Abstract
Background:
The prognostic value of Stathmin 1 (STMN1) in malignant solid tumors remains controversial. Thus, we conducted this meta-analysis to summarize the potential value of STMN1 as a biomarker for predicting overall survival in patients with solid tumor.
Methods:
We systematically searched eligible studies in PubMed, Web of Science, and EMBASE from the establishment date of these databases to September 2018. Hazard ratio (HR) and its 95% confidence interval (CI) was used to assess the association between STMN1 expression and overall survival.
Results:
A total of 25 studies with 4625 patients were included in this meta-analysis. Our combined results showed that high STMN1 expression was associated with poor overall survival in solid tumors (HR = 1.85, 95% CI 1.55, 2.21). In general, our subgroup and sensitivity analyses demonstrated that our combined results were stable and reliable. However, from the results of the subgroups we found that high STMN1 expression was not related to overall survival in colorectal cancer and endometrial cancer anymore, suggesting that much caution should be taken to interpret our combined result, and more studies with large sample sizes are required to further explore the prognostic value of STMN1 expression in the specific type of tumors, especially colorectal cancer and endometrial cancer.
Conclusions:
STMN1 could serve as a prognostic biomarker and could be developed as a valuable therapeutic target for patients with solid tumors. However, due to the limitations of the present meta-analysis, this conclusion should be taken with caution. Further studies adequately designed are required to confirm our findings.
Keywords
Introduction
Approximately 1,700,000 patients were newly diagnosed with cancers, and 600,920 patients died from cancer in the US in 2017. 1 In China, there were an estimated 4,000,000 new cancer patients and 3,000,000 cancer-linked deaths in 2015. 2 Cancer has emerged as one of the diseases featured with the highest morbidity and mortality around the world. So far, first-line therapies commonly recommended for cancer patients are still surgery, radiotherapy, and (or) chemotherapy. Although some novel therapeutic approaches, such as molecular-targeted therapy 3 and immunological therapy, 4 have been developed for some types of cancers, the majority of cancer patients still had poor long-term survival (5-year survival). Accordingly, it remains urgent to identify novel prognostic biomarkers and to develop more effective therapeutic strategies for cancer patients.
Stathmin 1 (STMN1) belongs to the Stathmin family and is a phosphoprotein of 18 kDa specifically located in cytoplasm, also known as Stathmin, Oncoprotein 18, metablastin or Leukemia-associated phosphoprotein p18. 5 All members of Stathmin family have a tubulin-binding domain, and a Stathmin-like domain with four serine phosphorylation sites at the N-terminal region, including residues 16, 25, 38, and 68. 5 STMN1 exhibits the key activity of a microtubule destabilizer, by contributing to the microtubule catastrophe or the sequestration of alpha/beta-tubulin heterodimers, functioning as a critical element of regulating microtubules dynamics.6,7 In particular, microtubule dynamics are essential for the success of cell cycle progression. In accordance with its function of regulating microtubule dynamics, STMN1 plays a pivotal role in cell division and proliferation in cancer cells.8,9 Moreover, STMN1 has been reported to be upregulated in several types of cancer tissues and correlated with tumor aggressiveness.10,11 Thus, an increasing number of recent studies focused on exploring the prognostic value of STMN1 cancer patients, and suggested that higher expression of STMN1 predicted worse survival in patients with several types of solid tumors, such as head and neck squamous cell carcinoma, 12 gallbladder carcinoma, 13 esophageal squamous cell cancer, 14 lung carcinoma,15,16 breast cancer, 17 and endometrial cancer. 18 However, because most of the published studies were limited by the small number of cases, the conclusions regarding the prognostic value of STMN1 expression in solid tumors remain indecisive. Therefore, we performed a systematic review of the literature and a meta-analysis to thoroughly assess the prognostic value of STMN1 expression in predicting the survival of human solid tumors.
Materials and methods
This meta-analysis was undertaken according to preferred reporting items for systematic reviews and meta-analyses statements. 19
Identification and selection of studies
PubMed, Web of Science, and EMBASE were searched for studies assessing the expression of STMN1 and survival in solid tumors from the establishment date of these databases to September 2018. The search terms included “Stathmin 1 or Stathmin or STMN1 or Oncoprotein 18 or Metablastin or Leukemia-associated phosphoprotein p18” (Title/Abstract) and “prognosis or prognostic or outcome” (Title/Abstract).
