Abstract
Keywords
Introduction
ALI can occur during cardiopulmonary bypass surgery, lung transplantation, shock, sepsis, among others, which can cause damage to alveolar epithelial cells and vascular endothelial cells. 1
Lung parenchymal stem cells (which promote epithelial proliferation or re-epithelialization through exogenous growth factors or cytokines) and extrapulmonary stem cell transplantation are two promising new treatment methods for ALI and ARDS. 1 The stem cells of the lung parenchyma patients, that is, their own stem cells, immediately replenish the damaged cells locally after injury but their proliferation potential is low and the repair effect is very limited. Meanwhile, the stem cells outside the lungs enter the lung tissue to repair the damaged lung, and are reserved for repairing more serious damage, which has become the focus of current research and the application of ALI and ARDS stem cell therapies. MYDGF is a growth factor secreted by bone marrow cells. Studies have shown that it can promote the growth of endothelial cells and the proliferation of liver cancer cells, as well as inhibit cardiomyocyte apoptosis and promote the proliferation of vascular endothelial cells.2,3 Pretreatment of ALI using MYDGF may improve the survival and growth of the transplanted MSCs, thereby improving the efficacy of cell transplantation and lung function. The underlying mechanism may be that MYDGF promotes microangiogenesis and provides an aerobic microenvironment for MSCs to survive; secondly, MYDGF may increase the survival rate of MSCs through anti-apoptotic effects. This experiment explored the therapeutic effects in mice treated with BMSCs or MYDGF alone and the combination of the two after ALI.
Methods
Materials
Recombinant human MYDGF (N-6His) (Novoprotein Scientific (Shanghai) Inc.), LPS (biosharp, BS904-10 mg), Bcl-2 antibody (Affinity Biosciences, AF6139), Bax antibody (Affinity Biosciences, AF0120), NF-κB p65 antibody (Affinity Biosciences, AF5006), p-Akt antibody (Affinity Biosciences, AF0016), and beta actin Polyclonal antibody (Affinity Biosciences, E-AB-20058) were purchased. A TNF-α ELISA kit (Wuhan Genemei Technology Co, Ltd), an IL-6 ELISA kit (Wuhan Elisa Lab Technology Co, Ltd), an IL-10 ELISA kit (Wuhan Elisa Lab Technology Co, Ltd), a BCA protein concentration determination reagent box (Elabscience Biotechnology), BMSCs (Procell Life Science and Technology Co, Ltd), and MLE-12 cells (Procell Life Science&Technology Co, Ltd) were bought.
Cells
For the cell experiment, five groups were created according to the results of the preliminary experiment. Group A: MLE-12; group B: MLE-12 + LPS; group C: MLE-12 + LPS + BMSCs; Group D: MLE-12 + LPS + MYDGF; group E: MLE-12 + LPS + BMSCs + MYDGF. MLE-12 and BMSCs cells from mice grown in the logarithmic phase were inoculated into 24-well plates, i.e., transwell chambers, at a density of 5 × 104/ml, according to the conditions of each group, with six replicate wells per group.
Proliferation and toxicity of CCK-8 cells
The cell experiment was divided into five groups: group A: MLE-12; group B: MLE-12 + LPS; group C: MLE-12 + LPS + BMSCs; group D: MLE-12 + LPS + MYDGF; group E: MLE-12 + LPS + BMSCs +MYDGF. Mouse MLE-12 cells grown in the logarithmic phase were used to inoculate a 24-well plate at a density of 5 × 104/ml. In groups B, C, D, and E, 400 μg/mL LPS was used to stimulate MLE-12 cells. In groups D and E, MYDGF (3000 ng/mL) was added, and in group A, the same amount of PBS was added. In groups C and E, BMSCs (5 × 104/ml) were added to a Transwell chamber with a pore size of 0.4 μm for indirect co-culture. In groups A, B, and D, no BMSCs were added. After culturing cells for 24 h, the chambers were removed, the supernatant was carefully aspirated, and LPS and MYDGF were added as described previously. After 24 h of culture, the activity of MLE-12 in each group was detected by using a Cell Counting Kit-8 (CCK-8).
