Abstract
Background:
Circulating microRNAs (miRNAs) are proposed as promising non-invasive diagnostic biomarkers for many cancers. However, the diagnostic value of circulating miRNAs in ovarian cancer is inconsistent in different studies. Thus we performed this meta-analysis to systematically evaluate the diagnostic value of circulating miRNAs in ovarian cancer.
Methods:
Eligible studies that were published prior to 30 June 2017 were searched from the PubMed, EMBASE, Cochrane Library, and Chinese National Knowledge Infrastructure. All analyses were performed using STATA 12.0 software. A bivariate regression was used to calculate pooled diagnostic accuracy estimates.
Results:
A total of 36 studies from 16 publications were included in this meta-analysis. The pooled sensitivity, specificity, positive likelihood ratio, negative likelihood ratio, and diagnostic odds ratio of circulating miRNAs for ovarian cancer diagnosis were 0.76 (95% confidence intervals (CI): 0.69, 0.81), 0.81 (95% CI 0.74, 0.87), 4.00 (95% CI 2.70, 5.30), 0.30(95% CI 0.24, 0.37) and 13.00 (95% CI 9.00, 19.00), respectively. The area under the summary receiver operating characteristic curve was 0.85 (95% CI 0.82, 0.88). Subgroup analyses showed that multiple miRNA assays yielded better diagnostic characteristics than a single miRNA assay, and plasma miRNAs were better than serum miRNAs for ovarian cancer detection.
Conclusion:
Circulating miRNAs, especially the combination of multiple circulating miRNAs, are promising biomarkers for the diagnosis of ovarian cancer. However, further large-scale prospective studies are necessary to validate the applicability of the miRNAs in the early detection of ovarian cancer.
Introduction
Ovarian cancer (OC) is the seventh leading cause of death in women and the most lethal form of common gynecological malignancies, 1 with over 238,700 new cases and 151,900 deaths worldwide annually, accounting for 4.93% of all cancer cases and 4.28% of all cancer deaths in women. The high mortality rate of OC is markedly correlated with atypical symptoms and a lack of effective screening methods. 2 Approximately 75% of OC patients were diagnosed with stage III or IV cancer. 2 Late detection is the direct cause of poor prognosis with less than 30% having a five-year survival rate. 3 Therefore, efficient diagnostic methods or biomarkers for early detection of OC are urgently needed to improve the prognosis and to reduce the mortality of OC patients.
Currently, a bimanual pelvic examination, cancer antigen (CA) 125, and transvaginal ultrasound are the most commonly used diagnostic methods for OC. 2 However, studies have shown that these methods have various inherent limitations, such as being time consuming, costly, invasive, inconvenient, and limited specificity and sensitivity.2,4–7 In this regard, the development of minimally invasive and maximally accurate diagnostic methods or biomarkers for early detection of OC is urgently needed.
MicroRNAs (miRNAs) are recently discovered endogenous small non-coding RNAs with a fundamental role in the regulation of gene expression. 8 MiRNAs regulate the expression of approximately 30% of human proteins that control various biological processes, such as cellular development, apoptosis, proliferation, differentiation, and tumorigenesis. 9 More importantly, aberrant expression of miRNAs leads to the development and progression of many types of diseases, particularly cancers, in human and animal studies.10,11 Studies have also shown that circulating miRNAs are remarkably stable and tolerant to RNase in serum and plasma samples.12,13
During the past few years, growing evidence has shown the potential of using circulating miRNAs as novel diagnostic markers for OC.14–29 However, due to the diversity in study design, sample size, race, sample type, and miRNA profiling, different studies yielded inconsistent conclusions regarding the diagnostic values of circulating miRNAs.30,31 Therefore, we performed this meta-analysis to systematically evaluate the clinical applicability of circulating miRNAs as non-invasive biomarkers for the diagnosis of OC.
Materials and methods
Literature search
We conducted a literature search using PubMed, Embase, the Cochrane Library, and the Chinese National Knowledge Infrastructure (CNKI) for related articles that evaluated the diagnostic value of circulating microRNAs in OC. The literature search was last updated on 30 June 2017. The following medical subject headings (MeSH) and keywords employed in the literature search included: (“ovarian neoplasms” OR “ovarian carcinoma” OR “ovarian tumor” OR “ovarian cancer”) AND (“microRNA” OR “miRNA” OR “miR*”) AND (“blood” OR “serum” OR “plasma” or “circulating”) AND (“diagnosis” OR “sensitivity and specificity” OR “receiver operating characteristics” OR “ROC curve” OR “ROC analyses”). There were no restrictions in language or publication date. In addition to the literature searched from the above sources, the articles listed in the reference lists of the reviewed articles were also manually reviewed by independent investigators to obtain additional eligible studies. Ethical approval was not necessary as this study is a systematic review and meta-analysis.
Inclusion and exclusion criteria
This meta-analysis was carried out following the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement (Supplement S1). The inclusion criteria of the studies in this meta-analysis were: (a) the study evaluated the diagnostic value of circulating miRNAs in OC; (b) all OC patients were diagnosed by gold standard assessments (e.g. histological examinations); and (c) sufficient data for 2×2 contingency table construction (true positive (TP), false positive (FP), false negative (FN), and true negative (TN)). The exclusion criteria were: (a) studies without sufficient data, or data that could not be retrieved or reconstructed to 2×2 tables; (b) duplicate publications; and (c) publications without original data, such as case reports, letters, editorial and expert opinions, comments, and reviews. Two reviewers (Q Zhou and Z He) cross-checked the eligibility of all studies. The disagreement in decisions was solved through discussion and subsequent consensus. For the same patient population that was used in several studies, only the most recent and largest study with the best quality was included.
