Abstract
Introduction
This study aimed to evaluate the oncological and prognostic significance of adjuvant chemotherapy (CT) in patients with adult granulosa cell tumors of the ovary (AOGCT).
Methods
We searched the Chinese National Knowledge Infrastructure, Clinical Trials, Wanfang Database, Web of Science, Cochrane Library, and MEDLINE electronic databases for articles published up to May 2023. Reference lists of the enrolled studies, Google Scholar, and scientific meeting reports were also manually searched.
Results
We enrolled 5641 patients with AOGCT from 33 eligible studies, including 1674 (29.7%) and 3967 (70.3%) patients in the CT and non-CT groups, respectively. Based on the cumulative results, adjuvant CT did not affect the risk of recurrence or progression (R/P) (odds ratio [OR]: 1.67, 95% confidence interval [CI]: 1.01–2.78, I2 = 63%, P = .05) and 5-year overall survival (OR: .86, 95% CI: .70–1.04, I2 = 0%, P = .12) of patients with AOGCT. However, adjuvant CT might reduce the 5-year disease-free survival (OR: 2.90, 95% CI: 1.19–7.08, I2 = 55%, P = .02). Sub-group analysis revealed that adjuvant CT did not affect the risk of R/P in patients with early-stage AOGCT (OR: .78, 95% CI: .43–1.43, I2 = 2%, P = .43) and advanced or recurrent AOGCT (OR: .78, 95% CI: .43–1.43, I2 = 2%, P = .43).
Conclusion
This meta-analysis suggests that patients with AOGCT might not benefit from adjuvant CT, even those with advanced or recurrent disease. The results should be interpreted with caution because of the inherent limitations of retrospective studies.
Keywords
Introduction
Granulosa cell tumor of the ovary (OGCT), a major sex cord–stromal neoplasm, is considered a low-grade malignancy, accounting for only 2% to 5% of all ovarian tumors. 1 It has an estimated incidence of approximately .47 to 1.6 per 100 000. 2 It is classified into the juvenile and adult types according to the differences in age of onset, clinical features, and histopathologic characteristics. Nearly 95% of cases belong to the adult subtype of OGCT (AOGCT), which occurs in the peri-menopausal and postmenopausal periods, with a peak prevalence at approximately 5 years of age. 3 It was first reported by Rokitansky 4 in 1855, who described it as a tumor with cells morphologically similar to granulosa cells of the ovary. It is characterized by indolent aggressive behavior, a prolonged natural course, and a tendency towards late recurrences, even in early-stage tumors. 5
Most AOGCTs are diagnosed at an early stage due to hyperestrogen-related clinical symptoms such as menorrhagia, amenorrhea, postmenopausal bleeding, or irregular menstruation. Surgery is the recommended treatment for resectable lesions. Patients with stage IA/B disease have a relatively favorable prognosis, and no additional adjuvant treatment other than surgery is required. Surgery remains the cornerstone treatment for advanced AOGCT (stage III/IV), and all visible lesions should be removed owing to the uncertainty of adjuvant treatment. AOGCT tends to recur late; up to 25% of all patients develop recurrences and require further treatment. 6 Although the most suitable therapeutic strategy for recurrent AOGCT is unknown, re-cytoreductive surgery is a common practice to relieve symptoms, prolong survival, or improve quality of life. 7 Whether surgery is more effective than chemotherapy or radiotherapy is unknown.8-12
Cytotoxic platinum-based chemotherapy (CT) is recommended for treating metastatic or recurrent AOGCT. The objective response rates (RRs) to multiple combined chemotherapy regimens have been reported to range from 50% to 92%;13-15 however, durable remission is infrequent.
16
Commonly, adjuvant CT is recommended for advanced AOGCT or high-risk early-stage tumors.
