Abstract
Objective:
The aim of this study is to establish the reference interval for serum pro-gastrin-releasing peptide (proGRP) determined by electrochemiluminescence immunoassay (ECLIA) in healthy Chinese Han ethnic adults.
Methods:
After screening, 9932 healthy Chinese Han adults (age range 18–95 years) were enrolled in this study, including 6220 men and 3712 women. Serum proGRP levels were measured by ECLIA. The reference interval was defined by non-parametric 95th percentile interval.
Results:
Serum proGRP levels conformed to a non-Gussian distribution. The reference interval for healthy Chinese Han adults calculated by the non-parametric method was 0–73.90 ng/mL in this study. Since serum proGRP levels were significantly correlated with age (r=0.226, P<0.001), the participants were divided into six age groups: 18–39, 40–49, 50–59, 60–69, 70–79, and ⩾80 years. No significant difference for serum proGRP levels was found between the sexes at each of six age groups. The reference intervals were gradually increased with age (65.35 ng/mL, 68.65 ng/mL, 74.10 ng/mL, 77.65 ng/mL, 84.57 ng/mL, and 98.03 ng/mL in 18–39, 40–49, 50–59, 60–69, 70–79, and ⩾80 years, respectively).
Conclusions:
We established the reference interval for serum proGRP, which was determined by ECLIA in the healthy Chinese Han population. Furthermore, our study suggests that it is necessary to establish the age-specific reference intervals for serum proGRP.
Introduction
Gastrin-releasing peptide (GRP), which was originally isolated and characterized from porcine stomach, 1 is a gut hormone, although it has been well recognized that GRP is widely present in neuroendocrine cells, nervous system cells, lung fibroblasts, and bronchial epithelial cells.2,3 The duration of the half-life of circulating GRP is so short (only 2 minutes or so) that it is not useful in laboratory medicine to detect the serum GRP levels. 4 proGRP is a more stable precursor of GRP. Miyake et al. 5 found, for the first time, that proGRP may be a specific tumor marker of small-cell lung cancer (SCLC). To date, it has been accepted that proGRP has a great value in the differential diagnosis, prediction, and prognosis of SCLC.2,6-8 Serum/plasma proGRP has been widely used as a tumor marker in clinical practice.
The appropriate interpretation of test results requires that they be compared with reference interval (RI). Accordingly, the provision of reliable RI determined from a healthy population is an essential task for clinical laboratories. At present, there are three major automated techniques for the determination of serum/plasma proGRP: (i) electrochemiluminescence assay (ECLIA) in cobas E601 analyzer from Roche Diagnostics GmbH; (ii) time-resolved immunofluorometric assay (TR-IFMA) in the AutoDELFIA1235 instrument from PerkinElmer company; and (iii) chemiluminescence assay (CMIA) in the ARCHITECT i2000 analyzer from Abbott Laboratories. 2 Nordlund et al. 9 reported the RI for serum proGRP using TR-IFMA in the healthy Nordic population. Zhang et al. 10 established the RIs for plasma ProGRP by CMIA on the Abbott ARCHITECT i2000 system in healthy adults of Chinese Han ethnicity. However, no data are available regarding the RI in healthy Chinese individuals for serum proGRP determined by ECLIA, which is also widely used in clinical laboratories.
The aim of the present study is to establish the RI for serum proGRP using ECLIA in the Roche cobas E601 system.
Subjects and methods
Subjects
A total of 11,998 adults participated in a routine physical examination in the Taizhou First People’s Hospital from March 2017 to February 2018. A health questionnaire and physical examination were given to each individual. Exclusion criteria included previous cancer, cardiovascular, liver, respiratory, gynecologic, or kidney diseases, infectious diseases, hypertension, impaired glucose tolerance, abnormal blood fat, pregnancy or lactation, and drug intake in the previous 2 weeks. After screening, 9932 apparently healthy Chinese Han adults (6220 males and 3712 females; age range 18–95 years) were finally included in this study. The detailed screening procedures are shown in Figure 1. This study was approved by the ethics committee of the Taizhou First People’s Hospital, and all participants gave their signed informed consent.