The eligibility criteria were: (a) the studies that explored the association between STMN1 expression and overall survival (OS) in solid tumor and hazard ratio (HR) assessing the association between STMN1 expression and OS, which could be extracted; (b) the STMN1 expression must be detected in the protein level; and (c) the studies must be written in English.
The exclusion criteria were: (a) studies that explored the association between STMN1 expression and OS in non-solid tumor; (b) STMN1 expression detected in the mRNA level; and (c) animal or cell experiments. Citation lists of retrieved articles were manually screened for identifying the eligible studies that were likely missed in the database search. Disagreement was resolved by consensus.
Data collection process
Two authors (Dan Zhang and ZengXi Yang) independently extracted data using predefined data abstraction forms. The following details were extracted: first author’s name, publication year, origin of population, sample size, tumor type, survival analysis method, the definition of high STMN1 expression, and follow-up time. Software Engauge Digitizer 4.1 (https://markummitchell.github.io/engauge-digitizer/) was used to extract HR and confidence interval (CI), when the survival data were only presented in the Kaplan–Meier curve.
Study quality assessment
We assessed the quality of the included study using the Newcastle–Ottawa scale (NOS). 20 The upper limit of score of the NOS system is 9. In this study, we defined a study with score of 6 or more as being of sound quality.
Statistical analysis
HR and its corresponding 95% CI were used to assess the association between STMN1 expression and OS in patients with malignant solid tumors. When both HR and the lower limit of 95% CI were > 1 (low expression as reference), it indicated that patients with high STMN1 expression had shorter OS than those with low expression. Data were analyzed using RevMan 5.3 analysis software (Cochrane Collaboration, Copenhagen, Denmark). Estimates of HRs were weighted and pooled using the random effects model if the heterogeneity was significant. If there was no significant heterogeneity, HR was pooled using the fixed effects model. Statistical heterogeneity was evaluated using Cochran’s Q and I 2 statistics. Sensitivity analysis was used to explore the possible sources of heterogeneity. On one hand, we performed sensitivity analyses by sequentially omitting one included study at a time to explore the effect of this individual study on the pooled result. On the other hand, we also performed sensitivity analyses, according to tumor type, region of patients, the definition of high STMN1 expression, survival analysis type, sample size, and NOS, to explore whether these factors were responsible for the heterogeneity. The Begg’s and Egger’s tests were applied to evaluate publication bias.21,22 If the Begg’s funnel plot appeared asymmetrical or the P value of any of the two tests was less than 0.05, it indicated that significant publication bias may exist. When significant publication bias existed, the trim-and-fill method was applied to explore whether publication bias substantially affected the stability of the combined HR. 23 All statistical tests were two-sided, and the statistically significant difference was defined as P < 0.05.