17
However, due to their indolent aggressive behavior, low incidence, protracted natural course, and tendency for late recurrences, it has been difficult to conduct prospective randomized studies to evaluate the prognostic significance of adjuvant CT. The uncertain benefits of adjuvant CT complicate its clinical selection. Some retrospective studies have suggested that adjuvant CT may be associated with prolonged disease-free survival (DFS) and possibly overall survival (OS) in AOGCT. However, most studies, including the latest study with the largest sample size based on the National Cancer Database (NCD)
18
and a preliminary narrative review by our team,
19
found that CT was not associated with improved survival rates. Whether adjuvant CT can improve the survival of patients with AOGCT remains unknown. We designed the present meta-analysis to quantitatively synthesize data from existing studies that compared oncologic and/or prognostic outcomes between CT and non-CT groups to solve the following clinical confusions: 1. Whether adjuvant CT could reduce the risk of recurrence or progression (R/P) of AOGCT? 2. Whether adjuvant CT could improve the 5-year DFS/OS of AOGCT? 3. Whether adjuvant CT could reduce the risk of R/P of early AOGCT? 4. Whether adjuvant CT could reduce the risk of R/P of advanced and/or recurrent AOGCT?
Materials and Methods
Literature Search
First, a normative protocol was established according to the standardized process of a systematic review and meta-analysis. The present study was approved and registered in the International Prospective Register of Systematic Reviews (PROSPERO; ID: CRD42023430867). 20 Our systematic review was performed step by step in accordance with the Preferred Reporting Items for Systematic Reviews and Meta examinations (PRISMA) statement. 21 Ethics/review board approval was not required because the present study was based exclusively on published literature. A comprehensive literature search was performed up to May 2023 using multiple mainstream databases with no limitations on region or language, including the Chinese National Knowledge Infrastructure (CNKI), Clinical Trials, Wanfang Database, Web of Science, Cochrane Library, and MEDLINE electronic databases. We sought to identify all studies that compared oncologic and prognostic outcomes between the CT and non-CT groups. We also searched BIOSIS Previews, the NLM Gateway, SIGLE, and GreyNet to avoid omitting unpublished negative results. Google Scholar, scientific meeting reports, and references of the enrolled studies were also searched to avoid omitting newly published data and internet resources.
Systematic literature retrieval was performed using MeSH using the following search terms: Granulosa Cell and (Tumor or Neoplasm or Tumors or Neoplasia or Neoplasias or Cancer or Cancers or Malignant Neoplasm or Malignancy or Malignancies or Malignant Neoplasms or Neoplasm, Malignant or Neoplasms, Malignant or Benign Neoplasms or Benign Neoplasm or Neoplasms, Benign or Neoplasm, Benign) and (Drug Therapy, Adjuvant or Adjuvant Chemotherapy or Adjuvant Drug Therapy) and ovary. Clinical studies comparing oncologic and prognostic outcomes between the adjuvant CT and non-CT groups of AOGCT were enrolled without time, territory, or language restrictions. We attempted to enroll prospective randomized controlled trials (RCTs) or quasi-RCTs; unfortunately, no RCTs were retrieved. Therefore, we included case–control, cohort, and observational studies. We gathered the relevant key factors for each study from the medical records and files, including the first author, research duration, sample size, FIGO stages, demographic characteristics, surgical data, methods and effects of adjuvant therapy, adverse events, time to recurrence or progression, DFS, and OS, if possible.
Two reviewers (YZ and HY) independently filtered all searched citations according to the predetermined inclusion and exclusion criteria. First, we excluded unmatched studies, including in vivo and/or in vitro studies, reviews, case reports, retrospective studies, observational studies, comments, letters by titles, and abstracts of citations. Second, full-text versions were obtained and further filtered according to the following inclusion criteria: a) original studies on AOGCT; b) outcomes included 5-year DFS, 5-year OS, recurrence, progression, or hazard ratio (HR) for survival; c) comparative outcomes between the CT and non-CT groups; and d) availability of full text. Any disagreements were resolved through mutual consultation.
