Abstract
The platelet-lymphocyte ratio (PLR) has been assessed in some studies on renal cell carcinoma (RCC), but the results have been inconsistent. This meta-analysis aims to review and report the latest data regarding the prognostic role of the PLR in RCC patients. Articles were searched in the PubMed, EMBASE, and Cochrane Library electronic databases. Studies were filtered according to a selection strategy, and data corresponding to the index of interest were extracted. A fixed-effects model or random-effects model was selected based on heterogeneity. The sensitivity analysis was carried out by eliminating the studies one by one. Finally, funnel plots and Egger's test were used to assess publication bias, and the trim and fill method was used to assess the impact of bias on the results. In total, 15,193 patients with RCC from 44 studies were included in this meta-analysis. The pooled analysis indicated that the higher the PLR was, the poorer the prognosis for RCC patients in terms of overall survival (hazard ratio (HR) = 1.01 (95% confidence interval (CI) 1.00, 1.02), P = 0.010), cancer-special survival (CSS) (HR = 1.21 (95% CI 1.00, 1.46), P = 0.05), progression-free survival (HR = 1.44 (95% CI 1.28, 1.62), P < 0.00001), recurrence-free survival (HR = 1.73 (95% CI 1.11, 2.71), P = 0.02), disease-free survival (HR = 1.63 (95% CI 0.91, 2.94), P = 0.01) and metastasis-free survival (HR = 1.223 (95% CI 0.712, 2.099), P = 0.466). In the subgroup analysis of high PLR, targeted treatment, TKI use, nivolumab use, surgical treatment, clear cell RCC, metastasis, Asian race, and high PLR were related to poor prognosis. This study showed that a high PLR was associated with the poor prognosis of RCC patients, but more studies are needed to confirm the value of the PLR.
Introduction
Although the treatment of renal cell carcinoma (RCC) has seen substantial improvements, it still accounts for 2%–3% of adult cancers, and 102,000 patients die from it every year. The number of RCC patients has increased over the past several years. 1 Among them, 30% develop metastasis, which is difficult to cure. Surgeons used to believe that only a small portion of patients were sensitive to immunotherapy, and that toxicity from immunotherapy occurs. Therefore, targeted therapies have been the main treatment method applied in clinical work. 2 However, as research progresses, substantial changes have been made in the medical management of mRCC (metastatic RCC). 3 Several meta-analyses based on phase III clinical trials assessed the rationality of immunotherapy. Compared with sunitinib monotherapy, nivolumab plus ipilimumab combination (N + I) treatment represents an immune-based combination that does not appear to present significantly higher toxicities. In addition, immune-based combination therapies were considered to reduce the risk of death by 26% compared to sunitinib4 and led to better health-related quality of life with higher cost effectiveness. 4 Moreover, the complete response rate based on immune combinations showed a triple increase, 5 and immune checkpoint inhibitors plus tyrosine kinase inhibitors (TKIs) have been proven to be beneficial to patients. 3 However, reliable and validated predictive biomarkers are urgently needed to evaluate the prognosis of patients treated with immunotherapy. 4
A recent study confirmed that the inflammatory response increases with tumor development. 6 Inflammation markers, such as C-reactive protein (CRP), neutrophil-to-lymphocyte ratio (NLR), monocyte-to-lymphocyte ratio (MLR), and platelet-to-lymphocyte ratio (PLR), have been studied extensively. Because the PLR is easy and inexpensive to determine, an increasing number of studies have assessed its value for predicting prognosis. 7 The PLR has been proven to have value in other solid tumors in recent years, such as esophageal squamous cell carcinoma, 8 oral cancer, 9 and small-cell lung cancer. 10 An increasing number of studies on the PLR of RCC patients have been published, but the results have been inconsistent. Wang et al. 11 evaluated the prognostic significance of the PLR, and Wang et al. 12 also performed meta-analyses including seven retrospective studies that proved that a higher PLR indicated poor overall survival (OS) and progression-free survival (PFS) in RCC patients. Nevertheless, due to some limitations in their studies, the value of the PLR in determining the prognosis of RCC patients needs to be assessed further.
Materials and methods
Search strategy and selection criteria
We searched articles in the PubMed, EMBASE, and Cochrane Library electronic databases. All English studies published up to 9 June 2021 were searched without any country or article type restrictions. All selected articles were independently screened to avoid missing additional studies in the initial search. Finally, we selected 44 records according to the workflow in Figure 1.
