Abstract
Background:
Previous studies have evaluated the diagnostic value of serum glypican-3 in patients with hepatocellular carcinoma. However, the results remain inconsistent and even controversial. Thus, the aim of the present meta-analysis was to clarify the diagnostic accuracy of serum glypican-3 for hepatocellular carcinoma.
Methods:
A meta-analysis including 22 studies was performed with 2325 cases and 2280 controls. Relevant studies were searched in the EMBASE, PubMed, and Web of Science databases, covering relevant papers published until November 1, 2017. The quality of the studies was assessed by revised QUADAS tools. Sensitivity, specificity, and other measures were pooled and determined to evaluate the accuracy of serum glypican-3 in the diagnosis of hepatocellular carcinoma by random-effects models. Summary receiver operating characteristic curve (sROC) analysis was performed to summarize the overall test performance.
Results:
The results showed that the pooled overall diagnostic sensitivity, specificity, and 95% confidence interval (CI) for serum glypican-3 in the diagnosis of hepatocellular carcinoma were 68% (56-79%) and 92% (82-96.0%), respectively. Besides, the summary diagnostic odds ratio and 95% CI for glypican-3 were 23.53 (8.57-64.63). In addition, the area under sROC and 95% CI was 0.87 (0.84-0.90). The major design deficiencies of included studies were differential verification bias, and a lack of clear exclusion and inclusion criteria.
Conclusions:
The results of this meta-analysis suggested that serum glypican-3 was acceptable as a moderate diagnostic marker in the diagnosis of hepatocellular carcinoma compared with healthy individuals, which could elevate the sensitivity and specificity of diagnosis. Furthermore, more well-designed studies with large sample sizes are needed to show the effectiveness of glypican-3 in the differential diagnosis of hepatocellular carcinoma.
Introduction
Hepatocellular carcinoma (HCC) is the sixth most frequently diagnosed malignant neoplasm, and the second leading cause of tumor death in the world.1-3 The prognosis of patients with HCC was generally poor, with a 5-year overall survival rate of less than 10%–15% worldwide, because of lack of effective and timely diagnostic methods.3,4 Therefore, early detection of HCC at an early stage has become increasingly important, to reduce HCC-associated mortality, improve the survival rate of patients and increase the opportunity of effective medical interventions.5,6
The European Research Association for the Study of the Liver suggested that liver imaging (including computed tomography, magnetic resonance imaging, and ultrasound) and serum alpha-fetoprotein (AFP) level tests have been widely used in clinical practice for the diagnosis of HCC. 7 However, the diagnostic performance of AFP is not satisfactory with the range of low sensitivity only ranging from 20% to 65% at the commonly used cut-off of 20 ng/mL.8-10 Imaging examination, as an indirect diagnostic method with good accuracy, completely depended on skills and experience of the radiologists and physicians to distinguish HCC from non-neoplastic nodules. Hence, a novel and reliable biomarker to complement AFP is needed to achieve high diagnostic accuracy of HCC. 11
As a glypican family of heparan sulfate proteoglycans, glypican-3 (GPC-3) plays an important role in cellular growth, and cell differentiation and migration.12,13 Previous studies have proved that GPC-3 is normally expressed in fetal liver and placenta; has negligible normal expression in healthy individuals and hepatitis patients; and is highly expressed in hepatocellular cancer cells.14-16 Many studies have reported that the expression of GPC-3 was increased in the tumor tissues of HCCs compared with healthy people with hepatitis. Immunostaining for GPC-3 in biopsy or resected tissue specimens has been regarded as a useful tool for diagnosing HCC.16-17 However, other studies on the diagnostic performance of serum GPC-3 for HCC have reported unclear and even conflicting results.18-20
Based on a systematic review and meta-analysis, we collected relevant research in recent years about the diagnostic performance of serum GPC-3, with the aim of exploring the diagnostic value of GPC-3 in HCC.
Materials and methods
Studies were searched in the electronic databases EMBASE, PubMed, and Web of Science up to November 1, 2017. Available publications were identified using the following keywords or text words: “GPC-3,” “glypican-3,” or “serum” and “blood,” or “HCC” and “hepatocellular carcinoma,” or “liver cancer,” or “liver cell carcinoma.” The most eligible literature was retrieved by assessing all relevant studies. Also, we hand searched the relevant research from reference lists of selected articles to identify the more relevant publications and to avoid those with irrelevant information.