Statistical Analysis
Heterogeneity among primary studies was assessed using Higgins I2, which measured the degree of dissimilarity among individual study results. 22 I2 was evaluated as follows: I2 = (Q−df)/Q×100%. Q-value was calculated by summing the squared deviations of the estimate of each study from the overall estimate, and df was degrees of freedom. I2 <50% was considered insignificant heterogeneity, and a fixed-effect model was adopted to poll the results. I2 >50% was considered substantial heterogeneity, and random-effect models were used. Recurrence was defined as radiological reappearance after the complete disappearance of the lesion due to previous treatment. Progression was defined as the emergence of new lesions or >20% enlargement on the sum measurement of the target lesions using the radiological method. Disease-free survival (DFS) was defined as the period between diagnosis and radiological recurrence or DFS reported in the original article. OS was defined as the period between diagnosis and death from any cause or the OS reported in the original article.
A funnel plot, a scatterplot of the odds ratio (OR) of individual studies on the x-axis against the standard error (SE) of the log OR on the y-axis, was generated to assess publication bias. 23 A symmetrical inverted funnel indicated the absence of publication bias, whereas asymmetry indicated potential publication bias. Sensitivity analysis was conducted for all outcomes to determine whether the conclusions were stable. Sensitivity analysis was performed by eliminating a single study step-by-step to survey whether the cumulative results changed significantly.
The meta-analysis was performed using Review Manager 5.3, and statistical significance was set at P < .05.
Results
Evidence Acquisition
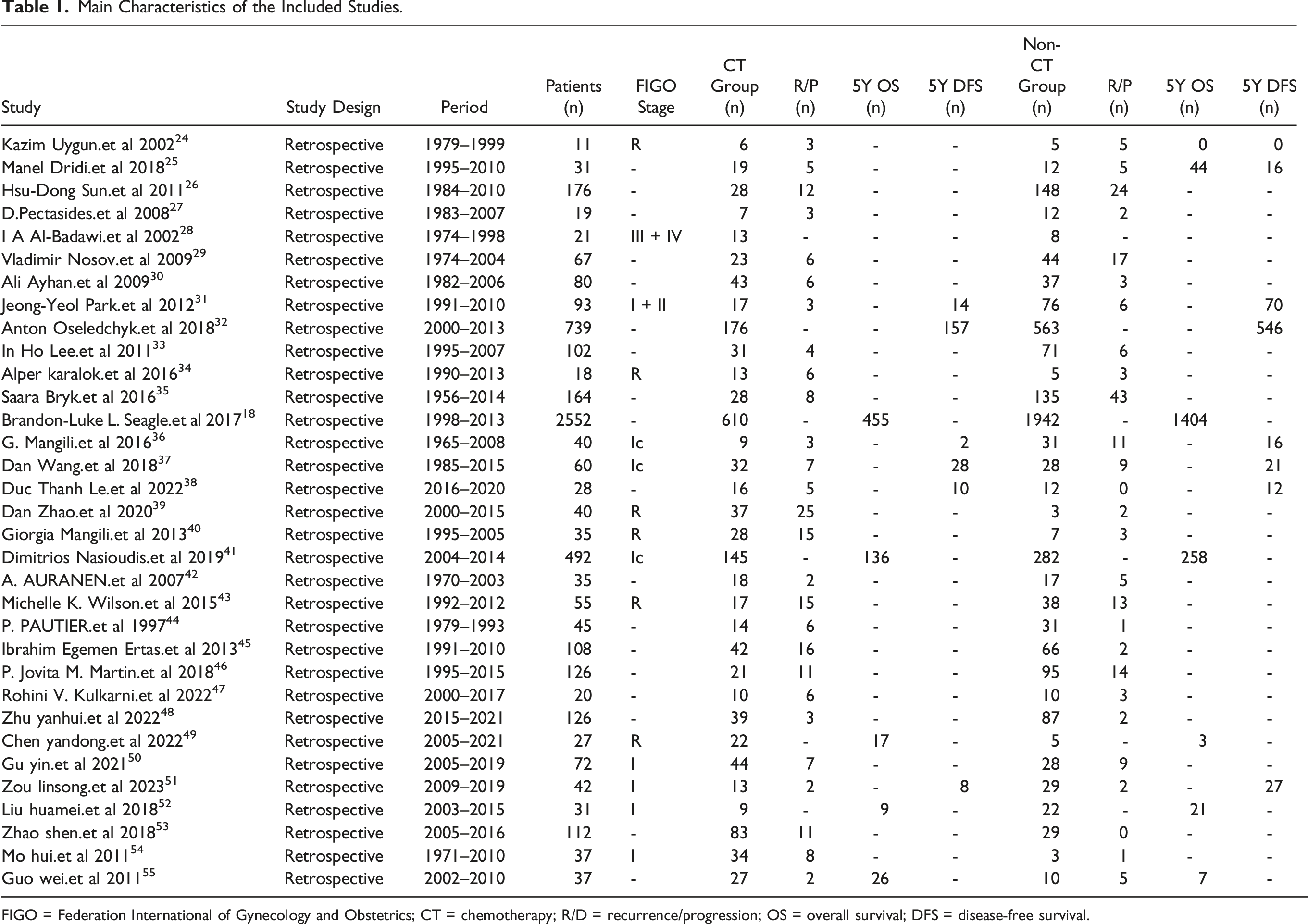
By systematically searching multiple databases, we retrieved 1792 literature, and 33 retrospective comparative studies were independently enrolled after filtering according to predetermined inclusion and exclusion criteria. The detailed evidence acquisition course is shown in Figure 1. The main features of the included studies are presented in Table 1. Overall, 5641 patients with AOGCT were