Abstract
Background:
Reliable reference intervals for serum progastrin-releasing peptide (ProGRP) in healthy Chinese adults with electrochemiluminescence immunoassay (ECLIA) are still lacking in the Chinese population.
Objectives:
The study aims to establish reference intervals for ProGRP with ECLIA in apparently healthy Chinese adults.
Methods:
A total of 384 apparently healthy individuals from six representative geographical regions in China were enrolled: 200 males and 184 females with a mean age of 43.4±12.2 years, and an age range from 21 to 85 years. Serum ProGRP levels were analyzed on Cobas e601 automatic immunoassay analyzer with ECLIA. Reference intervals for serum ProGRP with ECLIA were determined following CLSI C28-A3 guidelines using a nonparametric method.
Results:
In an apparently healthy Chinese population, the reference intervals for serum ProGRP with ECLIA were ⩽53.92 ng/L for adults aged 21–70 years and ⩽75.69 ng/L for adults aged >70 years, respectively.
Conclusions:
The reference values for serum ProGRP with ECLIA in an apparently healthy Chinese population were established according to the CLSI C28-A3 document, providing a reference for the clinical work.
Keywords
Introduction
Lung cancer is one of the most common cancers in the world and in China, both in terms of incidence and mortality. On the basis of different clinical behavior and sensitivity to chemo- and radiotherapy, lung cancers can be grouped into two major histological types; that is, non-small-cell lung cancer (NSCLC) and small-cell lung cancer (SCLC). SCLC, which comprises about 15–20% of all lung cancers, is extremely aggressive and cure rates are extremely low. Detection at an early stage of the disease as well as adequate monitoring of treatment response is vital in order to improve the outcome. As SCLC is highly sensitive to radiotherapy and chemotherapy, we differentiate it from NSCLC to ensure a specific therapeutic intervention. Tumor markers of SCLC have thus been studied in patients with lung cancer as tools to detect SCLC and improve SCLC diagnosis and monitoring as well as treatment selection.1,2
Progastrin-releasing peptide (ProGRP) is the precursor for gastrin releasing peptide (GRP), a gut hormone that is also present in nerve and monitoring fibers, brain, and neuroendocrine cells of the fetal lung. 3 ProGRP production significantly increases in SCLC and thus it has become a beneficial tumor marker in SCLC.4,5 Moreover, the electrochemiluminescence immunoassay (ECLIA), 6 which unites the specificity of the immunoassay and the sensitivity of the electrochemical analysis, is now a widely implemented detecting method and is gradually used to detect ProGRP.
The identification of the appropriate reference intervals (RIs) for a given tumor marker are an important basis for interpreting the test results. They help to define whether human bodies are in a normal condition or not, and thus are essential for clinical decision making. As RIs of clinical laboratory analytes were always significantly related to gender, age, methodology, and race, 7 the Clinical & Laboratory Standards Institute (CLSI) C28-A3 guidelines recommend to establish specific RIs for each gender, age, assay method, and reference population. However, there is no literature regarding the RIs for serum ProGRP in healthy Chinese adults with ECLIA. Therefore, we determined the serum ProGRP levels in healthy Chinese adults with ECLIA and established the RIs rigorously according to the CLSI C28-A3 guidelines.
Materials and methods
Subjects
This study was approved by the ethics committee of the Second Xiangya Hospital of Central South University and was designed rigorously according to CLSI C28-A3 guidelines, the authoritative document of establishment of RIs. Candidate participants were randomly recruited from six representative geographical regions in China: Northeast China (Shenyang), North China (Beijing), East China (Shanghai), South China (Guangzhou), Southwest China (Chengdu), and Northwest China (Xi’an). Informed consent was obtained from all participants. To select those apparently healthy individuals, all the participants completed a questionnaire, and detailed physical examinations and clinical laboratory tests were performed.
The exclusion criteria from the CLSI C28-A3 guidelines were: the presence of impaired glucose tolerance (fasting plasma glucose ⩾7.0 mmol/L); arterial hypertension (blood pressure ⩾140/90 mm Hg); the presence of cardiovascular, liver, respiratory, gynecologic, or kidney disease (as detected by ultrasonic wave, electrocardiogram, and chest X-ray film); body mass index (BMI) ⩾28 Kg/m2 or ⩽18.5 Kg/m2; TG ⩾2.26 mmol/L; TC ⩾6.22 mmol/L; smoking (⩾20 cigarettes per day); alcohol consumption (⩾30 g per day); pregnancy or lactation; surgery in the previous 6 months; acute or chronic infections; a history of malignancy; and drug intake within the previous 2 weeks.