Data extraction and quality assessment
Eligible publications were reviewed by two investigators independently (HR Li and W Li) and the relevant data were extracted in a standardized table. The extracted data included the following information: (a) basic characteristics of studies, such as the first author’s surname, publication year, country, sample size, assay methods, study design, and tumor stage and grade; (b) data needed for diagnostic meta-analysis, including studied miRNAs profiles, types of specimens, cut-off value, sensitivity, specificity, TP, FP, FN, and TN; and (c) information needed for methodological quality assessment. The disagreements were resolved by consensus among the investigators.
The quality of each included study was assessed independently by two investigators following the revised Quality Assessment of Diagnostic Accuracy Studies (QUADAS-2). 32 The quality of the studies was assessed by QUADAS-2 tool, which is composed of four key domains: patient selection, index test, reference standard, and flow and timing. A seven-item checklist was used to indicate the quality of the studies. The answer “yes,” “unclear,” or “no” were given to each item to judge the risk of bias and applicability. In the case of conflicting evaluations, a third investigator (Q Zhou) was consulted, and disagreement was resolved by multilateral discussion.
Statistical analysis
All data were analyzed using STATE 12.0 software (State Corporation, College Station, TX, USA). The bivariate regression model was used to estimate the pooled sensitivity (SE), specificity (SP), positive likelihood ratio (PLR), negative likelihood ratio (NLR), and diagnostic odds ratio (DOR), and to generate the bivariate summary receiver operator characteristic (SROC) curve with 95% confidence intervals (CIs). 33 Chi-square and I2 tests were used to assess the heterogeneity between studies. P<0.1 or I2>50% indicated the existence of significant heterogeneity.34,35 The potential sources of between-study heterogeneity were investigated by subgroup and meta-regression analyses according to the characteristics of the included studies. The Deeks’ funnel plot was generated for DOR to explore the potential publication bias, and P<0.01 was considered a significant publication bias. 36
Results
Literature retrieval
The literature retrieval procedure was illustrated in Figure 1. The initial search returned a total of 299 articles (296 from the database search, three from other sources), of which 43 duplicate publications were excluded. The remaining 256 articles were subjected to the next evaluation. After reviewing titles and abstracts, 231 articles were excluded due to the following reasons: 45 articles were reviews or commentaries, nine were not human studies, 114 were not diagnosis studies, and 63 did not use blood samples. Subsequently, the full text of the remaining 25 articles was carefully reviewed to assess the eligibility of these articles, and nine articles were further excluded because two studies had significant overlap, three were prognostic studies, and four lacked the necessary data. Finally, 16 articles were used for the meta-analysis.
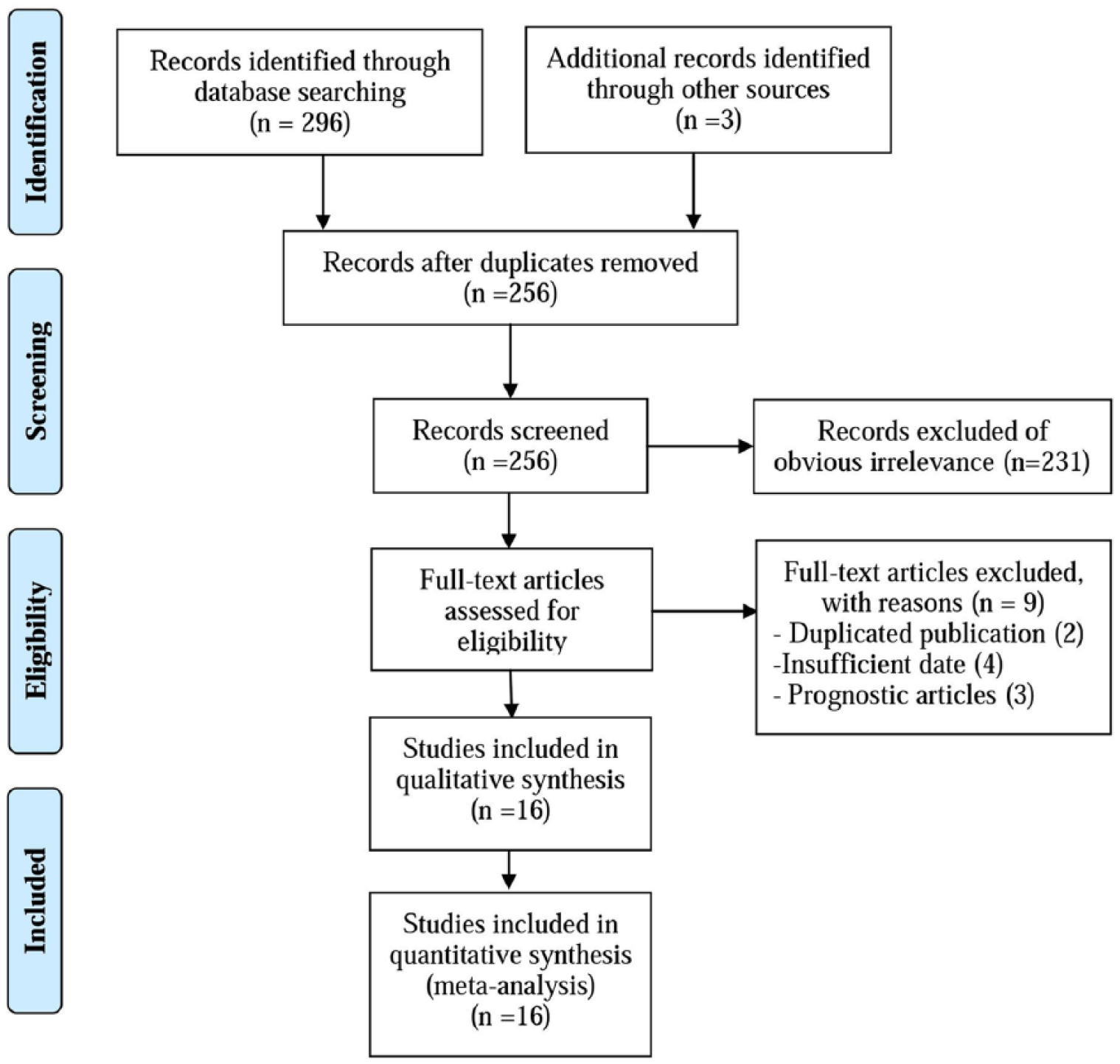
Flow chart showing the process for selecting eligible studies in the meta-analysis.
