Abstract
The platelet-to-lymphocyte ratio (PLR) is a useful prognostic factor in several cancers. However, the prognostic role of PLR in esophageal cancer remains controversial. The aim of this study is to evaluate the association between PLR and the oncologic outcome of esophageal cancer patients through a meta-analysis. Relevant articles were researched from Embase, PubMed, and Web of Science databases. The meta-analysis was performed using hazard ratio (HR) and 95% confidence intervals (CIs) as effect measures. Finally, 19 articles with 6134 patients were included in our study. The summary results indicated that the elevated PLR was negatively related to overall survival (HR= 1.263; 95% CI 1.094, 1.458). The subgroup analysis revealed that increased PLR was associated with poor overall survival in esophageal cancer patients for Asians (HR=1.252; 95% CI 1.141, 1.373) but not for Caucasians (HR=1.463; 95% CI 0.611, 3.502). When the patients were segregated by pathological type, sample size, and HR estimate method, high PLR was also significantly correlated with poor overall survival. In contrast, elevated PLR was not statistically associated with disease-free survival or cancer-specific survival. High PLR is associated with poor overall survival in patients with esophageal cancer. PLR may be a significant predictive biomarker in patients with esophageal cancer. Further large-cohort studies are needed to confirm these findings.
Introduction
Esophageal cancer (EC) is one of the most common and life-threatening types of cancer worldwide, with 477,900 new cases and 375,000 deaths in China, 2015. 1 Squamous cell carcinoma (SCC) and adenocarcinoma (AC) are the two common pathologic categories. SCC is the predominant histological subtype in Asia, while in Western countries, AC is more common, which covers more than 90% of all EC cases. 2 In recent decades, there have been significant improvements in diagnosis and therapeutic strategies. Neoadjuvant treatment and surgical resection have gradually been accepted as the mainstay of treatment for local and locoregional EC, but the prognosis is still not ideal due to the rapid recurrence or metastasis. 3 Generally, the tumor stage determined by the tumor-node-metastasis (TNM) staging system is the most important predictor of survival. However, heterogeneous clinical outcomes are frequently observed even within the same tumor stage, which implies the existence of other prognostic indicators.
Increasing evidence has indicated that the inflammatory response may play a key role in tumor development and progression.4,5 The systemic inflammation markers could indicate the prognosis in tumor patients. The Glasgow Prognostic Score, lymphocyte-to-monocyte ratio, neutrophil-to-lymphocyte ratio (NLR), and platelet-to-lymphocyte ratio (PLR) are common inflammation factors 6 and the prognostic roles of these factors have been evaluated in various types of cancers.7-9 PLR, calculated as platelet counts divided by lymphocyte counts, has been found with worse survival in gastric, colorectal, and lung cancers.10-12 Recently, several retrospective studies have evaluated the prognostic significance of PLR in patients with EC.13-15 However, according to their results, the current opinion on the prognostic role of PLR in EC has yet to be clearly defined. A previous meta-analysis on PLR in patients with EC found that high PLR is associated with tumor progression and were predictive of poorer survival. 16 However, this meta-analysis only included four retrospective studies. Increasing studies on the association between PLR and EC have been recently reported. Therefore, it is necessary to perform a meta-analysis to comprehensively and systematically evaluate the prognostic value of PLR in the oncologic outcomes of EC patients.
Material and methods
Search strategy
This study was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2009 guidelines. 17 We conducted a literature search via PubMed, Embase, and Web of Science databases for relevant articles from January 1990 to January 2017. Both full text and MeSH search for keywords were used. The main search terms included: “PLR” (e.g. “platelet lymphocyte ratio,” “platelet to lymphocyte ratio,” “platelet-to-lymphocyte ratio”) AND “esophageal neoplasm” (e.g. “esophageal cancer,” “esophageal carcinoma,” “EC”) AND “prognosis’’ (or ‘‘survival’’). Related articles and reference lists in each identified publication were also reviewed.
Inclusion and exclusion criteria
Articles were included if they met the following criteria: (a) patients with esophageal cancers were histopathological confirmed; (b) pretreatment PLR were reported; and (c) associations between pretreatment PLR and survival outcomes (overall survival (OS) and/or progression-free survival (PFS) and/or disease-free survival (DFS) and/or cancer-specific survival (CSS)) were reported.
Articles were excluded if they met the following criteria: (a) abstracts, letters, editorials, reviews, or case reports; (b) the studies were not available in English; (c) the studies had overlapping or repeat data; (d) the studies concerned non-human or non-clinical research; and (e) studies without sufficient data for estimating hazard ratio (HR).
