Abstract
Background
Recent studies revealed that various inflammatory and nutritional indexes were associated with prognosis in esophageal cancer (EC). However, these studies only evaluated one or two indexes, and the prognostic value of these indexes individually or in combination is unclear. This study aimed to construct an integrative score based on various inflammatory and nutritional indexes for prognosis in resectable
Methods
A total of 421 consecutive patients were randomly divided into either a training or validation cohort at a ratio of 7:3 for retrospective analysis. Using logic regression analyses, independent risk factors from peripheral blood indexes were screened to construct an integrative score. The associations regarding the integrative score, clinical characteristics, cancer-specific survival (CSS), and overall survival (OS) were analyzed.
Results
Out of 20 indexes, hemoglobin (HB), C-reactive protein to albumin ratio (CAR), and platelet to lymphocyte ratio (PLR) were independent risk factors based on logical regression analyses. Then, an integrative score with the optimal cut-off value of .67 was established according to the Combination Of HB, CAR, and PLR (COHCP). The area under the curve (AUC) indicated higher predictive ability of COHCP on prognosis than other indicators. Multivariate analyses revealed that COHCP serves as an independent prognostic score. Patients with COHCP low group (≤.67) had better 5-year CSS (57.3% vs 13.5%, P < .001) and OS (51.1% vs 12.3%, P < .001) than those with high group, respectively. Finally, the nomogram based on COHCP was established and validated regarding CSS and OS, which can accurately and effectively predict individual survival in resected ESCC.
Conclusion
The COHCP was a novel, simple, and useful predictor in resectable ESCC. The COHCP-based nomogram may accurately and effectively predict survival.
Keywords
Introduction
According to the 2018 Global cancer statistics, a total of 572 034 new cases of esophageal cancer (EC) were diagnosed and 508 585 cases died from EC. 1 Esophageal squamous cell carcinoma (ESCC), in terms of pathology, is the main type of EC, especially in China and other highest risk area of Asia and Africa. 2 The prognosis of ESCC is extremely poor because of the inability of early detection the disease. Despite the progress of medical science and the improvement of treatments in recent years, the survival of EC remains poor. 3 Therefore, the rising incidence and poor prognosis for this disease highlight the need for improving more predictive indicators that are essential prior to treatment.
Recent studies revealed that inflammatory and nutritional status are associated with tumor prognosis. 4 Therefore, a large number of peripheral inflammatory and/or nutritional indexes, such as hemoglobin (HB), platelet (PLT), monocyte (MONO), lymphocyte (LYMPH), neutrophil (NEUT), albumin (ALB), prealbumin (PALB), C-reactive protein (CRP), CRP to ALB ratio (CAR), NEUT to LYMPH ratio (NLR), PLT to LYMPH ratio (PLR), prognostic nutritional index (PNI), and systemic immune-inflammation index (SII), have been reported to be associated with tumor prognosis.5-12 However, nutritional and/or inflammatory status may influenced by various non-cancer-related conditions, which may lead to biased results. Moreover, these peripheral indexes are deficient in some respects, and the results for some indexes are still controversial.13,14 In addition, these studies only evaluated one or two indexes, and the value of these indexes individually or in combination is still unclear.
We hypothesized that the combination of these indicators could reduce the potential bias and improve the prognostic value. In this study, therefore, we constructed an integrative score based on various inflammatory and nutritional indexes for predicting prognosis in resectable ESCC. In addition, two cohorts were used to verify the predictive value of the integrative score. Finally, a predictive nomogram based on the integrative score was also constructed and validated to predict individual survival.
Methods
Patient Selection
This study was conducted in accordance with the Declaration of Helsinki and approved by the ethics committee of our hospital (IRB.2021-6). The clinical data in the current study was retrospective and anonymous. Therefore, the informed consent was waived. The current retrospective cohort study included 685 consecutive stage I–III resected ESCC patients in our hospital from January 2011 to August 2013. The inclusion and exclusion criteria were shown in Figure 1. Finally, a total of 421 patients were randomly divided into either a training cohort (n = 294) or validation cohort (n = 127) at a ratio of 7:3 for retrospective analysis. The reporting of this study conforms to STROBE guidelines.
15
Flow diagram of selection of eligible patients. According to the inclusion and exclusion criteria, 421 patients were randomly divided into either a training cohort (n = 294) or validation cohort (n = 127) at a ratio of 7:3 for further analysis.