Characteristics of selected studies
The main characteristics of the included 16 articles are listed in Table 1. In total, 36 studies from 16 articles with 3470 OC patients and 1606 healthy controls were included in the meta-analysis. All of the selected studies were published over seven years (2010–2017). Among the 36 studies, 21 were conducted in Asian populations and the remaining 15 were performed in Caucasian populations, including the following geographical regions: Korea (n=4), China (n=9), Germany (n=11), Australia (n=5), USA (n=2), Italy (n=3), and India (n=2). The diagnostic value of single-miRNA in OC was investigated in 29 studies, and the remaining seven studies focused on the diagnostic value of multiple-miRNAs in OC. Six of the studies used plasma miRNAs while the other 30 studies used serum miRNAs. Twenty-two studies used sequencing technology or miRNA microarray-based approaches for the screening of the miRNA biomarkers, whereas the other 14 studies used the frequently reported miRNAs as candidates for miRNA assays. All studies used quantitative real-time reverse transcription-polymerase chain reaction (qRT-PCR) to evaluate the expression of circulating miRNAs.
Main characteristics of the studies included in the meta-analysis.
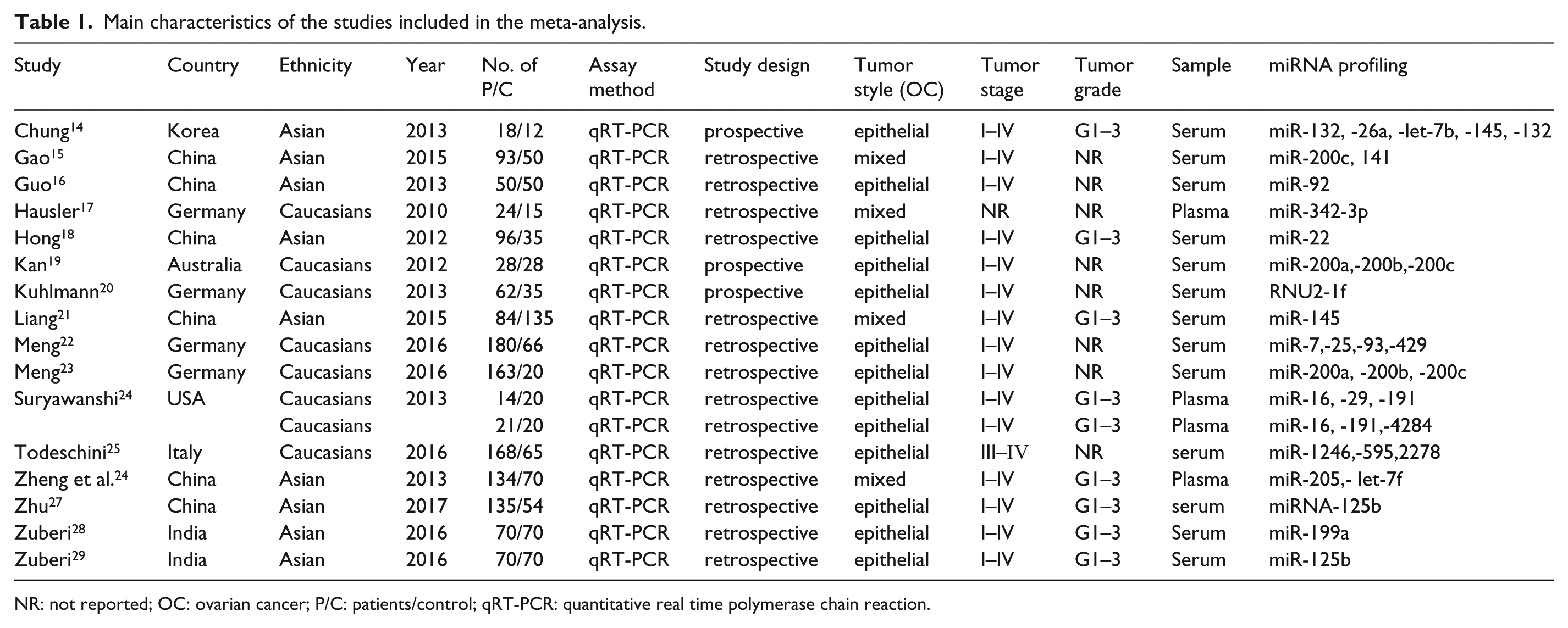
NR: not reported; OC: ovarian cancer; P/C: patients/control; qRT-PCR: quantitative real time polymerase chain reaction.
