Abstract
Introduction
Paraquat (PQ) is a herbicide, which is extremely toxic to humans and animals. 1 It is very fast acting agent that is used to control various grasses as well as broad leave weeds. 2 China is one of the leading PQ producing country, with daily production of 100,000 tons. 3 Due to the unavailability of effective antidotes, PQ intoxication can be fatal to humans. Animals as well as humans exposure to PQ occurs via ingestion, skin contact, or inhalation. PQ excessive exposures may result in death within 3.5 h. 4 However, the fatality rate of PQ poisoning has been observed to range from 50 to 90% when the pesticide is taken either accidentally or intentionally. 3 PQ intoxication generates excessive free radicals.5,6 That disrupt the anti-oxidant defense mechanism and function of cells. 7
The intoxication of PQ induces multiple organ damage including pulmonary toxicity, neurotoxicity, hepatotoxicity, cardiac toxicity, gastrointestinal toxicity as well as nephrotoxicity.8–13 Kidney is an important organ that is responsible for the excretion of waste materials. PQ accumulates in kidneys and leads to acute kidney injury (AKI) 14 with an immediate decline in renal filtration rate that causes significant morbidity and mortality.15,16 The renal injury and reduced renal functioning lead to inadequate elimination of PQ, which results in multi organ damage. 17 PQ exposure induces renal toxicity via the production of reactive oxygen species (ROS). Consequently, these ROS result in cell disruption by instigating lipid peroxidation (LP), apoptosis, mitochondrial damage and inflammatory response. 18
Anti-oxidants have crucial role in the treatment of oxidative stress related disorders.19–21 Plants are a potent source of medicines. 22 During the last few years, plant-derived anti-oxidants have been studied as possible medicines for treating a variety of OS-related disorders and toxicities. 23 Flavonoids are polyphenolic substances, which are abundant in plants, leaves, vegetables and fruits and show various pharmacological properties 24 i.e., anti-oxidant, anti-apoptotic and anti-inflammatory. 25 Sinensetin (SNS), a polymethoxylated flavonoid extracted from Orthosiphon aristatus and numerous citrus fruits. It is reported to display anti-cancer, 24 anti-bacterial, 26 hepato-protective, 27 anti-oxidant, 28 anti-inflammatory 29 and gastro-protective 30 activities. Nevertheless, the role of SNS in mitigating the renal damage has not been described yet. To overcome PQ-induced renal damage, an investigation on plant-based anti-oxidants was required. Thus, based on the abovementioned pharmacological potentials of SNS, the present study was designed to assess the attenuative effect of SNS on PQ-instigated renal toxicity in rats.
Materials and methods
Chemicals
PQ (CAS No: 75365-73-0, purity: 98%, Molecular Weight: 257.16,) and SNS (CAS No: 2306-27-6, Molecular Weight: 372.37, purity: ≥98%) were acquired from Sigma-Aldrich (Merck GaA, Darmstadt, Germany).
Animals
Forty eight albino rats (180–200 g weight) were used to perform the study. Animals were kept in animal house of University of Agriculture Faisalabad (UAF), at room temperature, humidity 45 ± 5% and 12 h. light/dark cycle. Free access to water and standard feed was given. The rats were acclimatized to the laboratory conditions for 7 days. All the rats were treated according to the guidelines of European Union of Animals Care & Experimentation that were further approved by the UAF ethical committee (16969-72/24-05-2023).
Experimental protocol
Forty eight rats were separated into four equal groups (n = 12/group). Control group, PQ administered (5 mg/kg), PQ + SNS co-administered (5 mg/kg and 20 mg/kg) and only SNS supplemented group (20 mg/kg). In co-administered group first PQ was administered then after the interval of 1-h SNS was provided. All the doses were administered orally with the help of oral gavage. The dose of PQ was selected according to the previous study, 31 whereas SNS dose was used according to the study of Kong et al. 32 Metabolic cages were used to collect the urine sample. The experimental trial was performed for 30 days. At last, rats were sedated by using xylazine (6 mg/kg) and ketamine (60 mg/kg) as described by Hamza et al. 33 beheaded and cardiac blood was gathered in sterile containers. The collected blood underwent centrifugation for 15 min at 3000 rpm. After separation, plasma was preserved at – 20°C till further assessment. Both the kidneys were removed and the left kidney was immersed in formaldehyde (10%) for histopathological examination. On the other hand, right kidney was stored at – 80°C for detailed biochemical analysis.