R‑esults
Search results
The search results are presented in Figure 1. The primary literature research identified 509 records. After screening the titles and abstracts, 412 records were excluded due to duplication and irrelevance to the topic. We then reviewed the full text of the remaining records at length; ultimately, 25 studies were determined eligible for this meta-analysis.12–14,16,24–44
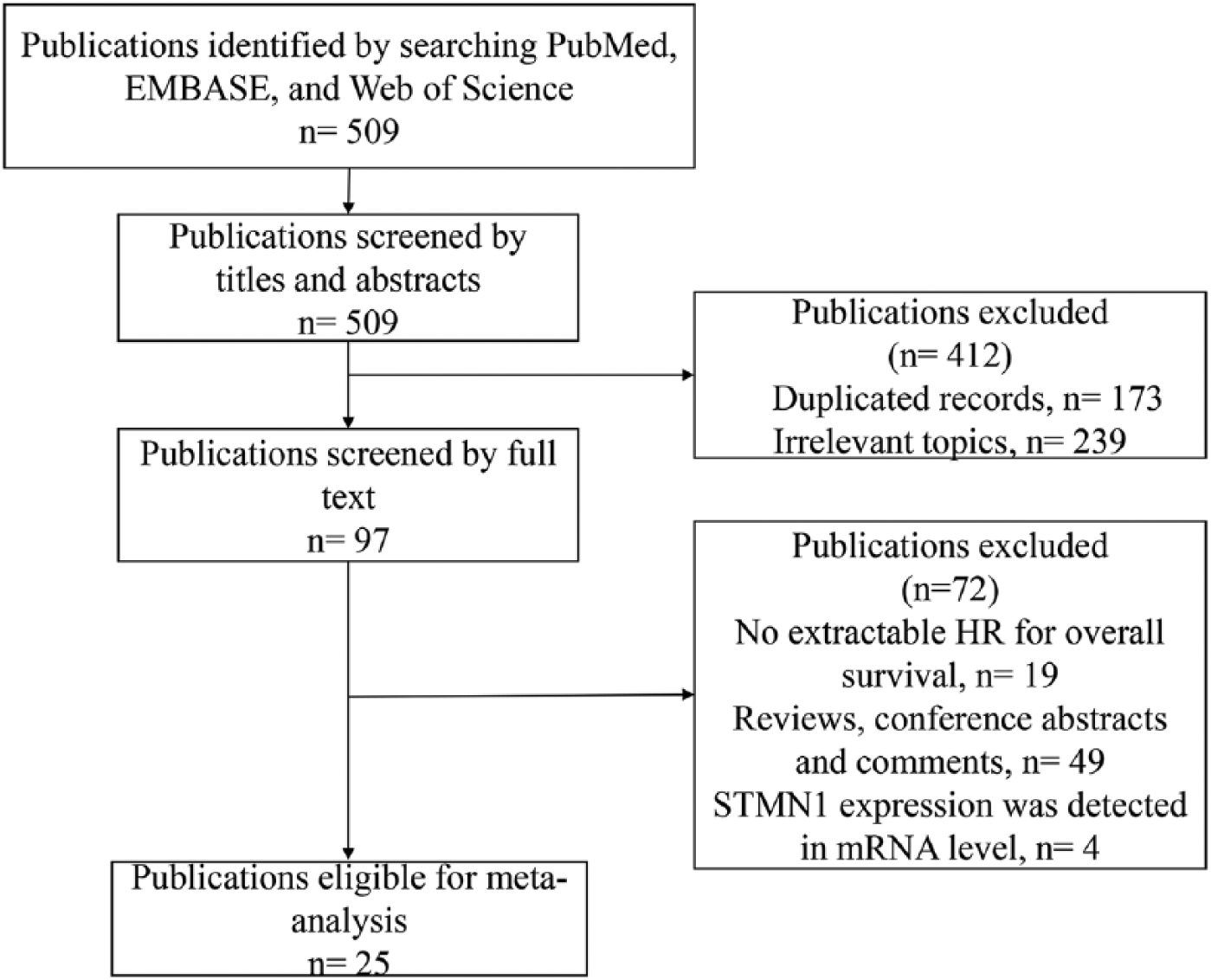
Flow diagram of identifying the eligible studies.
Description of studies
The main characteristics of the included studies are presented in Table 1. A total of 25 studies with 4625 patients were included in this meta-analysis. The mean sample size of the included studies was 185. Four studies evaluated esophageal cancer, four colorectal cancer, two gastric cancer, four oral cancer, three lung cancer, two gynecological cancer, and one each for osteosarcoma, gall bladder cancer, hepatocellular carcinoma, pancreatic cancer, and head and neck cancer. All included studies detected the expression level of STMN1 using the immunohistochemistry method. Only a few of the included studies reported the definitions of high STMN1 expression, which were inconsistent.
The main characteristics of the included studies.