Data extraction and quality assessment
Two researchers (Xiangwei Zhang and Yang Wang) reviewed the eligible articles independently. Articles that could not be categorized based on title and abstract alone were retrieved for full-text review. The following items were recorded for each study: first author, year of publication, country, total number of cases and gender, follow-up time, cut-off value, treatment strategy, cancer type and HR with 95% confidence interval (CI). HRs and 95 % CIs were obtained directly from individual articles or were calculated from indirect data according to the methods presented by Parmar. 18 HRs were extracted preferentially from multivariable analyses where available. Otherwise, HRs from univariate analyses were extracted.
The Newcastle–Ottawa Quality Assessment Scale (NOS) was used to assess the quality of the included studies, which consisted of three parts: (a) the selection of patients (0–4 points); (b) the comparability of groups (0–2 points); and (c) outcome assessment (0–3 points). 19 The maximum score is 9 points and NOS scores ≥7 were assigned as high-quality studies. Any conflicts in data extraction or quality assessment were resolved by a third reviewer (Linping Zhao).
Statistical analysis
Pooled HRs and 95% CIs were used to analyze the relationship between PLR and prognosis. A pooled HR > 1 indicated a worse prognosis in EC patients with high expression of PLR. Cochran’s Q test and Higgins I2 statistic were undertaken to assess the heterogeneity of the included studies. A P < 0.10 for Q test or I2 > 50% for I2 test suggested significant heterogeneity among the study and then a random-effect model (DerSimonian–Laird method) was used to combine the effect of PLR. 20 Otherwise, the fixed-effect model (Mantel–Haenszel method) was adopted. 21 Subgroup analyses using variables, such as ethnicity, histology, cut-off value, sample size, HR estimated method, and treatment strategy, were conducted to find reasons for heterogeneity among studies. Sensitivity analysis was conducted to test the reliability of outcomes by removing one single study in sequence. Publication bias was assessed by Begg’s or Egger’s test.22,23 All P-values were two-sided. A P < 0.05 was considered statistically significant. All the statistical analyses were performed using STATA statistical software version 12.0 (STATA, College Station, TX, USA).
Results
Study characteristics
The flow diagram of literature selection is summarized in Figure 1. Nineteen studies with a total of 6134 patients published between 2011 and 2017 were included in our meta-analysis.13-15,24-39 All the included studies were retrospectively designed. Among them, patients in 16 studies were Asian and in the other three were Caucasian. Fourteen studies were from China, three studies from the UK, and two from Japan. Twelve studies involved patients with SCC, two with AC, one with small-cell cancer, and four studies contained both SCC and AC patients. None of these studies included patients treated with non-curative intent. Eighteen studies included patients who underwent surgical resection with or without chemoradiotherapy. Only one study included patients who underwent curative chemoradiotherapy alone. The cut-off values applied in the studies were not consistent, ranging from 103 to 300. Ten studies had a PLR cut-off value of 150 or less, while six studies used a PLR cut-off value greater than 150. Two cut-off values were reported in two studies,24,28 and one study did not report the cut-off value. 26 Fifteen studies reported the relationship between EC and OS; six showed the association between EC and DFS; two reported the relationship between EC and RFS; one reported the relationship between EC and PFS; four reported the relationship between EC and CSS; and one reported the relationship between EC and disease-specific survival (DSS). HRs and 95% CIs were reported directly in 19 studies, 12 of which calculated HRs by the univariable analysis and seven via multivariate analysis. The HR of the high versus the low group was adopted in the study. 28 One study only reported relative risk (RR); thus we used the RR to replace HR when the data was pooled. 32 The characteristics of the included studies are shown in Table 1.
