Abstract
Keywords
Introduction
Sudden sensorineural hearing loss (SSNHL) is characterized by an acute hearing loss of more than 30 dB in three consecutive frequencies within three days.1–10 Viral infection, vascular ischemia, and immune mediation have been proposed as possible etiologies of SSNHL.2–6, 8–13 Recently, SSNHL has been investigated in terms of chronic inflammation.1,3,5,10,13 Chronic inflammation can cause microvascular injury and atherogenesis, which contribute to the risk of ischemia.3,10 Immunologic inflammatory factors, such as C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR), and hematologic inflammatory factors, such as white blood cell (WBC) count, neutrophil count or percentage, and lymphocyte count or percentage, have been used as risk and prognostic factors for SSNHL in many studies.1,4,6,8,11–14
Neutrophil-to-lymphocyte ratio (NLR) was reported to be a predictive marker in cardiovascular diseases, and platelet-to-lymphocyte ratio (PLR) was reported to be associated with atherosclerosis and atherothrombosis in peripheral arterial occlusive disease.3,13 NLR and PLR have also been used as predictive and prognostic factors for SSNHL in many studies.1–7,9,10,13,14 For other otologic sensorineural diseases, NLR and PLR were reported to be predictive parameters for vestibular neuritis, 15 and NLR was reported to be a useful predictive and prognostic indicator of Bell’s palsy. 16
In this study, the associations of immunologic inflammatory factors, such as CRP and ESR, and of hematologic inflammatory factors, such as WBC count, neutrophil percentage, lymphocyte percentage, NLR, and PLR, with prognosis of SSNHL were evaluated.
Materials and methods
Patient selection
This study was prospective and longitudinal. The inclusion criteria were acute hearing loss greater than 30 dB in three consecutive frequencies within three days resulting in diagnosis of unilateral SSNHL and no associated pathology such as chronic ear disease, otologic surgery, head trauma, ototoxic drug use, noise exposure, Meniere’s disease, autoimmune disease, or acoustic tumor.
Fifteen patients who were diagnosed with unilateral SSNHL were prospectively enrolled and followed from July 2016 to December 2016. The hearing threshold of the unaffected side was normal or mild hearing loss (23.2 ± 9.6 dB). Within seven days from the onset of SSNHL, the subjects were treated with the high dose oral steroid prednisolone 1 mg/kg/day for five days followed by a five-day taper. If the subject did not recover from the hearing loss after the initial high-dose oral steroid treatment, an additional intratympanic dexamethasone 5 mg injection was performed five times with a three-day interval between each injection with the initial injection performed within three days from the end of oral steroid treatment.
The subjects had no symptoms of vertigo and no history of other chronic diseases, such as hypertension, hypercholesterolemia, heart disease, kidney disease, or diabetes. Exclusion criterion was acute inflammatory disease such as upper respiratory infection because an acute inflammation could influence the values of inflammatory markers obtained from blood sampling. There were no patients excluded from enrollment.
This study was performed in accordance with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The Institutional Review Board of Seoul Medical Center approved this study (IRB 2016-077). Written informed consent was obtained from each participant.
Analysis of inflammatory biomarkers
CRP, ESR, WBC count, neutrophil percentage, lymphocyte percentage, NLR, and PLR were measured by blood sampling at the subject’s initial visit before the start of steroid treatment. Audiologic evaluations including pure tone audiometry and speech audiometry were performed at the initial visit and after treatment with oral or intratympanic steroid, and the final hearing test was performed three months after completion of treatment. The hearing threshold of patients at each ear was calculated with a weighted four-frequency average ([500 Hz + 1000 Hz × 2 + 2000 Hz × 2 + 4000 Hz]/6).
For the analysis of prognostic factors for SSNHL, the patients were divided into two groups according to the final hearing threshold of the affected ear based on Siegel’s criteria (complete recovery, final hearing better than 25 dB; partial recovery, more than 15 dB of gain and final hearing between 25 and 45 dB; slight improvement, more than 15 dB of gain and final hearing worse than 45 dB; and no recovery, less than 15 dB of gain or final hearing worse than 75 dB), 17 which were ‘complete or partial recovery group’ and ‘slight improvement or no recovery group’. CRP, ESR, WBC count, neutrophil percentage, lymphocyte percentage, NLR, and PLR were compared between the two groups.
Statistical analysis
Demographic characteristics and the values obtained from blood sampling were analyzed between the groups using Fisher’s exact test and nonparametric Mann–Whitney U test with IBM SPSS software, version 23 (IBM, Armonk, NY, USA).