Treatment and Follow-Up
The main standard procedure in the current study consisted of subtotal esophagectomy with two-field lymphadenectomy via right thoracotomy, including either McKeown procedure (for patients in the upper third) or Ivor Lewis procedure (for patients in the middle or lower third).16,17 Neoadjuvant therapy may affect the preoperative hematological indicators and thus affect the results of this study, so patients receiving neoadjuvant treatments were excluded from this study. At that time, postoperative adjuvant treatment was still uncertain. For ESCC patients with radical resection, NCCN guidelines only recommend regular follow-up. Thus, not all ESCC patients in China have received postoperative adjuvant therapy, which is mainly carried out according to the doctors' recommendations based on postoperative pathological results as well as the physical and financial status of each patient.18,19 Similar to previous studies, postoperative adjuvant treatments were performed for ESCC patients with T3-T4 stage and those with positive lymph node (LN) metastasis.20,21 The adjuvant treatments were performed, but not mandatory, including cisplatin-based chemotherapy and/or radiotherapy with a median irradiation dose of 50 Gy. The patients were followed up with regular checks. The last time was completed in December 2019.
Data Collection and Analyses
Data regarding clinical characteristics and preoperative indexes of systemic inflammation and nutrition (within one week before surgery) were retrospectively extracted from the medical records. The tumor node metastasis (TNM) stage regarding ESCC in the current study based on the seventh AJCC/UICC TNM staging system. 22 Preoperative indexes were from daily blood routine examination and biochemical test. The automated blood cell counter (Sysmex XE-2100, Kobe, Japan) and automated biochemical analyser (Hitachi 917, Mannheim, Germany) were used to measure the levels of indexes in blood routine examination and biochemical test, respectively. The definitions of PNI [10 × ALB (g/dl) + .005 × LYMPH(/mm3)] and SII (PLT × NEUT/LYMPH) referred to the previous published studies.6,12
Statistical Analysis
R 3.6.0 software, Medcalc 17.6 (MedCalc Software bvba, Ostend, Belgium) and SPSS 20.0 (SPSS Inc., Chicago, IL, USA) were used to perform statistical analyses. Logical regression was carried out for all inflammatory and nutritional indicators. Then independent risk factors from peripheral blood indicators were screened to construct an integrative score. Categorical variables were analyzed by chi-square or Fisher’s exact tests, while continuous variables were analyzed by Student’s t-tests. The ROC curves were carried out to identify the optimal cut-off values and explore the predictive accuracy of inflammatory and nutritional indicators for the areas under the curve (AUCs). Univariate and multivariate analyses were performed to analyze independent factors with hazard ratios (HRs) and 95% confidence intervals (CIs). A nomogram was established and validated by measuring discrimination and calibration in both training cohort and validated cohort. 23 All statistical tests were two-side, a P value < .05 was considered to be statistically significant.
Results
Patient Characteristics
Baseline Characteristics of ESCC Patients in the Training and Validation Cohorts.

ESCC: esophageal squamous cell carcinoma; TNM: tumor node metastasis; LN: lymph node; OE: open esophagectomy; MIE: minimally invasive esophagectomy; NEUT: neutrophil; LYMPH: lymphocyte; MONO: monocyte; PLT: platelet; HB: hemoglobin; CRP: C-reactive protein; ALB: albumin; PALB; prealbumin; LDH: lactate dehydrogenase; CAR: CRP to ALB ratio; CPR: CRP to PALB ratio; CHR: CRP to HB ratio; CLR: CRP to LYMPH ratio; NLR: NEUT to LYMPH ratio; NHR: NEUT to HB ratio; PLR: PLT to LYMPH ratio; PHR: PLT to HB ratio; LMR: LYMPH to MONO ratio; LAR: LDH to ALB ratio; LPR: LDH to PALB ratio.
