Abstract
Background
An Increasing number of studies in the literature have shown that microRNAs (miRNAs) can be used as early diagnostic markers for esophageal carcinoma (EC), but their conclusions remain controversial. Hence, we performed this meta-analysis to evaluate the diagnostic accuracy of using miRNAs in EC and to provide an experimental basis for early diagnosis of the disease.
Methods
This meta-analysis included 39 Asian studies from 18 articles, which covered 3,708 EC patients and 2,689 healthy controls. We used a bivariate random-effects model, the chi-square test and the I2 test to assess sensitivity and heterogeneity.
Results
Pooled sensitivity, specificity, positive likelihood ratio, negative likelihood ratio and diagnostic odds ratio of miRNAs for diagnosis of EC in Asians reached 0.798, 0.785, 3.705, 0.257 and 14.391, respectively. Additionally, the area under the summary receiver operating characteristic curve was 0.86. Subgroup analysis based on research country (China vs. Japan), sample types (plasma vs. serum) and miRNAs (single vs. multiple; singly reported miRNAs vs. repeatedly reported miRNAs) showed no significant difference in accuracy of diagnosis for each subgroup.
Conclusions
MiRNAs can distinguish EC patients from healthy controls. Blood-based miRNAs have better diagnostic value in detecting EC than saliva-based miRNAs, whereas both serum and plasma are recommended for clinical specimens for miRNA detection.
Introduction
Esophageal carcinoma (EC) represents the eighth most common carcinoma and the sixth leading cause of cancer death worldwide according to the International Agency for Cancer Research Globocan 2012 (1); it mainly includes esophageal squamous cell carcinoma (ESCC) and esophageal adenocarcinoma (EAC), based on pathological characteristics (2). EC distribution presents remarkable regional characteristics: ESCC occurs mainly in Asia, whereas EAC is mainly observed in Western countries. ESCC is the most common type of EC, accounting for about 90% of EC cases, and is one of the most aggressive carcinomas of the gastrointestinal tract (3). Given the absence of typical clinical symptoms and lack of tumor markers with sufficient sensitivity (SEN) and specificity (SPE), most EC patients are diagnosed at a relatively late stage of the disease (4). At present, although surgical techniques and chemotherapy and/or radiotherapy regimens have been improved, the overall survival rate of ESCC remains poor, ranging from 3% to 5% (5). However, the rate of 5-year survival can increase up to 90% when tumors are detected at early stages (6). Therefore, early diagnosis is important and is urgently needed for ESCC patients.
Currently, endoscopic biopsy remains the primary method for diagnosis of EC; however, its invasiveness and expense limit its widespread use (7, 8). The potential for sample errors with random endoscopic biopsy decreases the effectiveness of this procedure (9). With the developments in medical research and technology, more tumor markers have been discovered; these markers can be used to distinguish EC patients from the healthy population. Conventional serum tumor markers, which include carcinoembryonic antigen, carbohydrate antigen 19-9 and squamous cell carcinoma antigen, are used in early diagnosis and monitoring tumor dynamics of ESCC (10-12). However, insufficient SEN and SPE seriously restrict clinical application of these markers. Therefore, further studies should center on exploring novel molecular biomarkers with accurate SEN and SPE and noninvasiveness to detect early-stage EC.
Considering the molecular biomarkers, attention has been drawn to microRNAs (miRNAs) due to their potential as diagnostic biomarkers of cancers. MiRNAs are small (19-24 nucleotides in length), well conserved and noncoding RNAs, regulating expression of more than 30% of human human genes by inducing mRNA degradation or inhibiting posttranscription (13, 14). MiRNAs play vital roles in cell growth, proliferation, apoptosis, migration and invasion (15-17), and at present, increasing numbers of studies have indicated that abnormal miRNA expression takes part in the initiation and progression of various cancers, including EC. For example, miRNA-203 is down-regulated in human ESCC, and it can inhibit cell proliferation, migration and invasion (18, 19). MiRNAs are potential biomarkers for early diagnosis of cancers, including ESCC; miRNA-205 is expressed at a low level in ESCC and is a good indicator for early diagnosis, with 76% SEN and 86% SPE (20). MiRNA expression profiles are cell-specific or tissue-specific; these specificities may help diagnose different cancer types and predict progression of malignancies (21, 22). Furthermore, miRNAs exist in a stable form in the blood or other tissue fluids, and their expression levels are reproducible (23, 24). In conclusion, these findings provide a promising, high-efficiency, nonaggressive, early-stage diagnosis method for ESCC patients using miRNAs.