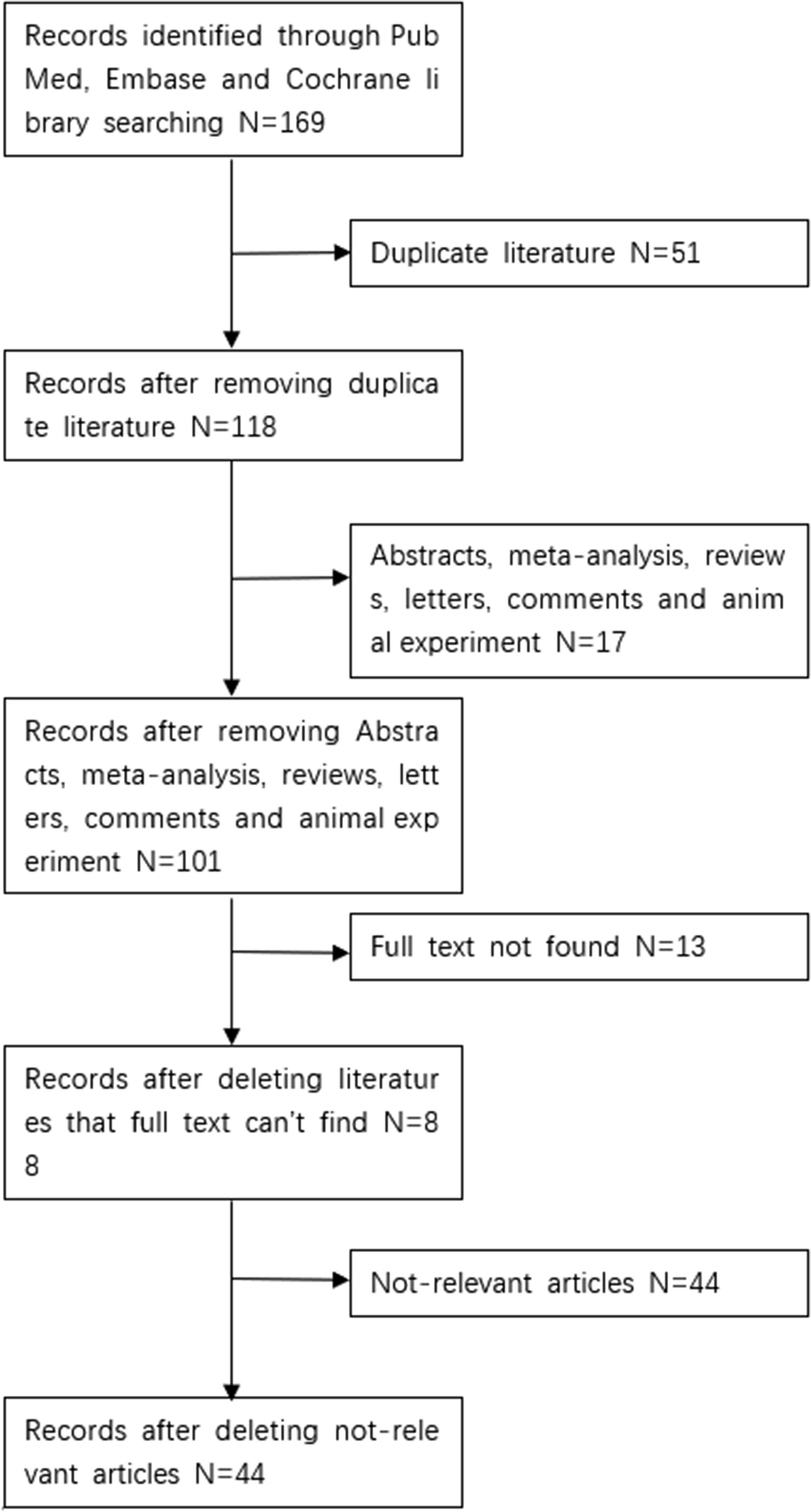
Flowchart of study selection.
Two authors independently searched and selected the studies. Any disagreements were resolved by discussion until a consensus was reached. The eligible studies met the following criteria: (a) confirmed RCC; and (b) OS, cancer-special survival (CSS), PFS, recurrence-free survival (RFS), disease-free survival (DFS), and metastasis-free survival (MFS) based on hazard ratios (HRs) and 95% confidence intervals (CIs) were reported.
Data extraction and quality
Two authors independently extracted the data. Any disagreements were resolved by discussion until a consensus was reached. Then, the Newcastle–Ottawa Scale (NOS) was used to assess the quality of the studies, and the studies were defined as high quality when the NOS score was ≥5. The following data were extracted: author, year of publication, country in which the study was conducted, tumor stage, statistical approach, total number of people included in the study, pathological characteristics, therapies, and PLR. We used Excel with the method proposed by Tierney to extract HRs and 95% CIs when studies only provided Kaplan–Meier curves.
Statistical analysis
We used RevMan 5.3 software to perform the meta-analysis. I2 <50 and P > 0.1 indicated that the heterogeneity was not significant, and a fixed effects model was used. I2 >50 and P < 0.1 indicated significant heterogeneity, and a random effects model was used. Subgroup analysis was performed to ascertain the results of the meta-analysis and to identify the source of heterogeneity. Sensitivity analysis was performed to ascertain the results of the meta-analysis by excluding each of the individual studies. Then, we used a funnel plot generated using RevMan 5.3 and Egger’s test via STATA 15.1 to assess publication bias, and used the trim and fill method to assess the impact of the bias on the result. The protocol for this systematic review was registered on INPLASY (Unique ID number) and is available in full on inplasy.com (https://inplasy.com/inplasy-2021-9-0064/).
Results
Included studies
According to the flow chart in Figure 1, we identified 169 potential studies that were potentially eligible for inclusion in our research based on our search formula. After excluding 51 duplicate studies and 17 unrelated study types—including abstracts, meta-analyses, reviews, letters, comments, and animal experiments—101 studies remained. Then, 13 studies that did not have full texts available were excluded. We excluded 44 unreliable articles after reading the full articles, and ultimately, 44 studies were included.7,13–55
Characteristics of the studies
As shown in Figure 2, most of the studies were retrospective and published between 2013 and 2021. In total, 15,193 patients with RCC were included in this meta-analysis, and the sample size of each study ranged from 26 to 1505. Overall, 15 studies included non-metastatic RCC, 20 included metastatic RCC and 12 included mixed RCC. There was 1 article on papillary RCC , 1 on non-clear cell RCC , 1 on Xp11.2 tRCC, 9 on clear cell RCC , and 30 on clear cell RCC and non-clear cell RCC. The median age of the patients was 60 years, and the median follow-up time ranged from 12 to 100 months. There were 29 studies that included surgery, 2 included combination therapy, and the others included chemotherapy. The results were reported as OS, CSS, PFS, RFS, DFS, and MFS, and the quality scores of these studies ranged from 5 to 9, identified with the NOS.
