Abstract
Objective
Many studies have focused on correlations between forkhead box protein C2 (FOXC2) and various tumors but discrepant results have been reported. Thus, we conducted this meta-analysis to assess the prognostic role of FOXC2 in tumors.
Methods
Four electronic databases (PubMed, Embase, Web of Science, and SinoMed) were screened through September 2019.
Results
The final analysis included 15 reports and 2115 patients; results suggested that cancer patients with FOXC2 had worse overall survival (hazard ratio 2.14, 95% confidence interval (CI) 1.74–2.64), cancer-specific survival (hazard ratio 2.65, 95% CI 1.44–4.89), and disease-free survival (hazard ratio 1.93, 95% CI 1.49–2.50) than patients lacking FOXC2.
Conclusions
The presence of FOXC2 was associated with poor survival in cancer patients. FOXC2 could be a promising prognostic marker in the future.
Introduction
Members of the forkhead box (FOX) transcription factor family share a common DNA binding domain. To date, 17 FOX gene subfamilies, FOXA to FOXR, have been identified in humans. 1 Forkhead box protein C2 (FOXC2), previously called “mesenchyme forkhead 1” (MFH-1), is a member of the FOX protein family. It is an important regulator that controls the generation of vascular and lymphatic tissue. 1 Epithelial–mesenchymal transition (EMT) is considered a crucial molecular mechanism inducing tumor development. 1 FOXC2 has been reported to be an EMT inducer and to be involved in tumor invasion and metastasis. 1
Recently, assessment of the prognostic role of FOXC2 has been evaluated in different types of cancers but the results have been variable. Published studies have indicated that high expression of FOXC2 is negatively correlated with overall survival (OS) for tumors, including pancreatic cancer, esophageal squamous cell carcinoma, human gliomas, colon cancer, hepatocellular carcinoma, and pulmonary neuroendocrine tumors.2–7In contrast, some researchers have reported that FOXC2 has a lesser association with OS in human gliomas, non-small-cell lung cancer, and hepatocellular carcinoma.8,9 Therefore, we conducted this meta-analysis to illuminate the prognostic value of FOXC2 in various tumors.
Patients and methods
Data sources and search strategy
This meta-analysis was conducted according to the guidance provided in the Cochrane Handbook. Four electronic databases, PubMed, Embase, Web of Science, and SinoMed, were searched through September 2019. Search terms included “forkhead box protein C2 OR FOXC2 OR MFH-1” and “prognosis OR survival OR outcome”. Ethical approval was not needed because this secondary study was a meta-analysis of previously published studies.
Selection criteria
Inclusion criteria were as follows: (1) studies of tumor patients that reported the prognostic impact of FOXC2; (2) articles published in English or Chinese with full text; and (3) availability of hazard ratio (HR) or Kaplan–Meier curve. Exclusion criteria were as follows: (1) a letter, conference abstract, case report, laboratory study, or review article; (2) lack of necessary data; or (3) multiple publications and duplicate records.
Data extraction
A total of 2115 patients in 15 studies were recruited.2–16 The following details were extracted: the first author’s last name, publication year, study country, pathology type, patient age, sex ratio, tumor stage, detection method, numbers of patients, source of HR, analysis model, follow-up and HRs with 95% confidence interval (CIs) for OS, cancer-specific survival (CSS), or disease-free survival (DFS). The HRs and corresponding 95% CIs were extracted preferentially from multivariate analyses when available. Otherwise, univariate results were acceptable. If the included studies lacked HRs, we extrapolated them from Kaplan–Meier curves using Engauge Digitizer version 11.3, as previously reported. 17 Additionally, relative ratios (RRs) and corresponding 95% CIs were calculated to determine the relationships between FOXC2 and some clinicopathological features.
Quality assessment
Two independent reviewers quantified the quality of each article using the Newcastle–Ottawa Quality Assessment Scale (NOS). 18 In the NOS, studies are awarded stars based on various quality parameters. Studies with ≥7 stars are priority quality studies, those with ≥5 stars are high quality studies, and <5 indicates low quality studies.
Statistical analysis
Heterogeneity was evaluated by using the I2 statistic and p-values, with a predefined significance threshold of I2 < 50% or p > 0.1. 19 If the heterogeneity test showed that there was less significant heterogeneity (I2 < 50% or p > 0.1), a fixed-effects model was applied to pool the outcome data. Otherwise, if significant heterogeneity (I2 ≥ 50%; p ≤ 0.1) existed, a random-effects model was used. Subgroup analyses were performed to investigate causes of heterogeneity. The robustness of the combined results was further assessed by sensitivity analysis. The Begg’s and Egger’s tests were used to evaluate publication bias. All statistical analyses were carried out using Stata version 12.0 software (Stata Corp., College Station, TX, USA). A p-value < 0.05 was considered to indicate a significant difference.
Results
Selection and characteristics of studies
A total of 332 records were screened and identified. After applying the inclusion and exclusion criteria, 2115 patients in 15 studies were assessed.2–16 A flow diagram of the study selection process is shown in Figure 1.

