Abstract
Background
Lupus enteritis (LE) is prone to incorrect and missed diagnoses. LE primarily occurs during the active stage of systemic lupus erythematosus (SLE), which often manifests with alterations in peripheral blood cell that may serve as indicators of disease activity and organ damage. This study aims to investigate the alterations and predictive value of routine blood parameters for LE diagnosis.
Methods
This exploratory study retrospectively analyzed the medical records of 36 patients with SLE who were admitted to Suining Central Hospital between January 2006 and April 2023. Additionally, a control group consisting of 72 SLE patients without LE, matched for sex and age, was enrolled. A comparison was made between the two groups regarding clinical characteristics and changes in routine blood parameters. Binary logistic regression and receiver operating characteristic (ROC) curve analyses were conducted to identify independent risk factors and evaluate their diagnostic performance for LE.
Results
The LE group exhibited significantly higher neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR), along with a lower lymphocyte count (LYM), compared to the control group (
Conclusion
Significant differences were observed in several routine parameters between patients with LE and the control group, which can be attributed to the occurrence of LE during the active stage of SLE. However, only the NLR emerged as an independent risk factor for LE,and its predicting vulue for was insufficient; no blood routine parameter has been identified as an reliable predictor for LE.
Keywords
Introduction
Lupus enteritis (LE) refers to vasculitis or inflammation of the intestines associated with systemic lupus erythematosus (SLE), supported by imaging and/or biopsy evidence. 1 LE predominantly occurs during the active stage of SLE, and patients often present with typical SLE symptoms such as rash, fever, and joint pain. However, some patients with LE may exhibit solely non-specific gastrointestinal symptoms without other manifestations of SLE, posing challenges in distinguishing it from other acute abdominal conditions. Consequently, This can lead to incorrect or missed diagnoses. Although commonly used clinical tests such as erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) can partially aid in detecting the activity of SLE, they lack diagnostic value for LE. Active SLE often presents with alterations in morphology, size, and count of peripheral blood cells, which may indicate disease activity and organ damage. Lee et al. 2 reported a significant decrease in white blood cell (WBC) count among patients with LE, which exhibited a strong correlation with the presence of the disease. In contrast, Wang et al. 3 observed no significant difference in WBC count between the LE and non-LE groups; however, they did report a notable reduction in lymphocyte count among patients with LE, indicating its potential diagnostic value. The red cell distribution width (RDW), Which is an indicator of chronic inflammation, has been demonstrated as a valuable marker for determining the activity of SLE. 4 furthermore, composite inflammatory indicators derived from routine blood parameters, such as the neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR), have shown superior predictive value compared to single indicators. Previous studies have not only established the utility of these two parameters as indicators for the active stage of immune rheumatic diseases,5,6 but they have also highlighted the association between NLR and renal involvement in SLE. 5 Howerever, there have been limited studies conducted on the alterations in routine blood parameters among patients with LE; nevertheless, their findings have exhibited inconsistency. Furthermore, there is a lack of reports regarding the predictive efficacy of blood routine parameters in LE. Therefore, this study aims to analyze the alterations in blood routine parameters among individuals with LE to identify potential indicators for early diagnosis.
Materials and methods
General information
A total of 36 inpatients or outpatients with LE, who were admitted to Suining Central Hospital between January 2006 and April 2023 and had complete data, were included in the study. The patients were matched for age and sex at a ratio of 1:2 with 72 SLE patients without LE serving as the control group. The inclusion criteria were as follows: each Patient with SLE met the 1997 classification criteria for SLE established by the American College of Rheumatology (ACR),and patients with LE were required to meet the following three conditions 7 : (i) presence of abdominal symptoms along with at least one of the following: nausea, abdominal pain, vomiting,diarrhea, or bloating; (ii) presence of at least one of these three imaging manifestations: mesenteric vasodilation, bowel wall thickening, or increased attenuation of mesenteric fat 1 ; (iii) lack of relief from digestive symptoms following use of mucosal protective agents, acid inhibitors, antibiotics, or gastrointestinal motility drugs, but successful alleviation after use of an increased dosage of glucocorticoids.