Laboratory methods
Blood samples from participants were collected by venipuncture into gel-separated vacuum test tubes in the morning after fasting for 10 hours and sitting for 30 minutes. The samples were kept at room temperature for 30 minutes and centrifuged at 3000 rpm for 10 minutes. Serum ProGRP levels were measured by ECLIA on the Cobas e601 automatic immunoassay analyzer (Roche Diagnostics GmbH, Penzberg, Germany) within 2 hours. The assay was calibrated using the ProGRP CalSet (Roche Diagnostics). Quality control was performed using two levels of PreciControl ProGRP (Roche Diagnostics).
Statistical analyses
The SPSS 17.0 software was used for the data analysis. The distribution of the data was detected using the Kolmogorov–Smirnov test. Outliers were removed with the Dixon test. After excluding outliers, the medians of variables were compared between the males and females by the standard normal deviate test (Z test), and the Kruskal–Wallis H test was applied to compare the variables among different age groups. Differences with a P value <0.05 were considered statistically significant. The 95% RIs were estimated according to recommendation by CLSI C28-A3 including nonparametric and robust method. 8 The Spearman correlation analysis was used to test for the correlation between age and ProGRP. Generally, we use the 2.5th and 97.5th percentiles as the lower and upper reference limits of the 95% RIs. However, for ProGRP, only the upper reference limit is of medical importance. On this occasion, we use 95th percentile as the 95% RIs. The 90% CI of each reference limit was also defined.
Results
Through the exclusion criteria, a total of 384 apparently healthy individuals (200 males and 184 females) were ultimately included in this study. The baseline characteristics of the individuals are presented in Table 1.
Serum ProGRP levels in healthy adults (ng/L).
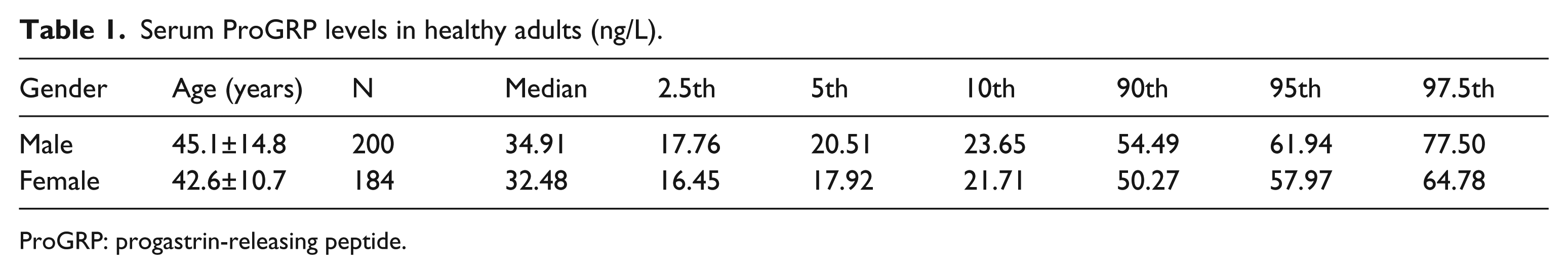
ProGRP: progastrin-releasing peptide.
The distribution of serum ProGRP levels in healthy adults
The levels of serum ProGRP in healthy adults were not in Gaussian distribution (P<0.05). No outlier was detected. A non-parametric method was thus used to calculate the 95% RIs.
Gender-dependent serum ProGRP levels
The distribution of serum ProGRP levels based on gender is shown in Table 1. The results of Z test showed there was no significant difference for serum ProGRP levels between males and females (P>0.05, Z>Z*). There was no need to separate the results into male and female groups.
Age-dependent serum ProGRP levels
Non-parametric spearman correlations were demonstrated between age and serum ProGRP levels and a significant correlation was observed. The serum ProGRP levels were positively correlated with age (r=0.347, P<0.05) (r, correlation coefficient).
The participants in our study were then divided into six age-dependent groups (21~30, 31~40, 41~50, 51~60, 61~70, and >70 years). The first five age groups did not significantly differ in their serum ProGRP levels (P>0.05), so they were combined into one group. However, the serum ProGRP levels in >70 years group were significantly higher than the 21~70 years group (P<0.05), so we separated them into two groups. The distribution of serum ProGRP levels based on age are shown in Figure 1 and Table 2.