Criteria for inclusion and exclusion of published studies
The included studies were required to meet this criteria: (i) a case-control, nested case-control, or cohort design; (ii) the diagnostic value of serum GPC-3 in patients with HCC was evaluated; (iii) genotype data was available for extraction to calculate sensitivity, specificity, and other measures; and (iv) when duplications existed or the same patients appeared in several publications, we only chose the most recent or complete study in the meta-analysis. The major exclusion criteria were: (i) no available genotype frequency data; (ii) non-case-control studies, case reports, letters, reviewed editorial articles; and (iii) duplicated publications with previous studies.
Data extraction
Two researchers independently inspected the studies and extracted appropriate information and data with a standard protocol to ensure the reliability and the accuracy of the results. The results were then reviewed by a third investigator. We extracted the following information from each study: name of first author, publication date, country, ethnicity, characteristics of HCC, characteristics of controls, assay type, cut-off value, HCC value (ng/mL), controls value (ng/mL), HCC/non-HCC, and raw data including true positive (TP), true negative (TN), false positive (FP), and false negative (FN) results. The quality of each reference was also evaluated by two investigators independently, according to the Cochrane Handbook. 21
Statistical analysis
The sensitivity, specificity, positive likelihood ratios (LR+), and negative likelihood ratios (LR−), as well as their corresponding 95% confidence intervals (95% CIs), were presented to assess the diagnostic value of serum GPC-3 in patients with HCC. The data were visualized as forest plots and receiver operating characteristic (ROC) curves. The