The following exclusion criteria were applied: (1) Patients with primary gastrointestinal lesions、adverse drug reactions, and other acute abdominal conditions; (2) Patients with co-morbid circulatory system diseases such as rheumatic valve disease or heart failure); (3) Patients who had received blood transfusions, steroid hormones, immunosuppressants or drugs affecting WBC count in the past 3 months; (4) Patients with missing key clinical data. The initial test data performed at admission were extracted for all enrolled patients. Patients in the LE group consisted of 33 females and three males, aged between 20 and 66 years, with a mean age of (39.22 ± 11.48) years. The disease durations ranged from 0.2 to 18.0 years, with a mean duration of (7.28 ± 4.65) years. The control group comprised 67 females and five males, aged between 21 and 73 years, with a mean age of (39.47 ± 12.19) years. The disease durations ranged from 0.9 to 19.0 years, with a mean duration of (7.85 ± 4.80) years. No statistically significant differences were observed between the two groups regarding age, sex or disease duration (
Study measures
General information, clinical symptoms, physical signs, laboratory test findings, imaging findings, SLE Disease Activity Index (SLEDAI) score, various routine blood parameters, NLR, and PLR were assessed in both groups.
Statistical methods
Statistical analyses were conducted using SPSS 15.0 and GraphPad Prism 5.0 software packages. Normally distributed measurement data were presented as
Results
Comparison of general information
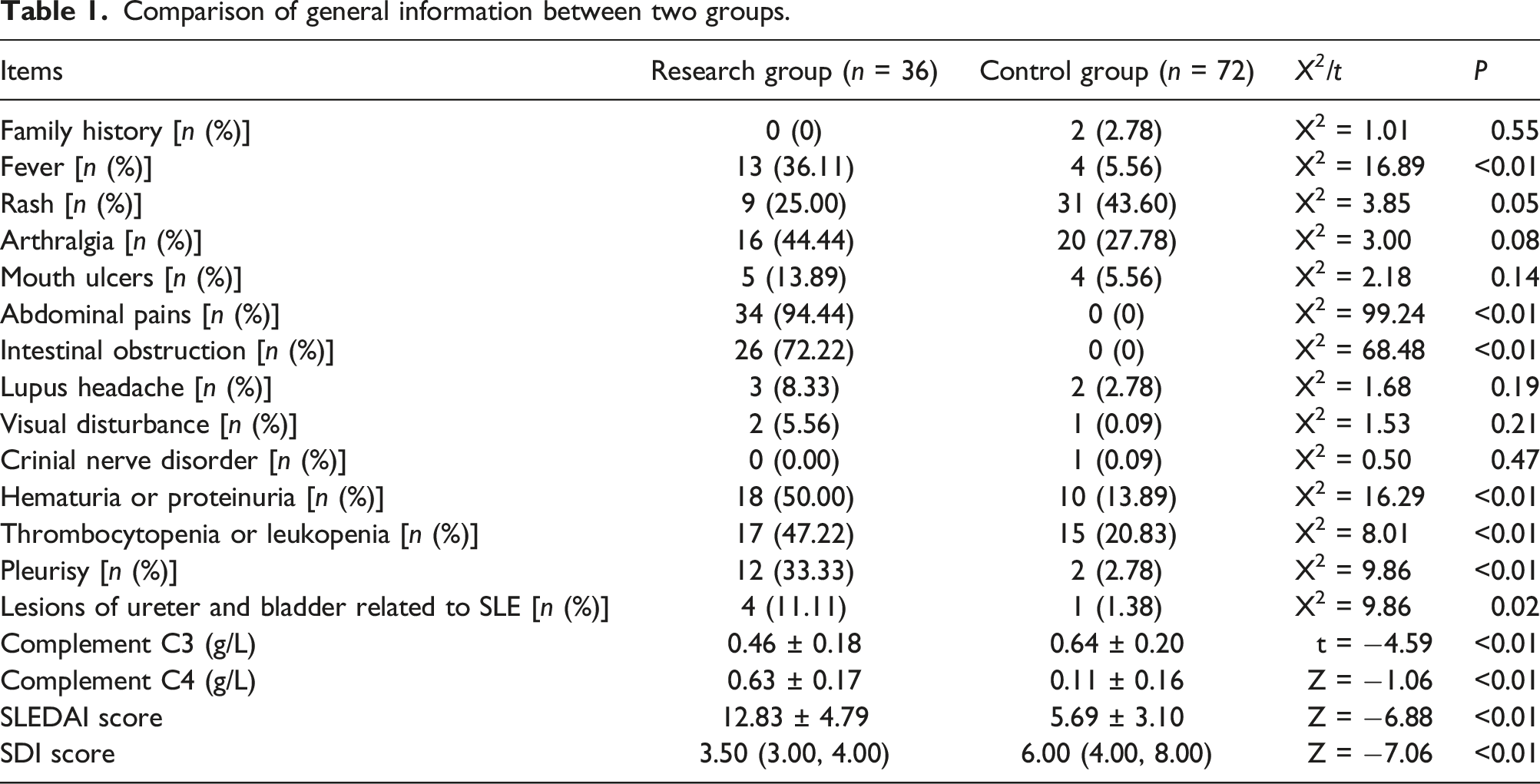
Comparison of general information between two groups.
Comparison of hematological parameters
Comparison of hematological parameters between two groups.