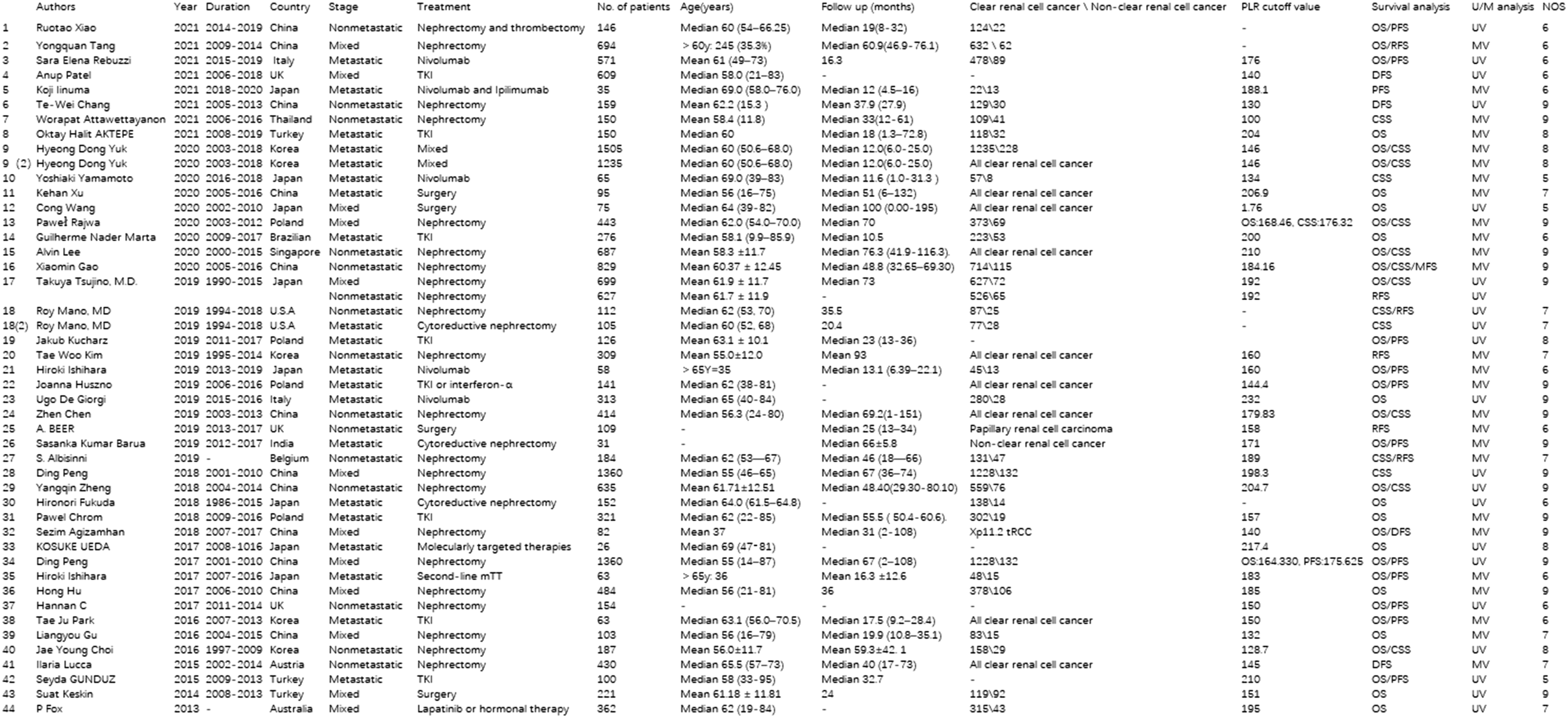
Characteristics of the studies.
OS of RCC patients
General overview
Among the studies, 34 were related to the OS of 11,566 RCC patients. Due to the significant heterogeneity, we used a random-effects model (I2 = 90%, P < 0.00001). We found that a high PLR was related to low OS (HR = 1.01 (95% CI 1.00, 1.02), P = 0.010).
Subgroups
To evaluate OS in different subgroups and determine the sources of heterogeneity, we performed subgroup analysis based on therapy, pathological characteristics, statistical approach, year of publication, tumor stage, country in which the study was conducted, sample size, and PLR (Table 1).
Subgroups for OS analysis.