Results
There were 10 males and five females, and the mean age was 54.5 ± 18.1 years. There were five affected right ears and 10 affected left ears. The mean interval between onset and treatment was 3.0 ± 1.4 days. The initial hearing threshold at the affected ear was 57.3 ± 26.6 dB. All patients showed flat or descending audiograms on pure tone audiometry, which means that hearing thresholds were almost the same at all frequencies or higher at high frequencies than at low frequencies.
Inflammatory biomarkers as prognostic factors of sudden sensorineural hearing loss
Demographic characteristics of patients according to assessment of final hearing threshold.

ap-value < 0.05.
There were no significant differences in CRP, ESR, WBC count, and PLR between the complete or partial recovery group (0.26 ± 0.26 mg/dL, 11.6 ± 7.5 mm/hr, 7.13 ± 1.33 103/μL, and 125.12 ± 27.94, respectively) and the slight improvement or no recovery group (1.91 ± 3.82 mg/dL, 16.2 ± 16.4 mm/hr, 8.02 ± 2.73 103/μL, and 106.78 ± 46.97). Neutrophil percentage (57.8 ± 4.0%) and NLR (1.94 ± 0.34) were lower, and lymphocyte percentage (30.3 ± 3.7%) was higher in the complete or partial recovery group than in the slight improvement or no recovery group (65.4 ± 4.2%, 2.80 ± 0.74, and 24.3 ± 4.7%, respectively). There were significant differences in neutrophil percentage, lymphocyte percentage, and NLR between the two groups (p = 0.005, 0.019, and 0.013, respectively) (Figure 1). Inflammatory biomarkers in the complete or partial recovery group and the slight improvement or no recovery groups. There were no significant differences in CRP, ESR, WBC counts, and PLR between the complete or partial recovery group and the slight improvement or no recovery group. Neutrophil percentage and NLR were significantly lower and lymphocyte percentage was significantly higher in the complete or partial recovery group than in the slight improvement or no recovery group. CRP: C-reactive protein; ESR: Erythrocyte sedimentation rate; WBC: White blood cell; NLR: Neutrophil-to-lymphocyte ratio; PLR: Platelet-to-lymphocyte ratio.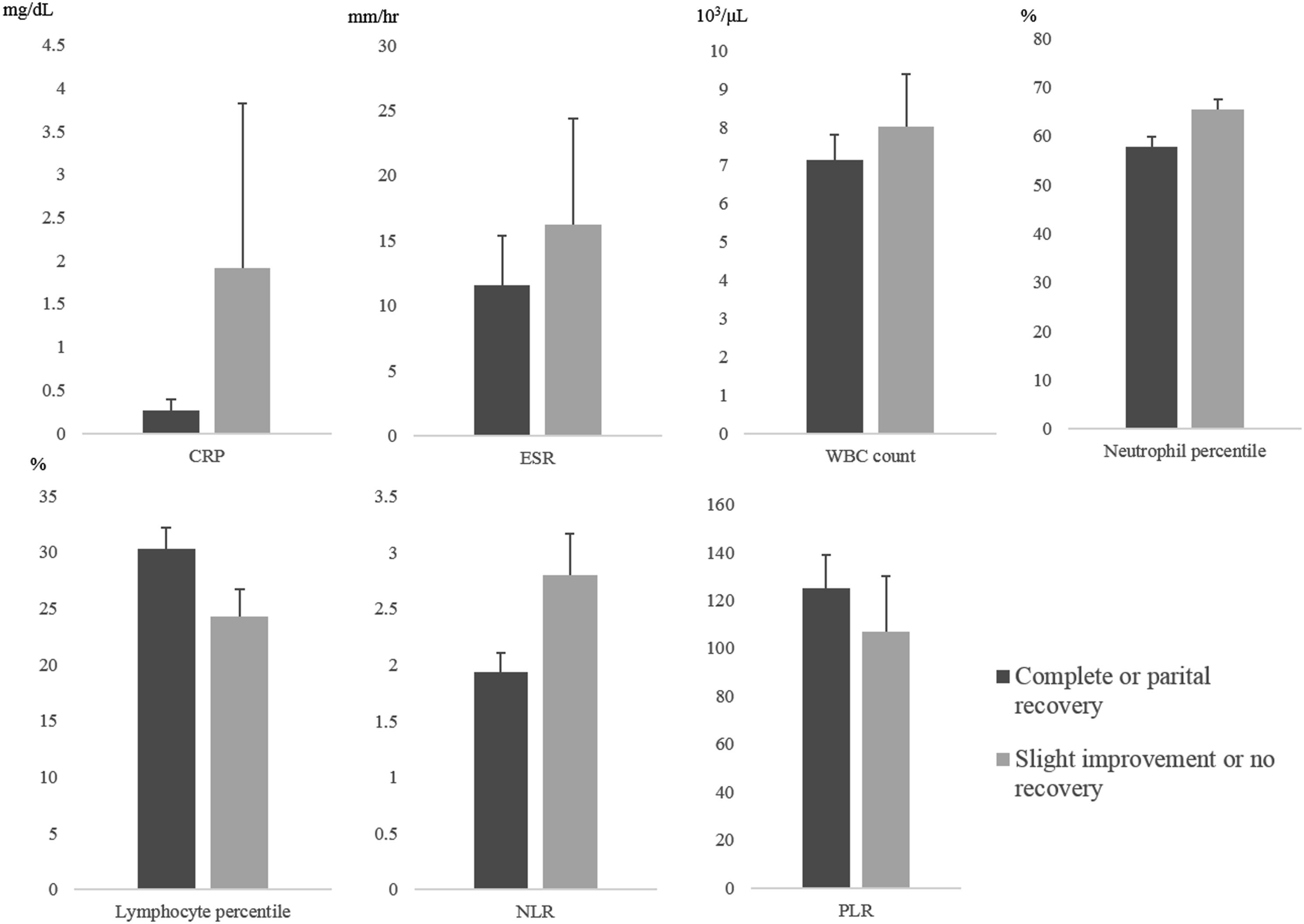
Discussion
In terms of chronic inflammation, SSNHL has been evaluated through the analysis of immunologic and hematologic inflammatory factors by blood sampling, an easy, simple, and inexpensive assessment method. Many studies have used these markers as predictive or prognostic factors of SSNHL, although there have been controversial results between several reports.
WBC count and its subtypes, such as neutrophil and lymphocyte count, have been used to evaluate the prognosis of inflammation in various diseases, such as cardiovascular disease, diabetes mellitus, and hypertension.1,9 Neutrophil count reflects inflammation, and lymphocyte count reflects general stress and nutrition. 1 In many studies, WBC count or neutrophil count were significantly higher and lymphocyte count was significantly lower in SSNHL or poorer recovery groups than in control or better recovery groups.1,6,9,12,14