Methodological quality assessment
The quality of the included studies was evaluated using QUADAS-2 quality assessment. The majority of included studies in this meta-analysis satisfied at least three of the seven items in QUADAS-2, indicating that the included studies were of moderate-high quality. The graph of risk of bias and applicability concerns of the included studies are shown in Figure 2.
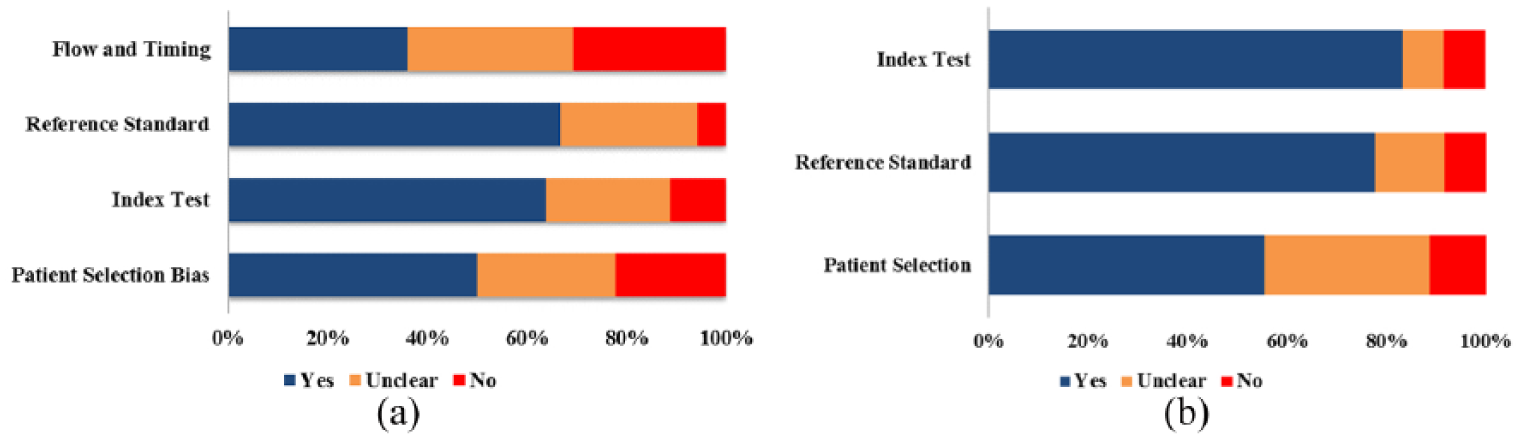
Overall quality assessment of included studies using the QUADAS-2 criteria ((a) risk of bias; (b) applicability).
Diagnostic accuracy of miRNAs
The diagnostic accuracy of miRNAs pooled from all studies in detecting OC is summarized in Table 2. The 36 studies used 30 different miRNAs, which were tested in 3470 participants for the diagnostic values of miRNAs as the biomarkers of OC. I2 values for sensitivity and specificity were 94.12% (95% CI 89.94, 94.20; P<0.001) and 85.46% (95% CI 83.83, 91.42; P<0.001), respectively, indicating the existence of statistical heterogeneity between studies (Figure 3). Thus, we used the random-effects model to calculate the pooled estimates. The overall sensitivity and specificity were 0.76 (95% CI 0.69, 0.81) (Figure 3(a)) and 0.81 (95% CI 0.74, 0.87) (Figure 3(b)). In addition, the overall PLR was 4.00 (95% CI 2.70, 5.30), the NLR was 0.30 (95% CI 0.24, 0.37), and the diagnostic odds ratio was 13 (95% CI 9, 19). Simultaneously, diagnostic accuracy was further assessed by SROC plotting and area under the curve (AUC) value, which was 0.85 (95% CI 0.82, 0.88). This indicates that circulating miRNAs tested in the studies were relatively highly accurate as differentiation markers to diagnose OC patients from healthy controls. The SROC curve for the included studies is shown in Figure 4.
Summary estimates of diagnostic criteria and the 95% confidence intervals.
