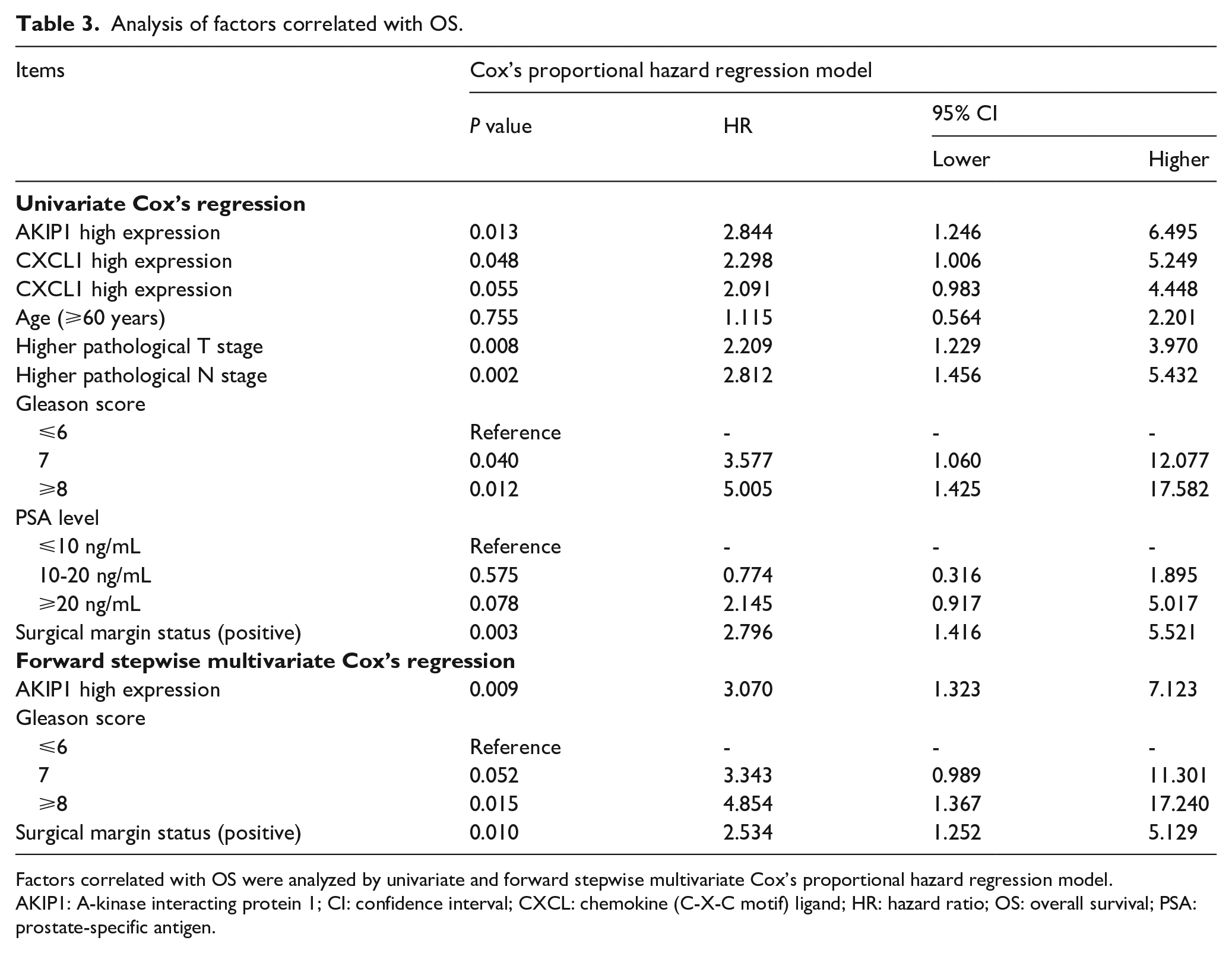
Factors correlated with OS were analyzed by univariate and forward stepwise multivariate Cox’s proportional hazard regression model.
AKIP1: A-kinase interacting protein 1; CI: confidence interval; CXCL: chemokine (C-X-C motif) ligand; HR: hazard ratio; OS: overall survival; PSA: prostate-specific antigen.
Discussion
In this study, we found that in tumor tissues of prostate cancer patients who underwent surgery: (a) the proportions of AKIP1, CXCL1, and CXCL2 high expression in tumor tissue were increased compared to AKIP1, CXCL1, and CXCL2 low expression in tumor tissue; (b) AKIP1 expression was positively correlated with CXCL1 and CXCL2 expression, and CXCL1 expression was positively associated with CXCL2 expression; (c) AKIP1 and CXCL1 levels were both positively correlated with pathological T stage and pathological N stage, and CXCL2 was positively associated with pathological T stage; (d) AKIP1 high expression was associated with worse DFS and OS, and CXCL1 high expression correlated with shorter DFS; and (e) AKIP1 high expression was an independent predictive factor for worse OS.