Evaluation of biochemical parameters
The renal homogenate was used to check the activities of antioxidant enzymes as well as the level of ROS and MDA. CAT activity was estimated in accordance with the method outlined by Aebi. 34 GSR activity was appraised by using the approach of Carlberg and Mannervik, 35 GPx activity was appraised via the procedure of Lawrence and Burk. 36 SOD activity was estimated with the procedure of Sun et al. 37 While the content of GSH and the activity of GST were evaluated by using the protocol demonstrated by Jollow et al. 38 and Habig et al. 39 respectively. The method of Hayashi et al. 40 was used to assess the ROS content, while Placer et al. 41 method was used to measure MDA content.
Evaluation of kidney function markers
Kidney function markers i.e., urea, urobilinogen, creatinine, urinary protein, creatinine clearance & albumin protein were measured using Randox standard laboratory kits (Crumlin, UK) as per the company’s instructions. The levels of urea, creatine and albumin protein were measured from the blood whereas, urinary samples were used to estimate the urobilinogen and urinary proteins.
Evaluation of kidney injury molecule-1 and neutrophil gelatinase-associated lipocalin
Neutrophil gelatinase-associated lipocalin (NGAL) and Urinary kidney injury molecule-1 (KIM-1) levels were determined using NGAL Quantikine ELISA Kit and KIM-1 Quantikine ELISA kits in accordance with the company’s instructions (R & D Systems China Co. Ltd, China). The levels of NGAL and KIM1 were measured from the urinary samples.
Evaluation of inflammatory markers
Renal homogenate was used to estimate Inflammatory indices i.e., IL-1β, TNF-α, IL-6, NF-κB level and COX-2 activity by using an ELISA kit (BioTek, Winooski, USA).
Evaluation of apoptotic markers
Renal homogenate was used to determine Bax, Caspase-9, Bcl-2, and Caspase-3 levels were appraised by using ELISA kit (from Cusabio Technology Llc in Houston, Texas, USA), according to the manufacturer’s guidelines.
Histopathology
For histopathological observation, hepatic tissues were kept in 10% formaldehyde for 24 h, dehydrated in the increasing concentrations of alcohol and then embedded in the blocks of paraffin wax. With the help of a microtome 5 μm sections were cut and stained using Hematoxylin-Eosin. Finally, photomicrography was performed to observe the slides under light microscope at 400X.
Statistical analysis
Data were shown as Mean ± SEM. One-way ANOVA and Tukey’s test was applied to the whole data. The level of significance was set at p < 0.05.
Results
Protective role of sinensetin on anti-oxidant enzyme activity
Effect of PQ and SNS on oxidative stress parameters.
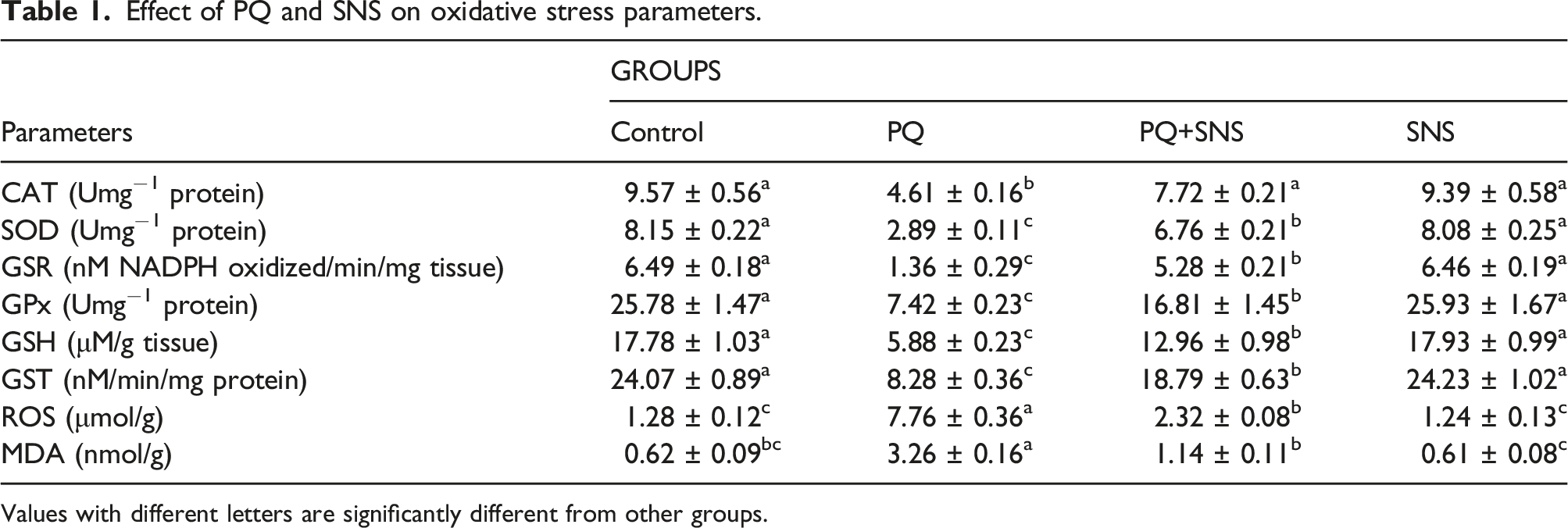
Values with different letters are significantly different from other groups.