X2-based Q-statistic was utilized to assess the Between-study heterogeneity, and P<0.05 was deemed statistically significant. The I2-square value, a quantitative measurement of inconsistency across different studies, 22 was also calculated. I2-square value typically ranges from 0% (no observed heterogeneity) to 100% (maximal heterogeneity). If heterogeneity across studies was not identified, the fixed-effects model was used for the meta-analysis. Otherwise, the random-effects model (DerSimonian-Laird method) was used. Also, publication bias was investigated using Deeks’ funnel plot asymmetry test. 23 When the P value of the Egger test was <0.05, statistical significance was defined. All statistical analysis was conducted by the statistical software STATA version 12.0 (StataCorp LP, College Station, TX, USA).
Results
Studies characteristics
As shown in Figure 1, 762 records of GPC-3 and HCC were retrieved. After screening titles and abstracts of relevant articles, 740 articles were excluded because they were not related to the inclusion criteria. Finally, 22 retrospective case-control studies published between 2003 and 2017 were included in the meta-analysis.24-43
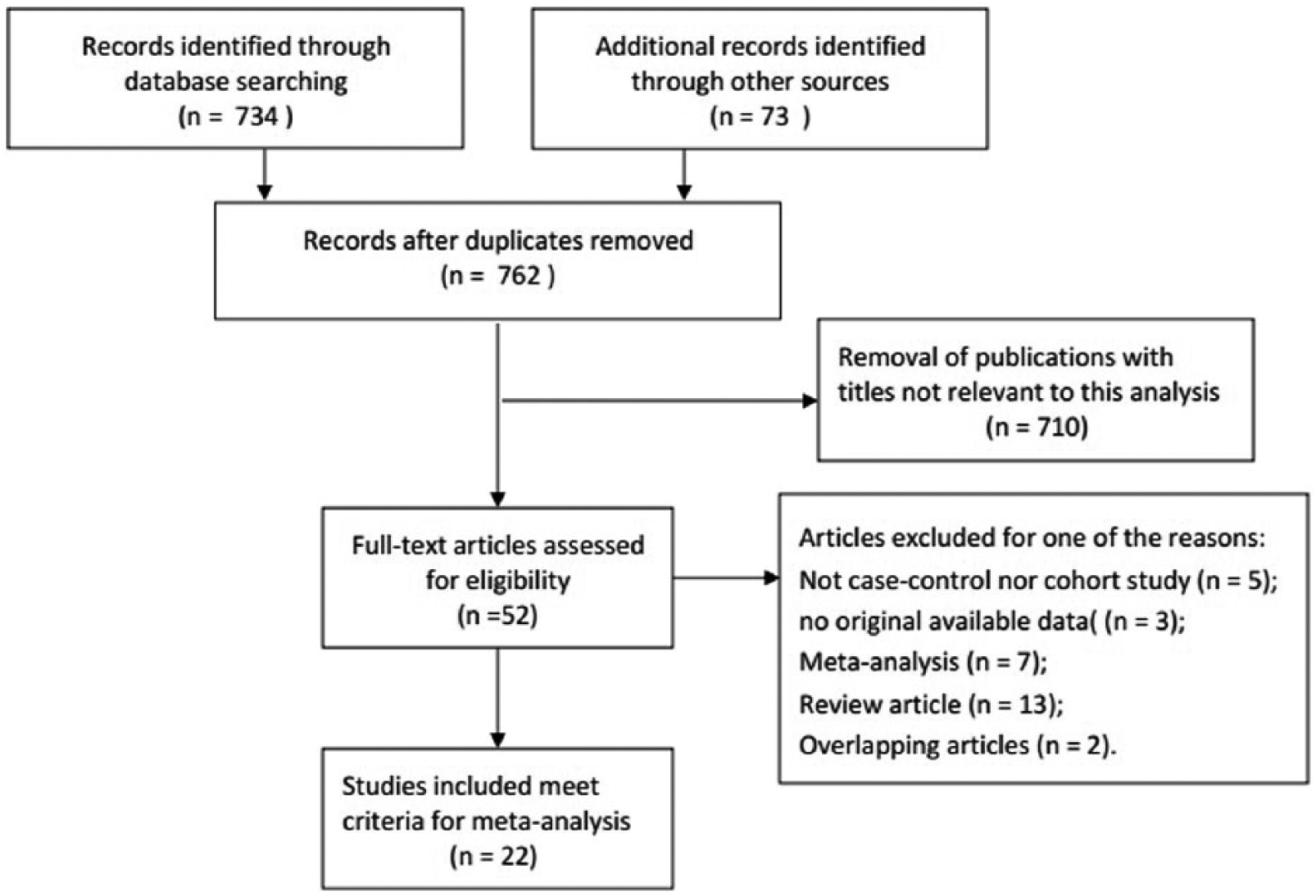
Flow diagram of selection process.
The present meta-analysis included 2325 cases of patients with HCC and 2280 controls from a total of 22 case-control studies that evaluated the diagnostic value of serum GPC-3 in patients with HCC; the detailed data of each study are listed in Table 1. The flowchart of the literature search and selection process is shown in Figure 1. In all the literature, HCC patients with or without the hepatitis B virus (HBV) or hepatitis C virus (HCV), and liver cirrhosis patients with or without HBV or HCV in the control group were included. In these studies, assay types were applied, such as enzyme-linked immunosorbent assay, chemiluminescent immunoassay, and immunoassay, to assay the diagnostic value of serum GPC-3 in patients with HCC. Also, 14 studies were conducted on the Asian population, 2 on the Caucasian population, and the remaining 6 studies on the African population.