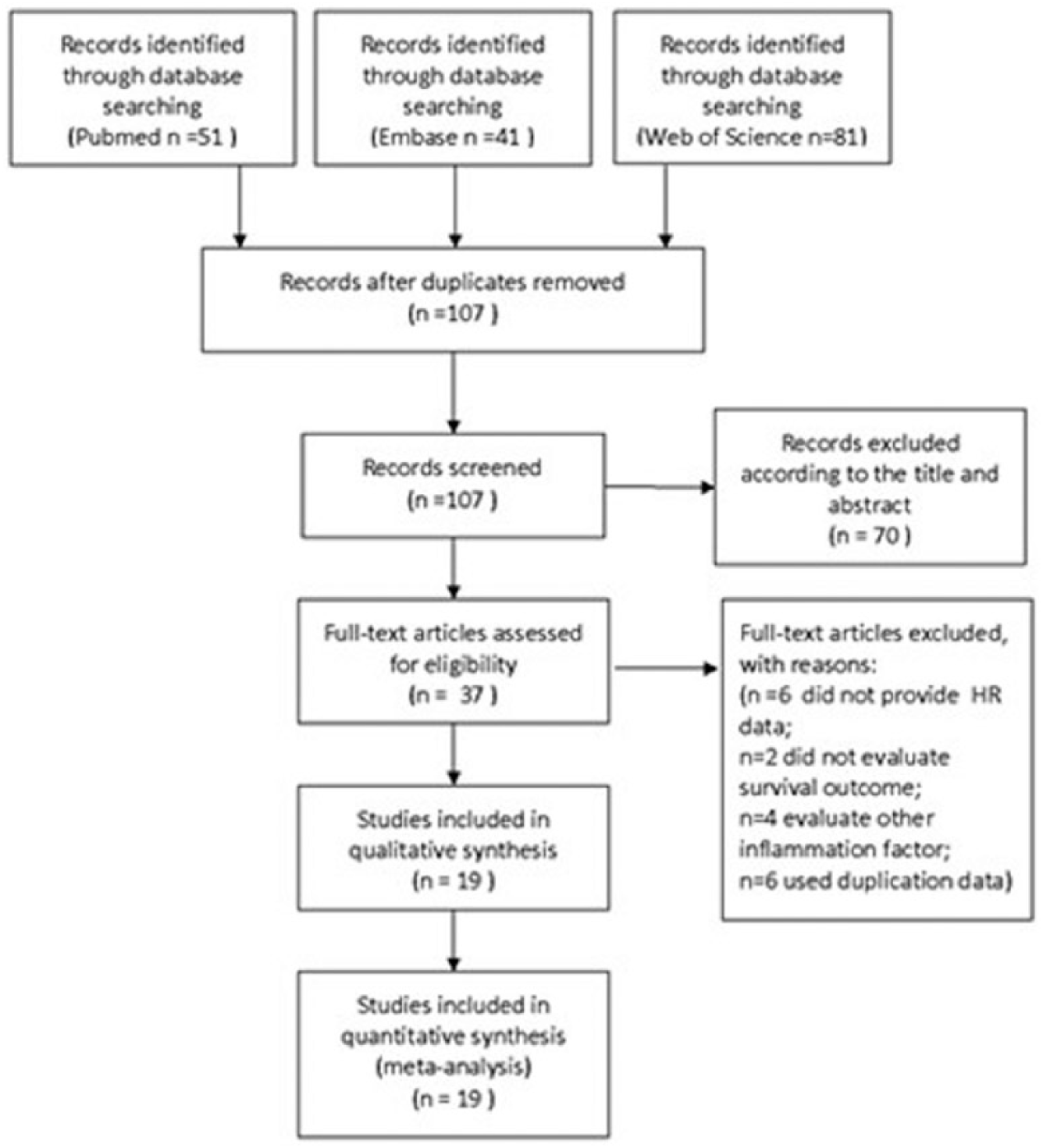
Flow chat of literature search and selection.
Main characteristics of all the studies included in the meta-analysis.
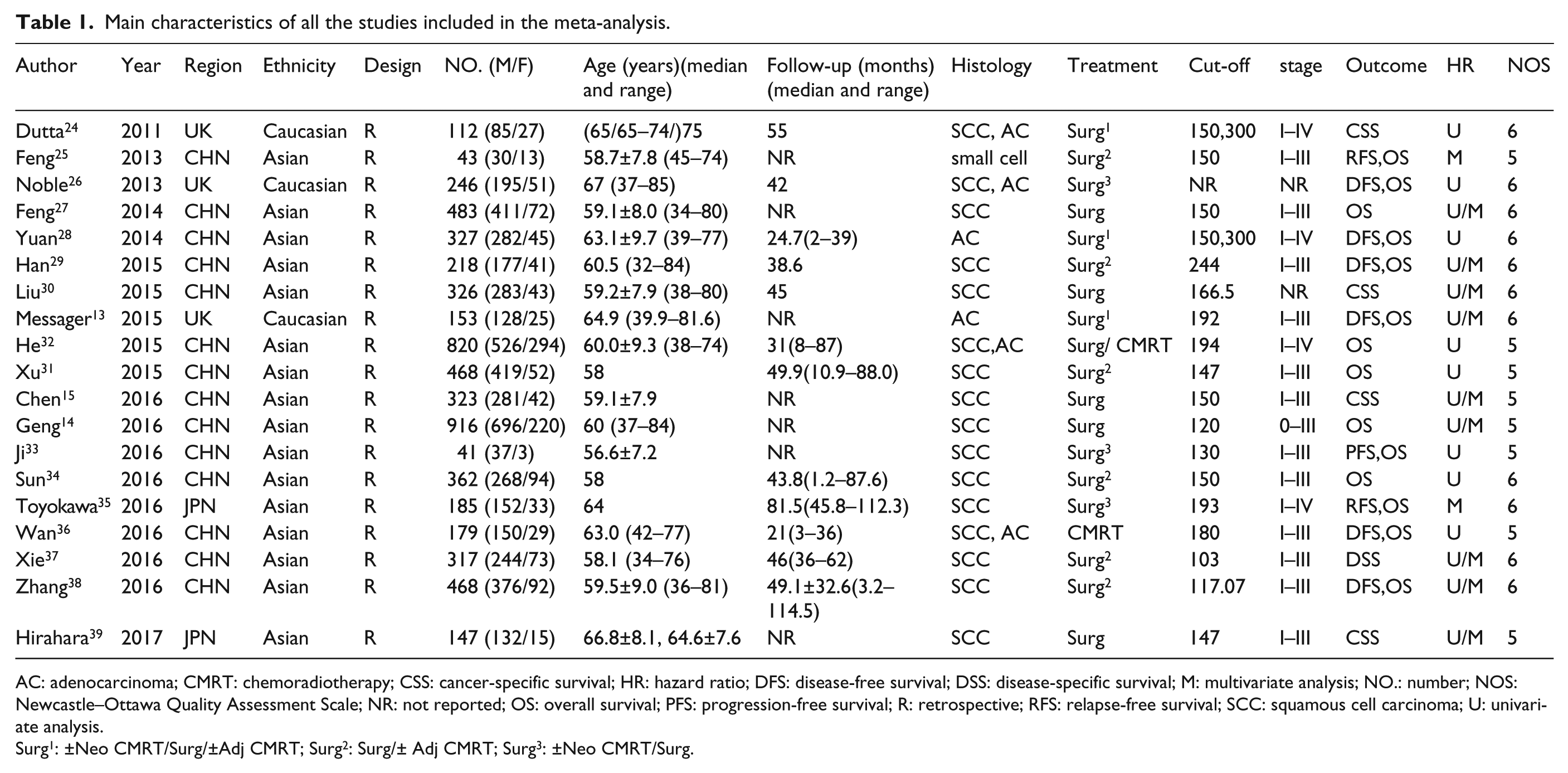
AC: adenocarcinoma; CMRT: chemoradiotherapy; CSS: cancer-specific survival; HR: hazard ratio; DFS: disease-free survival; DSS: disease-specific survival; M: multivariate analysis; NO.: number; NOS: Newcastle–Ottawa Quality Assessment Scale; NR: not reported; OS: overall survival; PFS: progression-free survival; R: retrospective; RFS: relapse-free survival; SCC: squamous cell carcinoma; U: univariate analysis.
Surg1: ±Neo CMRT/Surg/±Adj CMRT; Surg2: Surg/± Adj CMRT; Surg3: ±Neo CMRT/Surg.
PLR and OS
There was significant heterogeneity among studies for categorized PLR (I2=72.20%; P < 0.001) in the 14 studies evaluating OS. The random-effect model was performed to calculate the pooled HR and its 95% CI. The result showed that PLR was significantly associated with OS with the pooled HR 1.263 (95% CI 1.094, 1.458) (Figure 2(a)).
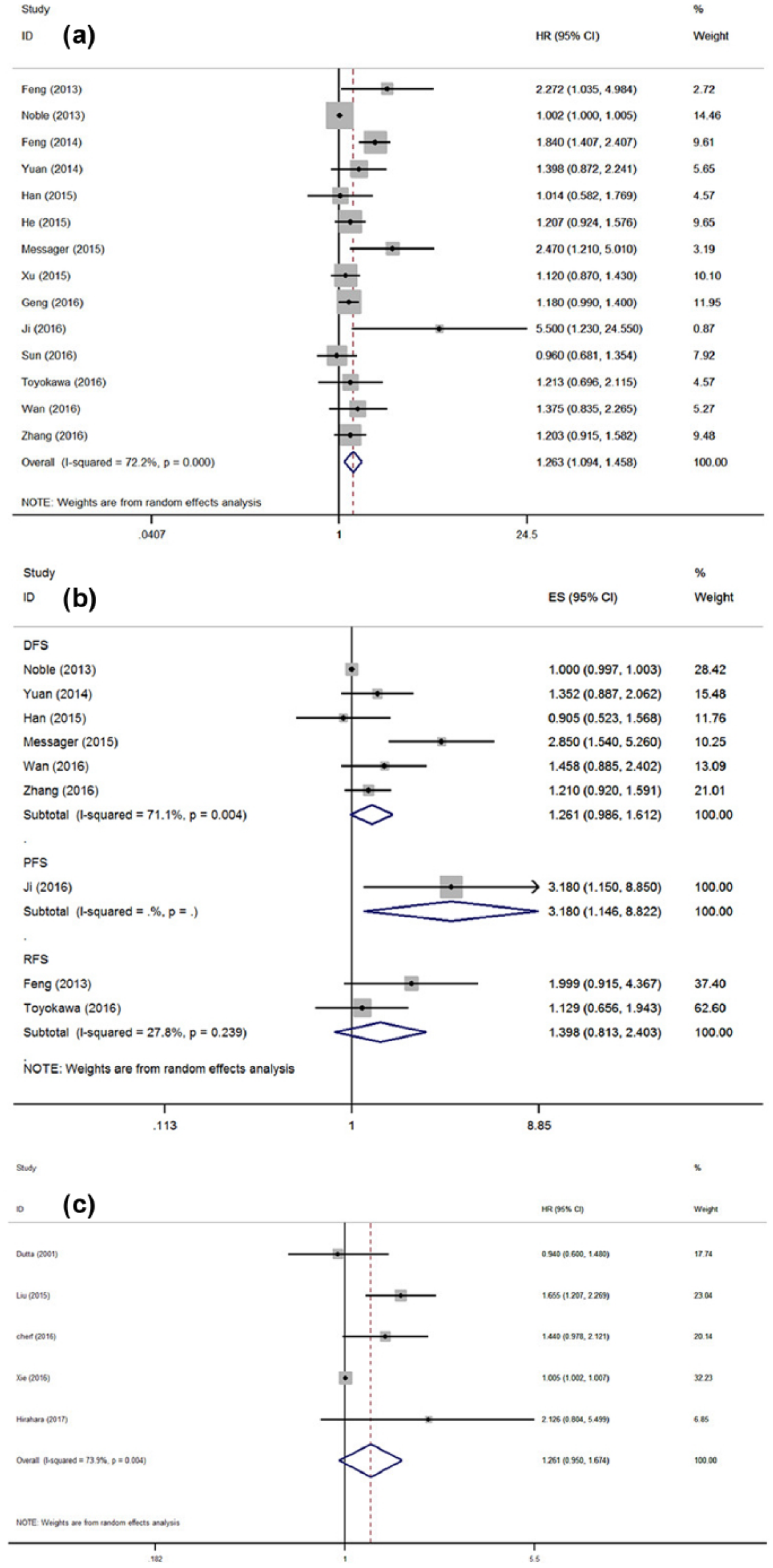