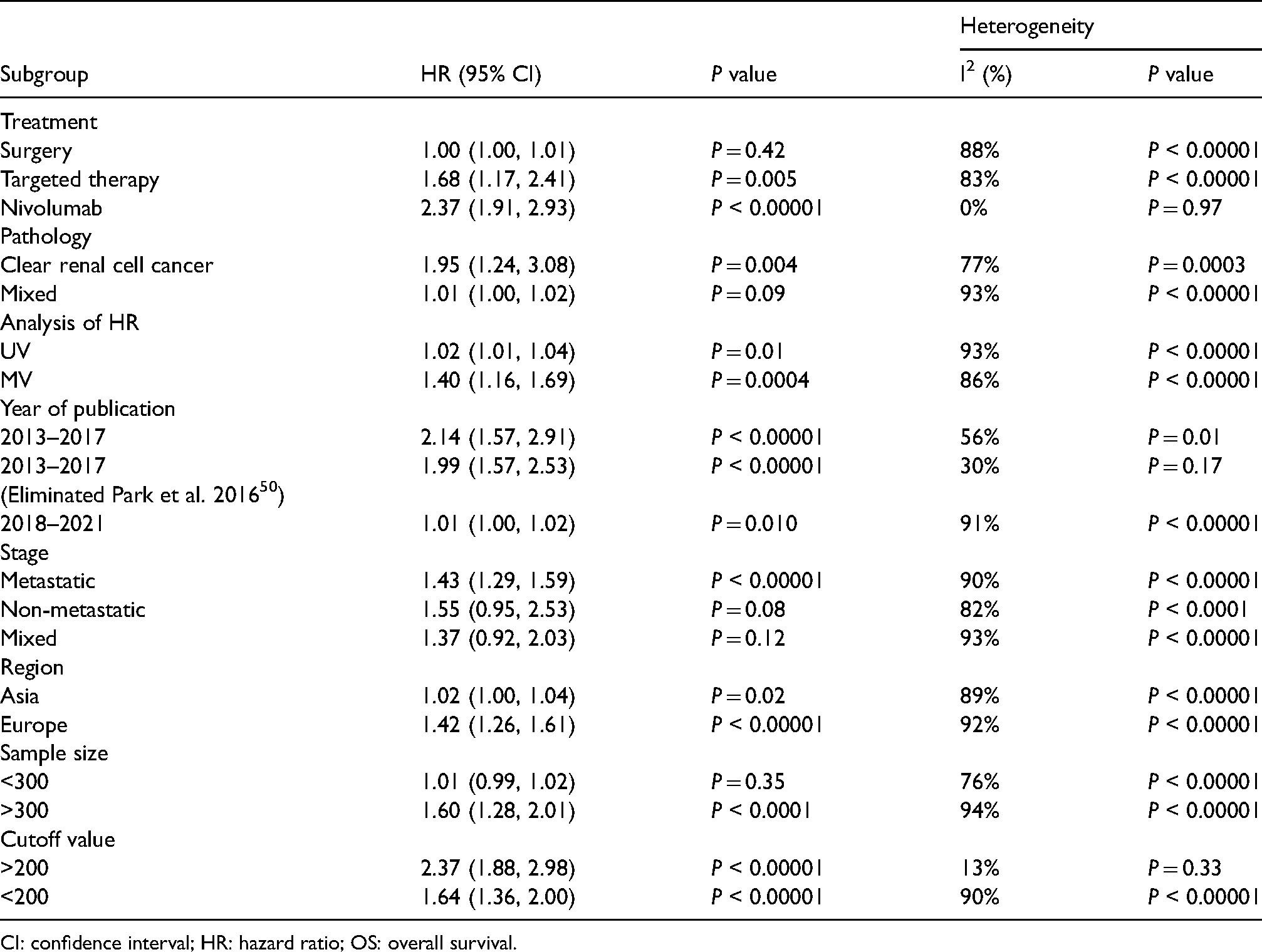
CI: confidence interval; HR: hazard ratio; OS: overall survival.
As shown in Table 1, we found that patients treated with nivolumab with a high PLR had a low OS rate (HR = 2.37 (95% CI 1.91, 2.93), P < 0.00001). Regarding the year of publication, in studies published in 2013–2017, excluding the study by Park et al. in 2016, 50 OS was decreased with high PLR (HR = 1.99 (95% CI 1.57, 2.53), P < 0.00001). High PLR was associated with low OS (HR = 2.37 (95% CI 1.88, 2.98), P < 0.00001) when PLR>200. In the subgroup analysis stratified by pathological characteristics, clear cell RCC was associated with OS (HR = 1.95 (95% CI 1.24, 3.08), P = 0.004). In the subgroup analysis stratified by the statistical approach, multifactor analysis was associated with OS (HR = 1.40 (95% CI 1.16, 1.69), P = 0.0004). In the subgroup analysis stratified by tumor stage, OS was associated with metastatic disease (HR = 1.43 (95% CI 1.29, 1.59), P < 0.00001). Studies published in Europe were associated with OS (HR = 1.42 (95% CI 1.26, 1.61), P < 0.00001). Finally, in the subgroup analysis stratified by sample size, a sample size >300 was associated with OS (HR = 1.60 (95% CI 1.28, 2.01), P < 0.0001).
CSS of RCC patients
General overview
In this meta-analysis, after eliminating the study by Peng et al. in 2018, 41 we obtained 14 studies on CSS in 6015 RCC patients. We used a random-effects model because of the significant heterogeneity (I2 = 55%, P = 0.007). We found that a high PLR was related to low CSS (HR = 1.21 (95% CI 1.00, 1.46), P = 0.05).
Subgroups
For the same reason as before, we proceeded with subgroup analysis (Table 2).
Subgroup analysis (excluding Peng et al. (2018)41) for CSS.