Characteristics and methodology assessment of the individual studies included in the meta-analysis.
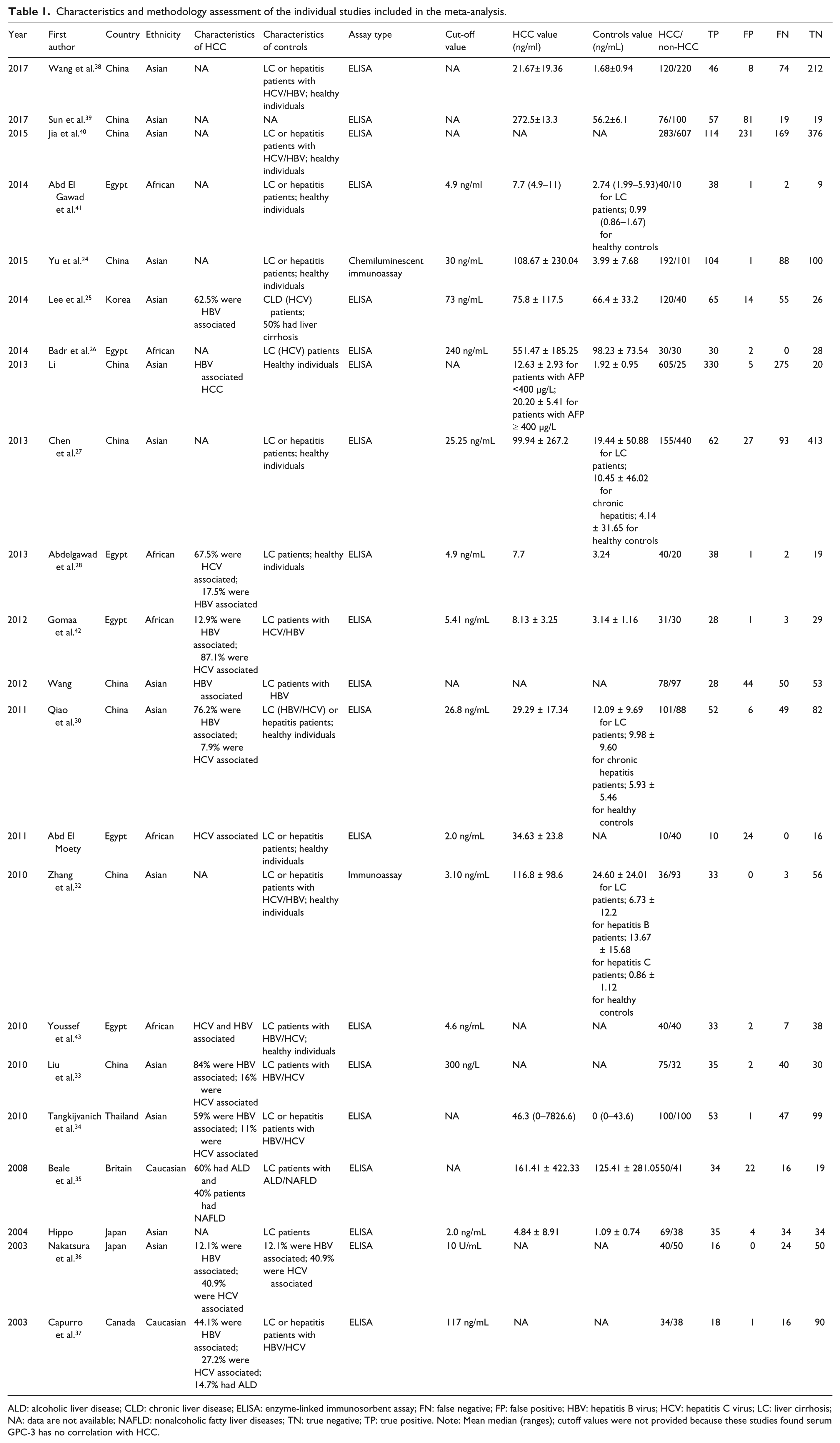
ALD: alcoholic liver disease; CLD: chronic liver disease; ELISA: enzyme-linked immunosorbent assay; FN: false negative; FP: false positive; HBV: hepatitis B virus; HCV: hepatitis C virus; LC: liver cirrhosis; NA: data are not available; NAFLD: nonalcoholic fatty liver diseases; TN: true negative; TP: true positive. Note: Mean median (ranges); cutoff values were not provided because these studies found serum GPC-3 has no correlation with HCC.
Quantitative synthesis results
In this meta-analysis, the random-effects model was selected to calculate the sensitivity, specificity, LR+, and LR− with corresponding 95% CIs, because the between-study heterogeneity was obvious (P<0.05). The meta-analytic results showed that the pooled overall diagnostic sensitivity, specificity, and 95% CI regarding serum GPC-3 in the diagnosis of HCC were 68% (56-79%) and 92% (82-96.0%), respectively (Figure 2). The summary diagnostic odds ratio (DOR) and 95% CIs for GPC-3 were 23.53 (8.57-64.63). In addition, the area under summary receiver operating characteristic (sROC) curves and 95% CI was 0.87 (0.84-0.90) (Figure 3). The major design deficiencies of the included studies were (i) differential verification bias; and (ii) a lack of clear exclusion and inclusion criteria. In total, in order to evaluate the diagnostic value of serum GPC-3 in patients with HCC, serum GPC-3 was acceptable as a moderate diagnostic marker in the diagnosis of HCC.