Combination of HB, CAR, and PLR Definition and Baseline Characteristics Analyses
All 20 variables, as continuous variables, were calculated according to the logistic equation. According to the logical regression, HB, CAR, and PLR were significant independent risk factors from all peripheral inflammatory and nutritional indicators. Subsequently, the logistic regression equation was as follows: Y = −.045*HB + .013*PLR + 5.033*CAR. Therefore, an integrative score Combined Of HB, CAR, and PLR (COHCP) was established. According to the regression equation, the continuous variable of COHCP = 111.8*CAR+.29*PLR-HB. The scatter diagrams and correlation diagrams about COHCP and its components of HB, CAR, and PLR were shown in Figure 2. The optimal cut-off points, according to the ROC curves, for COHCP and its components of HB, CAR, and PLR were .67, 120.5 g/L, .06, 152, respectively. Similarly, the optimal cut-off values for other conventional score of PNI and SII were 47.5 and 566, respectively. The baseline characteristics grouped by COHCP were shown in Table 2. However, there was no statistical difference between the surgical procedures grouped by COHCP (P = .118). Interestingly, COHCP was associated with postoperative pneumonia (P = .004) but not with AL (P = .248). Scatter and correlation diagrams of HB, CAR, PLR, and COHCP. Negative correlations between HB and COHCP (r = −.397, P < .001). Positive correlations between HB and PLR (r = .174, P = .003), CAR and COHCP (r = .582, P < .001), and PLR and COHCP (r = .536, P < .001), respectively. Comparison of Baseline Clinical Characteristics based on COHCP in ESCC. ESCC: esophageal squamous cell carcinoma; LN: lymph node; OE: open esophagectomy; MIE: minimally invasive esophagectomy; COHCP: Combined of HB, CAR, and PLR; HB: hemoglobin; CAR: C-reactive protein to albumin ratio; PLR: platelet to lymphocyte ratio; PNI: prognostic nutritional index; SII: systemic immune-inflammation index; TNM: tumor node metastasis.