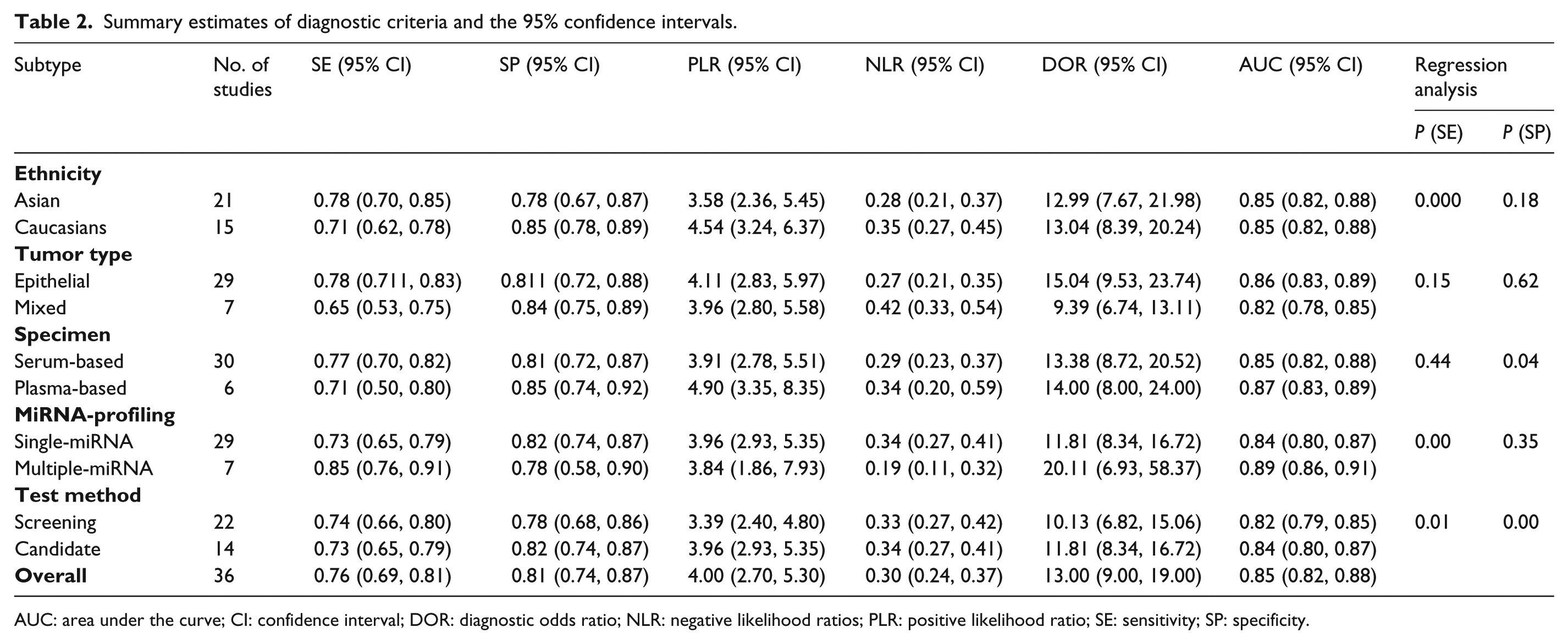
AUC: area under the curve; CI: confidence interval; DOR: diagnostic odds ratio; NLR: negative likelihood ratios; PLR: positive likelihood ratio; SE: sensitivity; SP: specificity.
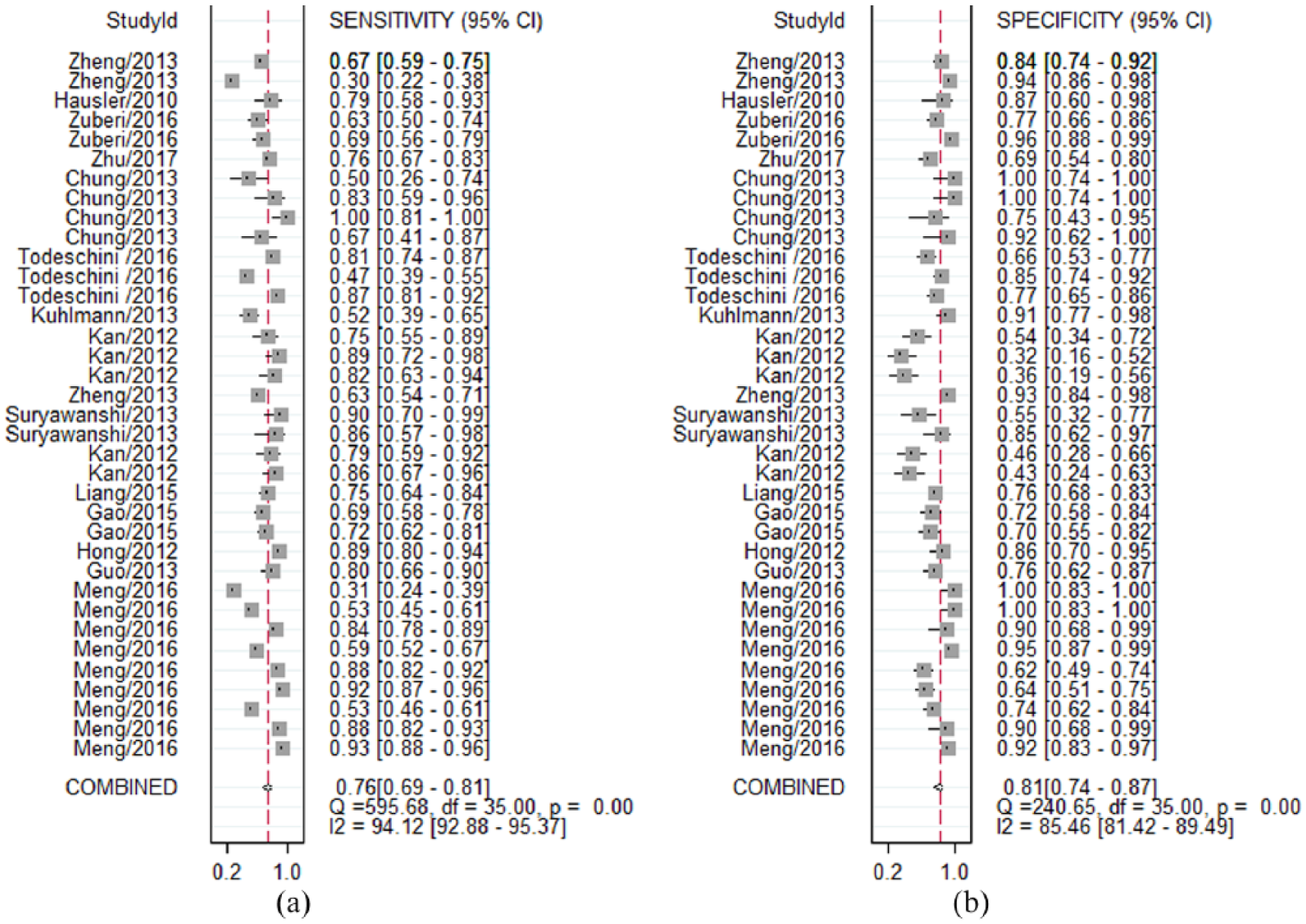
Forest plots of meta-analyses for sensitivity (a) and specificity (b).