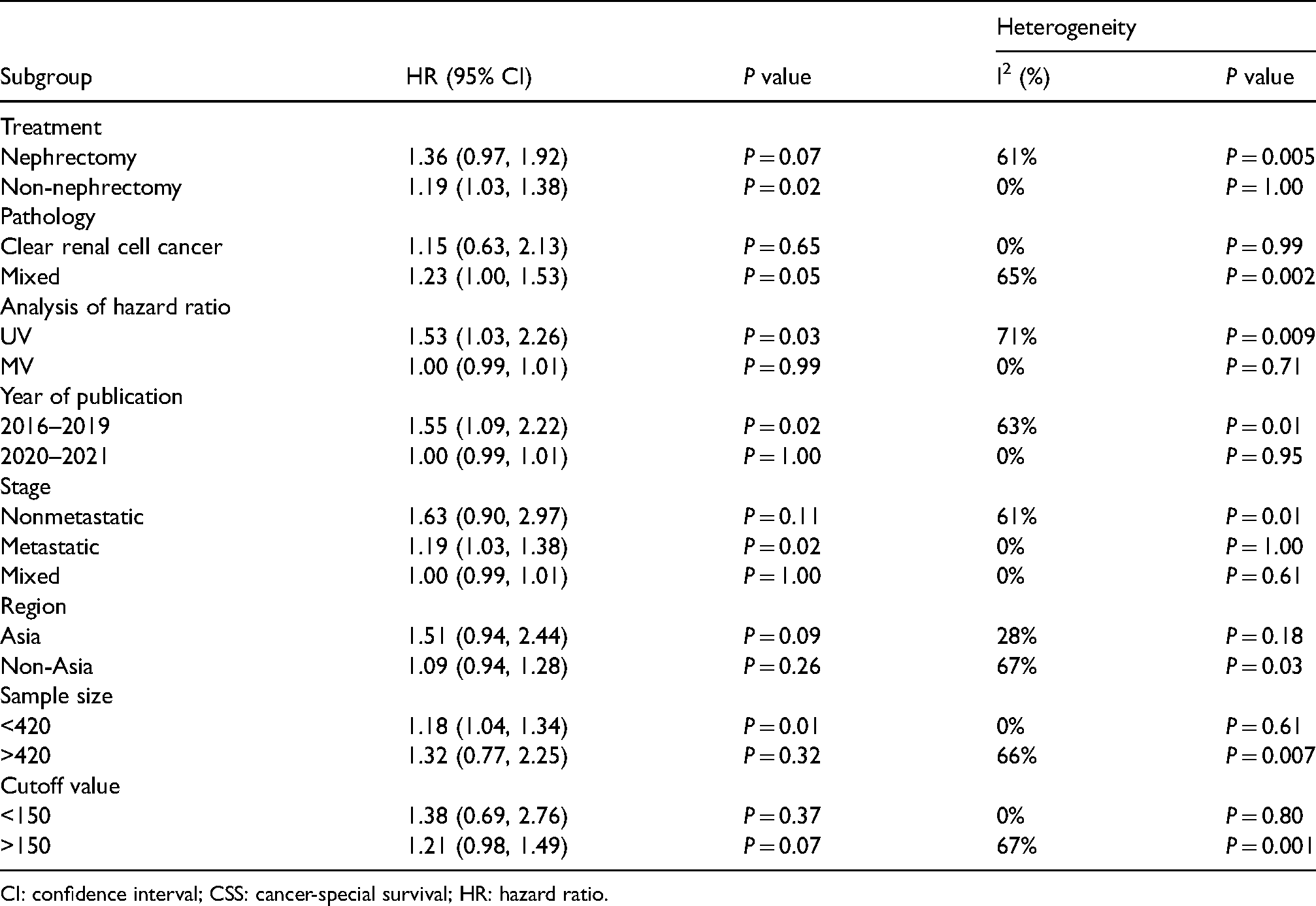
CI: confidence interval; CSS: cancer-special survival; HR: hazard ratio.
As shown in Table 2, after excluding the study by Peng et al. in 2018, 41 increased PLR was associated with CSS of non-nephrectomy patients (HR = 1.19 (95% CI 1.03, 1.38), P = 0.02), clear cell RCC (HR = 1.15 (95% CI 0.63, 2.13), P = 0.65), univariate analysis (HR = 1.53 (95% CI 1.03, 2.26), P = 0.03), publication in 2016–2019 (HR = 1.55 (95% CI 1.09, 2.22), P = 0.02), metastatic RCC (HR = 1.19 (95% CI 1.03, 1.38), P = 0.02), study publication in Asia (HR = 1.51 (95% CI 0.94, 2.44), P = 0.09), sample size<420 (HR = 1.18 (95% CI 1.04, 1.34), P = 0.01), and PLR >150 (HR = 1.00 (95% CI 0.99, 1.01), P = 0.07).
PFS of RCC patients
General overview
For PFS, we assessed 12 studies including 2848 RCC patients. We used a random-effects model due to the high heterogeneity (I2 = 93%, P < 0.00001). Ultimately, we found that the higher the PLR was, the lower the PFS (HR = 1.44 (95% CI 1.28, 1.62), P < 0.00001).
Subgroups
We also performed subgroup analysis for the PFS of RCC patients (Table 3).
Subgroup analysis for PFS.