Area under the curves Comparison Between COHCP and Other Indicators According to ROC Analyses
To better understand the predictive value, we compared the AUCs between COHCP and its components (HB, CAR, and PLR) and other conventional scores (PNI and SII). Area under the curves for COHCP, HB, CAR, PLR, SII, and PNI were shown in Figure 3 (A for continuous variables and B for categorical variables). According to the ROC curves, COHCP had the largest AUC (.771 for continuous and .744 for categorical) compared with other indicators, which indicated that higher predictive ability of COHCP on prognosis than other indicators. ROC analyses regarding AUC comparison between COHCP and HB, CAR, PLR, SII, and PNI. COHCP had the largest AUC (.771 for continuous (A) and .744 for categorical (B)) compared with other prognostic scores, which indicated that high predictive ability on prognosis.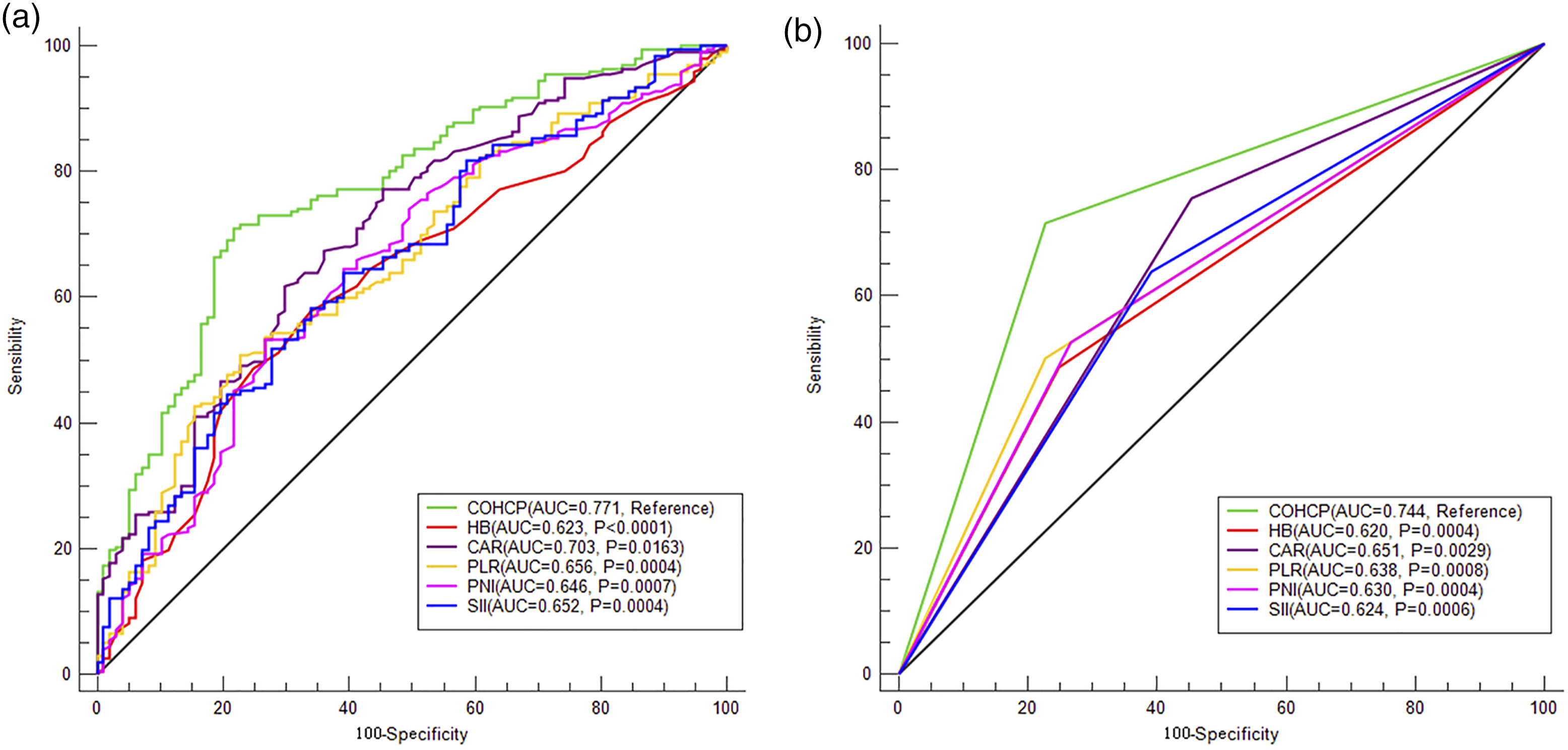
Univariate and Multivariate Analyses for Independent Prognostic Factors
Univariate Cox Analyses of CSS for ESCC in the Training Cohort.

ESCC: esophageal squamous cell carcinoma; LN: lymph node; OE: open esophagectomy; MIE: minimally invasive esophagectomy; COHCP: Combined of HB, CAR, and PLR; HB: hemoglobin; CAR: C-reactive protein to albumin ratio; PLR: platelet to lymphocyte ratio; PNI: prognostic nutritional index; SII: systemic immune-inflammation index; TNM: tumor node metastasis; HR: hazard ratio; CI: confidence interval; CSS: cancer-specific survival.
Univariate Cox Analyses of OS for ESCC in the Training Cohort.

ESCC: esophageal squamous cell carcinoma; LN: lymph node; OE: open esophagectomy; MIE: minimally invasive esophagectomy; COHCP: Combined of HB, CAR, and PLR; HB: hemoglobin; CAR: C-reactive protein to albumin ratio; PLR: platelet to lymphocyte ratio; PNI: prognostic nutritional index; SII: systemic immune-inflammation index; TNM: tumor node metastasis; HR: hazard ratio; CI: confidence interval; OS: overall survival.