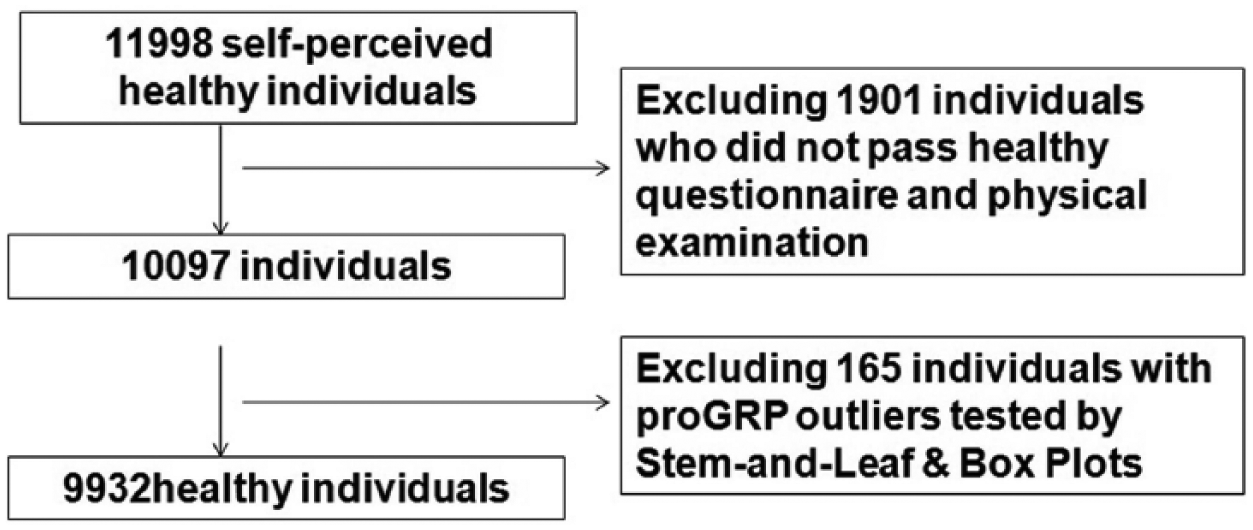
A flowchart of selection procedures of study subjects.
Laboratory methods
Venous blood of each participant was drawn and collected into gel separator tube. They were left to clot at room temperature for at least 30 minutes, then centrifuged for 10 minutes at 3000 rpm to separate for a serum sample. Serum proGRP levels were measured by ECLIA in Roche Cobas E601 system (Roche Diagnostics GmbH, Mannheim, Germany). The analyzer was routinely maintained according to the manufacturer’s instructions. Standard methodologies and dedicated reagents were used. Two levels of quality controls (as advised by the manufacturer) were run every day. Westgard alert rules were used to evaluate internal quality controls (IQC) during the entire period of the study. The total coefficient of variations (CV) of both levels of the quality control samples were below 8%.
Statistical analysis
Data were analyzed on SPSS 17.0 software (IBM SPSS STATISTIC, Chicago, IL, USA). The Kolmogorov–Smirnov test was used for testing the distribution of serum proGRP concentrations. Stem-and-Leaf & Box Plots were used to remove the outliers after logarithmic transformation. In accordance with CLSI C28-A3, the RI were defined by non-parametric 95% percentile interval. The difference between sexes was analyzed by the Mann–Whitney U test. The correlation between age and serum proGRP was assessed by Spearman’s rank correlation coefficient. If there was a significant correlation, age partitioning was required. After age grouping, the serum proGRP levels among different age groups were compared by non-parametric Kruskal–Wallis test. P values less than 0.05 were considered significant.
Results
Serum proGRP levels showed a skewed distribution by Kolmogorov–Smirnov test (P<0.05) (Figure 2). After logarithmic transformation, 165 outliers were removed by Stem-and-Leaf & Box Plots, and the remaining 9932 individuals (6220 men and 3712 women) were included in this study (Figure 1). The mean age of the 9932 participants was 49.8±15.3 years (age range 18–95years). According to the non-parametric method recommended by CLSI C28-A3, one-sided upper 95th percentile was set as the RI. Therefore, the RI for healthy Chinese Han adults was 0–73.90 ng/mL.