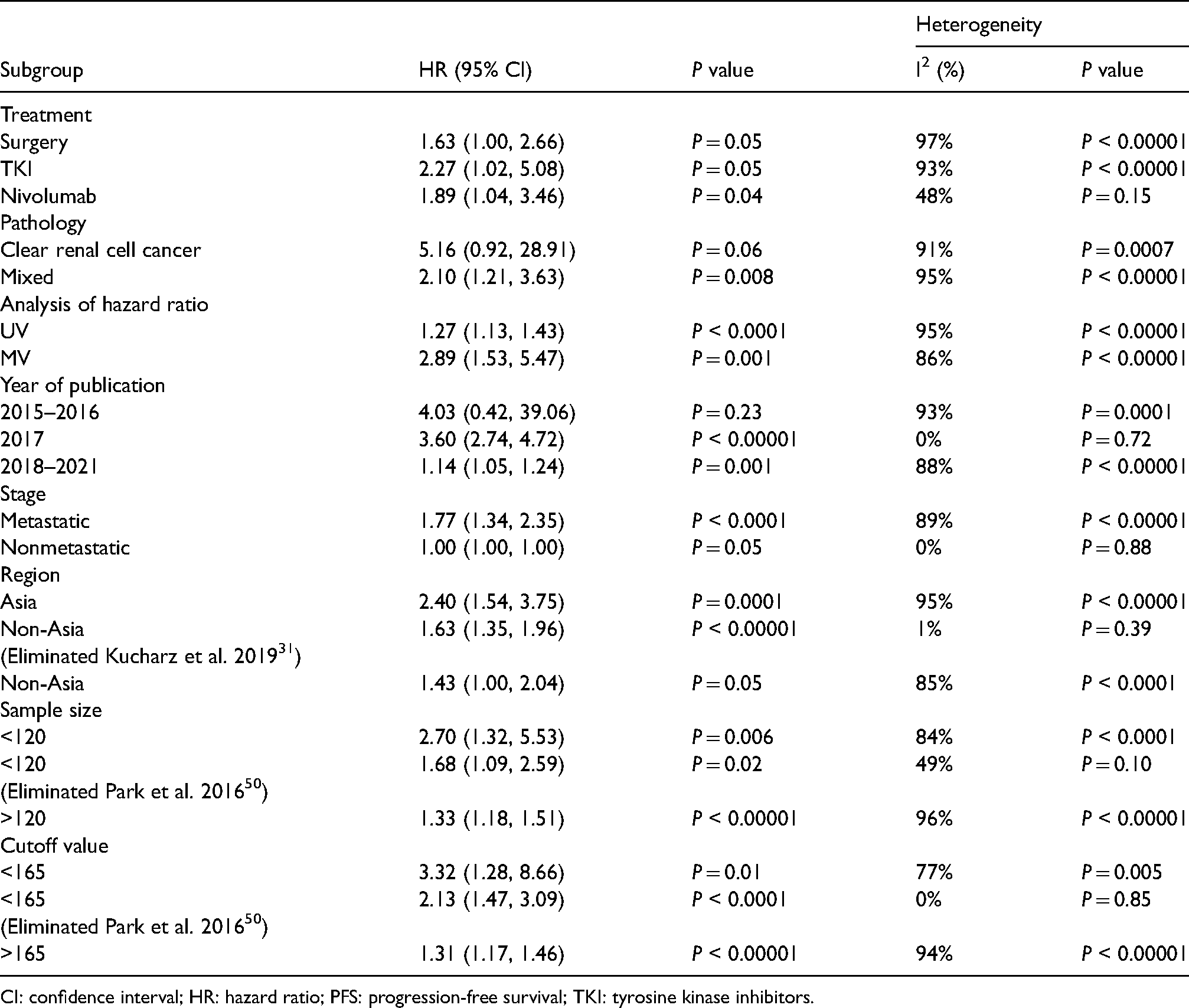
CI: confidence interval; HR: hazard ratio; PFS: progression-free survival; TKI: tyrosine kinase inhibitors.
As shown in Table 3, RCC patients with a high PLR had low PFS. Nivolumab treatment was associated with patient PFS (HR = 1.89 (95% CI 1.04, 3.46), P < 0.00001). Mixed pathology was associated with PFS (HR = 2.10 (95% CI 1.21, 3.63), P = 0.008). Multivariate analysis was associated with PFS (HR = 2.89 (95% CI 1.53, 5.47), P = 0.001). Studies published in 2017 were associated with PFS (HR = 3.60 (95% CI 2.74, 4.72), P < 0.00001), and non-metastatic RCC was associated with PFS (1.00 (95% CI 1.00, 1.00), P = 0.05). After exclusion of the study by Kucharz et al. in 2019, 31 studies published outside of Asia were associated with PFS (1.63 (95% CI 1.35, 1.96), P < 0.00001). Based on both the sample size and cutoff value, the study by Park et al. in 2016 50 needed to be excluded, and there was an association with PFS (sample size <120: HR = 1.68 (95% CI 1.09, 2.59), P = 0.02; cutoff value: HR = <165, P < 0.0001).
RFS of RCC patients
The higher the PLR, the lower the RFS (HR = 1.73 (95% CI 1.11, 2.71), P = 0.02), as indicated by the meta-analysis of five studies comprising 1342 RCC patients. A random-effect model was used (I2 = 71%, P = 0.009).
DFS and MFS of RCC patients
We used a fixed-effects model (I2 = 0%, P = 0.67) to assess DFS (HR = 1.63 (95% CI 0.91, 2.94), P = 0.01), and one study included an association of MFS (HR = 1.223 (95% CI 0.712, 2.099), P = 0.466).
Sensitivity analysis
Excluding studies one by one had no effect on the stability of OS and PFS. For CSS, excluding the study by Peng et al. in 2018,41 the result was stable.
Publication bias
Through funnel plot analyses and Egger’s test, we found that publication bias affected the OS, CSS, and PFS results. Thus, we performed the trim and fill method, and the results indicated that publication bias impacted OS and CSS, while PFS was stable.
Discussion
We included 44 studies including 15,193 patients in our meta-analysis, and the results indicated that the higher the PLR, the poorer the OS, CSS, PFS, RFS, DFS, and MFS, suggesting that the PLR is associated with the prognosis of RCC patients.