Animals
Forty healthy male BALB/c mice aged 7-8 weeks and weighing 24 ± 3 g were provided by and raised in the Experimental Animal Center of Anhui Medical University, and our study has been approved by the Experimental Animal Ethics Committee of Anhui Medical University (LLSC20201143). The mice were randomly divided into five groups, with eight mice in each group, including group A: control group (Did nothing); group B: LPS group (the ALI model); group C: LPS + BMSCs group (treatment with BMSCs); group D: LPS + MYDGF group (treatment with MYDGF); and group E: LPS + BMSCs + MYDGF group (treatment with BMSCs and MYDGF). Anesthetized mice: a 5–8 mm “I”-shaped incision was made on the front of the neck of each mouse, parallel to the trachea, to expose it. The insulin injection needle was parallelly inserted into the trachea to inject the corresponding drugs (group A was injected with PBS, and theother groups were injected with LPS). After the injection of the drugs, the mice were tilted and rotated several times so that the drugs could enter the left and right main bronchi more evenly. The mice in the A, B, D group were injected with DMEM through the tail vein. The order of drug injection was the following: tail vein injection of BMSCs or DMEM at 1 h after intratracheal injection of LPS or PBS, and tail vein injection of MYDGF 1 h before the intratracheal injection of the drugs. The drug injection doses used were: LPS (concentration of 10 mg/2 mL, dose of 10 μg/g), PBS (dose of 2 μl/g), BMSCs (density of 1 × 106/ml, dose of 1 × 107/kg), and MYDGF (concentration of 50 μg/500 μl, dose of 200 μg/kg). A total of 24 h later, serum, alveolar lavage fluid, and lung tissue samples were collected for further testing. The samples were detected by single blind method. All animal experiments were carried out in accordance with institutional Policies and Guidelines for the Care and Use of Laboratory Animals and that all efforts were made to minimize animal suffering.
Tissue preparation
1% (m/V, or 5 mg/kg) Pentobarbital was I.P to anesthesia mice, Depth of anesthesia was determined by the pedal reflex test, the animals were euthanized by decapitation. Tissue proteins were extracted using 10% RIPA lysate on ice, and the supernatant was centrifuged at 4°C and stored in liquid nitrogen for subsequent imprinting experiments.
Ccontent of BALF
The alveolar lavage fluid was collected, the supernatant was collected after centrifugation, diluted, and the protein concentration was determined using the BCA method. First, the standard protein solution was prepared. Second, the test solution was prepared. The BCA reaction took place and then the data was interpreted and the protein concentration was calculated.
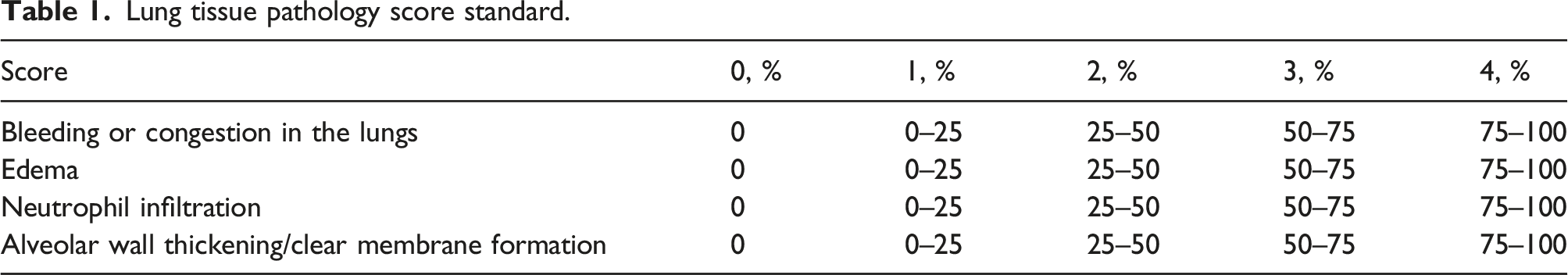
Hematoxylin-eosin staining
Lung tissue pathology score standard.
Western blot
After preparing the lung tissue protein sample, the protein concentration was determined. Then, a protein loading buffer was added and the solution was heated to 100°C for 5 min. SDS-PAGE electrophoresis was performed and the proteins were transferred to a PVDF membrane. The membrane was blocked, and incubated with the primary antibodies (NF-κB, p-Akt, Bax, and Bcl-2, 1:2000), and then with the secondary antibodies. Visualization of the membrane was performed successively. Blots were detected using an electrochemiluminescence system (GE Healthcare Biosciences) and qualified with Quantity One (Bio-Rad) software. We also detected β-actin as the protein loading control. Each target band was analyzed, the relative expression of the target protein was calculated, and statistical analysis was performed.
Enzyme linked immunosorbent assay
Whole blood samples were stored at 4°C for 2 h, centrifuged at 4°C and 3000 r/min for 10 min, and the levels of TNF-α, IL-6, and IL-10 were detected according to the operating procedure using an ELISA kit.