(a) Meta-analysis of the association between elevated PLR and OS in patients with EC. (b) Forest plot of studies evaluating the association between PLR and DFS/PFS/RFS in EC patients. (c) Forest plot of studies evaluating the association between PLR and CSS/DSS in EC patients.
PLR and DFS/PFS/RFS
There were nine studies with 1774 patients presenting the HR and 95% CI of PLR on DFS/PFS/RFS. The combined data showed that PLR was not statistically associated with DFS (HR= 1.261; 95% CI 0.986, 1.612; P= 0.065) or RFS (HR= 1.398; 95% CI 0.813, 2.403; P= 0.239) (Figure 2(b)).
PLR and CSS/DSS
The results showed that PLR was not significantly associated with CSS/DSS (HR obtained from the random-effects model: 1.261; 95% CI 0.950, 1.674; P=0.109. Figure 2(c)) with obvious heterogeneity (I2 = 73.9%; Ph = 0.004).
Subgroup analysis
In consideration of the high heterogeneity, we performed a subgroup analysis to identify the possible source of heterogeneity. Subgroup analysis by ethnicity revealed that PLR was a negative predictor of OS for Asian (HR= 1.252; 95% CI 1.141, 1.373; Ph= 0.072) but not for Caucasian populations (HR = 1.463; 95% CI 0.611, 3.502; Ph= 0.013). A negative association between PLR and OS was found in SCC, AC, and small-cell cancer. Because the PLR cut-off values were different among the included studies, we performed a subgroup analysis based on the different cut-off values. The data demonstrated that the pooled HR was 1.315 (95% CI 1.065,1.624; I2 = 65.6%; Ph=0.008) for studies with a cut-off value ≤ 150 mm and 1.290 (95% CI 1.078, 1.543; I2 = 0%; Ph= 0.49) for studies with a cut-off value > 150 mm. The results of other exploring subgroup analyses by the sample size (< 200 and ≥ 200), HR estimated method (univariate analysis and multivariate analysis), and treatment strategies (surgery or not) are shown in Table 2.There were only six studies providing data for PLR and DFS, and only five studies providing data for PLR and CSS. The forest plot suggested that there was no significant relationship between PLR and DFS or CSS. Therefore, it was unnecessary to conduct a subgroup analysis for DFS or PFS.
Subgroup analysis of pooled hazard ratios (HRs) reflecting the association between PLR and OS in EC patients.