Multivariate Cox Analyses of CSS and OS in ESCC.
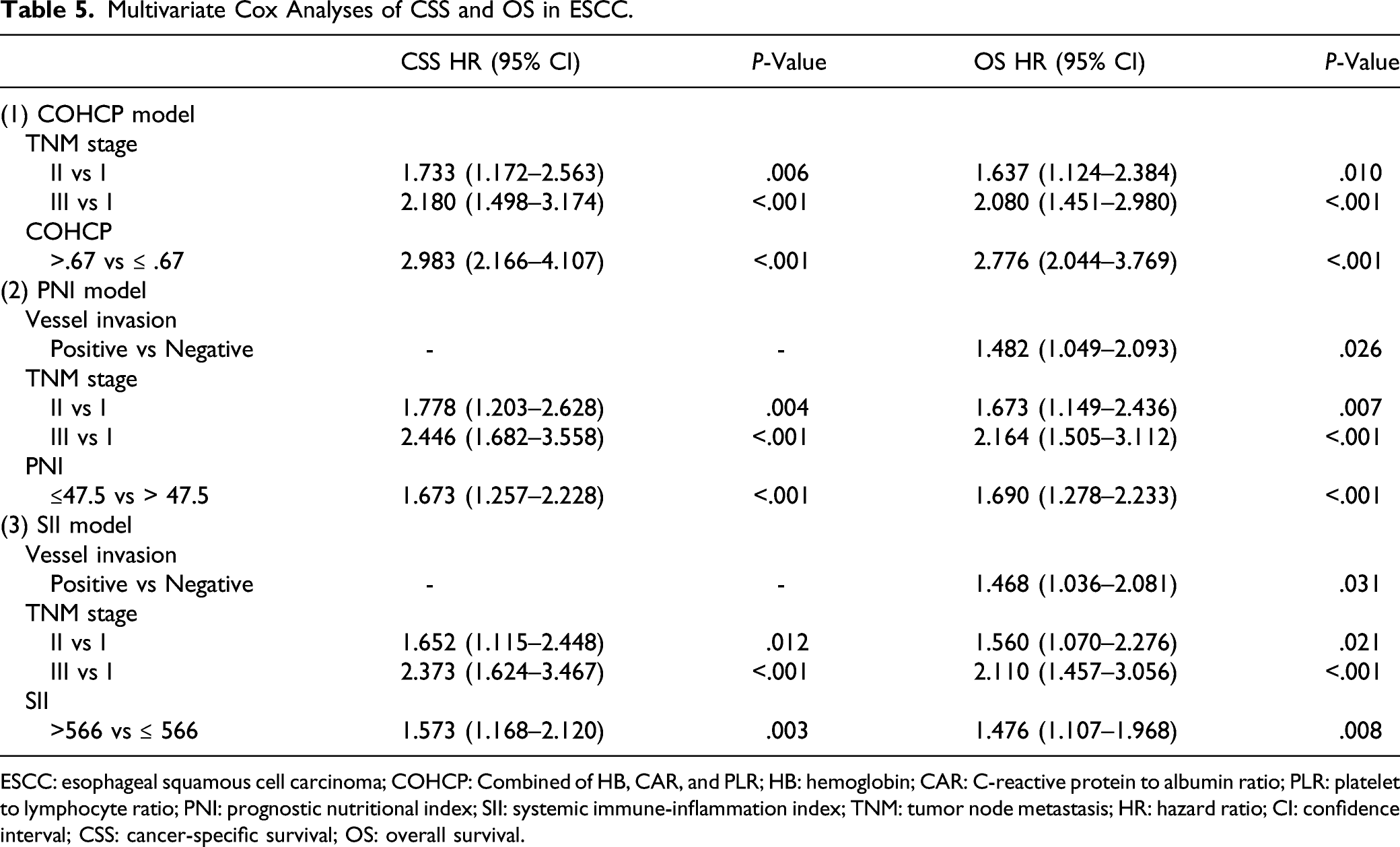
ESCC: esophageal squamous cell carcinoma; COHCP: Combined of HB, CAR, and PLR; HB: hemoglobin; CAR: C-reactive protein to albumin ratio; PLR: platelet to lymphocyte ratio; PNI: prognostic nutritional index; SII: systemic immune-inflammation index; TNM: tumor node metastasis; HR: hazard ratio; CI: confidence interval; CSS: cancer-specific survival; OS: overall survival.
Cancer-specific survival and OS Analyses and Subgroup Analyses
The survival curves of CSS and OS grouped by COHCP were shown in Figure 4A-B. Patients in high group (COHCP > .67) had worse 5-year CSS (13.5% vs 57.3%, P < .001) and OS (12.3% vs 51.1%, P < .001) than those in COHCP low group, respectively. In order to better explore the prognostic value of COHCP, subgroup analyses based on different TNM stages were performed. The results in the current study also demonstrated that poor 5-year CSS and OS in COHCP high group in subgroup analyses based on different TNM stages (Figure 4C-H). In addition, our study revealed that patients with MIE have better 5-year CSS (42.2% vs 27.6%, P = .013) and OS (36.7% vs 25.4%, P = .028) than those with OE, respectively. Patients with postoperative pneumonia had worse 5-year CSS (24.4% vs 36.5%, P = .013) and OS (22.1% vs 32.7%, P = .016) than those without pneumonia, but AL was not associated with prognosis. Kaplan–Meier for CSS and OS grouped by COHCP. Patients in high group had worse 5-year CSS (13.5% vs 57.3%, P < .001; A) and OS (12.3% vs 51.1%, P < .001; B) than those in low group. Subgroup analysis between COHCP and CSS and OS based on different TNM stages demonstrated good stratification significances (CSS: C-E; OS: F-H).