Currently, a growing number of studies have investigated that abnormal expression of miRNAs can be good diagnostic biomarkers of early-stage EC. However, published studies on miRNAs in EC still present inconsistent conclusions. For example, Hirajima et al reported good diagnostic characteristics, with 86.8% SEN and 100% SPE, for miR-18a (25) in ESCC, whereas Xie et al reported a lower SPE of 47.4% for miR-144 (26) in EC. Additionally, Komatsu et al (27) found that miR-21 has poor diagnostic accuracy, with 60% SEN in ESCC, whereas Ye et al found a significantly higher SEN of 97% for miR-21 in ESCC. Up to now, most published meta-analyses have evaluated the diagnostic accuracy of miRNAs in patients with EC. In these meta-analyses, Wan et al insisted that sample types (saliva-, serum- and plasma-based) showed no difference in diagnostic accuracy in EC (28); Wang et al suggested that blood-based miRNA assay displayed higher diagnostic accuracy than saliva-based miRNA assay in detecting ESCC (29), and Fen et al further analyzed diagnostic differences between serum-based and plasma-based assays in ESCC and found no discrepancy (30). In other carcinoma meta-analyses, investigators have shown better accuracy for plasma-based specimens than serum-based ones in breast cancer and gastric cancer (31-33). To further explore the clinical applicability of miRNA in EC patients, this present meta-analysis was performed by expanding the sample size.
Materials and methods
Search strategy
For this meta-analysis, we searched electronic databases, including PubMed, Embase, China National Knowledge Infrastructure and other sources, for studies that had assessed the diagnostic value of miRNA in EC until October 2016. Keywords used were as follows: “(miRNA or microRNA)” and “(esophageal or esophageal carcinoma or esophageal cancer)” and “(diagnosis or sensitivity or specificity or receiver operating characteristic [ROC] curve)”. Reference lists of various articles were also manually examined to find additional eligible studies. No language restriction was imposed on the search criteria.
Inclusion and exclusion criteria
Inclusion criteria for this meta-analysis consisted of the following: (i) patients were diagnosed with EC; (ii) miRNAs were used to diagnose EC; (iii) sufficient data were presented to generate 2 × 2 tables, including true positives, false positives, true negatives and false negatives, which were used to calculate values of SEN and SPE. Exclusion criteria were the following: (i) focus on survival or prognosis of EC; (ii) articles that were reviews, case reports or meta-analyses; and (iii) insufficient data.
Data extraction
Two authors independently screened the full texts of the included articles and extracted information as follows: lead author, year of publication, country of publication, sample size, patient spectrum, miRNA profiles, SEN, SPE and data of 2 × 2 tables. When a study contained both training and validation groups, each group was regarded an independent study in this meta-analysis. Table I presents the details of the studies included.