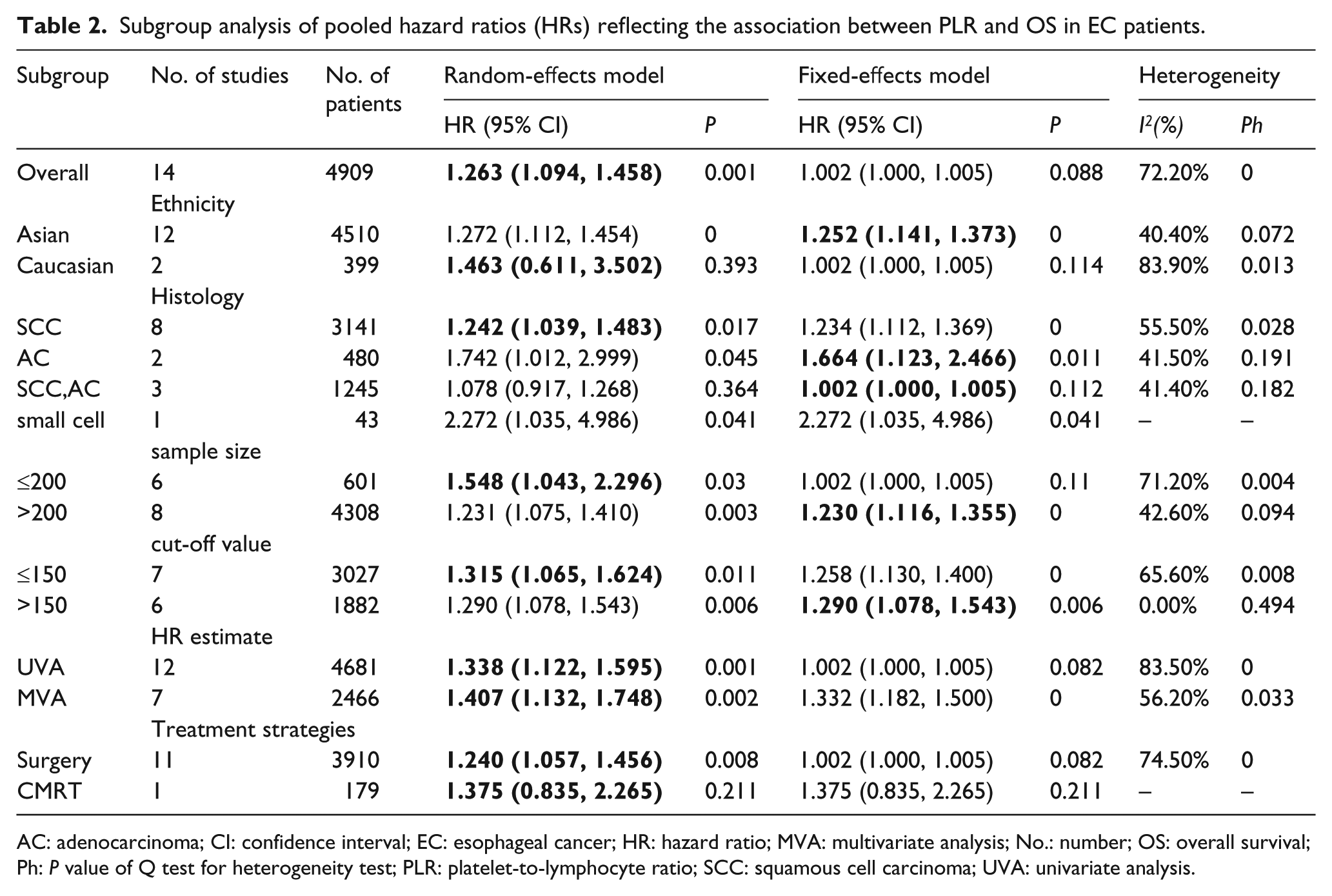
AC: adenocarcinoma; CI: confidence interval; EC: esophageal cancer; HR: hazard ratio; MVA: multivariate analysis; No.: number; OS: overall survival; Ph: P value of Q test for heterogeneity test; PLR: platelet-to-lymphocyte ratio; SCC: squamous cell carcinoma; UVA: univariate analysis.