AKIP1 was initially found to aberrantly expressed in several cancer cell lines, and accumulating studies have revealed its critical role in various cancers. For instance, a recent study elucidated that AKIP1 increases proliferation while it represses apoptosis in non-small cell lung cancer (NSCLC) cells. 14 Another study revealed that AKIP1 advances proliferation and invasion of gastric cancer cells through Slug-induced epithelial-mesenchymal transition (EMT). 6 Moreover, AKIP1 stimulates cell invasion and colony formation in hepatocellular carcinoma (HCC) cells, and enhances intrahepatic as well as lung metastasis in a mouse model of HCC by mediating Wnt/β-catenin/CBP signaling pathway. 7 In addition, it has been reported that AKIP1 enhances cell proliferation and invasion, promotes EMT by activating Zinc Finger E-Box Binding Homeobox 1 (ZEB1) in NSCLC cells. 15 Additionally, a study reveals that BCA3 (an alternative name of AKIP1) increases tumor growth, metastasis, and angiogenesis in vitro and in vivo by stimulating protein kinase B (AKT) translocating nuclear factor-kappa B (NF-κB) in HCC. 16 To point out, a study elucidates that AKIP1 downregulation suppresses cell motility and invasion by decreasing the expression of Akt/glycogen synthase kinase-3β (GSK-3β)/Snail pathway in breast cancer cells. 17 As for the regulatory role of CXCL1 and CXCL2 in cancers, a study reports that AKIP1 enhances proliferation in vitro and elevates tumor growth as well angiogenesis in vivo through increasing the expression of CXCL1, CXCL2, and CXCL8 in cervical cancer. 12
In our study, we found that AKIP1 expression was positively correlated with both CXCL1 and CXCL2 expressions, and AKIP1 high expression was associated with more advanced tumor characteristics, worse DFS and OS, and independently predicted unfavorable OS. These results could be explained by the following possible reasons: (a) AKIP1 probably promotes tumor progression via regulating cancer cell functions through multiple factors as reported by the previous studies of other cancers, such as regulating Wnt/β-catenin/CBP signaling pathway and EMT. Hence, the AKIP1 high expression was correlated with advanced tumor characteristics; (b) AKIP1 overexpression could result in more advanced tumor characteristics via enhancing cancer cell proliferation, invasion, and migration while inhibiting apoptosis through modulating various factors, which contributed to invasion, metastasis, and growth of the tumor and resulted in worse survival of patients.6, 7, 12, 14-17 Nevertheless, evidence illustrating the detailed mechanism of the interaction between AKIP1 and CXCL1/2 is still scarce, which needs further experiments for validation. Additionally, no correlation between tumor AKIP1 and Gleason score was found in our study, which might be caused by the following reasons: our sample size was relatively small; and the Gleason score was a staging system that focused more on histological features of prostate tumor, which might indicate that the association of AKIP1 with prostate tumor histological features was relatively weak.
CXCL1 and CXCL2 are two chemokines intimately related to immunity and inflammation, and in recent decades, they have been reported to be oncogentic factors in various cancers. For CXCL1, one study elucidated that the CXCL1-lipocalin2 (LCN2) axis enhances prostate cancer cell migration by activating Src and EMT. 11 Also, CXCL1 is reported to elevate migration and invasion of breast cancer cells through provoking the extracellular signal-regulated kinase (ERK)/matrix metalloproteinase (MMP)2/9 signaling pathway. 9 Another study revealed that tumor-associated macrophages secreted CXCL1 increases the metastasis in a breast cancer mouse model and enhances migration as well as invasion of human breast cancer cells by modulating the NF-κB/SRY-related HMG-box 2 (SOX2) pathway. 18 In terms of CXCL2, it has been illustrated that CXCL2/CXCR2 axis leads to stem cell characteristics of CPT-11-resistant LoVo colon cancer cells through mediating Gαi-2 and Gαq/11. 10 In addition, the signaling of CXCL2/MIF-CXCR2 contributes to the myeloid-derived suppressor cells recruitment in tissues of bladder cancer, which indicates the progression of bladder cancer. 19 These studies all suggest the roles of CXCL1 and CXCL2 in promoting malignancy in various cancers, including prostate cancer.
In this study, CXCL1 high expression and CXCL2 high expression both correlated with advanced tumor characteristics, and CXCL1 high expression was correlated with worse DFS, which might be explained by: (a) CXCL1 and CXCl2 were capable of promoting tumor progression by regulating cancer cell functions as elucidated by previous studies performed in other carcinomas, which might result in the correlations of CXCL1 and CXCL2 with advanced tumor characteristics; (b) as for the correlation of CXCL1 with shorter DFS, it was probably derived from that CXCL1 resulted in the progression of prostate cancer by promoting malignant behaviors in tumors, which often led to relapse in patients.
There were several limitations in this study. First, the sample size might not be large enough, which could cause a less satisfactory statistical power. Second, we did not include any patients with metastasis due to the fact that the prognosis of metastatic prostate cancer patients was notably different from localized patients, which suggested that the values of AKIP1, CXCL1, and CXCL2 in metastatic patients should be assessed in the future. Third, AKIP1, CXCL1, and CXCL2 expressions were not evaluated in the circulating samples, which were more applicable compared with the tissue samples in the clinical setting.
In conclusion, we discovered (a) positive correlations among AKIP1, CXCL1, and CXCL2, which are all correlated with advanced tumor characteristics, and (b) that AKIP1 is an independent predictive factor for prognosis in prostate cancer patients.
Supplemental Material
Supplementary_material – Supplemental material for A-kinase interacting protein 1, a potential biomarker associated with advanced tumor features and CXCL1/2 in prostate cancer
Supplemental material, Supplementary_material for A-kinase interacting protein 1, a potential biomarker associated with advanced tumor features and CXCL1/2 in prostate cancer by Danlan Wang, Yuanfang Luo, Yonglian Guo, Guohao Li and Fan Li in The International Journal of Biological Markers
Footnotes
Acknowledgements
None.
Author contributions
Danlan Wang and Yuanfang Luo contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