Nomogram Development and Validation
A predictive nomogram including two independent variables in multivariate analyses (TNM and COHCP) were established to predict 1-, 3-, and 5-year CSS (Figure 5A) and OS (Figure 5B). The C-indexes were .68, .70 for CSS, and .65 and .68 for OS in the training cohort and validation cohort, respectively. The calibration curves revealed acceptable agreements between these two cohorts regarding the individual 5-year CSS (Figure 6A-B) and OS (Figure 6C-D). The nomogram had higher overall net benefits of 5-year CSS (Figure 6E-F) and OS (Figure 6G-H) prediction than TNM stage based on the decision curve and time-dependent ROC curve analyses (Figure 6I-J for CSS and Figure 6K-L for OS, respectively). Therefore, the COHCP-based nomogram may accurately and effectively predict individual survival (CSS or OS) in resected ESCC. Nomogram model for CSS and OS prediction. Nomogram model predicts 1-, 3-, and 5-year CSS (A) and OS (B) in ESCC based on TNM stage and COHCP. Therefore, clinicians may use these nomograms to predict individual survival. Nomogram regarding calibration curves, decision curves, and time-dependent ROC curves. Calibration curves presented an acceptable agreement between the two cohorts in CSS (A-B) and OS (C-D). Decision curve analyses revealed nomogram model had higher overall net benefits than TNM stage for CSS (E-F) and OS (G-H). Time-dependent ROC curve analyses revealed survival prediction was significant higher in nomogram than TNM stage regarding CSS (I-J) and OS (K-L).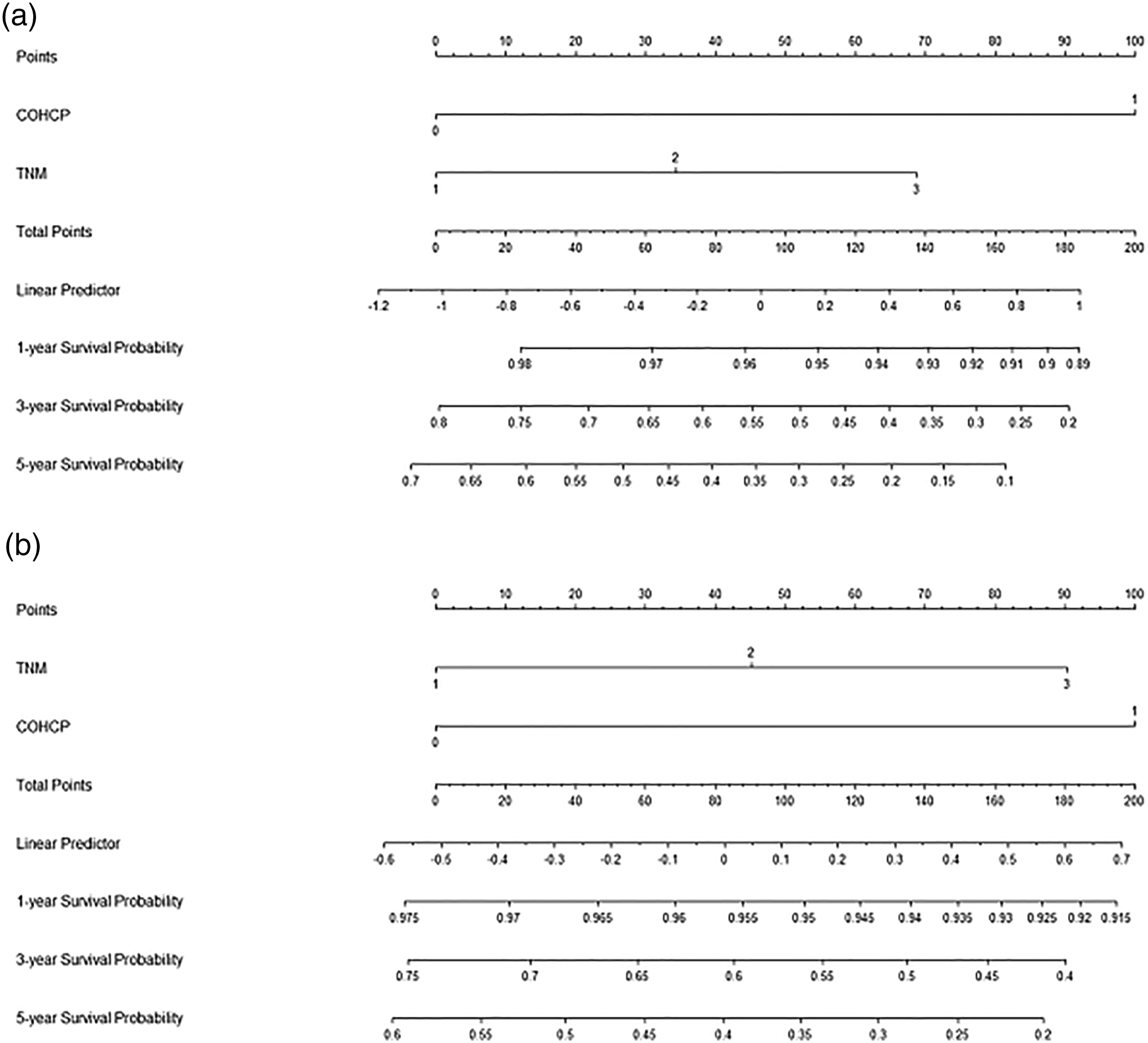

Discussion
The present study constructed an integrative score (COHCP) based on various inflammatory and nutritional indexes and confirmed the prognostic effect of the COHCP (combined with HB, CAR, and PLR) in multivariate analyses for patients with resectable ESCC. Finally, a new prognostic nomogram based on COHCP and TNM was firstly established and validated, which indicated that the nomogram can accurately and effectively predict individual survival in resected ESCC.
Nutrition and inflammation are associated with tumor prognosis. Decreased HB was the most common hematological abnormality in cancers. 24 An increasing evidences have revealed that decreased HB was associated with poor prognosis in cancers, including ESCC.9,25,26 C-reactive protein to albumin ratio and PLR were the most widely recognized indicators for prediction of prognosis in a number of cancers, including ESCC.5,7,10 Several meta-analyses have demonstrated that CAR and PLR were significantly related to prognosis in patients with ESCC.27-29 Several studies have reported the prognostic value of the combination use of HB, PLR and/or CAR with other potential markers.30,31