Statistical analysis
SPSS 23.0 was used for statistical analysis. All data are expressed as mean ± standard deviation (
Results
Proliferation and toxicity of cells
Comparison of OD value and cell survival rate detected by CCK-8 (

Group A: Control group, Group B: LPS group (model group), Group C: LPS + BMSCs group (treatment group), Group D: LPS + MYDGF group (treatment group), Group E: LPS + BMSCs + MYDGF group (treatment group).
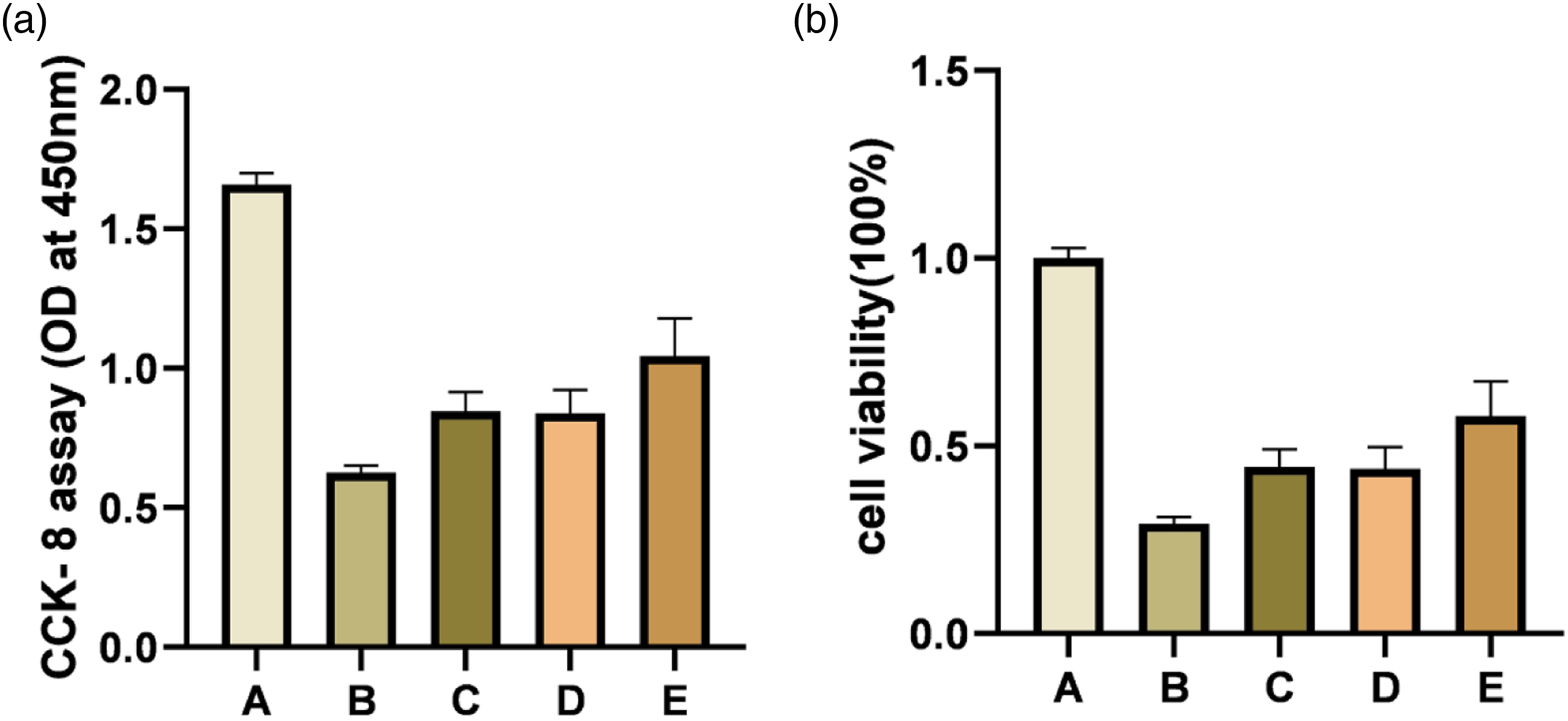
Proliferation and toxicity of CCK-8 cells. Note: Group A: MLE-12; Group B: MLE-12 + LPS; Group C: MLE-12 + LPS + BMSCs; Group D: MLE-12 + LPS + MYDGF; Group E: MLE-12 + LPS +BMSCs +MYDGF.