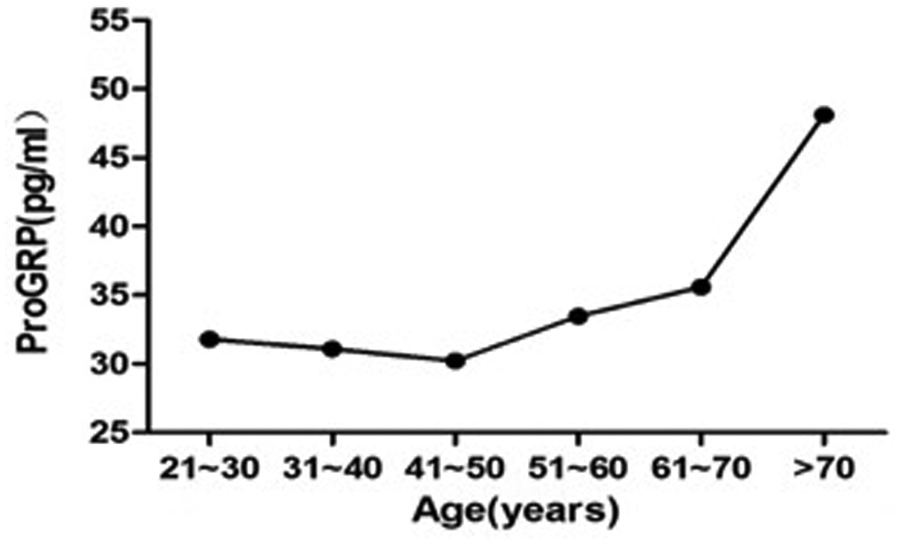
Serum ProGRP levels (median value) in six age groups.
Age-dependent serum ProGRP levels (ng/L).
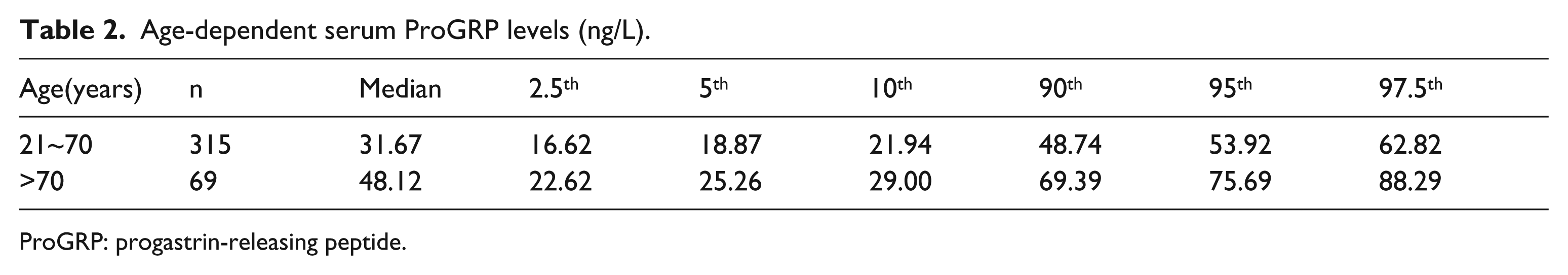
ProGRP: progastrin-releasing peptide.
Reference values of serum ProGRP levels
CLSI guidelines for determining laboratory reference ranges recommends a minimum of 120 subjects for a 95th percentile clinical reference range determination with 90% confidence intervals (CI) for each partition group. The reference values for 21~70 years were established using a non-parametric statistical method. Due to the insufficient number of participants (n=69) in >70 years group, a robust statistical method, which is recommended in the C28-A3 document to solve the problem, was applied to calculate reference values. The RIs of ProGRP in Chinese adults and the corresponding 90% CI are shown in Table 3. In an apparently healthy Chinese population, the RIs for serum ProGRP with ECLIA were ⩽53.92 ng/L for adults aged 21–70 years and ⩽75.69 ng/L for adults aged >70 years, respectively.
RIs and 90% CI of serum ProGRP levels (ng/L).

CI: confidence interval; ProGRP: progastrin-releasing peptide; RIs: reference intervals.