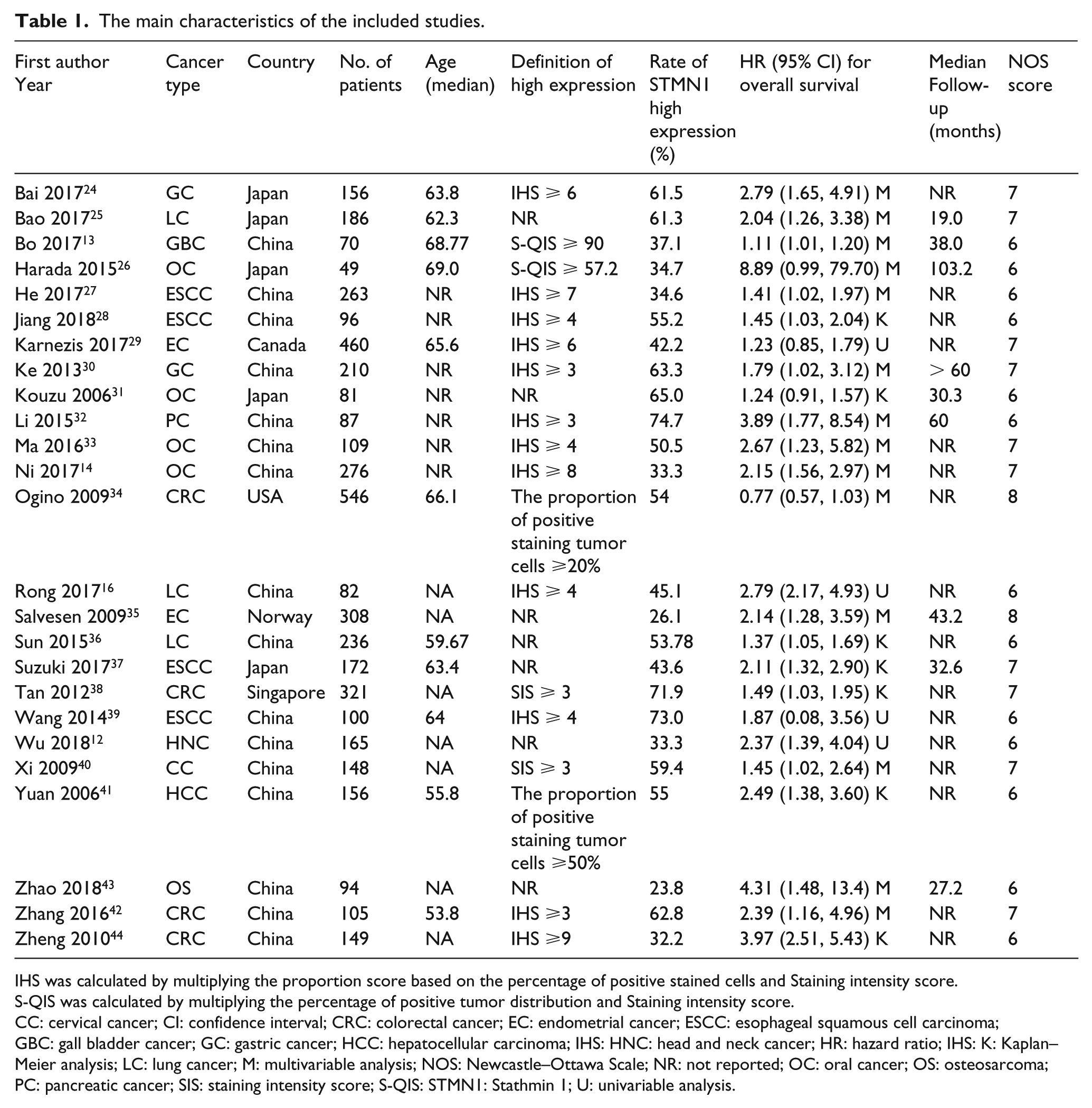
IHS was calculated by multiplying the proportion score based on the percentage of positive stained cells and Staining intensity score.
S-QIS was calculated by multiplying the percentage of positive tumor distribution and Staining intensity score.
CC: cervical cancer; CI: confidence interval; CRC: colorectal cancer; EC: endometrial cancer; ESCC: esophageal squamous cell carcinoma; GBC: gall bladder cancer; GC: gastric cancer; HCC: hepatocellular carcinoma; IHS: HNC: head and neck cancer; HR: hazard ratio; IHS: K: Kaplan–Meier analysis; LC: lung cancer; M: multivariable analysis; NOS: Newcastle–Ottawa Scale; NR: not reported; OC: oral cancer; OS: osteosarcoma; PC: pancreatic cancer; SIS: staining intensity score; S-QIS: STMN1: Stathmin 1; U: univariable analysis.