Protective role of sinensetin on malondialdehyde and reactive oxygen species level
PQ-administered rats showed a notable (p < .05) rise in MDA and ROS levels as compared to the control animals. However, PQ + SNS co-treated rats showed a remarkable decrease in MDA and ROS levels in contrast to PQ-administered rats. However, SNS (alone) treated rats displayed the levels of ROS and MDA close to the control animals (Table 1).
Protective role of sinensetin on renal function markers
Protective role of SNS on renal function markers.

The values with dissimilar superscripts are considerably different from the other groups.
Protective role of sinensetin on inflammatory markers
Protective role of SNS on inflammatory markers.

The values with dissimilar superscripts are significantly different from other groups.
Protective role of sinensetin on apoptotic markers
Protective role of SNS on apoptotic markers.

The values with dissimilar superscripts are significantly different from other groups.
Protective role of sinensetin on renal histology
PQ administration caused histopathological anomalies i.e., leukocyte infiltration, glomerular atrophy, enlargement of Bowman’s capsule, thickening of the Bowman’s capsule basement membrane, indicating nephrotoxicity as compared to the control animals. However, SNS + PQ co-administration significantly restored all these damages in comparison to PQ exposed animals. However, in SNS only treated animals the histological profile was comparable to the control animals (Figure 1). (a) Control group demonstrating regular histology. (b) PQ intoxicated group demonstrating PQ induced renal damages. (c) PQ + SNS group demonstrating recovered renal histological profile. (d) SNS group demonstrating regular histology as in control group. PQ: Paraquat; SNS: Sinensetin; RT: Renal tubules; BS: Bowman spaces.
Discussion
Paraquat (PQ) is a lethal chemical compound that is broadly applied as a herbicide, however it is very toxic to human as well as animals. 1 PQ intoxication leads to several organs failure as well as acute kidney injury (AKI)42,43 and death. 14 PQ induced oxidative stress (OS) causes DNA damage, lipid peroxidation, oxidation of cellular protein 44 and histopathological anomalies in the renal tissues. 45 Nowadays, phytomedicines are used to treat various disorders, including kidney damage. Sinensetin (SNS) is a polymethoxylated flavonoid, that is primarily isolated from the branches of Orthosiphon aristatus and numerous citrus fruits. SNS is reported to show significant antioxidant, 28 anti-inflammatory, 29 and anti-cancer 46 and anti-bacterial 26 properties. Therefore, the present research was designed to assess the protective effect of SNS on PQ induced kidney damage in rats.
Our findings revealed that PQ treatment decreased the activities of antioxidant enzymes (CAT, GST, GSR, SOD, and GPx) and GSH content in the renal tissues, while escalating the levels of MDA and ROS. The findings of our study are also confirmed by the study of Ijaz et al. 47 who demonstrated that PQ exposure decreased the activities of anti-oxidants and increased ROS and MDA levels. Antioxidants serve as a key defense mechanism against ROS. 48 CAT, SOD and GPx are the major anti-oxidants responsible for the removal of ROS. 49 SOD changes O−2 into hydrogen peroxide. CAT is a key anti-oxidant that aids GSH and GPx in converting H2O2 into oxygen and water. 50 GSR maintains reduced GSH levels in cells, via reducing ROS levels. 51 GST has a pivotal function in detoxification. 52 The imbalance between ROS and anti-oxidants activity leads to the deterioration of renal tissues. 53 PQ intoxication increases the concentration of ROS, such as H2O2 and OH, which results in lipid peroxidation (LP).54,55 MDA is a marker of LP, and its level directly indicates the level of LP. 56 However, SNS supplementation reduced OS and increased the activities of antioxidant enzymes, while lowering the levels of ROS and MDA due to its anti-oxidant property. Moreover, Procházková et al. 57 have demonstrated that flavonoids have multiple hydroxyl groups in their chemical structure that contribute to the antioxidant property.