Content of BALF
BCA protein concentration of BALF μg/μl) (
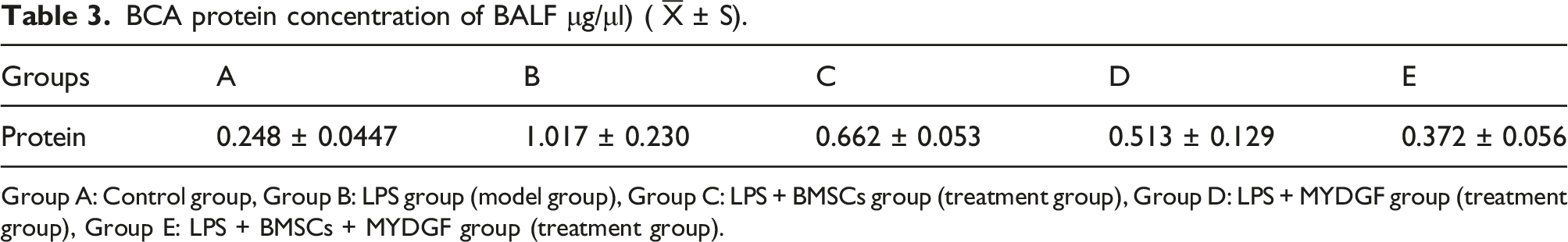
Group A: Control group, Group B: LPS group (model group), Group C: LPS + BMSCs group (treatment group), Group D: LPS + MYDGF group (treatment group), Group E: LPS + BMSCs + MYDGF group (treatment group).

Protein content of BALF. HE staining pathological pictures of the mouse lung tissues of each group (×200 times). Note: Group A: MLE-12; Group B: MLE-12 + LPS; Group C: MLE-12 + LPS + BMSCs; Group D: MLE-12 + LPS + MYDGF; Group E: MLE-12 + LPS +BMSCs +MYDGF.
Hexamination staining
The lung tissue sections of BALB/c mice in the blank control group were uniformly light red. No obvious bleeding or exudation points were found under the microscope, the alveolar structure was clear, there was no obvious inflammatory exudation in the alveolar cavity, and the alveolar walls were smooth and intact, with no thickening, no edema, or congestion in the lung interstium, and no obvious pathological damage was found. The microscopic findings of the lung tissue of BALB/c mice in the LPS-induced ALI model group included pulmonary interstitial edema, congestion, hyaline membrane formation, unclear alveolar boundaries, obvious alveolar wall thickening, blurring and disappearance of the normal physiological structure of alveoli, congestion of alveolar interstitial capillaries with obvious bleeding, and exudation points. Furthermore, there was obvious red blood cell and inflammatory cell infiltration in the alveolar cavities of mice. Microscopic observation of the treatment groups (group C: LPS + BMSCs; group D: LPS + MYDGF; group E: LPS +BMSCs +MYDGF) showed that the pathological damage was lower than that of the model group. Although the alveolar boundary was still unclear in part of the visual field, the alveolar structure was relatively complete under part of the visual field, and the alveolar wall was slightly thickened. Although edema, hemorrhage, and inflammatory infiltration were still observed in the lung tissues, they were reduced compared with the model group. According to the lung tissue pathology scoring standard (Table 1), the pathological damage score of the blank control group was significantly lower than that of the model group (p < .05). The pathological damage scores of the treatment groups (group C: group LPS + BMSCs; group D: LPS + MYDGF; group E: LPS +BMSCs +MYDGF) were significantly lower than that of the model group, and significantly higher than that of the blank control group (p < .05) (Figure 3). HE staining was used to analyze the pathological damage of mouse lung tissues. Group A: control group; Group B: LPS (model group); Group C: LPS + BMSCs (treatment group); Group D: LPS + MYDGF (treatment group); Group E: LPS + BMSCs + MYDGF (treatment group). Note: Group A: MLE-12; Group B: MLE-12 + LPS; Group C: MLE-12 + LPS + BMSCs; Group D: MLE-12 + LPS + MYDGF; Group E: MLE-12 + LPS +BMSCs +MYDGF.