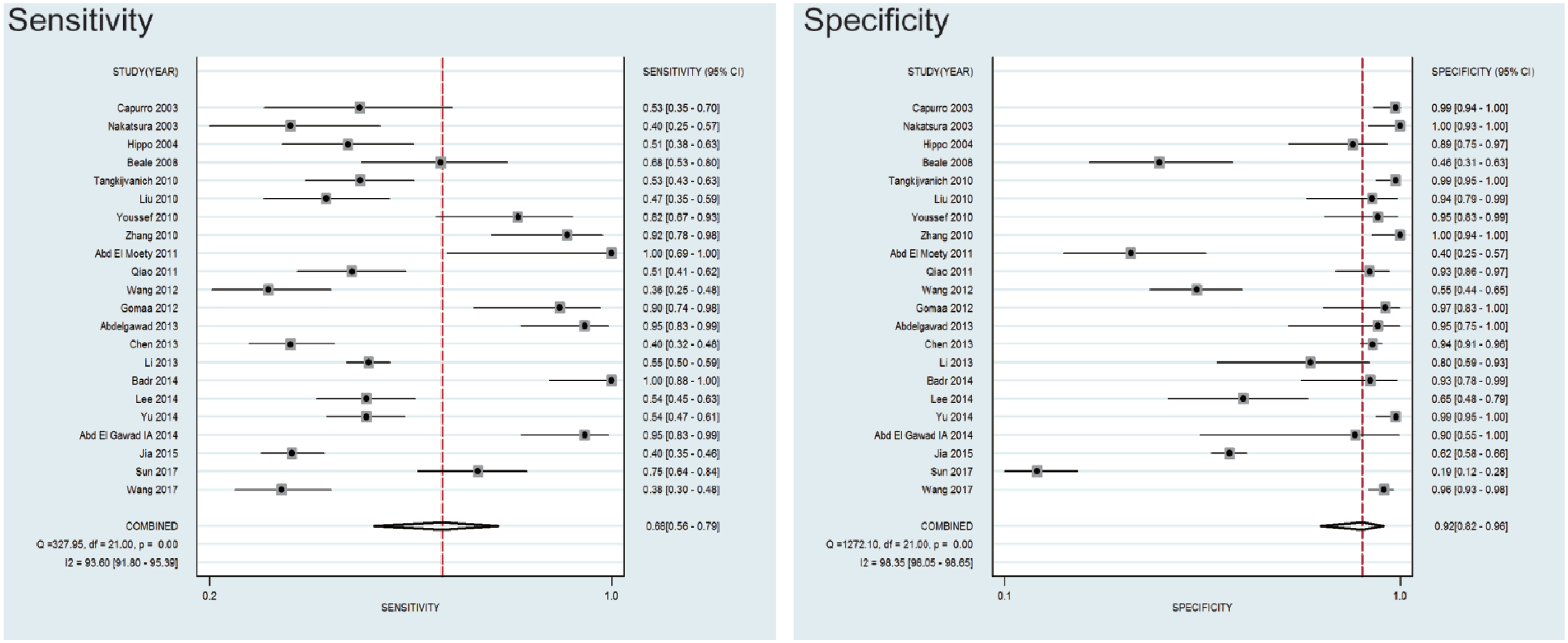
Forest plots of sensitivity and specificity of by GPC-3 as a diagnostic marker for HCC in the 22 studies included in the meta-analysis. Each solid circle represents an eligible study. The size of each solid circle reflects the sample size of each eligible study. Error bars represent 95% CI.