Meta-analysis of association between STMN1 expression and OS
HRs for the prognostic value of STMN1 extracted a total of 25 studies for synthesized analysis. Because there was significant heterogeneity among the included studies (Cochran’s Q, P < 0.0001, I2 = 82.0 %), the random effects model was used to perform this synthesized analysis. The combined results showed that high STMN1 expression was closely related to poor OS in patients with solid tumors (HR 1.85; 95% CI 1.55, 2.21) (Figure 2).
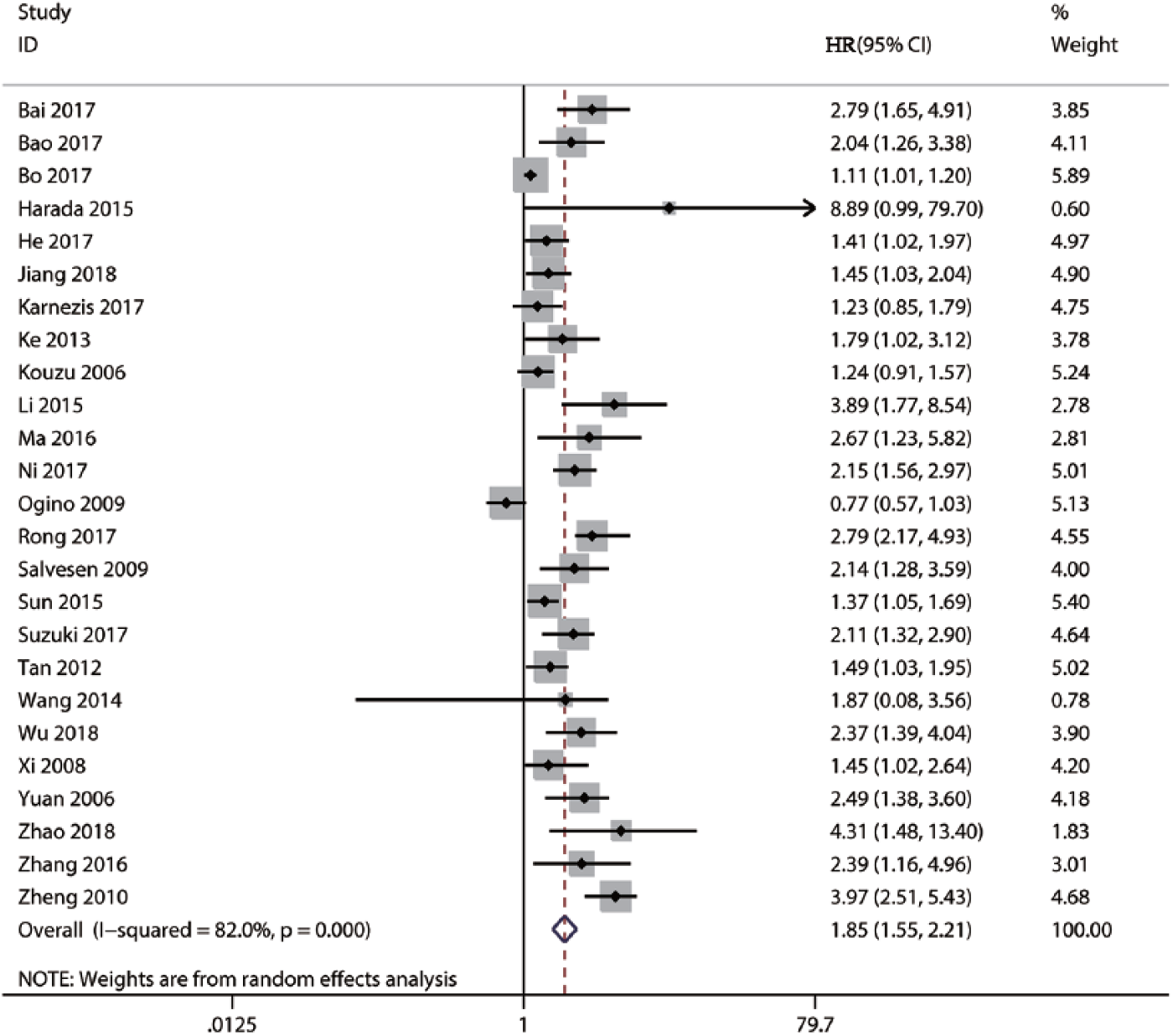
Forest plot of the hazard ratio (HR) assessing the association between Stathmin 1 (STMN1) expression and overall survival (OS) in patients with solid tumors.