enrolled, including 1674 (29.7%) and 3967 (70.3%) in the CT and non-CT groups, respectively. Flow diagram for evidence acquisition in this systematic review and meta-analysis. Main Characteristics of the Included Studies. FIGO = Federation International of Gynecology and Obstetrics; CT = chemotherapy; R/D = recurrence/progression; OS = overall survival; DFS = disease-free survival.
Evidence Synthesis
We enrolled 5641 patients with AOGCT from 33 retrospective studies. Twenty-seven studies compared the risk of R/P between CT and non-CT groups of AOGCT, 6 studies analyzed 5-year DFS between CT and non-CT groups, 5 analyzed 5-year OS between CT and non-CT groups, 7 compared the risk of R/P between CT and non-CT groups of early AOGCT, and 6 compared the risk of R/P between CT and non-CT groups for advanced and recurrent AOGCT.
Pooled results suggested that adjuvant CT did not reduce the risk of R/P (OR: 1.67, 95% CI: 1.01–2.78, I2 = 63%, P = .05) (Figure 2A). We performed a sensitivity test by culling individual studies separately and found insignificant changes in the pooled results and I2, indicating that the results were reliable. We found that adjuvant CT did not improve 5-year DFS of AOGCT (OR: 2.90, 95% CI: 1.19–7.08, I2 = 55%, P = .02) (Figure 2B); contrarily, we found that 5-year DFS in the CT group was significantly lower than that in the non-CT group, which might have been due to more number of poor prognostic patients in the CT group. A sensitivity test was performed after excluding the study by Wang et al; I2 reduced to 0% and the 5-year DFS in the CT group was still significantly lower than that in the non-CT group (OR: 4.30, 95% CI: 2.51–7.35, P < .00001), indicating that the pooled result was stable and reliable. We also found that adjuvant CT did not improve 5-year OS of AOGCT (OR: .86, 95% CI: .70–1.04, I2 = 0%, P = .12) (Figure 2C). We performed a sensitivity test by culling individual studies and found insignificant changes in the pooled results and I2, indicating that the results were reliable. Effect of adjuvant CT on AOGCT. (A) Forest plot of adjuvant CT for risk of R/P of AOGCT. (B) Forest plot of adjuvant CT for 5-year DFS of AOGCT. (C) Forest plot of adjuvant CT for 5-year OS of AOGCT.