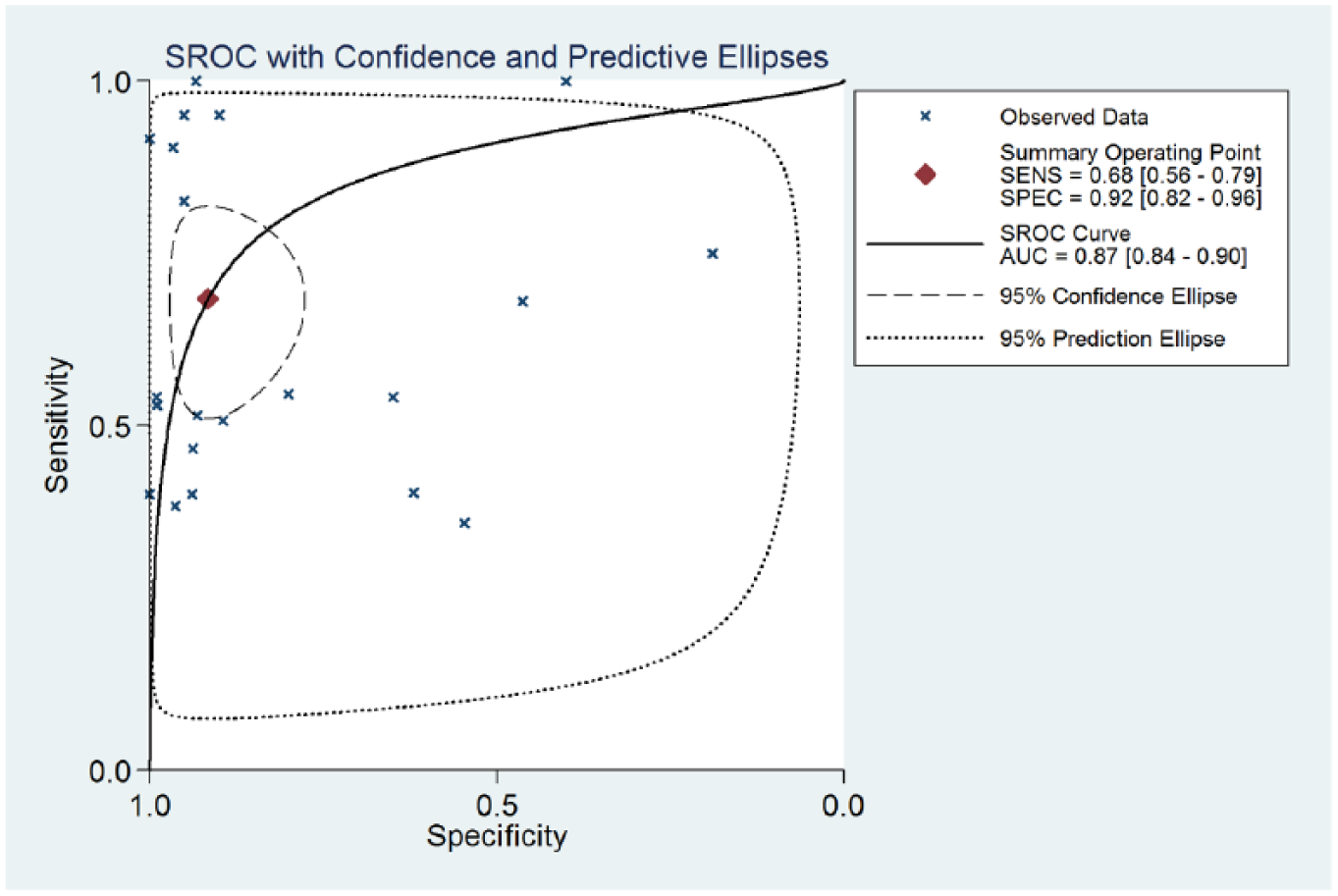
Summary receiver operating characteristic curves (sROC) from the hierarchical summary receiver operating characteristic model generated from the 22 studies that found that GPC-3 is a diagnostic marker for HCC. Each solid circle represents an eligible study. The size of each solid circle represents the sample size of each eligible study. The overall diagnostic efficiency is summarized by the regression curve.
Test of heterogeneity
For sensitivity, specificity, LR+, LR−, and DOR, the I2-square was 93.6%, 98.3%, 97.8%, 95.7%, and 100%, respectively, which indicated that significant heterogeneity between eligible studies was observed. To verify whether the heterogeneity could be interpreted by a threshold effect, the Spearman approach was applied by difference cut-off value, objective method, population, or others. In this meta-analysis, Spearman’s correlation coefficient between the logic of sensitivity and the logic of 1-specificity was 0.053, and the P value was 0.940, which indicated that heterogeneity was not explained by the threshold effect.
Publication bias
The potential publication bias for all the included studies was evaluated by the Deeks’ funnel plot asymmetry test. The slope coefficient of the regression line had a P value of 0.922, indicating that the data were symmetric and did not have a likelihood of publication bias (Figure 4).