Discussion
Lung cancer is one of the leading causes of death from malignancy worldwide. It can be categorized into two clinically relevant groups: SCLC and NSCLC. SCLC, comprising 15–20% of all lung cancers, is extremely aggressive and cure rates are extremely low. Therefore, early diagnosis and adequate treatment monitoring is essential to improve the outcome. As SCLC is highly sensitive to radiotherapy and chemotherapy, we differentiate it from NSCLC to ensure a specific therapeutic intervention.
The clinical utility of ProGRP as a biomarker for SCLC detection has been established in a number of studies.5,7,9-11 The Elecsys® ProGRP assay is an ECLIA that uses a biotinylated ProGRP-specific mouse monoclonal antibody and a ruthenium-labeled ProGRP-specific mouse monoclonal antibody to capture and detect ProGRP in human serum and plasma. The increased stability of the Elecsys® ProGRP assay in serum and plasma offers clear benefits over existing assays and thus it has gradually become a widely implemented detecting method in clinical laboratories.
The identification of the appropriate reference values for a given tumor marker is an important basis for the interpretation of test results. Reference values help defining whether human bodies are in normal condition or not and thus are essential for clinical decision making. The absence of appropriate reference values can directly affect medical interventions and clinical therapeutic strategies.7,11 To date there are no data available regarding the reference values for serum ProGRP levels with ECLIA in Chinese populations. As RIs of clinical laboratory analytes were always significantly related to age, gender, or race, 7 establish the corresponding RIs is an urgent task.
Therefore, with Roche Elecsys® ProGRP assay, we carefully established the RIs of serum ProGRP with ECLIA for Chinese adults. The RIs are ⩽53.92 ng/L for 21–70 years old and ⩽75.69 ng/L for 70–85 years old in both males and females.
Nordlund et al. 12 reported the reference values for serum ProGRP measured by a time-resolved immunofluorometric assay (TR-IFMA) in 806 healthy individuals from the Nordic Reference Interval Project Bio-bank and Database (NOBIDA). The 97.5th percentile for serum ProGRP on TR-IFMA was 58.9 ng/L. The result was similar to the 97.5th percentile of the 21~70 years in our study (⩽62.82 ng/L), lower than the 97.5th percentile of the >70 years (⩽88.29ng/L). The effect of a distinct reference population and an assay method will lead to discrepancy. Specific RIs should be established for a specific assay method and reference population. We believe our establishment of serum ProGRP reference ranges specifically for ECLIA and for the Chinese population will ensure a more reliable reference interval. Zhang et al. 13 reported the reference values for plasma ProGRP levels in healthy adults of Chinese Han ethnicity by using the chemiluminescent microparticle immunoassay (CMIA) in 578 healthy Chinese. The plasma ProGRP RIs for Chinese adults with CMIA established by them were calculated by two age subclasses. Their results (⩽57.26 ng/L for18–50 years and ⩽81.42 ng/L for >50 years) were similar to ours (⩽53.92 ng/L for 21–70 years and ⩽75.69 ng/L for 70–85 years). In fact, the elderly groups is a special group whose body conditions always present physiological changes that are diverse from those of young adults. Our study found that the ProGRP levels in serum were not affected by gender but increased along with age. The calculation of reference values separately by age subclasses (21~70 years and >70 years) is very necessary. Korse et al. 6 reported the reference ranges of the serum ProGRP with a cohort of 1085 healthy individuals from five European sites (50 males and 50 females in Amsterdam; 33 males and 67 females in Barcelona; and 4 males and 37 females in Bonn); two Chinese sites (31 males and 40 females in Peking Union Medical College Hospital; and 24 males and 51 females in Peking Xuanwu Hospital); and an additional German site (336 males and 362 females in Kiel) with the Elecsys® ProGRP assay. They concluded that the 95% reference values for serum ProGRP with Elecsys® ProGRP assay was ⩽66 pg/mL. Our distinctive result was age dependent and had a lower limit for adults aged 21–70 years (⩽53.92 pg/mL) and a higher limit for adults aged >70 years(⩽75.69 pg/mL). Although the Korse et al. 6 research was a multicenter study, they included only two Chinese sites, both of which were in Beijing. In comparison, our samples, which came from six representative geographical regions, Northeast China (Shenyang), North China (Beijing), East China (Shanghai), South China (Guangzhou), Southwest China (Chengdu), and Northwest China (Xi’an), are more representative of the Chinese population. On this occasion, we believe our study will present a more precise RI for serum ProGRP (Table 4).