Western blot
The Western Blot results in the lung tissues showed that the expression of NF-κB in the ALI model group was significantly higher than that in the control group, while that in the treatment group was significantly lower than that in the ALI model group (p < .05). There was no statistical significance between groups C and D, and between groups D and E (p > .05). The relative expression of NF-κB in group E was significantly lower than that in group C (p < .05) (Figure 4(a) and (e)). Western Blot was used to analyze the relative expression of NF-κB, p-AKT, Bax, and bcl-2 proteins in mouse lung tissues. Group A: control group; Group B: LPS (model group); Group C: LPS + BMSCs (treatment group); Group D: LPS + MYDGF (treatment group); Group E: LPS + BMSCs + MYDGF (treatment group). Note: Group A: MLE-12; Group B: MLE-12 + LPS; Group C: MLE-12 + LPS + BMSCs; Group D: MLE-12 + LPS + MYDGF; Group E: MLE-12 + LPS +BMSCs +MYDGF.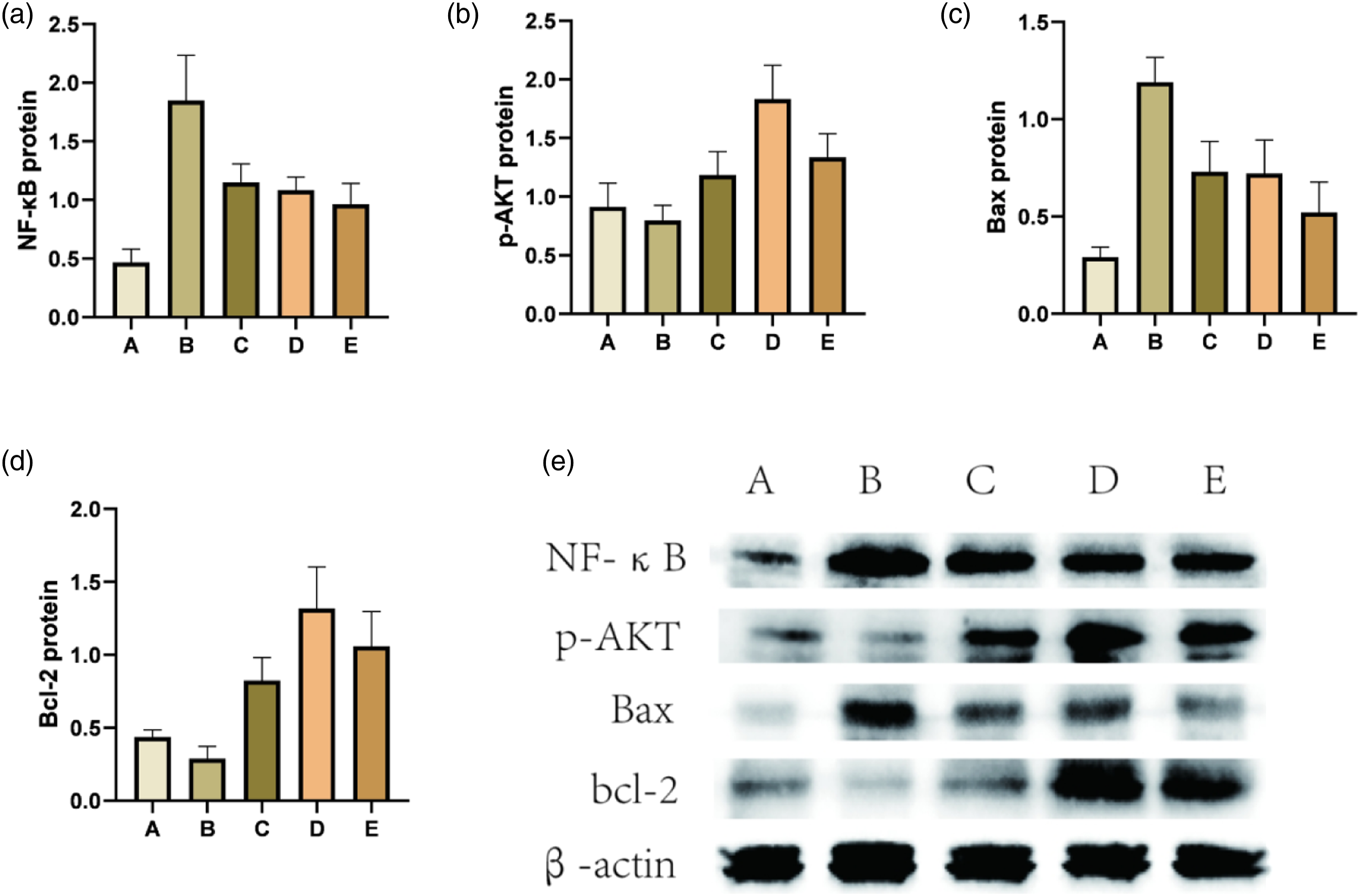
The relative expression of p-AKT in the lung tissues in the ALI model group and the control group was not statistically significant (p > .05), while that in the treatment group was significantly higher than that in the ALI model group (p < .05). There was no statistical significance between groups C and E (p > .05). The protein relative expression of group C was significantly higher than that of group D and group E (p < .05) (Figure 4(b) and (e)).