Sensitivity analyses
The results of the sensitivity analyses did not substantially change when any study was excluded, indicating the robustness of our findings. (Figure 3(a))
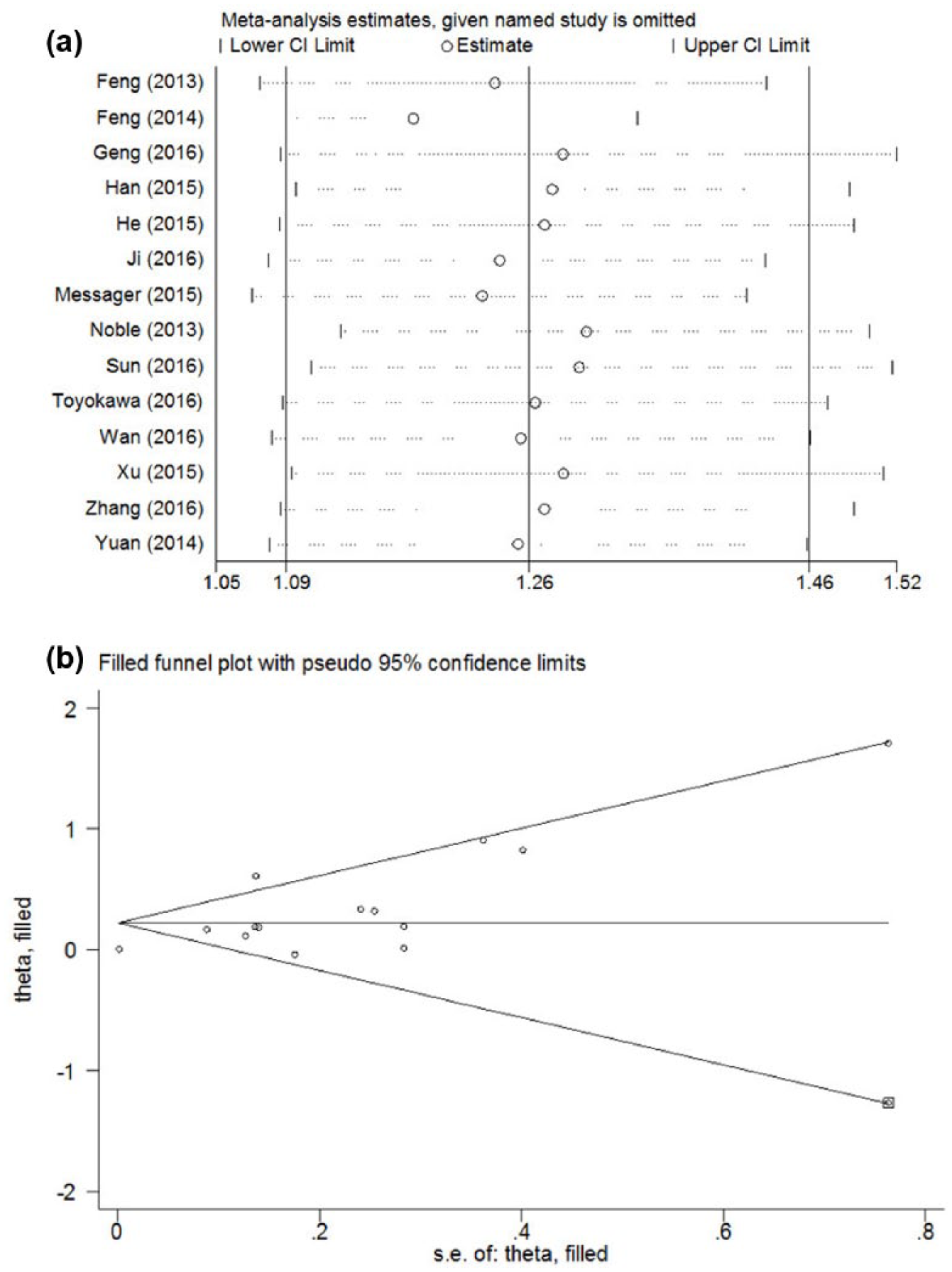
(a) Sensitivity analysis on the relationship between PLR and OS in EC. (b) Funnel plot adjusted using a trim and fill method for OS.
Publication bias
In the analysis of PLR and OS, the Begg’s test and Egger’s test were used to evaluate publication bias. There was significant publication bias on the studies exploring the prognostic value of PLR to EC (P = 0.352 for Begg’s test and P = 0.001 for Egger’s test). Therefore, we used a trim and fill method to estimate the effect of PLR to EC. The recalculated pooled HR of OS did not significantly alter by filling one unpublished study (HR, 1.249; 95% CI 1.080, 1.444; P = 0.003; Figure 3(b)).
Discussion
To the best of our knowledge, there were three meta-analyses summarizing the prognostic value of PLR in ECs, among which one meta-analysis included four articles, and the other two included only one article. Although the three studies consistently showed that elevated PLR was negatively associated with survival in EC, more research with controversial results concerning the prognostic role of PLR have been published. Therefore, it is necessary to conduct an updated meta-analysis to validate the conclusion. Our meta-analysis, including 19 individual studies enrolling 6134 patients, indicated that elevated PLR was significantly associated with poor OS (HR, 1.263; 95% CI1.094, 1.458) of EC patients (Figure 2(a)). A subgroup analysis indicated that PLR is significant in the prognosis of Asian cases, but not in Caucasian cases. More studies should be conducted to explore the prognostic value of PLR for Caucasian cases, because there were only two articles enrolling Caucasian cases in our analysis. Subgroup analyses revealed that poor OS with elevated PLR could be found in both SCC and AC. There was also interaction between PLR and the sample size (≤ 200 or>200). Cut-off values for PLR varied among the studies included. Therefore, we performed a subgroup analysis based on the cut-off values. We found that patients with low PLR had better OS, compared to those with an elevated PLR, regardless of the PLR cut-off values. A subgroup analysis stratified by an HR estimation method also revealed that the PLR had a negative effect on OS. However, we did not find that the elevated PLR was related to the combined DFS, PFS, or CSS, which may be attributed to the limited number of studies included. Taking all of these into consideration, PLR may be a significant biomarker in the prognosis of EC.