RCC is still one of the most common tumors, affecting 2%–3% of adults worldwide.1 With the development of therapies, prognostic markers are increasingly needed in clinical management to help doctors assess the condition of patients, especially for immunotherapy, due to the lack of appropriate biomarkers to evaluate effectiveness. 4 Currently, the postoperative histopathological type is the main prognostic marker, but it is not ideal 56 because histopathological type is assessed after surgery and is not predictive before treatment. Therefore, prognostic markers that can be used before surgery are necessary. A recent study confirmed the mutually reinforcing relationships of inflammation and cancer. An inflammatory tumor microenvironment, composed of tumor cells, inflammatory cells, and surrounding stromal cells, can continuously change its characteristics. 6 Inflammation markers, such as CRP, NLR, MLR, and PLR, have been studied extensively. These markers are inexpensive and routinely measured in clinical management. 7 Platelets can secrete many growth factors to promote the growth of tumors. Lymphocytes can participate in the antitumor immune response. 12 The PLR has been proven to have value in other solid tumors in recent years, such as esophageal squamous cell carcinoma, 8 oral cancer, 9 and small-cell lung cancer. 10 An increasing number of studies on the PLR in RCC patients have been published, but the results are inconsistent. Wang et al. 11 and Wang et al. 12 performed meta-analyses to explore the value of the PLR in RCC patients, but the outcomes of the prognosis of RCC patients still need to be assessed further.
Due to heterogeneity, we performed a subgroup analysis and found no significant heterogeneity when patients received nivolumab treatment or when the study by Park et al. in 2016 50 was removed from the analysis of studies published in 2013–2017 or studies with PLR >200. We believe these studies may be the source of heterogeneity for OS. Considering that its effect value was extracted through a Kaplan–Meier curve and that it obviously increased the heterogeneity in every subgroup, we believe that the study by Peng et al. in 2018 41 had inherent deviations and therefore removed it from our study.
In the subgroup analysis, subgroups with patients who received non-nephrectomy treatment, patients with clear cell RCC only, patients with metastatic disease, patients treated in Asian countries, and patients with PLR <150 had no heterogeneity, and there was no significant heterogeneity in studies with a sample size <420, published in 2020–2021, or with multivariate analysis. These studies may be sources of heterogeneity for CSS. For PFS, there was no significant heterogeneity in studies published in 2017 or in studies with patients with non-metastatic RCC. After eliminating the study by Kucharz et al. in 2019, 31 there was no significant heterogeneity (I2 = 1%) in the subgroup of patients treated outside of Asia. After excluding the study by Park et al. in 2016, 50 there was no significant heterogeneity in studies with both <120 patients and patient PLR <165. The study by Kucharz et al. in 2019 31 was eliminated because it obviously increased the heterogeneity of the subgroup. In addition, the study by Kucharz et al. in 2019 31 established two groups: the first group received two courses of sunitinib treatment, and the second group received three courses. We employed data from the second group, which may have caused heterogeneity. Because the study of Park et al. in 2016 50 increased the heterogeneity in multiple subgroups and included only 65 patients (without strict criteria for including patients), we believe that it is non-representative. Therefore, we surmise that this study was the source of heterogeneity in the PFS subgroup analysis. Wang et al. 12 proved the negative impact of a high PLR on OS in RCC patients receiving targeted treatment, patients with metastatic disease, or patients with PLR >195, and studies published in Asia or with a sample size >100 also showed an impact of PLR. Our results were similar to theirs, but the HR values in our study were not as high as theirs. In the metastatic subgroup, our result (HR = 1.43 (95% CI 1.29, 1.59), P < 0.00001) was similar to that of Wang et al. 11 (HR = 1.97 (95% CI 1.44∼2.70, P < 0.001). In our study, PLR >200 was associated with a poorer OS (HR = 2.37 (95% CI 1.88, 2.98), P < 0.00001), while PLR <200 showed the following results for OS: HR = 1.64 (95% CI 1.36, 2.00), P < 0.00001. CSS had the following results: PLR <150: HR = 1.38 (95% CI 0.69, 2.76), P = 0.37; and PLR >150: HR = 1.21 (95% CI 0.98, 1.49), P = 0.07. PFS had the following results: PLR <165: HR = 3.32 (95% CI 1.28, 8.66), P = 0.01; and PLR >165: HR = 1.31 (95% CI 1.17, 1.46), P < 0.00001. We believe that a high PLR has a negative effect on prognosis. The main reason for the differences among our study and the studies of Wang et al.11 and Wang et al.12 may be the number of studies included. The region, year of publication, and sample size may be other factors. In general, high PLR indicated poor prognosis.
In addition, we performed other subgroup analyses that had not been performed in other studies. In our results regarding high PLR, patients who received nivolumab treatment had poor prognosis in terms of OS (HR = 2.37 (95% CI 1.91, 2.93), P < 0.00001) and PFS (HR = 1.89 (95% CI 1.04, 3.46), P = 0.04). In addition, there were poor outcomes in patients with a high PLR who received targeted treatment in terms of OS (HR = 1.68 (95% CI 1.17, 2.41), P = 0.005) and PFS (HR = 2.27 (95% CI 1.02, 5.08), P = 0.05). However, this correlation was not significant in patients undergoing surgery in terms of OS (HR = 1.00 (95% CI 1.00, 1.01), P = 0.42). These are new findings because the included studies on nivolumab treatment were published in 2019–2021, and they suggest that surgeons should pay attention to PLR when patients receive nivolumab treatment.