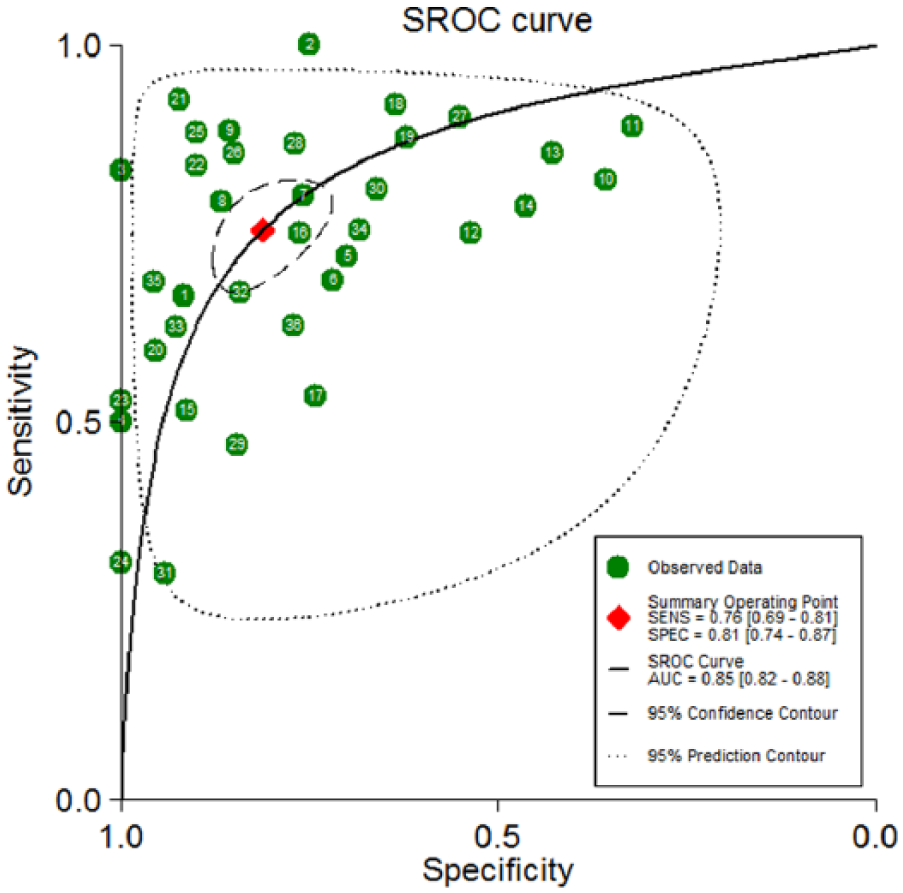
The SROC of the miRNA test for the diagnosis of OC.
Subgroup analyses and meta-regression
Our results showed that all diagnosis performance parameters (SE, SP, PLR, NLR, and DOR) had significant heterogeneity (P<0.1). Therefore, we did subgroup analyses and meta-regression to clarify a possible source of heterogeneity. Subgroup analyses were performed using different subtypes, including ethnicity (Asian or Caucasian), tumor type (epithelial or mixed), specimen types (plasma or serum), miRNA-profiling (single-miRNA or multiple-miRNA), and test method (screening or candidate). We found that the overall accuracy of the studies on Caucasian populations was similar when compared with that on Asian populations (AUC of 0.85 vs. 0.85). An analysis of tumor type indicated that the miRNA assay has superior diagnostic accuracy in epithelial OC patients than in patients with mixed pathologic type of OC (AUC of 0.86 vs. 0.82). An analysis of specimen-type-based subgroups suggested that a plasma-based study had a higher level of AUC than the serum-based assays (0.87 vs. 0.85), indicating that plasma miRNAs were relatively reliable diagnostic biomarkers compared with serum miRNAs. An analysis of miRNA profile subgroups suggested that the use of multiple miRNAs assays was a superior diagnostic method compared to a single miRNA assay (AUC of 0.89 vs. 0.84). In addition, we analyzed the test method subgroups. The results demonstrated that frequently reported miRNAs had better OC diagnostic accuracy than the miRNAs that were selected by sequencing technology or miRNA microarray-based screening approaches (AUC of 0.84 vs. 0.82). The pooled data, such as sensitivity, specificity, PLR, NLR, DOR, and AUC for each subgroup are shown in Table 2.