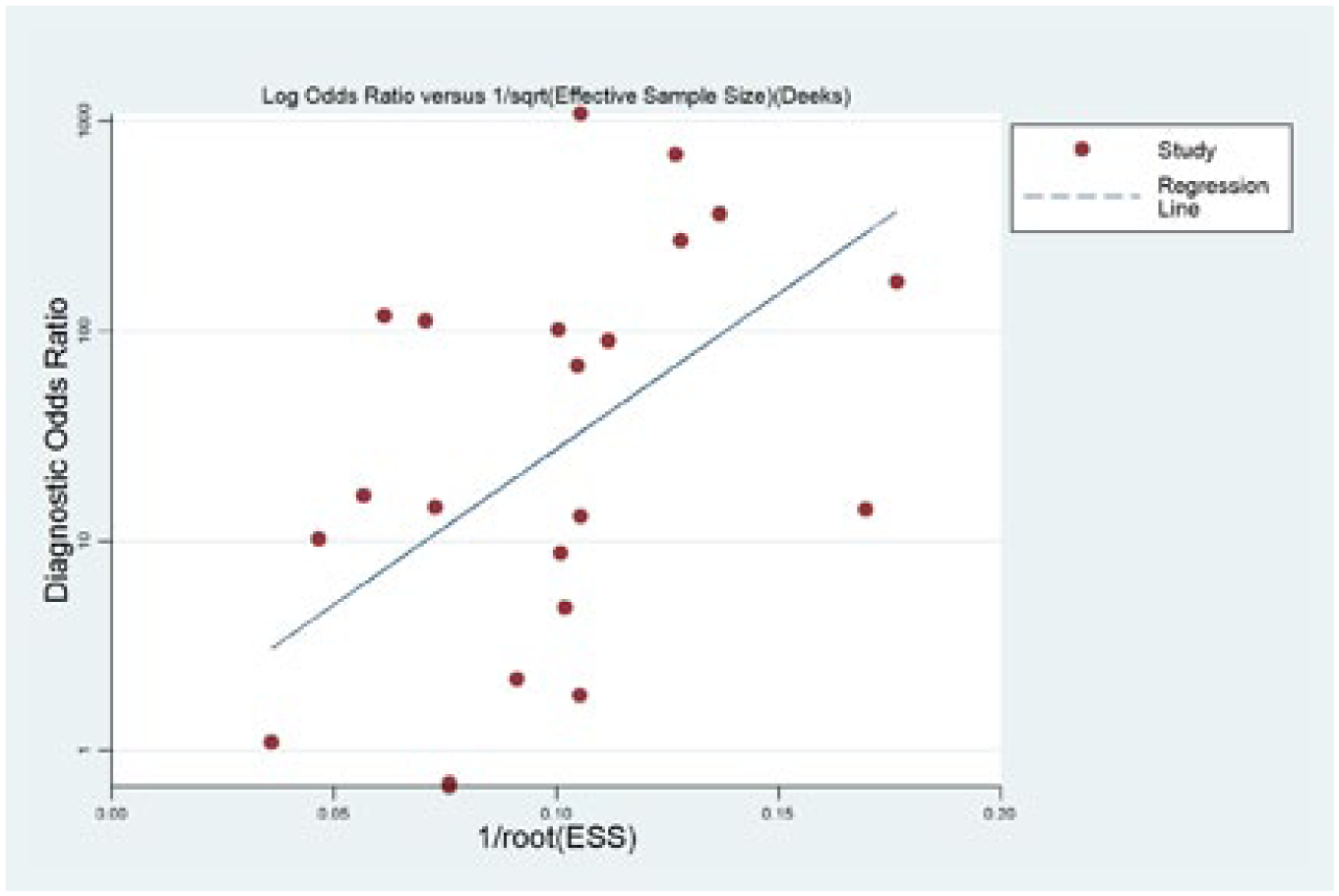
Linear regression test of funnel plot asymmetry. The statistically non-significant P value of the slope coefficient indicates symmetry of the data and a low likelihood of publication bias.