The Western Blot results showed that Bax expression in the ALI model group was significantly higher than in the control group, while that in the treatment group was significantly lower than that in the ALI model group (p < .05). There was no statistical significance between groups C and D (p > .05). The relative expression of Bax in group E was significantly lower than that in groups C and D (p < .05) (Figure 4(c) and (e)).
The expression of bcl-2 in the ALI model group was lower than that in the control group, while that in the treatment group was significantly higher than that in the ALI model group (p < .05). There was no statistical significance between groups D and group E (p > .05). The relative expression of bcl-2 in group E was significantly higher than that in group C (p < .05) (Figure 4(d) and (e)).
Enzyme linked immunosorbent assay
Serum IL-6 concentration (pg/mL) (
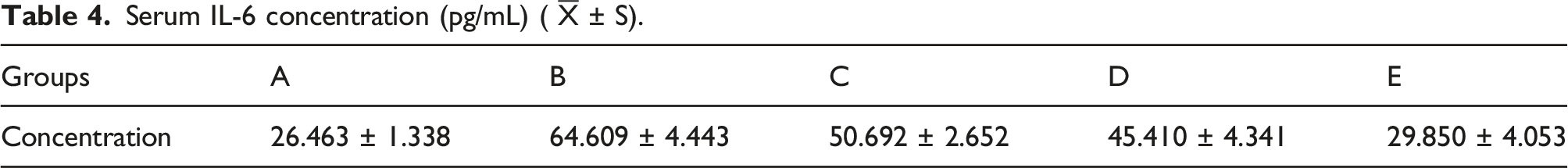

ELISA was used to analyze mouse serum IL-6, IL-10, and TNF-α concentrations. Group A: control group; Group B: LPS (model group); Group C: LPS + BMSCs (treatment group); Group D: LPS + MYDGF (treatment group); Group E: LPS + BMSCs + MYDGF (treatment group). Note: Group A: MLE-12; Group B: MLE-12 + LPS; Group C: MLE-12 + LPS + BMSCs; Group D: MLE-12 + LPS + MYDGF; Group E: MLE-12 + LPS +BMSCs +MYDGF.
Serum IL-10 concentration (μg/μl)(

Group A: Control group, Group B: LPS group (model group), Group C: LPS + BMSCs group (treatment group), Group D: LPS + MYDGF group (treatment group), Group E: LPS + BMSCs + MYDGF group (treatment group).
Serum TNF-α concentration (μg/μl)(

Group A: Control group, Group B: LPS group (model group), Group C: LPS + BMSCs group (treatment group), Group D: LPS + MYDGF group (treatment group), Group E: LPS + BMSCs + MYDGF group (treatment group).
Discussion
ARDS is pathologically characterized by increased infiltration of inflammatory cells into the lungs and increased permeability of the vascular endothelial cells, which further aggravates lung tissue damage, interstitial congestion, and pulmonary edema, 4 and can lead to apoptosis, further aggravating the clinical symptoms. 5 Many supportive therapies reduce the mortality of ARDS, including lung protective mechanical ventilation, neuromuscular blockade, and prone position. Conservative fluid therapy increases the number of days that patients with ARDS require a ventilator. Compared with supportive therapy, drug intervention has not significantly improved the clinical outcome of patients with ARDS. 6 Low tidal volume lung protective ventilation is still the basis for the treatment of patients with ARDS. 7
In in vivo and in vitro experiments, the dysfunction of the normal endothelial-epithelial barrier plays an important role in the development of ALI. 8 A common and important pathological change of ALI caused by various reasons is the damage of the alveolar epithelial cells. If the alveolar epithelial cells are repaired, lung injury can be effectively treated. With the development of stem cell biology research, the understanding of the role of lung-related stem cells/progenitor cells in the repair of lung injury is also increasing. Speeding up the repair process of damaged lung tissues and reducing inflammation are two important aspects in improving the prognosis of patients with ALI and ARDS. 9 Growth factors and anti-inflammatory cytokines are involved in the repair of ALI. 8
Lung parenchymal stem cells, which promote epithelial proliferation or re-epithelialization through exogenous growth factors or cytokines, and extrapulmonary stem cell transplantation are two promising new treatment methods for ALI and ARDS. 10 The stem cells of the lung parenchyma, that is, their own stem cells, immediately replenish the damaged cells locally after injury, but their proliferation potential is low and the repair effect is very limited. Meanwhile, the stem cells outside the lung enter the lung tissue to repair the damage, and are reserved for repairing more serious damage, which has become the focus of current research and application of ALI and ARDS stem cell therapies.