ProGRP reference intervals in different studies.
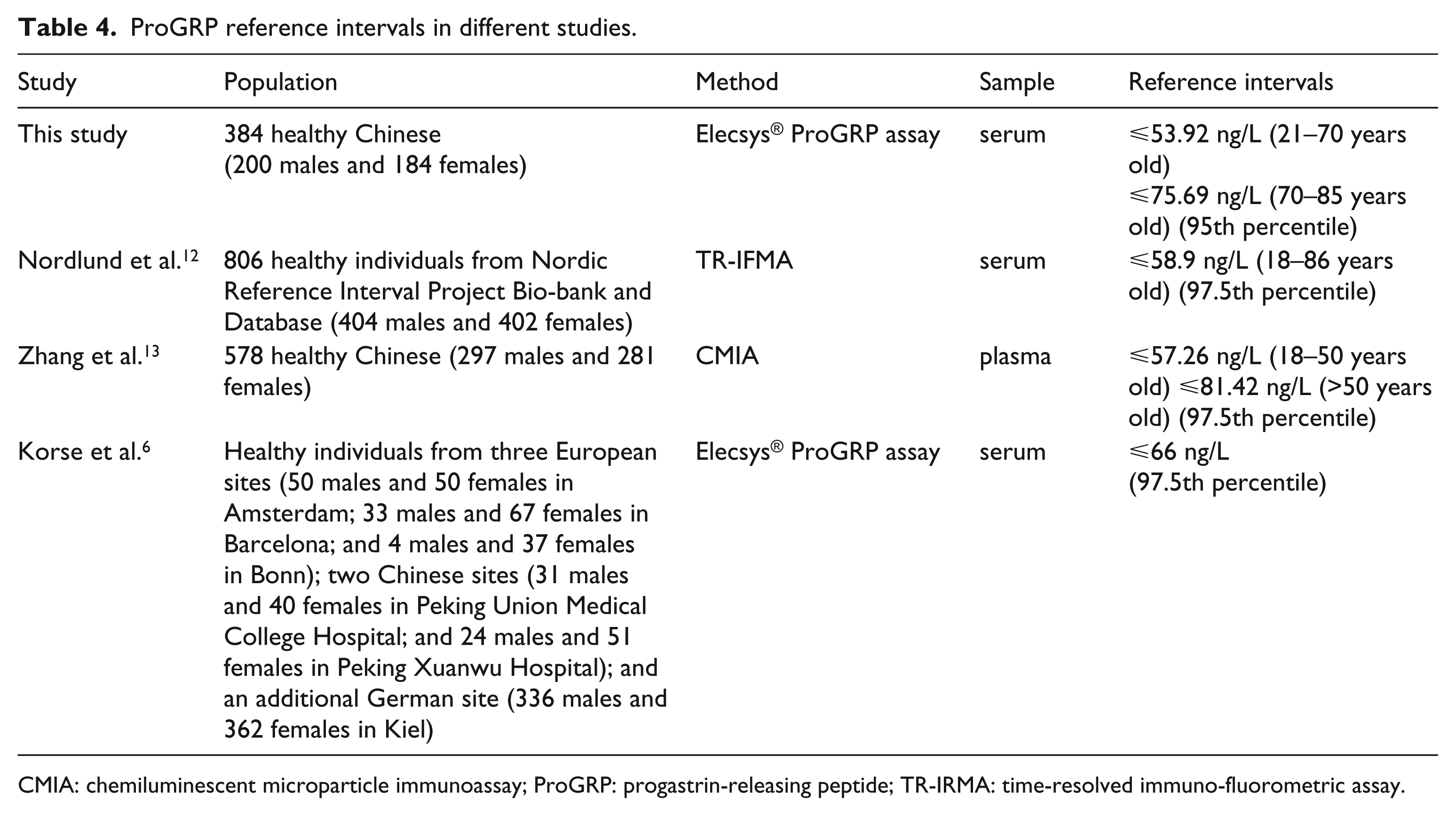
CMIA: chemiluminescent microparticle immunoassay; ProGRP: progastrin-releasing peptide; TR-IRMA: time-resolved immuno-fluorometric assay.
In summary, this systematic study established the reference values for serum ProGRP with ECLIA in an apparently healthy Chinese population according to the recommendation of CLSI C28-A3 guidelines. The results will provide a valuable and reliable reference for the clinical work and laboratory studies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