The exact mechanism between COHCP and cancer was still unknown. Study has reported that decreased HB leads to hypoxia of tumor cells, stimulates tumor growth, and increases the resistance to radiotherapy and chemotherapy. 32 Lymphocytes play an important role in the process of anti-tumor response, regulating the tumor angiogenesis, proliferation, apoptosis, and metastasis. 33 In contrast, PLTs can directly secrete a variety of tumor growth factors and angiogenic factors to promote tumor cells growth. 34 Study has also reported that elevated serum CRP can induce various inflammatory cytokines associated with cancers, such as interleukin-6. 35 As a common marker regarding nutritional status, ALB can activate a variety of cytokines, such as interleukin-1 and tumor necrosis factor-α. 36
It is well known that serum HB, CAR, and PLR are common clinical markers in daily clinical practice. Compared to previous studies, the current study had several advantages: First, most previous studies only evaluated one or two indexes. Second, the prognostic nomogram model based on the combination of inflammatory and nutritional score with TNM stage system was more accurate in predicting survival than that of the conventional TNM stage system. Third, our model offers a convenient method in predicting outcomes for surgical patients in ESCC. Patients with elevated COHCP may benefit from nutritional and/or inflammatory interventions. It is noted that preoperative nutritional support improved outcome. However, previous studies revealed that some anti-inflammatory drugs may represent a good strategy for cancer prevention and therapy in several Western countries. 37
Based on the key evidence from trials in Western countries, 38 NCCN guidelines recommend neoadjuvant treatments for locally advanced EC. However, a large number of ESCC patients with locally advanced stage in China tended to prefer surgery as the initial treatment.39,40 In a study of 11 791 patients who underwent radical resection of ESCC at 542 participating hospitals, 31.8% were stage II and 50.3% were stage III. However, only 18.5% of patients received neoadjuvant treatment. 41 For ESCC patients with radical resection, NCCN guidelines only recommend regular follow-up. Thus, not all ESCC patients in China have received postoperative adjuvant therapy, which is mainly carried out according to the doctors' recommendations based on postoperative pathological results as well as the physical and financial status of each patient.18,19 Several studies indicated that postoperative adjuvant therapy had survival benefits for those with T3-T4 stage, positive LN metastasis, and positive resection margin.20,21,42
In recent years, MIE has become a standard surgical method for EC. Compared with OE, MIE has a variety of advantages, such as decreased morbidity, shorter hospital stay, and rapid recovery and discharge. 43 Compared with OE, a meta-analysis including 55 relevant studies revealed that 18% lower 5-year all-cause mortality after MIE (HR = .82, 95% CI: 0.76–.88). 44 The similar results were also found in another study. 45 In the current study, our study revealed that patients with MIE have better 5-year CSS (42.2% vs 27.6%, P = .013) and OS (36.7% vs 25.4%, P = .028) than those with OE, respectively. However, MIE was not an independent prognostic factor in further multivariate analysis.