To further find out the potential sources of heterogeneity, meta-regression was conducted to illustrate the source of heterogeneity by five pre-specified covariates (ethnicity, tumor type, specimen type, miRNA profiling, and screening method). The bivariate model was used to assess the impact of the six covariates on the sensitivity and specificity of the study. Our results exhibited that ethnicity (P<0.01), test method (P<0.05), and miRNA profiling (P<0.001) were possible sources of heterogeneity in sensitivity, test method (P<0.01), and specimen type (P<0.05) was most likely responsible for the heterogeneity in specificity. These results were consistent with the results of the subgroup analyses. The results of multivariable meta-regression analyses for sensitivity and specificity are shown in Table 2.
Publication bias
The Deeks’ funnel plot was examined for DOR to find the potential publication bias in this meta-analysis. The funnel plot for publication bias was symmetry (Figure 5) for all included studies, and the P value for Deeks’ test was 0.586, indicating that no significant publication bias existed in the included studies.
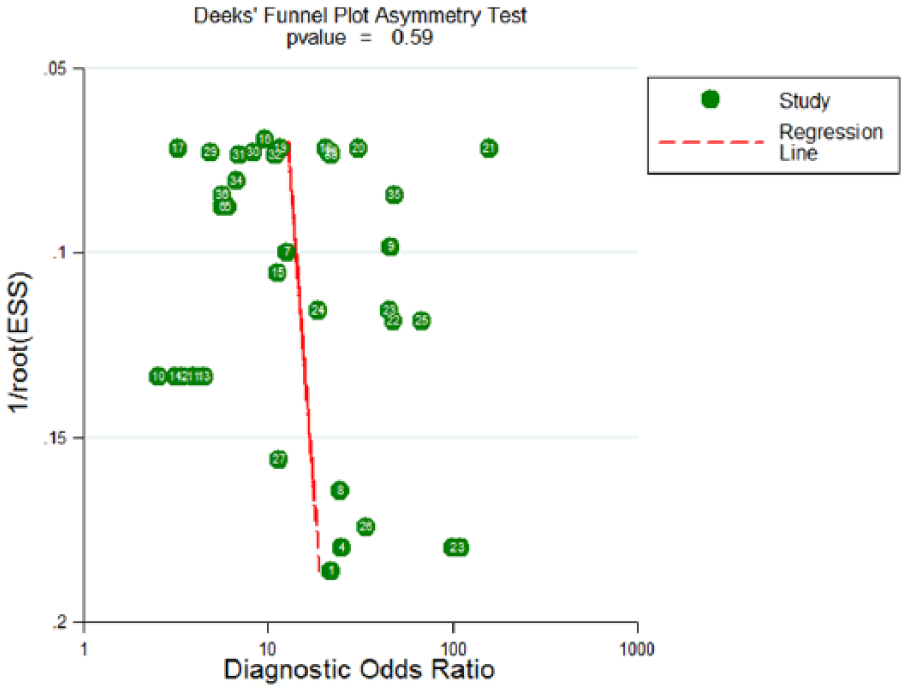
Deeks’ linear regression test of funnel plot asymmetry.
Discussion
Due to unclear symptoms in the early stages of OC and the lack of effective screening methods, OC is often diagnosed at a late stage with peritoneal dissemination and massive ascites. The five-year survival rate of patients is less than 30% despite combined treatments with aggressive cytoreductive surgery plus taxane-based and platinum-based chemotherapy. 3 Developing suitable biomarkers for the early detection and monitoring of OC has long been a hot spot in cancer research. 2 However, existing diagnostic methods for OC have their own inherent deficiencies, which limit their clinical applications. It is now urgent to find highly accurate and minimally invasive biomarkers for the early detection of OC.
Recently, circulating miRNAs have attracted considerable attention because of their high stability and their potential use as non-invasive diagnostic markers for cancers. Multiple studies have demonstrated that unique miRNA expression profiles in body fluids may contribute to the diagnosis of cancers, such as nasopharyngeal cancer, 37 osteosarcoma, 38 glioma, 39 esophageal squamous cell carcinoma, 40 prostate cancer, 41 breast cancer,42,43 hematologic cancer,44,45 gastric cancer,45,46 and lung cancer. In OC, the potential of circulating microRNAs as novel diagnostic biomarkers has gained increasing interest, leading to growing numbers of studies to evaluate the diagnostic accuracy of circulating miRNAs for early OC detection. 47 However, inconsistent conclusions were obtained in these studies because of the differences in ethnicity, study design, sample size, sample type, assay method, and miRNA profiles between the studies.14–29 Thus far, the results of these studies have not been systematically evaluated. Hence, we performed this meta-analysis to integrate all related publications to evaluate the overall accuracy of circulating miRNAs as detection biomarkers in OC diagnosis.