Flow diagram of study selection process.
The enrolled subjects were from China, Japan, Spain, and Norway. These studies were published from 2011 to 2019, and the follow-up time ranged from 3 to 242 months. The patients were diagnosed with various tumors, including esophageal squamous cell carcinoma (n = 2), oral squamous cell carcinoma (n = 2), hepatocellular carcinoma (n = 2), human gliomas (n = 2), and other tumor types (n = 7); TNM stages ranged from I to IV. There was no standardized cut-off value for all eligible studies. Immunohistochemistry (IHC) was conducted in 13 studies to detect FOXC2 expression, and the patients were dichotomized by staining intensity and extent. For the remaining two studies, quantitative reverse transcription polymerase chain reaction (qRT-PCR) was used, and the patients were divided into two groups, taking the median score value as cut-off point. Based on the NOS, all included studies were assessed as priority quality. Details of the basic study characteristics are summarized in Table 1.
General characteristics of the included studies.

NOS, Newcastle-Ottawa Quality Assessment Scale; SI, staining intensity; SE, staining extent; NR, not reported; OSCC, oral squamous cell carcinoma; HCC, hepatocellular carcinoma; EHCC, extrahepatic cholangiocarcinoma; IHC, immunohistochemistry; qRT-PCR, quantitative reverse transcription polymerase chain reaction. **Shown as median (range); ***shown as overall follow-up month.
FOXC2 and OS
Eight studies, including 1163 patients, reported the prognostic value for OS. As shown from the pooled results, patients with FOXC2 (+) were predicted to have worse OS (HR 2.14, 95% CI 1.74–2.64, p < 0.001) than patients lacking FOXC2 (−) (Figure 2). The effect of FOXC2 on OS among disease subgroups was further assessed. The prognostic effect of FOXC2 was greater in human gliomas (HR 2.78, 95% CI 1.79–4.32, p < 0.001), followed by hepatocellular carcinoma (HR 1.90, 95% CI 1.09–3.03, p = 0.024) and lung cancer (HR 1.87, 95% CI 1.14–3.05, p = 0.012). The HR of the group of other tumors was 2.07 (95% CI 1.51–2.83, p < 0.001) (Table 2).

Forest plots of studies evaluating HR of FOXC2 for OS, DFS, and CSS.
Subgroup analyses of results.
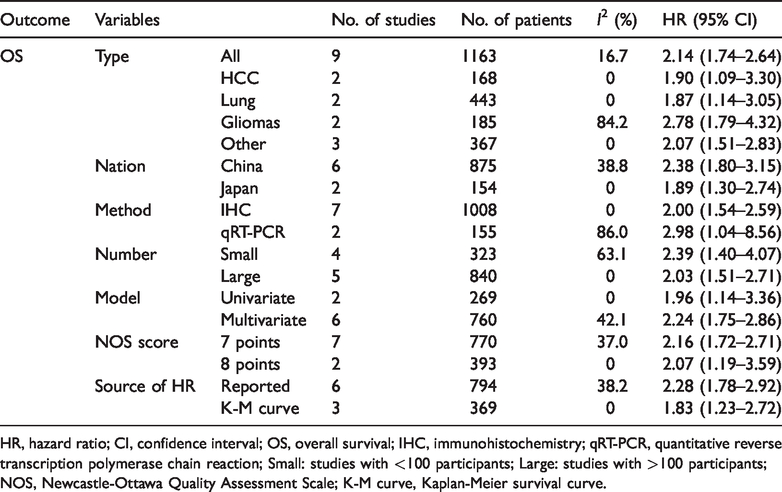
HR, hazard ratio; CI, confidence interval; OS, overall survival; IHC, immunohistochemistry; qRT-PCR, quantitative reverse transcription polymerase chain reaction; Small: studies with <100 participants; Large: studies with >100 participants; NOS, Newcastle-Ottawa Quality Assessment Scale; K-M curve, Kaplan-Meier survival curve.
To explore possible sources of heterogeneity, we performed subgroup analyses. As shown in Table 2, the results indicated that FOXC2 remained a useful prognostic marker for OS regardless of study country, detection method, numbers of patients, statistical model, NOS score, or HR source. The heterogeneity may be partially caused by these parameters.
We performed a sensitivity analysis by sequentially removing studies to investigate the influence of an individual study on the pooled results. The pooled HRs for OS were not significantly changed, suggesting robustness of the results (Figure 3).