Regarding pathological characteristics, clear cell RCC patients with high PLR tended to have a poor prognosis (OS: HR = 1.95 (95% CI 1.24, 3.08), P = 0.004; CSS: HR = 1.15 (95% CI 0.63, 2.13), P = 0.65; PFS: HR = 5.16 (95% CI 0.92, 28.91), P = 0.06). Based on this result, it is better to explore the value of PLR after confirming pathological characteristics in the clinic. We also found a stronger association between poor OS and high PLR in the studies published in 2013–2017 (HR = 2.14 (95% CI 1.57, 2.91), P < 0.00001) than in the studies published in 2018–2021 (HR = 1.01 (95% CI 1.00, 1.02), P = 0.010). The same pattern was seen for other effective markers: CSS (2016–2019 HR = 1.55 (95% CI 1.09, 2.22), P = 0.02; 2020–2021: HR = 1.00 (95% CI 0.99, 1.01), P = 1.00) and PFS (2015–2016: HR = 4.03 (95% CI 0.42, 39.06), P = 0.23; 2017: HR = 3.60 (95% CI 2.74, 4.72), P < 0.00001; 2018−2021: HR = 1.14 (95% CI 1.05, 1.24), P = 0.001). Advances in the clinical management of RCC patients have led to better prognosis, which may cause this difference. This could also explain why the HRs of our study were lower than those of the studies by Wang et al.11 and Wang et al.12, as we included a large number of studies published in recent years (the data in the studies of Wang et al.11 and Wang et al.12 were older). RCC with metastasis (HR = 1.43 (1.29, 1.59), P < 0.00001) was not an obvious factor compared with that in the study of Wang et al. 12 (mixed therapies in metastatic patients: HR = 6.05, 95% CI: 1.34, 27.37, P = 0.019). This difference may be caused by the number of studies, as only four were included in the study of Wang et al. 12 The result of our study is likely more credible.
In the PFS group with a high PLR, TKI treatment had an HR of 2.27 ((95% CI 1.02, 5.08), P = 0.05), and nivolumab treatment had an HR of 1.89 ((95% CI 1.04, 3.46), P = 0.04). We calculated the HRs for CSS in RCC patients, while the studies of Wang et al. (2017) 11 and Wang et al. (2018) 12 did not. The PLR also had value in predicting RFS, DFS, and MFS in our study, which was not found in the studies of Wang et al. 11 and Wang et al. 12 It is worth mentioning that the PLR had more clinically significant value in predicting the outcome for metastatic patients than non-metastatic patients.
We recommend that doctors detect the PLR value, especially in patients treated with non-surgical therapies, because the PLR showed stronger predictive power in patients who received chemical treatment, such as nivolumab and TKIs, than in patients who underwent surgery.
The PLR has been studied as a marker for predicting prognosis in RCC patients for several years. Wang et al. 11 and Wang et al. 12 proved its value in RCC patients. However, there were some differences between our results and theirs. We believe these differences may be due to the number of studies included and the year of publication of the studies. We believe our study is the most recent and complete meta-analysis. Our results further support the value of the PLR for RCC prognosis, especially in patients with non-surgical treatment or metastasis, which can provide advice for the clinical management of mRCC patients.
In addition, our results provide a direction for assessing the prognosis of immunotherapy despite the limited number of records. According to these findings, PLR values may be helpful in the selection of treatment for RCC patients. Nevertheless, additional high-quality randomized controlled trials RCTs focusing on immunotherapy are still needed. The cutoff value of the PLR is important but has not yet been determined because it is a continuous variable. If identified, the cutoff value of PLR may contribute to risk stratification in patients with RCC in the future. Moreover, more studies focusing on different histological types are needed to explore the effectiveness of the PLR value to predict unfavorable pathologic results (sarcomatoid features and tumor necrosis). Due to the difficult clinical management and personalized treatment for mRCC, 57 relevant prognostic factors are needed to guide clinical treatment. With the development of research on the relationship of inflammation and the advancement of tumors, we believe there will be a larger stage for inflammatory biomarkers of RCC, such as the PLR.
There are some limitations to our study. First is high heterogeneity, which resulted from a large number of studies with various factors being included, as discussed above. Second, there was publication bias for OS and CSS because positive results tend to be submitted and published, and further research is needed. We believe our results are representative because we included the newest and largest number of studies thus far. Third, the cutoff value of the PLR varied from study to study, which is a difficult problem that needs additional study. Fourth, the results of our studies may have potential bias because they were calculated by pooled HRs and 95% CIs, and according to the NOS, some of the included studies had a low or moderate risk of bias. Fifth, we did not analyze a subgroup of cytoreductive nephrectomy, which is an important treatment for mRCC patients, and the records of nivolumab are still scarce, so more studies need to be performed. Due to the characteristics of the tumor itself, histological types other than clear cell RCC were not studied.
Conclusion
Overall, based on the evidence thus far, the PLR is a credible and inexpensive predictive marker that is easy to determine. A higher PLR is related to poorer OS, CSS, PFS, RFS, DFS, and MFS in RCC patients. Nevertheless, more high-quality studies need to be conducted to determine the PLR cutoff value to assess RCC patient prognosis due to the limitations above.
Footnotes
Acknowledgments
We acknowledge the School of Medicine, Xiamen University and Zhongshan Hospital of Xiamen University for their support.
Author contributions
ZX designed the study. GCL and ZX collected data, performed the statistical analysis, and drafted the manuscript. All authors read and approved the final manuscript.
Funding
The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
None declared.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