Note: RDW is the red cell distribution width, MPV is the mean platelet volume, PLR is the platelet-to-lymphocyte ratio, and NLR is the neutrophil-to-lymphocyte ratio.
Analysis of LE risk factors
Logistic regression analysis of LE risk factors.

Diagnostic performance of NLR for LE
The analysis of the receiver operating characteristic (ROC) curves revealed that the area under the curve for diagnosing LE using NLR was 0.68 ( ROC curve of NLR for diagnose LE.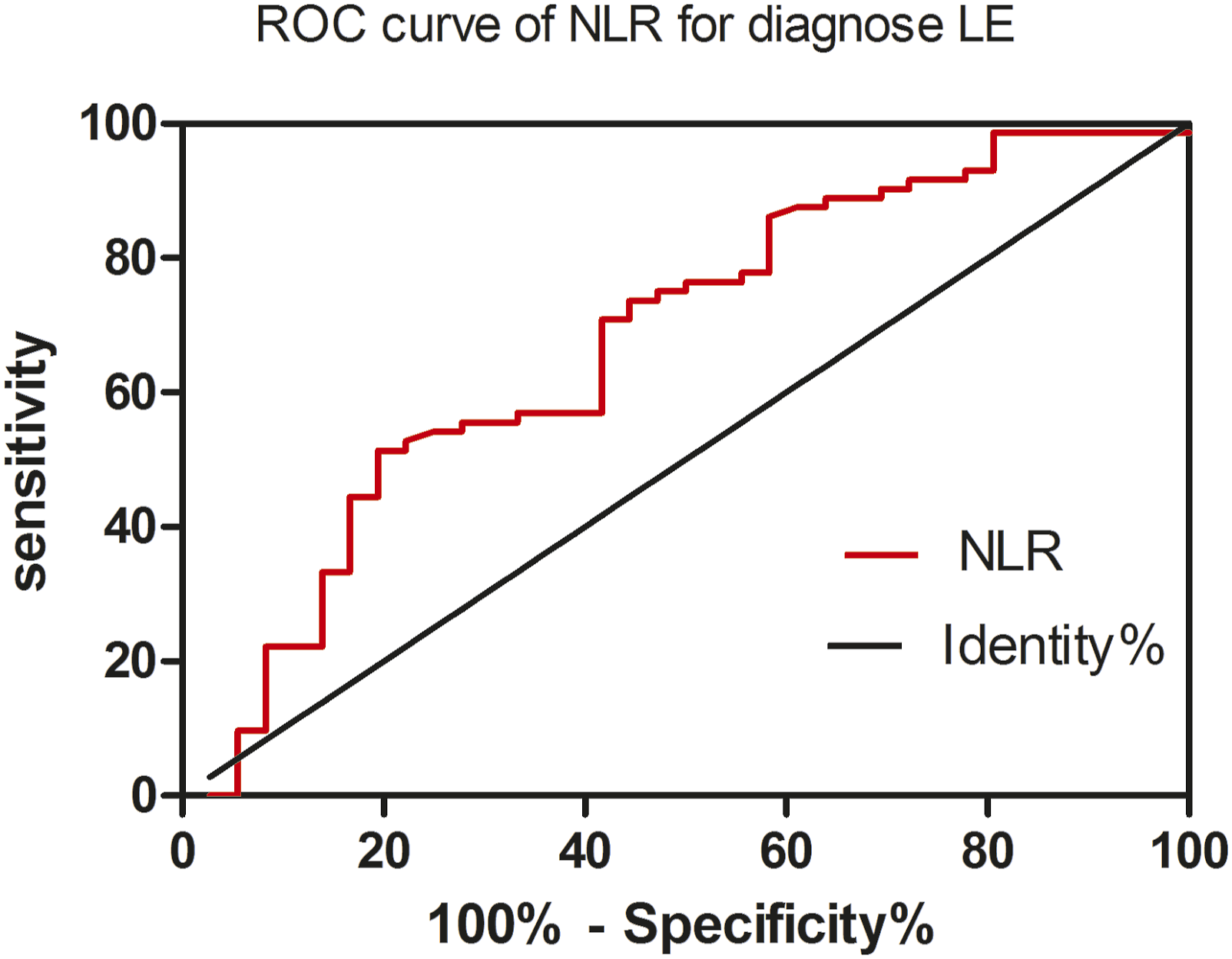
Discussion
Systemic lupus erythematosus (SLE) is a highly heterogeneous autoimmune disease characterized by significant variations in clinical manifestations due to the involvement of different organs. Studies have demonstrated that approximately 40%–50% of SLE patients present with symptoms related to the digestive system, 8 most commonly associated with other gastrointestinal disorders (such as small intestinal malabsorption, autoimmune liver disease, and inflammatory bowel disease), drug side effects or infection; However, it should be noted that some of these symptoms may also arise directly from SLE itself. The underlying pathophysiology of LE involves vasculitis, leading to local ischemia of the gastrointestinal tract and subsequent manifestation of symptoms such as nausea, vomiting, diarrhea, and abdominal pain.9,10 However, some patients with LE only exhibit intestinal involvement during new-onset11–13 or recurrent LE. 14 In highly rare cases, patients with LE may present with intestinal emphysema 15 or acute gastric dilatation. 16 Renal involvement can also coexist with Some cases of LE.17,18 These non-specific symptoms cannot be used to differentiate between LE and other gastrointestinal diseases easily, which can easily lead to incorrect and missed diagnosis. Patients with LE who do not receive prompt diagnosis