As mentioned previously, the FIGO stage seemed to be the most important predictor of recurrence or progression. To avoid possible confounding factors, we performed a subanalysis of tumors with similar FIGO stages. We found that adjuvant CT did not affect the risk of R/P of early (stage I or II) AOGCT (OR: .78, 95% CI: .43–1.43, I2 = 2%, P = .43) (Figure 3A). We performed a sensitivity test by culling individual studies and found insignificant changes in the pooled results and I2, indicating that the results were reliable. We also found that adjuvant CT did not affect the risk of R/P of advanced (stage III or IV) and recurrent AOGCT (OR: 1.69, 95% CI: .46–6.25, I2 = 55%, P = .43) (Figure 3B). A sensitivity test was performed by culling individual studies and the pooled results and I2 showed insignificant changes, indicating that the results were reliable. Given the importance of surgery, we tried to restrict our analysis to similar surgical outcomes; unfortunately, only 2 studies compared the risk of R/P between the CT group and non-CT group in AOGCT with optimal cytoreduction. The pooled result suggested that adjuvant CT did not reduce the risk of R/P (OR: 3.74, 95% CI: 1.55–8.98, I2 = 0%, P = .003) (Figure 3C). We could not extract oncologic and prognostic outcomes in AOGCT with unresectable lesions because of incomplete outcome data from the original studies. Subgroup analysis of the effect of adjuvant CT on AOGCT. (A) Forest plot of adjuvant CT for risk of R/P during early stage AOGCT. (B) Forest plot of adjuvant CT for risk of R/P during advanced and recurrent AOGCT. (C) Forest plot of adjuvant CT for risk of R/P during unresectable AOGCT.
Methodological Quality and Risk of Bias Assessment