Subgroup and sensitivity analyses
Considering that there was a significant heterogeneity in our combined result, we performed subgroup and sensitivity analyses to explore the sources of the heterogeneity. We performed subgroup analyses, according to tumor type, region of patients, the definition of high STMN1 expression, survival analysis type, sample size, and NOS, to explore whether these factors accounted for the majority of the heterogeneity. The results showed that the heterogeneity in all subgroup analyses did not significantly decrease, suggesting that these factors were not the main sources of the heterogeneity (Table 2). Overall, the combined results in the subgroup analyses still indicated that high STMN1 expression was associated with poor OS, suggesting the robustness of our combined result. However, from the results of the subgroups, we also observed that high STMN1 expression was not related to OS in colorectal cancer and endometrial cancer anymore. On one hand, the limitation of the sample size in the two subgroups may explain the negative result. On the other hand, the tumor heterogeneity itself resulted in the inconsistent prognostic significance of STMN1 expression in different tumors. Therefore, our results should be taken with much caution, and more high-quality studies are required to further explore the prognostic value of STMN1 expression in the specific type of tumors, especially colorectal cancer and endometrial cancer. Additionally, we performed sensitivity analyses by sequentially omitting one study at a time to determine whether some individual studies were responsible for the majority of the heterogeneity. When any of the included studies was omitted, the heterogeneity was still obvious, suggesting that no individual study could explain the majority of the heterogeneity alone. However, the result of the sensitivity analysis showed that the combined result did not significantly change (Supplement 1), implying that our combined result was stable and reliable.
Subgroup analysis of pooled hazard ratios for overall survival.
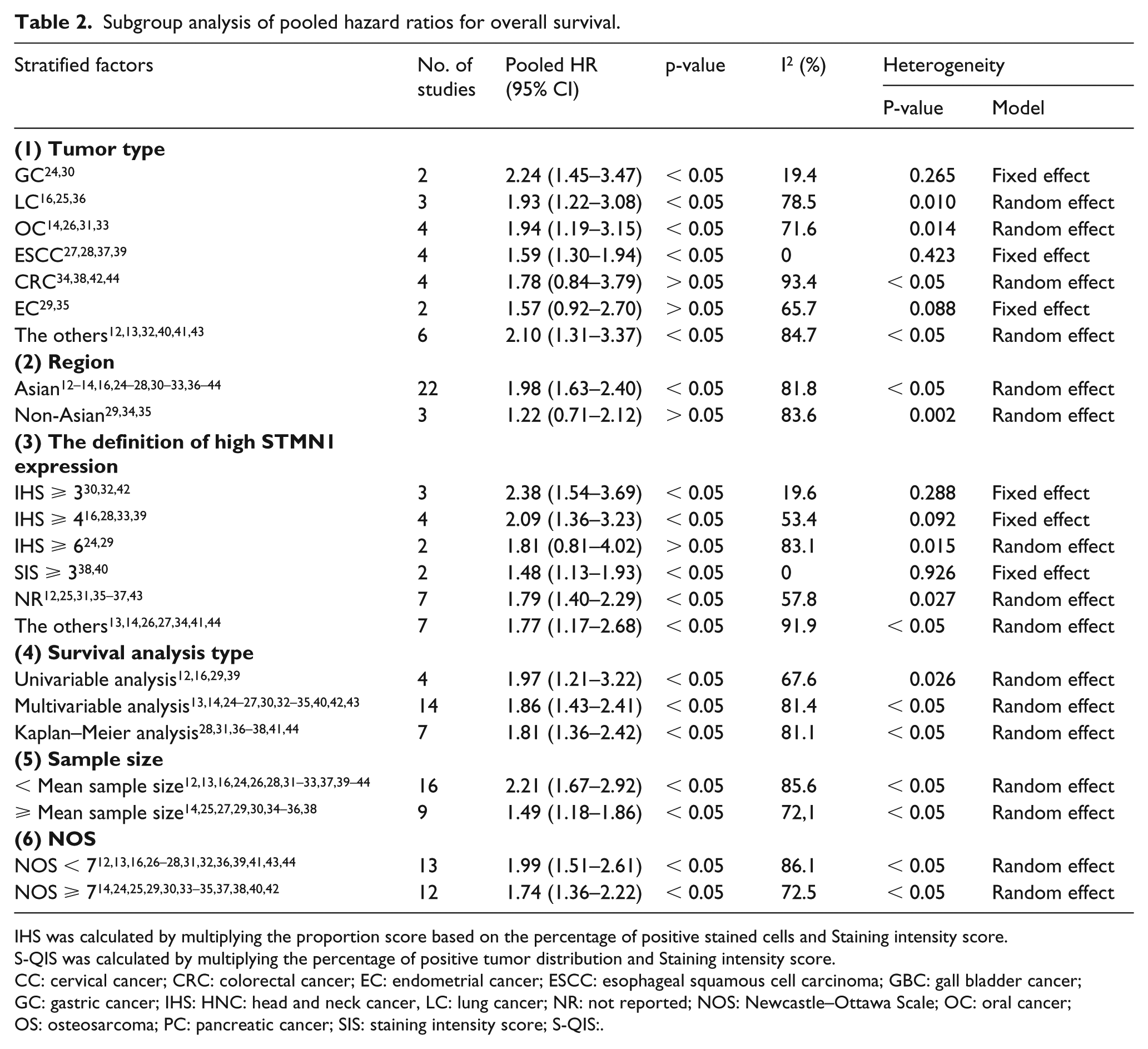
IHS was calculated by multiplying the proportion score based on the percentage of positive stained cells and Staining intensity score.
S-QIS was calculated by multiplying the percentage of positive tumor distribution and Staining intensity score.