AL and pneumonia were the two main postoperative complications in ESCC. A study including 434 ESCC patients who underwent radical resection revealed that complication had no effect on long-term survival, despite an increasing immediate postoperative outcome and hospital mortality. 46 Another research including 1100 EC patients with resection indicated that AL did not adversely affect survival (the incidences of AL was 9.6%). 47 While another study indicated an opposite result. 48 In the current study, the incidence of AL was 8.2% in the training cohort and 9.4% in the validation, respectively. Several research studies have revealed that postoperative pneumonia was a risk factor for a decreased survival in EC patients with radical resection.49,50 The similar result was found in the current study. Several possible reasons why postoperative pneumonia affects the long-term outcome of EC indicated that patients who developed postoperative pneumonia may have affected pathways that led to decreased host immunity against the tumor.51,52
The results determined that COHCP was a novel, simple, and useful predictor in resectable ESCC. First, our results indicated that COHCP has potential application in the clinical treatment of ESCC. We believe that patients with increased levels of COHCP in ESCC should be regarded with caution. Closer follow-up may be required for early-stage patients and more adjuvant therapy may be required for those with local advanced stage after surgical resection. Second, published research studies revealed that malnutrition was highly prevalent in patients with EC, which was associated with outcomes. Nutritional supplementation before surgery appeared to be an effective strategy for reducing postoperative complications and mortality and improving short-term survival. 53 Third, published studies demonstrated that anti-inflammatory drugs were associated with decreased cancer incidence and recurrence in several western studies.37,54,55 Therefore, a variety of anti-inflammatory drugs (such as aspirin, celecoxib, ibuprofen, and dexamethasone) targeting inflammation and the molecules (such as cyclooxygenase 2, vascular endothelial growth factor, and NF-κB) involved in inflammatory process may be used as adjuvants for conventional therapies, but additional studies are needed to better understand their potential in anticancer treatments. 55 Finally, the results of our study need further confirmation.
Several limitations in this study should be acknowledged. First, owing to retrospective study in a single center, studies with low numbers were usually underpowered to show any statistical significance. Therefore, the results for the current study were correlated to certain bias and inaccuracy. Second, the current study did not calculate sample size analysis, which may affect the statistical significance of the results. However, we believed that the current sample size was enough to enable a reliable statistical result. Third, neoadjuvant therapy is a recommended treatment for local advanced ESCC based on the current NCCN guidelines. However, unlike the neoadjuvant therapy recommended by NCCN, a large number of ESCC patients select surgery first in China, which is also recommended by the Chinese guidelines. Therefore, the results should be regarded with caution. Fourth, the data in the current study were relatively old. Therefore, the significance of the results of this study was limited for the current treatment and prognosis due to the new technologies and medical advances. Fifth, although the strict inclusion and exclusion criteria were adopted in the current study, serum markers may be affected by other conditions, and the results should be regarded with caution. Finally, these two cohorts were from the single center, which may reduce the generalizability. Despite the above limitations, the current COHCP-based nomogram may still accurately and effectively predict individual survival in resected ESCC.
Conclusion
In summary, we initially proposed a novel integrative score (COHCP) based on inflammatory and nutritional score. The results determined that COHCP was a novel, simple, and useful predictor in resectable ESCC. The current COHCP-based nomogram may accurately and effectively predict individual survival. The COHCP may allow for treatment stratification, thereby helping clinicians provide a more personalized approach to cancer treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from Zhejiang Medical and Health Science and Technology Project (2018KY290, 2019RC129). This study was also supported by Zhejiang TCM Science and Technology Project (2021ZB034).
Ethics Statement
This study was conducted in accordance with the Declaration of Helsinki and approved by the ethics committee of Zhejiang Cancer Hospital (IRB.2021-6). This study was a retrospective study with anonymous data, the informed consent was waived.