Discussion
In recent years, an increasing number of studies in cancer research have focused on the field of tumor diagnosis, which included serum marker detection, tissue pathology, imaging, and more. 44 It is common knowledge that the most ideal methods in the field of cancer screening should be specific, non-invasive, and convenient, especially at an early stage. 44 Considering that serum biomarkers play important roles in the diagnosis of cancer, it might be regarded as potential biomarkers in HCC diagnosis. 45 However, due to the limitation of low sensitivity alpha-fetoprotein (AFP), novel and reliable markers are widely used serum marker for diagnosis of HCC, there is an urgent need to find a new biomarker to improve the diagnostic accuracy for HCC. 11 As a candidate serum marker for HCC patients, several recent studies have shown that GPC-3 significantly inhibited the development of embryos; however, it was highly expressed in HCC tissue and may become one of the novel promising serum markers to promote cell growth by stimulating Wnt signaling. 8
Recently, a growing number of studies have investigated the relationship between the diagnostic value of serum GPC-3 and patients with HCC.14-20 However, the results were inconclusive and even conflicting. Yu et al. 24 found that an applicable chemiluminescent immunoassay with stable performance against GPC-3 in diagnosing HCC had been established, and the combination of GPC-3 with CK19 and AFP could improve the diagnostic sensitivity for HCC. Thus, we suggested that GPC-3 was an oncofetal antigen that showed great promise as a biomarker for the diagnosis of HCC. Capurro et al. 37 showed that GPC-3 was specifically overexpressed in most HCCs and was elevated in the serum of a large proportion of patients with HCC. However, studies from Wang et al. 29 revealed the opposite results, which showed that lower serum levels or lower sensitivity were found in patients with liver cirrhosis and HCC. Thus, we conducted this meta-analysis to evaluate the diagnostic value of serum GPC-3 in patients with HCC. The results indicated that serum GPC-3 was acceptable as a moderate diagnostic marker in the diagnosis of HCC compared with healthy individuals.
Compared to a single study especially in analyzing unexplained studies, meta-analysis, as a powerful and useful tool, could provide more sufficient results to clinical workers. 46 As a result, we suggest that a much stronger advantage exists to prove the association between the level of serum GPC-3 and the diagnosis of HCC. The present meta-analysis deems that serum GPC-3 might be a moderate diagnostic marker of HCC, compared with healthy individuals. In previous studies, this outcome has remained unclear. Therefore, a better method is required to analyze and understand the association between serum GPC-3 levels and the diagnosis of HCC. Also, for the first time, a meta-analysis was adopted to test whether our results were established on the firm evidence of effect. In the current meta-analysis, the summary DOR and 95% CIs for GPC-3 were 23.53 (8.57-64.63); and the area under the sROC curves and 95% CI were 0.87 (0.84-0.90). In order to evaluate the diagnostic value of serum GPC-3 in patients with HCC, serum GPC-3 has become an acceptable moderate diagnostic marker in the diagnosis of HCC compared with healthy individuals. Overall, sufficient statistical evidence, including the large sample size, were used to estimate such an association.
However, there are several limitations involved in our meta-analysis. First, the included studies were mainly conducted on Asians and Africans; only two studies were on Caucasian populations. Also, there are no reports available on other population groups. Thus, future research should focus on the influence of ethnicity factors. Second, only 22 studies were included in this meta-analysis, which might weaken its reliability. Therefore, more well-designed studies with large cases of each specific cancer should be conducted to validate the diagnostic value of serum GPC-3 in patients with HCC. Moreover, the threshold effect and obvious heterogeneity that existed in our study was probably due to the large difference in reagent resource, lifestyle, race, gender, age, the assay type, and the cut-off value. Hence, to improve the reliability of this meta-analysis, more studies are needed to evaluate the effectiveness of GPC-3 to differentially diagnose HCC in the future.
Conclusion
This meta-analysis suggested that serum GPC-3 was acceptable as a moderate diagnostic marker in the diagnosis of HCC compared with healthy individuals, which could elevate the sensitivity and specificity of diagnosis. To further evaluate the diagnostic value of serum GPC-3 in patients with HCC, better designed studies with large sample sizes are needed to show the effectiveness of GPC-3 in differentially diagnosing HCC.
Footnotes
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical statement
This article does not contain any studies with human participants or animals performed by any of the authors.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Grand Program on Key Infectious Diseases (No. 2015ZX10004801), the Ministry of Science and Technology of the People’s Republic of China (grant no. 2012DFA30850), the Beijing Municipal Science and Technology Commission (grant no. D131100005313004), the Beijing Municipal Health System High Level Personnel Training Programme (grant no. 2013-3-074), the National Major Scientific and Technological Special Project during the Thirteenth Five-year Plan Period Beijing (2017ZX10203205-006-003), and the Collaborative Innovation Centre of Infectious Diseases.