CC: cervical cancer; CRC: colorectal cancer; EC: endometrial cancer; ESCC: esophageal squamous cell carcinoma; GBC: gall bladder cancer; GC: gastric cancer; IHS: HNC: head and neck cancer, LC: lung cancer; NR: not reported; NOS: Newcastle–Ottawa Scale; OC: oral cancer; OS: osteosarcoma; PC: pancreatic cancer; SIS: staining intensity score; S-QIS:.
Publication bias
The Begg’s funnel plot and Egger’s tests were utilized to evaluate publication bias in this meta-analysis. The results showed that Begg’s funnel plot appeared asymmetrical (Supplement 2A), and the P values of the two tests were less than 0.05, suggesting there was significant publication bias. We then used a trim-and-fill analysis to explore whether the publication bias significantly affected the credibility and stability of the combined result for OS. The result of the trim-and-fill analysis showed that the adjusted combined HR was still greater than 1 and its corresponding CI did not contain 1 (HR 1.42; 95% CI 1.16, 1.73), which demonstrated that the publication bias did not significantly affect the stability of our combined results (Supplement 2B).
Discussion
STMN1 is highly expressed in various malignant tumor tissues compared with matched non-tumor tissue, indicating the possibility of STMN1 as a prognostic factor. 10 The epithelial-mesenchymal transition (EMT) plays a critical role in promoting metastasis, progression, and recurrence of cancers. 45 Consistently, STMN1 has been reported to contribute to tumor progression by promoting the EMT of cancer cells via the regulation of microtubule dynamics.46–48 STMN1 also played an important role in regulating the other tumor cellular behaviors. For instance, downregulating STMN1 expression could induce tumor cell cycle arrest in the G2/M phase, and decrease their colony-forming ability, including esophageal carcinoma cells, gallbladder carcinoma cells, and glioma cells.49–51 Similarly, STMN1 downregulation could also inhibit the proliferation of glioblastoma cells and esophageal squamous cell carcinoma.14,52 Additionally, several previous studies revealed that silencing the expression of STMN1 promoted cellular apoptosis of glioma, 50 oral cancer, 53 prostate cancer, 54 non-small cell lung cancer, 55 and esophageal squamous cell carcinoma. 37 Furthermore, some researchers demonstrated that the downregulation of STMN1 expression was capable of inhibiting the migration and invasion of cancer cells.14,52,53,56 Overall, these previous studies support the notion that STMN1 may act as an oncogene and plays a pivotal role in promoting cancer progression, indicating that STMN1 may be a useful prognostic biomarker and therapeutic target.