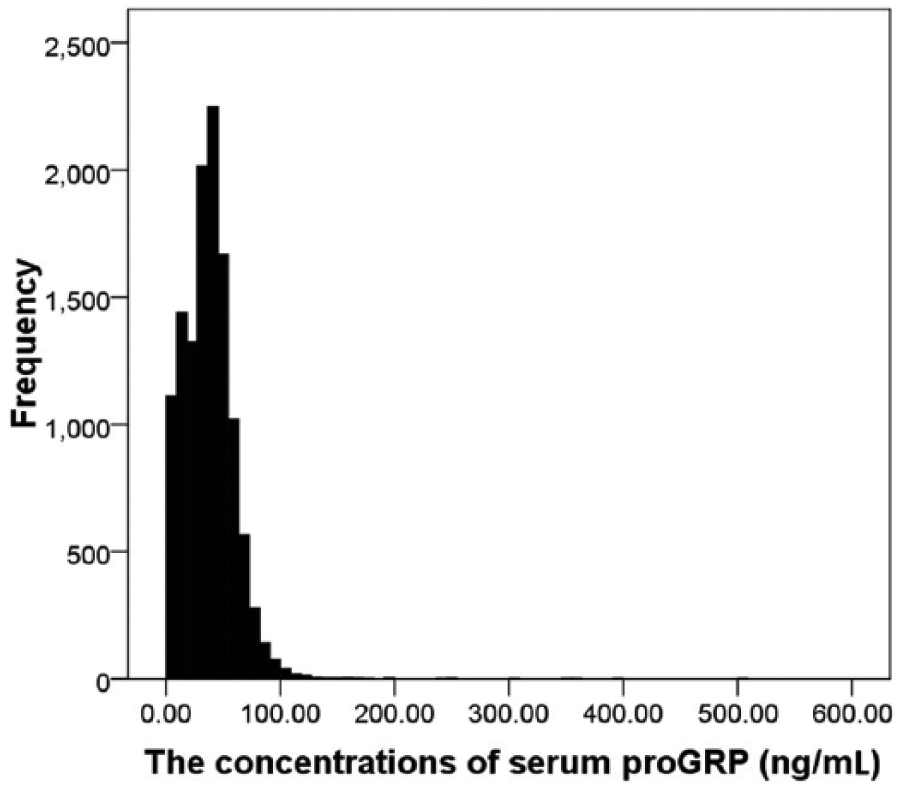
Frequency distributions of serum proGRP levels.
Spearman’s rank correlation analysis indicated that serum proGRP levels were significantly correlated with age (r=0.226; P<0.001) (Figure 3). Therefore, the participants were divided into seven age groups: 18–29, 30–39, 40–49, 50–59, 60–69, 70–79 and ⩾80 years. Since there was no significant difference for serum proGRP between the age groups of 18–29 and 30–39 years (P=0.826), the two groups were combined into one group (18–39 years). Between-group comparisons showed statistically significant differences between age partitions (P<0.001) (Figure 4 and Supplementary Table 1). Subsequently, between-sex comparisons were performed at each of the six age groups: 18–39, 40–49, 50–59, 60–69, 70–79 and ⩾80 years. No significant difference was found between sexes in any age group (Supplementary Table 1).
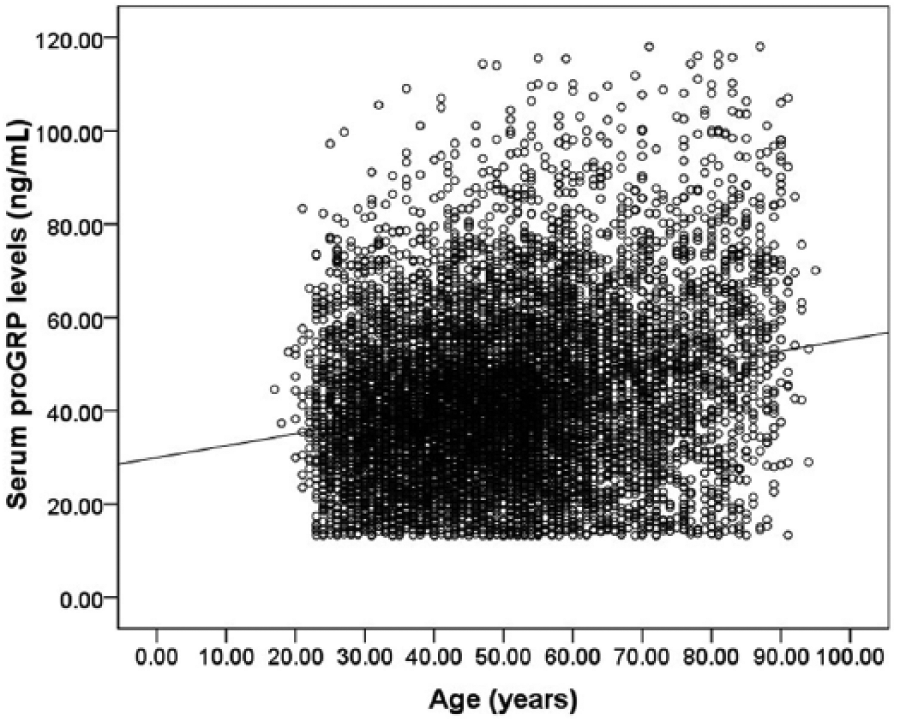
Scatterplot distributions for serum proGRP levels associated with age.