Mesenchymal stem cells (MSCs) are pluripotent stem cells. They have characteristics such as multi-directional differentiation potential, hematopoietic support, and promotion of stem cell implantation, as well as immune regulation and self-replication. MSCs can differentiate into a variety of tissue cells under specific induction conditions both in vivo and in vitro, which can be used to repair aging and diseased tissues and organs.11,12
Stem cells, especially MSCs, have shown therapeutic potential in lung diseases. 13 More and more pieces of evidence show that stem cell therapy has great therapeutic prospects for ARDS. 14 MSCs can be isolated from most types of mesenchymal tissues, such as the bone marrow, umbilical cord blood, placenta, and adipose tissue. MSCs are considered non-immunogenic, and this characteristic theoretically allows allogeneic transplantation. 15 MSCs coordinate tissue repair capabilities by releasing soluble paracrine factors 16 and have anti-inflammatory effects on the host tissues. 17 The vascular endothelium is injured in ARDS, and MSCs may help protect the endothelial barrier function. 18 Alveolar epithelial cell injury leads to multiple damage pathways in the development of ARDS. BMSCs may protect epithelial cell function in ARDS. 10 The soluble factors secreted by BMSCs play a vital role in wound repair, probably through their anti-inflammatory, anti-apoptotic, angiogenic, and immunomodulatory properties.19,20 The potential beneficial mechanisms of MSCs include the production of paracrine factors, the release of microvesicles, and the transfer of mitochondria, which can reduce lung endothelial and epithelial damage and enhance the relief of pulmonary edema. 21 Prostaglandin E2 (PGE2), which is secreted by BMSCs, has a powerful immunomodulatory function, allowing macrophages to secrete IL-10 to accelerate the recovery of the damaged tissues. 22
At present, there is a considerable amount of preclinical evidence that bone marrow mesenchymal stem cell therapy may be effective in the treatment of ARDS, especially in ARDS associated with infection. 23 The most studied extrapulmonary adult stem cells include BMSCs, and multipotent adult progenitor cells. Among them, BMSCs have unique advantages, such as a high amplification ability and good gene stability after multiple in vitro passages, multi-differentiation potential, and the ability to repair various tissues and organs, and the fact that they can be imported into the body through various ways, not easily leading to immune rejection. They can also be conveniently sampled, are easy to separate and culture, and do not involve moral or ethical issues.24–26 However, there are some problems when using exogenous mesenchymal stem cell treatments. There are several reports showing that intravenous infusion of MSCs causes pulmonary embolism and right pulmonary infarction. 27 However, it has also been reported that intravenous injections of MSCs were safe in an injured lung model, and no acute adverse effects were observed on the cardiopulmonary function. 25
Growth factors seem to play an important role in the repair and resolution of ALI. A variety of growth factors can promote the repair of a damaged alveolar epithelium. The currently known ones include keratinocyte growth factor, hepatocyte growth factor, epidermal growth factor, acidic fibroblast growth factor, transforming growth factor-α (TGF-α), and vascular endothelial growth factor, among others. Growth factors use at least two main pathways in ALI: tyrosine kinase receptor mediation and serine-threonine kinase receptor mediation. 8
It has been confirmed that MYDGF inhibits cardiomyocyte apoptosis through the PI3K-Akt pathway. Studies have shown that it promotes the growth of endothelial cells through the MAPK1/3, STAT3, and cyclin D1 signaling pathways, that it has an angiogenic effect, and can be used in post-AMI treatment to protect and repair the heart. 3 In hepatocellular carcinoma, C19orf10 regulates the MAPK/Akt pathway and activates cell proliferation. 28
In this experiment, LPS induced ALI in mice and the therapeutic effect was evaluated by determining the alveolar lavage fluid protein concentration, via HE staining, by analyzing the serum inflammatory factor content, the expression of several lung tissue proteins, and by performing in vitro cell proliferation-toxicity experiments. Interleukin-10 is an anti-inflammatory cytokine and can protect from lung injury, 29 which involves the inhibition of TH1 differentiation, inhibition of neutrophil activation, and the downregulation of chemokines and it has prophylactic value for lipopolysaccharide-induced ALI. 30 Increased levels of interleukin-10 in lung tissues inhibit lipopolysaccharide-induced ALI by reducing the induction of pro-inflammatory mediators and chemokines, reducing mast cell activation and NF-κB activation, and downregulating the pathways that produce oxidative stress. 31 It has been reported that interleukin-10 has an anti-apoptotic effect and restores the damaged Bax/bcl-2 ratio in articular chondrocytes. 32