Characteristics of studies included in the meta-analysis
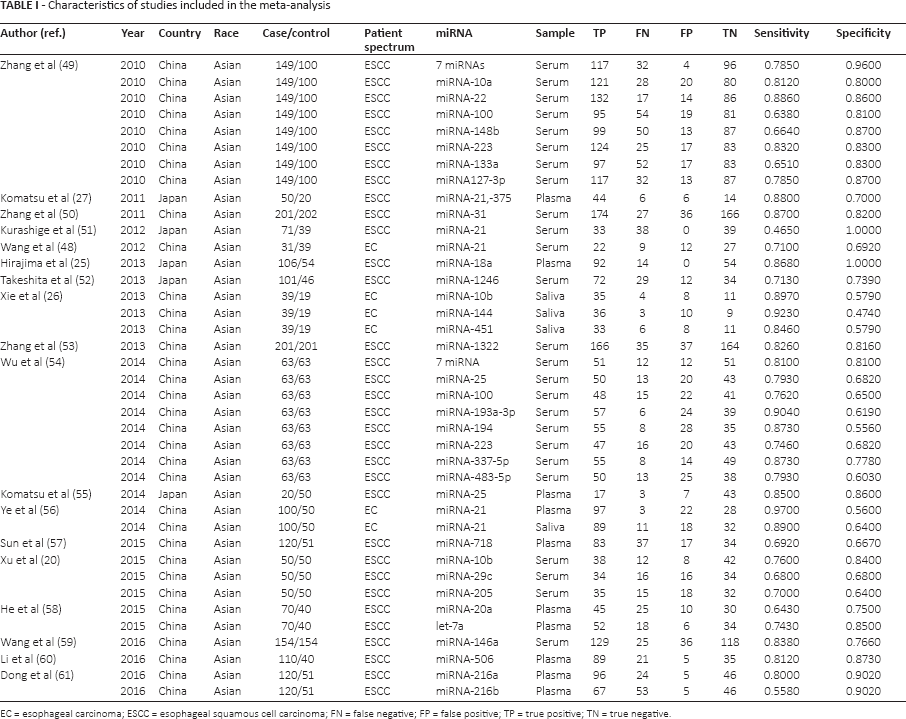
EC = esophageal carcinoma; ESCC = esophageal squamous cell carcinoma; FN = false negative; FP = false positive; TP = true positive; TN = true negative.
Statistical analysis
All data analyses were conducted using standard methods recommended for diagnostic accuracy in Stata software (version 12.0; Stata, College Station, TX, USA). We calculated SEN, SPE, positive likelihood ratio (PLR), negative likelihood ratio (NLR) and diagnostic odds ratio (DOR) with 95% confidence interval (95% CI) using a bivariate meta-analysis model (34). SEN and SPE of articles included were pooled to plot the summary receiver operating characteristic (SROC) curve and to calculate the area under the curve (AUC) (35). We used the chi-square and I2 tests to assess heterogeneity among studies. Values of p<0.1 or I2>50% indicated significant heterogeneity (36, 37). To evaluate heterogeneity between studies, we performed both subgroup and sensitivity analysis. Deeks' funnel plots were used to investigate publication bias (38).
Results
Search results and characteristics of studies
Through searching for keywords in databases, we found 231 articles, of which 50 were considered for further full-text review after checking titles and abstracts. In the end, we included 18 articles according to our above-mentioned inclusion and exclusion criteria (Supplementary Figure 1, available online at www.biological-markers.com – Flow diagram of the study).
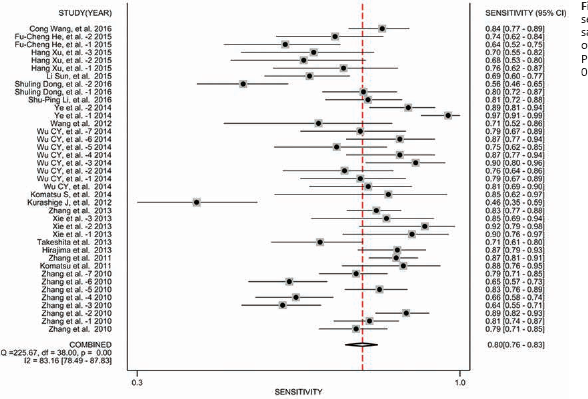
Forest plot of sensitivity of miRNA assays in the diagnosis of esophageal cancer. Pooled sensitivity was 0.80 (95% CI, 0.76-0.83).
Table I shows specific characteristics of the included literature. In 18 articles, 39 studies were included, covering 3,708 patients and 2,689 healthy controls. Among the studies included, 33 studies from 15 publications were related to miRNAs as ESCC diagnostic biomarkers, and 6 studies from 3 publications were related to miRNAs as EC diagnostic biomarkers (no clear description was observed on type of EC); 34 studies had been performed in China, 5 studies in Japan and all studies considered Asian populations. Twenty-five studies used serum samples, 10 studies depended on plasma specimens and 4 studies were based on saliva samples. This present meta-analysis included reports of 30 different miRNAs, with 5 miRNAs repeated in these reports. Two studies focused on panel miRNAs, and 37 studies discussed single miRNAs.