and treatment are at risk of developing severe complications, including intestinal bleeding, disseminated intravascular coagulation, intestinal perforation, and intestinal infarction, 7 which can endanger the lives of the patients. Therefore, when managing SLE patients presenting with digestive system symptoms, particularly in cases where initial symptoms involve gastrointestinal lesions, it is imperative to consider not only the potential for acute abdomen and infectious diseases (such as acute cholecystitis, acute appendicitis, acute pancreatitis, perforated peptic ulcer, and intestinal infection) but also the possibility of LE. Additionally, careful evaluation should be conducted to exclude abdominal pain caused by gastrointestinal ischemia associated with conditions like antiphospholipid syndrome (APS) and thromboembolism.
Due to the non-specific nature of its symptoms, LE is susceptible to incorrect and missed diagnoses which necessitates an urgent need for identifying effective markers or diagnostic models for it. Currently, numerous studies have been published on the risk factors associated with this condition. Aso et al. 19 discovered that antibodies against the α3 subunit of anti-ganglionic nicotinic acetylcholine receptors could potentially serve as biomarkers for LE. Yoshida et al. 20 reported that a lower CH50 level may indicate intractable or recurrent cases of lupus erythematosus (LE). Liu et al. 21 developed a highly accurate nomogram for predicting this condition accurately. Furthermore, APS, elevated peripheral blood CD19 cell count, urinary system involvement, and elevated d-dimer levels have also been identified as risk factors for LE. These prediction methods are either overly complexor costly, limiting their accessibility to all healthcare facilities.