Sensitivity analysis for overall survival.
To assess the publication bias of the main pooled result, we conducted Begg’s test and Egger’s linear regression test. As shown in Figure 4, Begg’s funnel plot for OS (p = 0.602) showed that the probability of publication bias was low. Confirmation using Egger’s regression test also failed to show evidence of obvious bias for OS (p = 0.635).

Publication bias analysis for overall survival.
FOXC2 and clinicopathological features
The relationship of FOXC2(+) with some clinicopathological features (those reported in more than two studies) was summarized for human gliomas. No significant correlation was observed between FOXC2(+) and sex (RR 1.19, 95% CI 0.94–1.52, p = 0.155) or WHO grade (RR 1.43, 95% CI 0.84–2.45, p = 0.191).
FOXC2, CSS, and DFS
Five studies (789 cases) reported HRs for CSS, and six studies (1015 patients) reported HRs for DFS. Overall, when CSS was analyzed, FOXC2(+) was correlated with a poor outcome (HR 2.65, 95% CI 1.44–4.89, p = 0.002). Similarly, for DFS, a significant relationship was found between FOXC2(+) and worse prognosis (HR 1.93, 95% CI 1.49–2.50, p < 0.001) (Figure 2).
Discussion
In the present meta-analysis, we analyzed 15 reports (2115 patients) and discovered that patients with high levels of FOXC2 had unfavorable OS, CSS, and DFS. Subgroup analyses showed that overexpression of FOXC2 was associated with poor OS in study populations in both China and Japan. Additionally, a high level of FOXC2 was a marker of poor prognosis of OS in cancer (human gliomas, hepatocellular carcinoma, lung cancer, and other cancers), regardless of the means of detection, number of participants, statistical model, NOS score, or HR source.
Currently, the exact mechanism underlying the prognostic value of FOXC2 in tumor patients remains largely unexplored. One potential mechanism may be an association of high FOXC2 expression with EMT. 5 It has been widely reported that EMT plays a critical role in cancer development and progression. 20 In addition, several EMT-inducing factors, such as E-cadherin, transforming growth factor-β, and FOX proteins, could help predict prognosis in patients with cancer.5,6,21,22 According to some previous studies, among these predictors, the FOX proteins, including FOXM1, FOXQ1, FOXF1, FOXC1, FOXA1/2, and FOXO3a, are involved in tumor metastasis by mediating EMT. 5 Indeed, the prognostic impact of FOXC2 has been well documented in different cancers.2–16 Further study found that FOXC2 might interact with the MAPK, AKT, and GLI1 pathways in cell proliferation in many cancer types. 1 FOXC2 also plays an essential role in tumor angiogenesis and lymph angiogenesis, which are crucial for tumor metastasis. 1 Thus, high expression of FOXC2 could promote cancer invasion and metastasis and be associated with poor survival in cancer patients.
Previous studies have reported that the level of FOXC2 expression is significantly linked to patient clinicopathological features.3,11–13,15 We propose that, by mediating cancer invasion and metastasis, FOXC2 might increase the cancer burden and thus be related to patients’ clinicopathological features. Unfortunately, owing to the limited information, we could not draw a clear association between FOXC2 expression and clinicopathological characteristics.
Several limitations should be acknowledged in this study. First, some articles reported no significant correlation between FOXC2 and OS. Second, heterogeneity was observed even though it was not significant. Heterogeneity might be partially caused by different definitions of FOXC2 overexpression. Third, the patients included were primarily from Asian countries; thus, our conclusions might only apply to Asian patients.
Despite the above limitations, our meta-analysis had the following merits. To the best of our knowledge, this is the first meta-analysis to specially evaluate the effect of FOXC2 on tumor prognosis in different types of cancer. Our goal was to gain insight into the prognostic value of FOXC2 in various tumors, rather than to compare FOXC2 expression in early- and late-stage tumors, as was reported in a review published in 2018. 23 The sensitivity analysis revealed the robustness of our meta-analysis findings, and the evidence indicated that publication bias was low.
In conclusion, our analysis demonstrated that high expression of FOXC2 was associated with poor survival in cancer patients. FOXC2 could be a promising prognostic marker and therapeutic target in the future. Additional large, multicenter prospective studies are needed to confirm our findings.