Diagnostic accuracy
To evaluate the diagnostic accuracy of miRNAs in detecting EC, we first constructed forest plots of SEN and SPE of miRNA assays for diagnosis of EC (Figs. 1 and 2). With the existence of significant heterogeneity between SEN and SPE data (I2 = 83.16%, p<0.01; and I2 = 79.77%, p<0.01, respectively), we used a random-effects model to calculate pooled estimates. Pooled results and their corresponding 95% CIs were as follows: SEN, 0.798 (95% CI, 0.763-0.829); SPE, 0.785 (95% CI, 0.739-0.824); PLR, 3.705 (95% CI, 3.078-4.458); NLR, 0.257 (95% CI, 0.221-0.300) and DOR, 14.391 (95% CI, 11.103-18.653). The AUC reached 0.86 (95% CI, 0.83-0.89), and Figure 3 shows the corresponding overall SROC curve. In summary, our findings suggested that miRNAs could be used to discriminate EC samples from healthy controls.
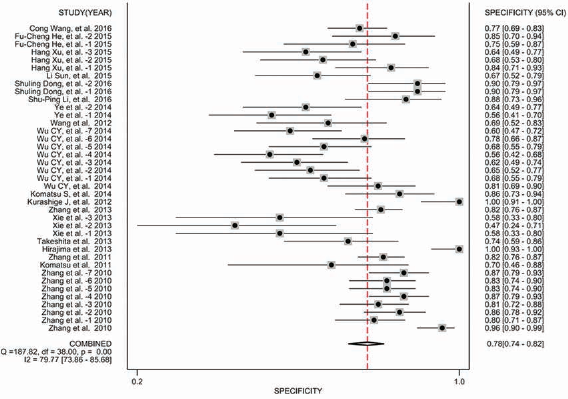
Forest plot of specificity of miRNA assays in the diagnosis of esophageal cancer. Pooled specificity was 0.78 (95% CI, 0.74-0.82).
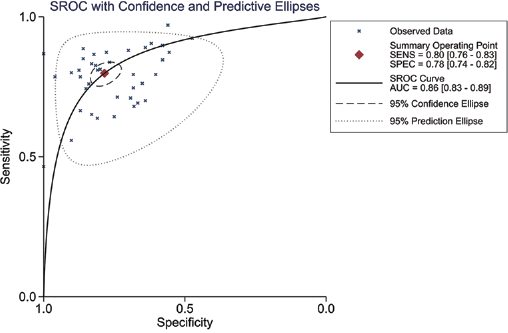
Summary receiver operating characteristic (SROC) curve for overall studies. The AUC was 0.86 (95% CI, 0.83-0.89). AUC = area under the curve; SENS = sensitivity; SPEC = specificity.
ESCC is the most common subtype of EC. Our study included 15 articles on ESCC. We performed an independent meta-analysis on miRNA diagnostic accuracy to discriminate ESCC patients from healthy controls. Pooled SEN, SPE, PLR, NLR and DOR totaled 0.777 (95% CI, 0.742-0.809), 0.809 (95% CI, 0.766-0.846), 4.072 (95% CI, 3.312-5.005), 0.275 (95% CI, 0.237-0.319) and 14.789 (95% CI, 11.027-19.833), respectively, and the AUC reached 0.86 (95% CI, 0.83-0.89). The miRNA diagnostic values obtained for ESCC were similar to those for EC.
Subgroup analyses
For the sake of exploring potential sources of heterogeneity, subgroup analysis was performed based on sample types (blood or saliva, serum or plasma). Table II lists the pooled diagnostic parameters for each group.
