Abstract
Objectives
Methods
Results
Conclusions
Introduction
The incidence as well as the disease-related mortality rate of lung cancer worldwide are the highest among all types of cancers. Approximately 20%-25% of lung cancer cases are represented by small cell lung cancer (SCLC), and the prognosis of this type of cancer is especially poor, accounting for about 25% of all lung cancer deaths (1). The pro-gastrin-releasing peptide (31-98) (ProGRP), a member of the bombesin family of peptides, has a mitogenic activity in SCLC and can be produced by SCLC in an autocrine fashion (2). ProGRP has been shown to be a reliable tumor marker in patients with SCLC, and its clinical use has shown high sensitivity and specificity for the diagnosis of SCLC (2-3-4-5).
In 1995, Aoyagi et al established a sensitive enzyme-linked immunosorbent assay (ELISA) method for the measurement of plasma ProGRP (6). This method has been commonly used in ProGRP research for more than 10 years, until in 2009 Yoshimura et al evaluated the analytical performance of a fully automated chemiluminescent microparticle immunoassay (CMIA) for the measurement of multiple sites of serum and plasma ProGRP. Since then, a convenient and automated method has been available for hospitals and clinical laboratories (7). ProGRP is found to be significantly more stable in plasma than in serum, and the use of plasma samples is normally preferred as it may improve the clinical reliability of ProGRP measurements by minimizing pre-analytical changes in ProGRP concentrations (8).
To date there is no data available regarding the reference intervals (RIs) for plasma ProGRP levels in Chinese populations, evaluated according to the recommendations of the Clinical and Laboratory Standards Institute (CLSI) C28-A3 guideline (9). Therefore, we determined the plasma levels of ProGRP in healthy adults of Chinese Han ethnicity with CMIA on Abbot ARCHITECT i2000 system and we thus established the RIs for plasma ProGRP levels in our laboratory.
Materials and Methods
Ethics Approval
This study was approved by the ethics committee of the First Affiliated Hospital of Medical College at Zhejiang University, China.
Study Participants
In this cross-sectional study 578 healthy individuals of Chinese Han ethnicity were enrolled from different geographical zones of the Zhejiang province. The mean age of the participants was 45.9±15.8 years (range 18-83 years); their health status was determined by a general health evaluation that included a physical examination and clinical laboratory tests, including fasting plasma glucose, lipids levels, liver function tests, kidney function tests, tumor markers tests, and urinalysis, all performed at the Health Care Centre of the First Affiliated Hospital of the Medical College of Zhejiang University. Informed consent was obtained from all participants, who completed the questionnaires according to the CLSI C28-A3 guideline recommendations (9).
Exclusion criteria were the following: presence of impaired glucose tolerance (fasting plasma glucose ≥7.0 mmol/L), arterial hypertension (blood pressure ≥140/90 mmHg), presence of cardiovascular, liver, respiratory, gynecologic, or kidney disease (as detected by ultrasonic wave, electrocardiogram, or chest X-ray film), body mass index (BMI) ≥28 Kg/m2 or ≤18.5 Kg/m2, abnormal triglycerides (≥2.26 mmol/L), high total cholesterol (≥6.22 mmol/L), smoking (≥20 cigarettes per day), alcohol consumption (≥30 g per day), pregnancy or lactation, surgery in the previous 6 months, acute or chronic infections, history of malignancy, and drug intake within the previous 2 weeks.
Sample Collection
In the morning, after a 12-hour fast, whole blood samples were collected by venipuncture into 5 mL vacutainers containing dipotassium ethylenediaminetetraacetic acid (EDTA-2K) (Becton Dickinson, Franklin Lakes, NJ, USA). Within 2 hours, the samples were sent to the laboratory and centrifuged (10 minutes, 3000 g); plasma was then isolated from red blood cells and stored at -80°C until the assay was performed.
ProGRP Measurement
Plasma ProGRP concentrations were measured using a fully automated CMIA running in ARCHITECT i2000 (Abbott laboratories, Wiesbaden, Germany). The ARCHITECT ProGRP assay is a 2-step, sandwich format CMIA (3, 7) by which analyte capture is performed with paramagnetic microparticles coated with 2 mouse anti-ProGRP(31-98) monoclonal antibodies. First, specimen, anti-ProGRP-coated paramagnetic microparticle solution, and assay buffer were mixed and incubated for 18 minutes at 37°C. The ProGRP present in the specimen bind to the anti-ProGRP coated microparticles. After washing, a solution containing anti-ProGRP acridiniun-labeled conjugate was added to the mixture and incubated for 4 minutes at 37°C. After another wash, the pre-trigger and trigger reagents were added to the mixture, and the chemiluminescent reaction was detected as relative light units (RLUs). The amount of ProGRP in the specimen and the RLUs detected are directly correlated parameters.
Statistical Analysis
All data were analyzed with the SPSS 16.0 statistical software (Chicago, IL, USA). Results were expressed either as mean ± standard deviation, or median (with range or interquartile range). The Dixon test was used to detect outliers (9). In order to assess the distribution of ProGRP among the RIs we used a test of normality, the one-sample Kolmogorov-Smirnov (K-S) test. The RIs were defined by nonparametric 95% percentile intervals, as recommended by the CLSI C28-A3 guideline. For ProGRP, only the upper limit was of medical importance, thus the RIs were regarded as the one-sided upper 95th-percentile limits of the distribution of the reference population. We used the Mann-Whitney U-test for the comparisons between 2 groups, and the Kruskal-Walls H test for the comparisons among 3 groups. The correlation between ProGRP levels and age was analyzed with the Spearman's correlation test. P values <0.05 were considered statistically significant.