All the included 33 retrospective studies had low bias. The score assessment based on the Newcastle-Ottawa Scale was high in the 33 enrolled studies (Figure 4A). Most studies had a relatively low quality of selection, high quality of comparability, and exposure. Overall, the heterogeneity in the enrolled studies was relatively low in terms of 5-year DFS/OS (I2 = 0%), whereas it was relatively high in the enrolled studies related in terms of the risk of R/P (I2 = 63%). There was an insignificant publication bias for the eligible studies in terms of the risk of R/P because the funnel plot was symmetrical (Figure 4B). However, there was a significant publication bias for eligible studies related to 5-year DFS/OS outcomes because the funnel plots were asymmetrical (Figure 4C and D). Assessment of quality and publication bias. (A) Quality assessment of NOS for cohort studies. (B) Funnel plot of adjuvant CT for risk of R/P of AOGCT. (C) Funnel plot of adjuvant CT for 5-year DFS of AOGCT. (D) Funnel plot of adjuvant CT for 5-year OS of AOGCT.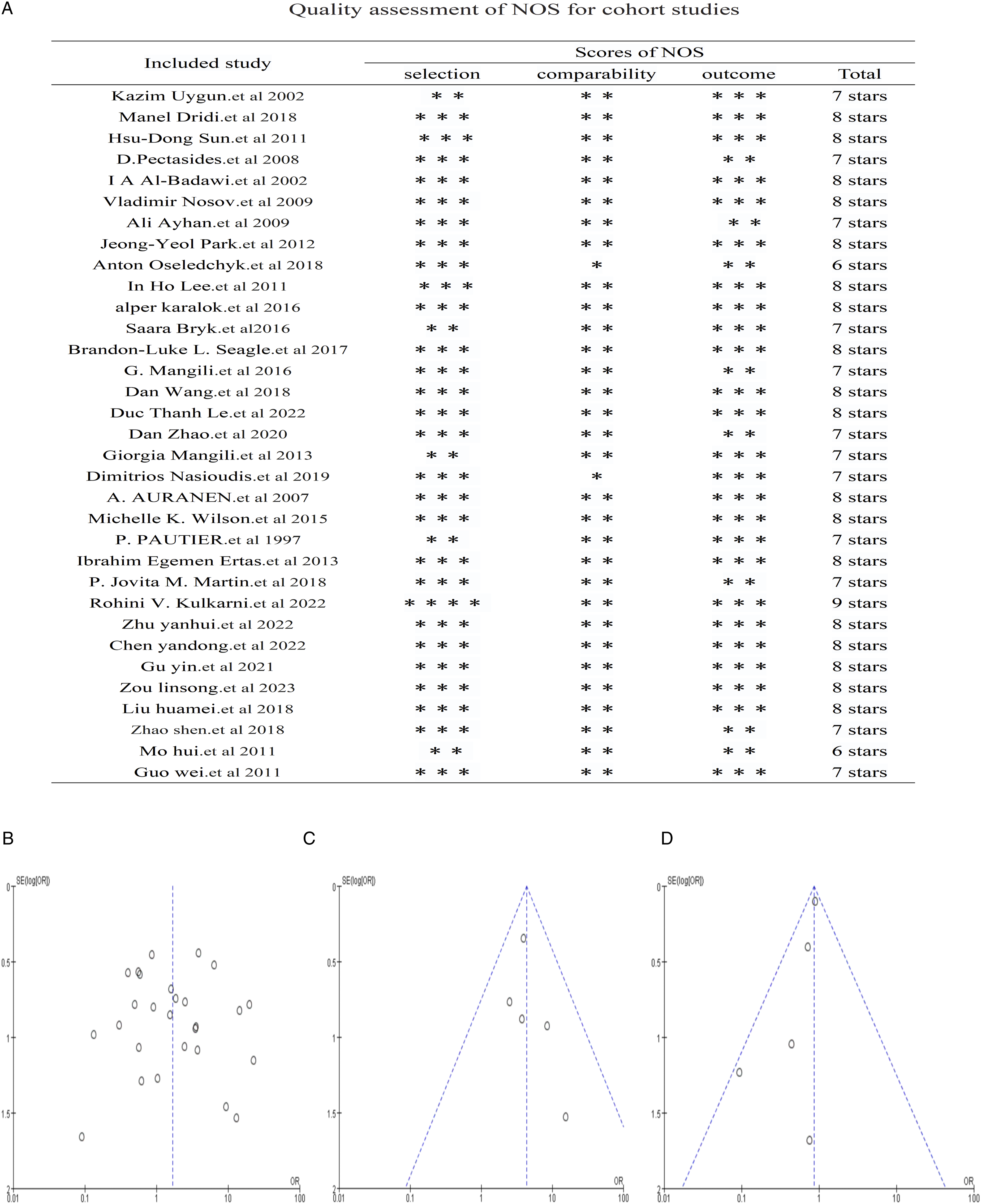
Discussion
The present meta-analysis quantitatively synthesized all existing studies that evaluated the effect of adjuvant CT on AOGCT and found noteworthy results. We reported reliable outcomes of the largest series (N = 5641 patients) of AOGCT till date. To the best of our knowledge, this is the first meta-analysis to evaluate the prognostic effect of adjuvant CT on AOGCT. We also found that adjuvant CT did not improve the oncological or survival outcomes of AOGCT. Third, owing to incomplete outcome data from the original studies, we could not evaluate the effect of CT on unresectable AOGCT.