Summary estimates of diagnostic criteria
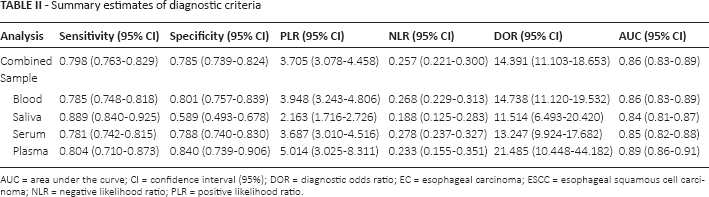
AUC = area under the curve; CI = confidence interval (95%); DOR = diagnostic odds ratio; EC = esophageal carcinoma; ESCC = esophageal squamous cell carcinoma; NLR = negative likelihood ratio; PLR = positive likelihood ratio
For blood samples, pooled values were SEN = 0.785, SPE = 0.801, PLR = 3.948, NLR = 0.268, DOR = 14.74 and AUC = 0.86, respectively. For saliva samples, pooled values were SEN = 0.889, SPE = 0.589, PLR = 2.163, NLR = 0.188, DOR = 11.514 and AUC = 0.84. For serum samples, the pooled values were SEN = 0.781, SPE = 0.788, PLR = 3.687, NLR = 0.278, DOR = 13.247 and AUC = 0.85. For plasma samples, pooled values were SEN = 0.804, SPE = 0.840, PLR = 5.014, NLR = 0.233, DOR = 21.485 and AUC = 0.89.
The above results suggest that sample type (blood or saliva) is a potential source of heterogeneity. Blood-based miRNA assays displayed better diagnostic accuracy than saliva-based ones. However, no significant difference was observed in diagnostic accuracy between serum and plasma. We also conducted subgroup analysis for countries (China vs. Japan) and miRNA (single vs. multiple miRNAs; singly reported vs. repeatedly reported miRNAs) and found no significant differences. The results are shown in Table III.
Summary estimates of diagnostic criteria
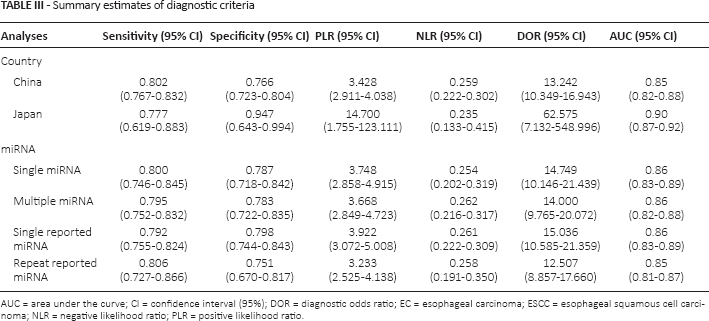
AUC = area under the curve; CI = confidence interval (95%); DOR = diagnostic odds ratio; EC = esophageal carcinoma; ESCC = esophageal squamous cell carcinoma; NLR = negative likelihood ratio; PLR = positive likelihood ratio
Sensitivity analysis and publication bias
To ensure that our findings were not significantly influenced by any individual study, we performed an SEN analysis and observed no significant heterogeneity. To further evaluate potential publication bias, Deeks' funnel plot asymmetry test (t = 1.54, p = 0.133) was conducted, and no significant publication bias was detected. These tests all confirmed the validity of our meta-analysis results.
Discussion
Our meta-analysis included 39 studies, and it suggested that miRNAs were ideal diagnostic markers of EC, with a pooled SEN of 0.789, pooled SPE of 0.785 and AUC of 0.86. Through subgroup analysis, a blood-based miRNA assay was found to display better diagnostic accuracy than a saliva-based miRNA assay in detecting EC, but no significant difference in diagnostic accuracy existed between serum and plasma. These results indicate that both serum and plasma can be recommended as clinical specimens for miRNA detection.
MiRNA was first discovered in 1993 by Lee while studying nematode development (39). In 2008, miRNA was detected in human serum for the first time by Lawrie et al (40). Then miRNAs were detected in different body fluids (including plasma, serum, urine, milk, semen and tears) (41), and they showed strong stability in these media (41-43). With research developments, miRNA was reported to participate in the occurrence of cancer in oncogenes or tumor suppressor genes (Supplementary Table I, available online at www.biological-markers.com – Expression of miRNA in esophageal carcinoma in meta-analysis).