Results
Tests of Normality and Statistical Description
The plasma ProGRP values (Z=1.685, p=0.007) did not conform to a Gaussian distribution according to the Kolmogorov-Smirnov test. Therefore, we used nonparametric statistical methods to calculate the RIs with one-sided upper 95th percentiles. To assess the potential relationship between ProGRP and gender, the 297 male participants were compared to the 281 female participants. Age did not differ between males and females (respectively 46.4±16.6 years vs 45.3±14.3 years, p=0.400). The results of the Mann-Whitney U-test showed the absence of a significant difference in ProGRP values between males and females (p=0.077).
Age-Dependent Reference Intervals
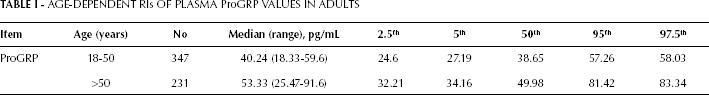
The distribution of the ProGRP levels based on age is shown in Figure 1. The ProGRP levels were positively correlated with age (r=0.45, p<0.001). To further study the role of age in ProGRP levels, all selected participants were divided into different groups according to their age: 18-30 years, 31-40 years, 41-50 years, 51-60 years, and >60 years. The 3 age groups of 18-30 years, 31-40 years, and 41-50 years did not differ in their plasma ProGRP levels (p=0.140) and were thus combined into 1 group. Similarly, the plasma ProGRP values between the groups of 51-60 years and >60 years were not statistically different (p=0.947), and were thus combined into 1 group. The ProGRP levels were significantly higher in the >50 years group compared to the 18-50 years group (Fig. 2, p<0.001). Therefore, we established the following age-dependent RIs: ≤57.26 pg/mL for young adults (18-50 years), and ≤81.42 pg/mL for old adults (>50 years) (Tab. I).
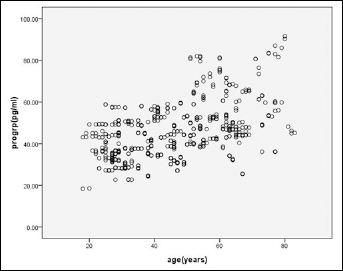
Distribution of plasma ProGRP values as a function of age.
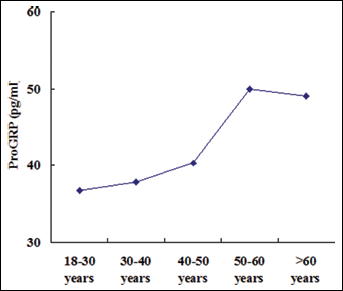
Plasma ProGRP values (median value) in the 5 age groups.
AGE-DEPENDENT RIs OF PLASMA ProGRP VALUES IN ADULTS
Discussion
The identification of the appropriate RI for a given tumor marker plays an important role in the diagnosis and treatment of cancer. The absence of information about age, gender, race, and region of origin for assay-specific RIs can directly affect medical interventions and clinical therapeutic strategies (10, 11). Most clinical laboratories in China use the RIs provided by the assay's manufacturers, reporting simple and economical validations of RIs performed with 20 healthy subjects. However, the manufacturer often provides little information regarding the details of their healthy subjects and sampling procedures, and this is also true for RIs quoted in the literature (11, 12). Therefore, we thought it was important to correctly establish appropriate RIs for our own clinical laboratory, according to the C28-A3 guideline.
A minimum of 120 healthy subjects is required to establish 95% RIs for a particular subgroup (9). However, a subclinical disease may be present in subjects defined as healthy, and this could decrease the accuracy of a research study (9, 13). In this study we enrolled a larger population (578 subjects) in order to enhance the statistical power and reliability of our results. In addition, healthy individuals selected for the study underwent a health evaluation and were selected according to specific exclusion criteria (10, 14).
When the reference values do not follow a Gaussian distribution, nonparametric statistical analysis is the method of choice for computing RIs (9, 15). Therefore, a nonparametric statistical test was here used to calculate the upper limit of the 95th percentile for ProGRP (as only the upper limit was of medical importance). We confirmed that age was the only evaluated factor that positively correlated with ProGRP levels. Age-dependent RIs for plasma ProGRP were thus established.
Our study is the first to report RIs of plasma ProGRP for a population in China. In 2008, Nordlund et al (16) reported the serum RIs for ProGRP as measured by a time-resolved immunofluorometric assay (TR-IFMA) in 806 healthy individuals from the Nordic Reference Interval Project Bio-bank and Database (NOBIDA) (17). The upper limit of the 97.5th percentile RI of ProGRP in that study was 58.9 pg/mL. Compared to our study, their serum RIs for ProGRP were similar to the plasma RIs for our group of 18-50 years old participants and lower than the plasma RIs for our >50 years old participants. In addition, the serum RIs of ProGRP reported in the Norwegian study (17) were both age and gender-independent. The reason for these discrepancies is unclear, but the main cause may be due to the different ProGRP assay method, operation analyzers, race, region of origin, and sample type.
In summary, we establish the RIs in healthy adults of Chinese Han ethnicity according to the CLSI C28-A3 document for clinical laboratory. We find that age, but not gender, is an important factor when determining RIs for plasma ProGRP. We are confident that these RIs may be reliably adopted to evaluate the diagnosis of SCLC in Chinese Han adults. Our study highlights the practical importance of identifying RIs for plasma ProGRP in adults of different ethnicity and to use multicenter-generated RIs.