AOGCT accounts for no more than 5.0% of ovarian tumors and is identified as a rare tumor. However, AOGCT is a major sex cord–stromal ovarian tumor. AOGCT has unique characteristics that differ from those of usual epithelial ovarian tumors and carries a unique molecular profile with missense mutation 402C-> G (C134 W) in the transcription factor forkhead box L2 (FOXL2). 56 It is characterized by the secretion of steroid hormones, indolent aggressive behavior, prolonged natural course, and a tendency towards late recurrences. It is usually diagnosed at an early stage and has a relatively good prognosis after complete resection, without any adjuvant treatment. Advanced and recurrent AOGCT has a poor prognosis. AOGCT tends to have late recurrences, even during the early stage. Almost 70% of patients with recurrent AOGCT die from the disease. 57 The optimal treatment for advanced and recurrent AOGCT is unknown, and there is no proven effective treatment except surgery. Chemotherapy is commonly recommended for recurrent, advanced, and metastatic AOGCT, although the optimal treatment regimen remains to be determined. However, the benefits of adjuvant CT have not been confirmed by reliable evidence from RCTs. Whether adjuvant CT can improve the survival of patients with AOGCT remains unknown.
In recent years, the proportion of AOGCT treated with CT has increased annually. Nasioudis et al 41 retrospectively analyzed high-risk early AOGCT (stage IC) from the NCD between 2004 and 2015 and found that the proportion gradually increased from 22.7% in 2007 to 39.2% in 2015. Early clinical studies found that AOGCT was chemotherapy-sensitive, with a high overall response rate to different combination CT regimens; however, durable remission appeared to be <50%. The effect of CT on the oncological outcomes or prognosis of AOGCT based on existing retrospective studies is inconsistent. A systematic literature review revealed that most studies did not support the benefits of adjuvant CT. Uygun et al 24 retrospectively analyzed the treatment results of 11 recurrent AOGCTs and found that the R/P ratio in the CT group was lower than that in the non-CT group, without a statistical difference. Similarly, Dridi et al, 25 Auranen et al, 42 Karalok et al, 34 Wang et al, 37 and others found that the R/P ratio in the CT group was lower than that in the non-CT group, without a statistical difference. Ayhan et al, 30 Pectasides et al, 27 Lee et al,33,54 and others found that the R/P ratio in the CT group was higher than that in the non-CT group, without a statistical difference. However, Wei et al 55 found the R/P ratio in the CT group was significantly lower than that in the non-CT group. Sun et al, 26 Ertas et al, 45 Wilson et al, 43 and others found that the R/P ratio in the CT group was significantly higher than that in the non-CT group. Similar confusion also existed when it came to the prognostic outcomes such as 5-year DFS/OS. Inconsistent results regarding the effect of CT make the treatment choice for AOGCT difficult. Considering the potential toxic effects of CT, reliable evidence is urgently required. The rarity, indolent aggressive behavior, prolonged natural course, and tendency towards late recurrence make it difficult to conduct prospective controlled studies. Our meta-analysis provides more reliable evidence than the individual studies.
By pooling the existing studies, we found that adjuvant CT did not reduce the risk of R/P, which was consistent with most previous studies; AOGCT had a tendency for late relapse, extraovarian spread, and recurrence might occur as long as 44 years after initial treatment. 58 The risk factors related to recurrence are unknown. Owing to the indolent behavior and prolonged recurrence interval, we speculated that inherent biological characteristics might be the major prognostic factors, which might not be altered by chemotherapy. To avoid possible confounding factors, we performed a subanalysis of similar FIGO stages. We found that adjuvant CT did not reduce the risk of R/P in early, advanced, and recurrent AOGCT. Given the mainstay importance of surgery, cytoreduction without complete excision may significantly reduce the prognosis. Therefore, we attempted to restrict our analysis to similar surgical outcomes; however, only 2 studies evaluated the effect of CT during AOGCT with optimal cytoreduction, and the pooled results suggested that adjuvant CT did not reduce the risk of R/P. Owing to the limitations of the original studies, we could not extract the oncological and prognostic outcomes of unresectable AOGCT. The effect of CT on AOGCT with gross residual lesions after surgery could not be determined in this meta-analysis.