The prognostic role of PLR in other gastrointestinal cancers, such as gastric cancer and colorectal cancer, has also been researched. Kim et al. 42 retrospectively analyzed 1986 gastric cancer patients with curative surgery. The results showed that the PLR was not an independent prognostic factor for OS. Similar results were also reported in a meta-analysis conducted by Xu et al., 43 which revealed that an elevated PLR may not act as a negative predictor for the OS, although the elevated PLR was correlated with a more advanced stage risk in gastric cancer. Ozawa et al. 44 retrospectively analyzed 234 patients with stage II colorectal cancer, who underwent a curative resection, and found that the PLR was an independent prognostic factor to DFS and CSS. Similarly, a meta-analysis conducted by Min et al. 45 indicated that pretreatment PLR might be a cost-effective and non-invasive serum biomarker for poor prognosis in colorectal cancer. In addition to PLR, the prognostic role of other systemic inflammation markers, such as NLR, has also been reached in EC. Gao et al. 46 retrospectively analyzed 1281 esophageal squamous cell carcainoma (ESCC) patients and showed that pretreatment NLR was independently associated with OS in these patients.
Tumor-associated systemic inflammatory response is believed to correlate with prognosis and survival outcomes in cancer patients.4,5 The mechanism between inflammation and tumor progression has not been worked out exactly. More and more evidence shows that the systemic inflammatory response may play a key role by promoting angiogenesis and distal spread, suppressing antitumor immunity and impacting the response to anti-cancer therapies. 47 As part of the inflammatory response, thrombocytosis is common in patients with solid tumors. Platelets can directly promote the growth of tumor cells by secreting several angiogenic and tumor growth factors, such as vascular endothelial growth factor, basic fibroblast growth factor, and platelet derived growth factor. Also, platelets could protect cancer cells from natural-killer-mediated lysis and promote distal metastasis by activating the Smad and the NF-kB pathways.48,49 In contrast, lymphocytes play a vital role in the process of T-cell-mediated anti-tumor response. The tumor infiltrating T cells could secrete interleukin (IL)-4, IL-5 and tumor necrosis factor, regulating the angiogenesis, proliferation, apoptosis, and metastasis of cancer. Lymphocytopenia has been demonstrated to predict a poor prognosis in terms of survival.50,51 Taken together, the relative value of a combined elevated platelet and decreased lymphocyte could predict the clinical outcomes of cancer patients more accurately. PLR is a promising prognostic marker for the clinical decision-making process regarding EC therapy and survival.
However, there were some limitations in our meta-analysis. First, all of the included studies were retrospective, which was more prone to some biases. Second, this study was limited to articles published in the English language, which could lead to publication bias. Third, the heterogeneity was significant, which could affect the interpretation of the results. The sources of heterogeneity might result from other factors, including the resource of patients, the research method, the duration of follow-up, the cut-off value of high PLR, the statistic method, and so on. Although we performed subgroup analyses, we could not completely explain the heterogeneity. Also, platelet and lymphocyte counts are non-specific biochemical markers, which may be influenced by concurrent conditions, such as infections, inflammation, and medications. However, most of the included studies did not explicitly control for such concurrent conditions; thus the confounding effect of concurrent inflammatory conditions cannot be completely excluded. Notably, the cut-off values varied among the studies. We could not identify a consistent cut-off value, so we conducted the meta-analysis based on the respective cut-off value of PLR and recognized the combined effect of PLR on the prognosis of EC. Thus the combined HR generally explained the effect of elevated PLR compared to lower PLR on the prognosis of EC. The combined HR may have no direct instructive value, but indicates that PLR may have an influence on the prognosis of EC, and that a certain cut-off value may exist, which still needs to be discovered. Further meta-analysis, including additional studies with large sample sizes, is needed to correct for publication bias and heterogeneity.
Conclusion
In summary, this meta-analysis demonstrated that elevated PLR might be a negative prognostic factor for patients with EC. In the future, more well-designed and large-scale studies are needed to confirm the conclusion.
Footnotes
Author contributions
Xiangwei Zhang and Yang Wang cotributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the Provincial Science and Technology Development Plan of Shandong(2016GSF201107).