Studies have shown that LE mainly occurs during active SLE22–24. Although LE cases have also been reported in patients with SLEDAI <4, these occurrences are relatively infrequent.2,25 Our study demonstrated that all 35 patients with lupus erythematosus (LE), except for one who had a SLEDAI score <5, exhibited SLEDAI scores >5. Additionally, the LE group showed significantly higher SLEDAI scores compared to the non-LE group, which supports the idea that LE mainly presents during the active stage of systemic lupus erythematosus (SLE). Simultaneously, alongside general symptoms and digestive tract symptoms, we observed a higher prevalence of systemic damage in the blood system, urinary system, nervous system, and other systems among patients in the LE group compared to those in the non-LE group. Additionally, the SDI score was significantly elevated in the LE group compared to the non-LE group. This observation can be attributed to the predominant occurrence of LE during SLE's active stage and its relatively severe manifestation.
Routine hematological parameters may exhibit alterations during the onset or active phase of the disease, and certain variations among these parameters could potentially serve as indicators for predicting the likelihood of LE onset. For instance, leukopenia 2 or lymphocytopenia 3 may suggest a higher probability of LE occurrence. NLR and PLR are indicators derived from routine blood parameters that offer greater utility in comprehending a patient's systemic inflammatory state due to their ability to integrate two distinct immune pathways. NLR and PLR can serve as indicators for determining the active stage of rheumatoid arthritis and spondyloarthropathy, while NLR has also been associated with renal involvement and pathological types in SLE. These studies suggest that routine blood parameters in patients with LE may undergo changes, some of which might be linked to organ damage and have predictive value for organ dysfunction. In this study, a combination of binary logistic regression analysis and ROC curve analysis was employed to identify risk factors for LE from routine blood parameters and determine their predictive value. Our findings suggest that NLR may serve as an independent risk factor for LE, while the other parameters did not demonstrate any association with LE. However, the ROC curve analysis further demonstrated that the practical utility of NLR in predicting LE is limited, as evidenced by its modest area under the curve and low Youden index.
In this study, we observed significant differences in the three parameters of NLR, PLR, and LYM between the LE group and non-LE group. Specifically, NLR and PLR were significantly increased while LYM was significantly decreased in the LE group, which is consistent with the active phase of SLE. Regression analysis revealed that only NLR was an independent risk factor for LE; however, its predictive value for LE was limited to promoting disease activity rather than predicting occurrence. Since no effective blood routine parameter has been found to predict LE, a comprehensive approach encompassing immunological, imaging, and pathological tests should be adopted in clinical practice to ensure a thorough evaluation of LE and mitigate the risks associated with misdiagnosis and inappropriate treatment.
Conclusion
Based on the available data, significant differences were observed in several parameters between patients with lymphedema (LE) and the control group, which can be attributed to the occurrence of LE during the active stage of SLE. Although we identified NLR as an independent risk factor for LE, its predictive value for LE was limited to promoting disease activity rather than predicting occurrence. Since no effective blood routine parameter has been found to predict LE, a comprehensive approach encompassing immunological, imaging, and pathological tests should be adopted in clinical practice to ensure a thorough evaluation of LE and mitigate the risks associated with misdiagnosis and inappropriate treatment.
Footnotes
Acknowledgements
The authors would like to thank the reviewers and authors of all the references, We also thank all the editors of the European Journal of inflammation.
Author contributions
Wen–Xin Zhang was the main contributor to this manuscript, and she wrote the initial version. Jin–He Xiong (corresponding author) finalized the manuscript. Wen–Xin Zhang downloaded the references and collected data, while Jin–He Xiong evaluated the data critically. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