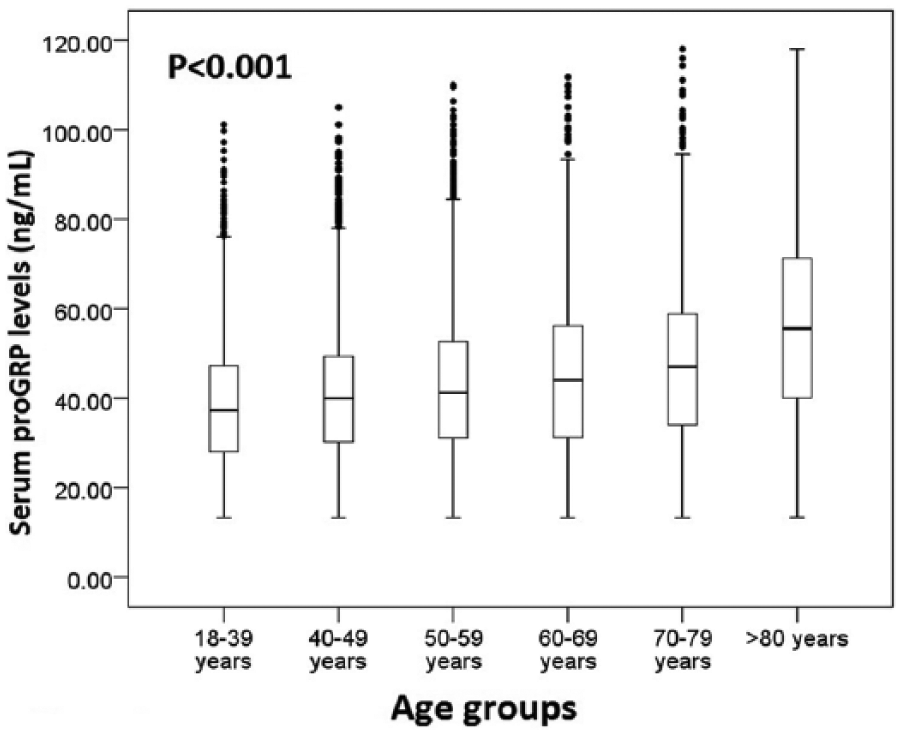
Serum proGRP levels at various age partitions.
In accordance with the aforementioned results, the RIs for serum proGRP needed to be calculated by six age partitions, 18–39, 40–49, 50–59, 60–69, 70–79 and ⩾80 years, but not by sex. The results showed that the RIs were gradually increased with age (65.35 pg/mL, 68.65 pg/mL, 74.10 pg/mL, 77.65 pg/mL, 84.57 pg/mL, and 98.03 ng/mL at 18–39, 40–49, 50–59, 60–69, 70–79, and ⩾80 years, respectively) (Figure 5).
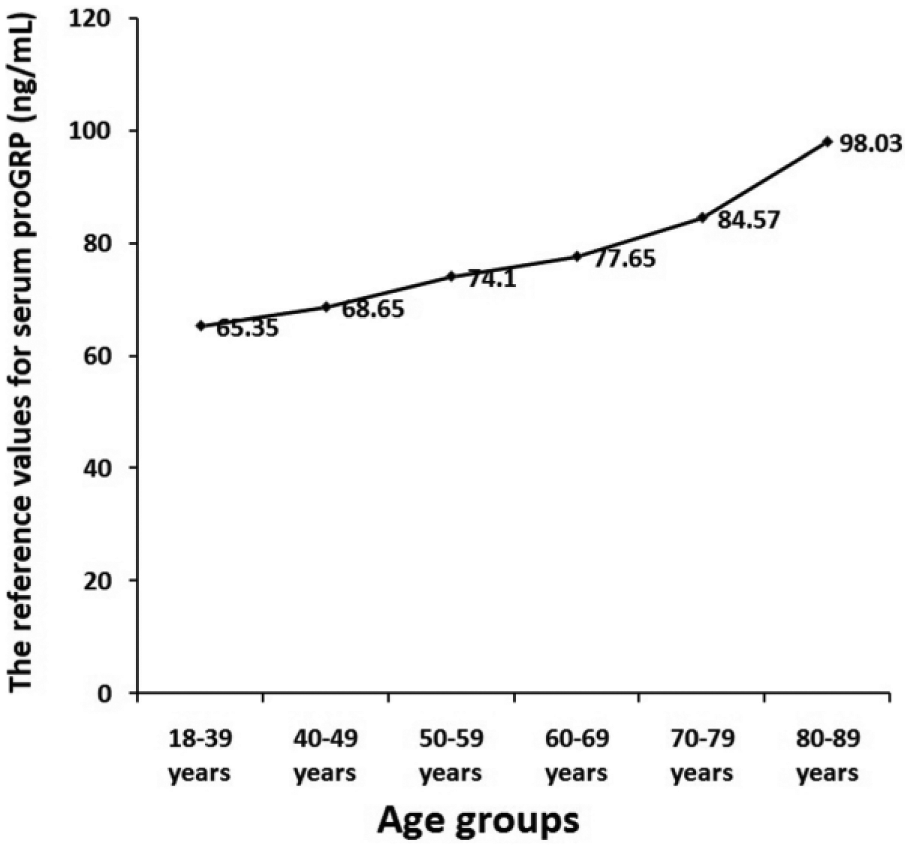
Values of upper reference limits for serum proGRP at various age partitions.