FIGO stage is the most important prognostic factor in AOGCT. Various factors are related to the prognostic outcomes including age, rupture of tumor, tumor size, residual tumor after surgery, non-staging surgery, mitotic activity, aneuploidy, over-expression of p53 and Ki-67, FIGO stage, and nuclear atypia. However, only stage had shown a consistent effect on the survival of patients with AOGCT in the existing literature. Early AOGCT had a relatively favorable prognosis with a 5-year OS of approximately 95%; however, it reduced to approximately 50% for advanced-stage AOGCT.59-61 Adjuvant CT was commonly recommended for high-risk and advanced AOGCT. However, the survival benefits of adjuvant CT were not supported by prospective studies. After a systematic review of the literature, we found that the results from existing retrospective studies were inconsistent. Oseledchyk et al 32 and Linsong et al 51 found that the 5-year DFS in the CT group was significantly lower than that in the non-CT group. Le et al, 38 Mangili et al, 36 and Park et al 31 found that the 5-year DFS in the CT group was lower than that in the non-CT group without statistical significance. The pooled result suggested that adjuvant CT did not improve the 5-year DFS of AOGCT. Contrarily, we found that 5-year DFS in the CT group was significantly lower than that in the non-CT group. By analyzing the original studies carefully, we found that patients who received adjuvant CT had poorer prognosis. Seagle et al, 18 Yandong et al, 49 Nasioudis et al, 41 Wei et al, 55 and Huamei et al 52 found that the 5-year OS in the CT group was higher than that in the non-CT group without statistical significance. The pooled result also suggested that adjuvant CT did not improve the 5-year OS of AOGCT.
The important aim of this meta-analysis was to quantitatively synthesize all existing studies to resolve key clinical questions. The key question that we focused on was whether the administration of CT could improve the oncological and prognostic outcomes of AOGCT. However, oncological outcomes are influenced by demographic characteristics, clinicopathological factors, and treatment details. There were highly variable prognostic factors between studies. This was the most important limitation of our meta-analysis. Another important limitation was that most of the enrolled studies were unicentric retrospective studies; the limited number of cases and selection bias may have significantly affected the final results.
The main strengths of our meta-analysis include being the first systematic review on the effect of adjuvant CT for AOGCT with the largest sample size and the evaluation of 5-year prognostic indicators. Additionally, 4 main points of our meta-analysis must be addressed. First, our meta-analysis did not support the benefits of adjuvant CT for AOGCT, even in advanced and recurrent cases. However, the results should be viewed with caution because all the enrolled studies were retrospective studies with inherent selective biases. Second, we could not extract the treatments and results for unresectable AOGCT. The effect of CT on AOGCT with a poor prognosis is unclear, and prospective studies in these patients are urgently needed. Third, various novel non-CT treatments have been increasingly adopted by patients with AOGCT during the past decades, such as hormonal therapies 62 and novel targeted approaches.63-65 These novel therapies might be effective and might improve survival. The present meta-analysis did not consider this because only a few patients received novel therapies. Fourth, it was impossible to evaluate the adverse events of CT because these data were not available for most of the included studies. We considered that adverse events might affect the prognosis of patients with AOGCT.
Conclusion
The present meta-analysis systematically reviewed and quantitatively synthesized the current evidence on the effect of adjuvant CT in AOGCT. AOGCT is characterized by the secretion of steroid hormones, indolent or aggressive behavior, a prolonged natural course, and late recurrences. AOGCTs have a relatively favorable prognosis during the early stages; however, the prognosis of advanced and recurrent AOGCT is poor. The present meta-analysis found that the administration of adjuvant CT did not improve the oncological and prognostic outcomes of AOGCT, regardless of whether the patients had early or advanced/recurrent disease. However, the effect of CT on unresectable AOGCT could not be determined. The results should be interpreted with caution because of the limited number of cases and selection bias of the enrolled studies. Considering the potentially toxic effects of CT, reliable evidence is urgently required to determine the effect of CT on AOGCT.
Footnotes
Authors’ Contributions
HY and YZ were major contributors in writing the manuscript and retrieving the literature. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Medical Science and Technology Research Foundation of Guangdong Province (A2022317).