Although interleukin-6 has a pro-inflammatory and anti-inflammatory dual effect, it mainly exerts pro-inflammatory effects. Interleukin-6 has been shown to be elevated in both plasma and BALF and indicates a poor prognosis in patients with ALI. 8
The functional importance of NF-κB in inflammation is based on its ability to regulate the promoters of a variety of inflammatory genes, including tumor necrosis factor-α, interleukin-1β, interleukin-6, and iNOs. 33 The inappropriate activation of NF-κB is related to the pathogenesis of ALI/ARDS. 34
In this animal pre-experiment, one mouse died in the BMSC treatment group on the second day after treatment, which was considered to be related to the high LPS concentration, individual differences, BMSCs, and other factors. For example, BMSCs could have not been completely digested, and the resuspension could have not been homogeneous, leading to the presence of residual small cell clumps and to the precipitation of BMSCs, which may have led to the active clumping of the EP tube before injection. This could have resulted in an excessively high concentration of BMSCs when entering the body or in the formation of tiny cell clumps, which eventually could have resulted in pulmonary embolism after the tail vein injection of BMSCs. The indicators tested in this experiment did not show completely better results in the E treatment group than in the C and D treatment groups. In the C and D treatment groups, the test indicators did not clearly show which treatment regimen was better. At present, a large number of animal experiments in China and abroad have confirmed that BMSCs have a clear therapeutic effect on ALI and have a protective effect on damaged lung tissues. In our previous experiments, we also confirmed that BMSCs can promote the formation of chick embryo chorioallantoic membrane blood vessels. In animal experiments, we found that tail vein injections of MYDGF or BMSCs alone have a certain therapeutic effect on LPS-induced ALI, which reduced inflammation and cell apoptosis in ALI, thereby reducing lung injury. Moreover, the therapeutic effect of an injection of BMSCs after pretreatment with MYDGF may be better than using MYDGF and BMSCs alone. Based on the results of the CCK-8 in vitro cell experiment, the combined action of MYDGF and BMSCs can reduce the damage of LPS to MLE-12 cells and promote the repair and regeneration of MLE-12 cells. LPS is used alone in vitro to directly stimulate MLE-12 cells to simulate the damage of lung epithelial cells in ALI. In vivo, ALI damages lung epithelial cells in various ways, which mainly results from a variety of inflammatory factors that are released by immune cells during an immune response to LPS. However, in an in vitro cell experiment, LPS can damage MLE-12 cells without involving the participation of immune cells. The state of MLE-12 cells after stimulation by LPS treatment was significantly worse than that of the blank control group and the treatment groups. The cell boundary was unclear, the growth on the bottom of the cell inoculation plate was not uniformly distributed, and cell were clumped. Although the growth status of the cells in each treatment group was worse than that of the blank control group, it was better than that in the model group, which was also confirmed by the follow-up CCK-8 experimental results. According to the results of in vitro cell experiment, combined treatment effect of MYDGF and BMSCs can reduce the damage effect of LPS on MLE-12 and reduce apoptosis
Although this study has proved the role of MYDGF and BMSCs, there is still a lack of in-depth mechanism research, which will be further explored in subsequent studies. And the power analysis for sample size calculation was not done, but the number of animals and the number of repetitions in this study are greater than or equal to 3. We conducted a frontal correlation design and study based on previous research examples and literature.
Conclusions
We found that the combined treatment effect of MYDGF and BMSCs was better than using MYDGF or BMSCs alone. We speculate that a pretreatment with MYDGF after ALI in mice may improve the survival and growth of transplanted MSCs, thereby improving the curative effect of cell transplantation. Furthermore, MYDGF has microangiogenesis-promoting and anti-apoptosis effects, thereby improving lung function. This experiment is only a preliminary exploration, but further experiments still need to verify this view.
Footnotes
Author contributions
Shenglin Ge, Hui-Wen Gong and Qian Gong formulated the basic hypotheses, experimental design and data analysis. Shi Jiang and performed the experiments. All of the authors edited the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors have completed the ICMJE uniform disclosure form.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This paper was funded by Ge’s Natural Science Foundation of Anhui Province of the First Affiliated Hospital of Anhui Medical University.
Ethical approval
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Experiments were performed under a project license (NO.: LLSC20201143) granted by the Experimental Animal Ethics Committee of Anhui Medical University, in compliance with the national or institutional guidelines for the care and use of animals.