An increasing number of recent studies have discovered that miRNAs can be used as markers for early diagnosis of tumors. For example, Tsujiura et al found that miR-106b was overexpressed in plasma of patients with gastric cancer, while 1 month after surgery, levels of miR-106b decreased rapidly. Thus, miR-106b could be used as a biomarker for early diagnosis of gastric cancer (44). Detection of serum miR-21 can be used in the early diagnosis of colorectal cancer (45). Low serum levels of miR-361-3p and miR-625 serve as markers for early diagnosis of lung cancer (46). Urinary miR-574-3p and miR-107 are markers for early diagnosis of prostate cancer (47). Some studies have suggested that miRNAs can be used as early diagnostic biomarkers for EC, presenting high SEN and SPE. Wang and Zhang discovered higher serum miR-21 in EC patients than in healthy controls and that this miRNA could serve as a diagnostic marker for EC, with 71.0% SEN and 69.2% SPE (48). However, conclusions remain contradictory. Therefore, more studies are needed to further confirm research results.
The results of our meta-analysis confirmed that miRNAs are noninvasive biomarkers that can be used to discriminate EC patients from healthy controls; these results are similar to those of Wan et al (28). In a study by Wang (29), a blood-based miRNA assay displayed better diagnostic accuracy than saliva-based miRNA assay in ESCC. Our results in EC were similar to those of Wang, but opposite to those of Wan et al (28), who insisted that saliva-, serum- and plasma-based miRNA samples showed no difference in diagnostic accuracy for EC. Our study also indicated that no difference exists between plasma-based and serum-based specimens in terms of diagnostic value for EC detection; whereas some previous studies have shown that plasma-based specimens exhibited higher accuracy than serum-based specimens in gastric cancer and breast cancer (28, 29).
The reasons for these differences remain unclear, and the mechanism of abnormal expression of miRNA in different sample species needs further studies. In spite of this uncertainty, our study results are significant regarding early diagnosis of EC. Detecting miRNA levels in serum and plasma presents a higher accuracy than in saliva.
In addition to the above-mentioned data, we also performed subgroup analyses based on number of miRNAs. A nonsignificant higher accuracy was observed in multiple miRNAs vs. individual miRNAs; this result was contrary to those of some previous studies (31-33), which stated that multiple miRNAs present significantly better accuracy than single miRNAs. As only 2 articles included multiple miRNAs in our analysis, these findings may be insufficient to support such a conclusion. Thus, further research should include more studies on multiple miRNAs.
In subgroup analyses between countries of patients (China vs. Japan), our results indicated that no difference exists between Chinese and Japanese populations in terms of accuracy of diagnosis with miRNA. For the first time, we conducted subgroup analyses between repeated and single reports on miRNA, and no statistical difference was observed. An independent meta-analysis of ESCC was also conducted because it is one of the most common types of EC. Pooled SEN, SPE and the AUC reached 0.777, 0.809 and 0.86, respectively. These results are similar to miRNA diagnostic values in EC, indicating that miRNAs are potential diagnostic biomarkers of EC regardless of their subtype.
To guarantee the reliability of our research, we performed SEN analysis to ensure that findings were not significantly influenced by any individual study. We also conducted Deeks' funnel plot asymmetry test to evaluate potential publication bias. In our meta-analysis, all diagnostic miRNAs were detected in body fluids (blood or saliva) and also in tumor tissues. However, detection of miRNA in tissues is not ideal for early diagnosis considering its invasiveness and high cost. A significant role of miRNAs in cancer research should not be ignored.
Despite using a relatively large sample size, and although our study showed that miRNAs are potential noninvasive early diagnostic markers of EC, several limitations to this meta-analysis must be noted. First, few studies included multiple miRNAs. Second, the type of EC was not clearly noted in some studies, hence, we could not perform a subgroup analysis on the type of EC. Finally, the research participants studied in Asia were mainly concentrated in China and Japan. These populations may all be potential sources of heterogeneity. Therefore, more high-quality research is needed to study the accuracy of using miRNAs in the diagnosis of EC.
Conclusions
Our meta-analysis provides evidence that miRNAs may serve as noninvasive biomarkers for distinguishing EC patients from healthy controls. Blood-based miRNA assays have a higher diagnostic value than saliva-based ones in detecting EC, and both plasma- and serum-based miRNA are recommended for clinical diagnosis of EC. More issues must be considered before these findings can be translated into clinical applications, and the exact roles of miRNAs in EC progression will need to be clearly elucidated in the future.
Footnotes
Financial support: This research was supported in part by the National Natural Science Foundation of China (grant nos. 81260301 and 81560399).
Conflict of interest: The authors declare that they have no competing interests.