In our meta-analysis, the pooled sensitivity and specificity of the circulating miRNA assays were 0.76 (95% CI 0.69, 0.81) and 0.81 (95% CI 0.74, 0.87), corresponding to an AUC up to 0.85 (95% CI 0.82, 0.88). Recent studies reported the diagnostic performance of some conventional blood biomarkers in OC, such as CA125 (optimal sensitivity, 0.74; optimal specificity, 0.83) 48 and HE4 (sensitivity, 0.74; specificity, 0.87).48–50 In comparison, the diagnostic accuracy of circulating miRNAs is at least similar to that of the conventional biomarkers for OC screening. However, this conclusion is based on only two studies that directly compared the diagnostic value of circulating miRNAs with other conventional markers. The DOR is the ratio of the odds of a true-positive to the odds of a false-positive, which is another important indicator in mirroring the discriminating power of a diagnostic test. DOR value ranges from zero to infinity, with higher values indicating better performance, and a value of less than 1.0 indicating low discriminating ability in the diagnostic test. 51 In our study, the pooled DOR was 13 (95% CI 9, 19), indicating good discriminatory performance of circulating miRNAs for OC. Likelihood ratios (LRs) are metrics that indicate the verity of sensitivity and specificity, and LRs of more than 10 or less than 0.1 generate large and often conclusive shifts from pretest to posttest probability. 51 In our study, the pooled PLR was 4.0, indicating that circulating miRNAs have four-fold higher chance of being tested positive than being falsely diagnosed in OC. The pooled NLR was 0.30, implying that the chance of the miRNA test being false-negative was 30%. In summary, our study demonstrated that circulating miRNAs are promising diagnostic biomarkers in OC.
Heterogeneity is a potential problem for result interpretation in any meta-analysis. In our study, significant heterogeneity was detected in the included studies. We explored the potential sources of heterogeneity by conducting subgroup analyses. Different racial expression profiles have been reported to affect circulating miRNA concentrations. In the ethnic subgroup analysis, we found that an Asian population-based miRNA test yielded similar accuracy against a Caucasian-based test (AUC of 0.87 vs. 0.84). An analysis of tumor type indicated that miRNA assays have superior diagnostic accuracy in epithelial OC patients than in patients with a mixed pathologic type of OC (AUC of 0.86 vs. 0.82), suggesting that a circulating miRNA assay may be more precise to detect epithelial OC than to detect mixed pathologic types of OC. Notably, different sample sources of miRNA contribute to the heterogeneity in the studies. A serum-based sample and a plasma-based sample had different DOR (13 vs. 14) and AUC (0.85 vs. 0.87), indicating that plasma may be a better source for miRNA assays in the diagnosis of OC. A possible explanation for this heterogeneity is that due to the influence of the coagulation process, samples from different sources may have different diagnostic accuracies for miRNA detection. Moreover, miRNA profile subgroup analyses showed that a multiple-miRNA assay is superior to a single-miRNA assay in OC detection, with an SE of 0.85 versus 0.73; an SP of 0.78 versus 0.82; and an AUC of 0.89 versus 0.84. In contrast, frequently reported miRNAs had better OC diagnostic accuracy than the miRNAs that were selected by sequencing technology or miRNA microarray-based screening approaches (AUC of 0.84 vs. 0.82). These results were consistent with previous studies and meta-analyses on the diagnostic accuracy of circulating miRNAs in the detection of solid tumors.19,22,24,26,44,52 Meta-regression analysis demonstrated that ethnicity, miRNA profiling, specimen type, and sample type were the potential sources of heterogeneity, which further confirmed the subgroup analysis results. Finally, Deeks’ funnel plot asymmetry tests showed no publication bias, and confirmed the robustness of our results.
Even with our best efforts to estimate the diagnostic value of circulating microRNAs in OC, there were several inevitable limitations that need to be addressed. First, due to insufficient data from the included studies, we were unable to analyze the potential relationship between the levels of circulating miRNAs and the clinicopathological features of OC, such as tumor size, FIGO stage, histological type, and lymph node metastasis. Second, heterogeneity was detected in the meta-analysis, but the sources of heterogeneity could not be fully identified by subgroup analyses and meta-regression based on several specific variables due to limited available data. Third, the cut-off definition of circulating miRNAs appears to be different in each study, which may be a reason for inconsistent results. Fourth, only a limited number of studies directly compared the diagnostic value of circulating microRNAs with other conventional markers (CA125, HE4, etc.), thus our study could not fully elucidate whether circulating microRNA assays can improve the diagnostic accuracy of commonly used tumor markers for OC screening. Finally, our meta-analysis was based on a limited population (3470 OC patients and 1606 healthy controls). Among all 30 miRNAs tested in the cited 36 studies, only six were discussed more than once. Therefore, large-scale studies are necessary to further validate the diagnostic value of circulating miRNAs in OC. Despite the limitations listed above, our study also has its merits: (a) it is the first meta-analysis to evaluate the diagnostic value of circulating miRNAs in OC detection; (b) subgroup and meta-regression analyses were performed to investigate sources of heterogeneity in our meta-analysis; (c) sensitivity analysis was also performed to confirm the robustness of our results, and no publication bias was detected in the meta-analysis.In conclusion, our meta-analysis results suggest that circulating miRNAs, especially multiple-miRNA assays, are promising biomarkers for the early diagnosis of OC; also, plasma miRNAs were superior to serum miRNAs for OC detection. However, large-scale prospective studies are needed to further validate the applicability of circulating miRNAs in the early detection of OC.
Supplemental Material
Supplement_S1---PRISMA_Checklist – Supplemental material for Identification of circulating microRNAs as diagnostic biomarkers for ovarian cancer: A pooled analysis of individual studies
Supplemental material, Supplement_S1---PRISMA_Checklist for Identification of circulating microRNAs as diagnostic biomarkers for ovarian cancer: A pooled analysis of individual studies by Quan Zhou, Man-Zhen Zuo, Ze He, Hai-Rong Li and Wei Li in The International Journal of Biological Markers
Footnotes
Acknowledgements
We thank all the authors whose publications were able to be adopted in our meta-analysis.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Yichang Medical and Health Research Project (A17-301-12).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