Discussion
It is well known that serum proGRP has been widely used as a tumor marker for SCLC. However, if there is no appropriate RI or medical decision limit, the test result for serum proGRP is of limited value. The RI is required for the interpretation of clinical laboratory results and patient care.11 -15 In fact, the RI for serum proGRP using TR-IFMA has been reported in a healthy Nordic population. 9 In healthy adults of Chinese Han ethnicity, the RIs for plasma ProGRP by CMIA on the Abbot ARCHITECT i2000 system also have been established. 10 ECLIA is another method widely used in clinical laboratories in China. However, no study has reported the RI for serum proGRP determined by ECLIA in healthy Chinese individuals.
In the present study, we established the RI for serum proGRP using ECLIA in healthy adults of Chinese Han ethnicity for the first time. Due to non-Gussian distribution for serum proGRP levels, we used the non-parametric statistical method for calculating the RI with the one-sided upper 95th percentile. The upper reference value was 73.90 ng/mL—slightly higher than that recommended by Roche Diagnostics GmbH (69.2 ng/mL), which was established with 698 apparently healthy Caucasian adults.
Similar to the study by Zhang et al., 10 we found that serum proGRP levels are closely associated with age, but not with sex. However, the results are different from the study by Nordlund et al., 9 which reported no association of serum proGRP levels with age and sex. The discrepancy may be due to the differences in assay method, race, and region of origin. In contrast to the study by Zhang et al. 10 in which they found a statistical difference for proGRP only between individuals over the age of 50 and individuals under the age of 50, our results showed that serum proGRP levels gradually increased with age. The reason for this difference may lie in the different assay method, sample type, and sample size. Their study sample size was relatively small (578 subjects) and may be insufficient for stratification analysis according to age, while ours was sufficient (9932 subjects).
Therefore, we established age-specific RIs for serum proGRP. Our results indicated that the RI gradually increases with age without significant differences between sexes at any age partition. These results suggest that it is necessary to establish age-specific RIs for serum proGRP, which will be helpful for better interpreting clinical laboratory results.
There are some limitations in this study. First, the nature of a single center and ethnic study may make our results less representative, although the Han ethnicity accounts for more than 91% of the Chinese population. However, further multicenter and multiethnic studies are needed. Second, the RIs established in this study are only suitable for serum proGRP determined by ECLIA in the Roche cobas E601 system, which may be different to those by other measurement methods, such as enzyme immunoassay, radioimmunoassay, chemiluminescence assay, etc.
In the current study, we established the RI for serum proGRP using ECLIA in the healthy Chinese Han population. Furthermore, our study has shown the necessity for establishing age-specific RIs of serum proGRP.
Supplemental Material
Supplementary_Table_1 – Supplemental material for Establishment of the reference interval for serum pro-gastrin-releasing peptide in healthy adults of Chinese Han ethnicity
Supplemental material, Supplementary_Table_1 for Establishment of the reference interval for serum pro-gastrin-releasing peptide in healthy adults of Chinese Han ethnicity by Bing Zhao, Miaomiao Zhang, Feng Lin, Jing Xie, Yan Liang and Zaixing Yang in The International Journal of Biological Markers
Footnotes
Author contributions
Bing Zhao, Miaomiao Zhang, and Feng Lin contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was obtained from all participants.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