According to our investigation, PQ intoxication remarkably elevated KIM-1, NGAL, urea, urobilinogen and creatinine levels, while reducing the level of creatinine clearance and albumin in PQ treated rats. Creatinine & urea are important indicators to assess renal dysfunction. NGAL and KIM-1 are the important indicators of AKI. KIM-1 is a trans-membrane protein that is used as an initial diagnostic indicator for AKI. It is not expressed in normal or healthy kidney, but it can be found during the early stages of nephrotoxicity. 58 NGAL level increases in the blood following the renal damage and it is eliminated through the urine. 59 Urobilinogen does not normally occur in urine, 60 but it is reported as a result of renal toxicity. High creatinine level along with urea shows reduced renal function and acute kidney damage. 61 However, SNS treatment reduced KIM-1, NGAL, urea, urobilinogen and creatinine levels, as well as increased creatinine clearance and albumin due to its nephroprotective nature.
The exposure to PQ increased the levels of NF-κB, IL-6, TNF-α, IL-1β and COX-2 activity in PQ treated animals as compared to the control animals. Previously it was described that PQ exposure increased the levels of inflammatory markers in rats. 47 NF-κB is a transcription factor, which stimulates TNF-α, IL-1β, COX-2, IL-6 production that are associated with sever inflammation and other ROS associated diseases. 62 COX-2 is another major inflammatory indicator, which has significant role in inflammatory response. 63 However, SNS supplementation to PQ exposed rats resulted in a significant decrease in the levels of inflammatory markers. This protective activity of SNS might be ascribed to its anti-inflammatory nature. Moreover, it is demonstrated that SNS has been shown to reduce lipopolysaccharide-induced inflammation in RAW 264.7 cells, by regulating the protein level of inhibitor κB-α. 64
In the present research, PQ administration increased the levels of Caspase-9, Bax and Caspase-3, while decreasing the Bcl-2 level. The outcomes of our study are in line with the study of Mustafa et al. 65 who described that PQ exposure disturbed the levels of apoptotic markers in rats. Bax acts as an apoptotic marker, which stimulates apoptosis; besides Bcl-2 averts the cells from apoptosis. 66 A decline in Bcl-2 and increase in Bax drastically change mitochondrial membrane permeability and subsequently leads to augmented eviction of cytochrome C into the cytoplasm. 67 The elevated level of cytochrome C instigates Caspase-9 that activates Caspase-3, leading to proteins degradation and apoptosis. 68 However, SNS administration led to a considerable reduction in Caspase-3, Bax, Caspase-9 levels and a significant increase in Bcl-2 level owing to its anti-apoptotic property.
The administration of PQ promoted severe histopathological anomalies in the renal tissues i.e., deterioration of renal tubules, presence of sinusoidal space, vacuolation, tubular dilation and destruction of focal epithelial cell, necrosis of hematopoietic tissue, as well as tubular necrosis. Our results are further confirmed by previous studies of Awadalla 69 and Meulenbelt and Vale, 70 who reported that PQ intoxication prompts significant histopathological anomalies in the renal tissues of rats. Nevertheless, SNS treatment in PQ + SNS co-administered group protected the renal tissues and renal tubules from all these anomalies. This protective nature of SNS may be credited to its anti-apoptotic, anti-oxidant and anti-inflammatory nature. Moreover, it is demonstrated that SNS has the ability to protect from osteoarthritis due to its chondroprotective nature. 71
The present study has some limitations such as, the power analysis was not performed and the study was conducted using rats as animal model. Therefore, we recommend that the clinical trials should be performed in future to check its efficacy on human being.
Conclusion
Our study revealed that SNS showed significant mitigative effects against PQ prompted nephrotoxicity. PQ exposure disturbed kidney function markers, increased OS, apoptotic and inflammatory markers levels. Moreover, PQ exposure prompted histomorphological anomalies in rat’s renal tissues. However, SNS supplementation improved all these damages and histopathological anomalies due to its anti-apoptotic, anti-oxidant & anti-inflammatory activities.
Footnotes
Author contributions
MUI and AM intended the study. AH and AM executed the experiment. AH and HAK assisted in statistically analyzing the data. MUI, AM and AH wrote the manuscript. The manuscript was read and approved by all of the authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Animal welfare
The present study followed international and University of Agriculture, Faisalabad institutional guidelines for humane animal treatment and complied with relevant legislation.