Consistently, accumulating evidence has suggested that elevated STMN1 expression is closely correlated not only with the development and progression of cancer,10,11 but also with poor survival.16,26,57–60 The evidence from a meta-analysis by Rong et al. 16 confirmed that the overexpression of STMN1 was positively associated with high tumor stage, poor differentiation, and lymph node invasion. 10 However, to date, the prognostic value of STMN1 expression in solid tumors has not been systematically assessed by meta-analysis. Thus, to further determine whether STMN1 could be used a prognostic biomarker and a potential therapeutic target in solid tumors, we performed this timely meta-analysis. We found that high STMN1 expression was associated with poor OS in solid tumors (HR 1.85; 95% CI 1.55, 2.21). Furthermore, in general, our subgroup and sensitivity analyses demonstrated that our combined result was stable and reliable. Taken together, our meta-analysis demonstrated that TMN1 could be used as a prognostic biomarker and a potential therapeutic target in solid tumors.
Nevertheless, several limitations should be considered with much caution in this meta-analysis, when interpreting our synthesized result. First, the most important limitation was that there was significant heterogeneity, and our sensitivity and subgroup analysis failed to identify its sources. Actually, several factors that may cause heterogeneity, such inconsistent follow-up time, patient age and tumor stage, were not analyzed in the subgroup analysis due to lack of available data in the included studies. Therefore, more homogeneous studies with high quality are required to validate the prognostic value of STMN1 in solid tumors. Second, although our meta-analysis included a total of 25 eligible studies, the number of eligible studies that referred to the specific type of tumor was limited; thus the small sample size may cause a degree of bias. Therefore, more studies with large sample sizes are required to further explore the prognostic value of STMN1 expression in the specific type of tumors. Third, all the included studies were performed based on retrospective design, and under this situation those studies with positive results were more likely to be published than those with negative ones, which may cause a degree of publication bias. Fourth, because of the inconsistent cut-off values of high STMN1 expression in the included studies, the utility of STMN1 as a prognostic indicator may be impaired in patients with solid tumors. Therefore, it may be imperative to standardize the detection methods and cut-off values in further studies. Fifth, the calculation errors could not be completely avoided when manually extracting data from the Kaplan–Meier curve using Engage Digitizer 4.1. Sixth, among the included studies, only two were from Western countries, with the majority from Asian populations. Therefore, more studies should be conducted to further confirm whether STMN1 could act as a prognostic biomarker in tumor patients in Western countries.
Conclusion
STMN1 could serve as a prognostic biomarker and could be developed as a valuable therapeutic target for patients with solid tumors. However, due to the limitations of the present meta-analysis, this conclusion should be taken with caution. Further studies, adequately designed, are required to confirm our findings.
Supplemental Material
Supplement_1 – Supplemental material for Association of STMN1 with survival in solid tumors: A systematic review and meta-analysis
Supplemental material, Supplement_1 for Association of STMN1 with survival in solid tumors: A systematic review and meta-analysis by Dan Zhang, Lizhen Dai, ZengXi Yang, XiChen Wang and Yin LanNing in The International Journal of Biological Markers
Supplemental Material
Supplement_2 – Supplemental material for Association of STMN1 with survival in solid tumors: A systematic review and meta-analysis
Supplemental material, Supplement_2 for Association of STMN1 with survival in solid tumors: A systematic review and meta-analysis by Dan Zhang, Lizhen Dai, ZengXi Yang, XiChen Wang and Yin LanNing in The International Journal of Biological Markers
Footnotes
Authors contribution
Dan Zhang and Lizhen Dai equally contributes to this article.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