NLR and PLR are parameters composed of two hematologic inflammatory values. Composite parameters more specifically reflect inflammatory status with less variation than single parameters alone. Moreover, single measures are more likely to be affected by various pathological and physiological conditions than composite parameters.7,10 Many studies have reported that NLR or PLR were significant biomarkers for the occurrence or prognosis of SSNHL, while single parameters were not significant factors.1,4,6,7,13 NLR and PLR are more reliable biomarkers of SSNHL than WBC count, neutrophil count, or lymphocyte count alone.
In the analysis of neutrophils or lymphocytes, not only the absolute count but also the percentage compared to WBC should be considered. In the study by Durmus et al. both absolute count and percentage of neutrophils and lymphocytes were analyzed, and low absolute count and a low percentage of lymphocytes were significant factors in the occurrence and prognosis of SSNHL. 6 In this study, the percentages of neutrophils and lymphocytes were significantly different between the two groups according to recovery. Because the absolute count of WBC subtypes may vary by individual, their percentages are more reliable and more applicable. NLR and PLR are ratios composed of two single parameters, so they reflect percentages of WBC subtypes and are more reliable biomarkers.
Many studies on the prognostic and predictive value of NLR and PLR have been reported. In some studies, both NLR and PLR were significant factors of SSNHL,3,5,6,13 but in other studies, either NLR or PLR was a significant factor of SSNHL.1,2,4,7 However, NLR has been more frequently reported as a significant factor than PLR. Seo et al. 3 reported that NLR and PLR in SSNHL and unrecovered groups were significantly higher than in control and recovered groups, but after adjustment in a binary logistic regression model, only NLR was a significant prognostic factor of SSNHL 3 Lee et al. 7 reported that NLR was significantly higher in an SSNHL group than in a control group, but PLR was not. 7 Qiao et al. 13 reported that 15 days after treatment, both NLR and PLR were significantly higher in an ineffective treatment group than in an effective treatment group, but before treatment, only NLR was significantly higher in the ineffective treatment group than in the effective treatment group. 13 In the meta-analysis by Chen et al. 10 NLR seemed to be a more reliable biomarker of SSNHL than PLR. 10 From the results of this study and these reports, NLR at the initial visit is thought to be a reliable and suitable prognostic factor of SSNHL.
Higher NLR suggests microarterial vascular inflammation, and higher PLR indicates pathological injury to peripheral vascular endothelium. 13 Neutrophils are associated with active nonspecific inflammation, whereas lymphocytes are associated with regulation or protection against inflammation. 10 In inflammatory diseases, neutrophils are important for cytokine production, and lymphocytes are depleted due to early apoptosis during inflammation. 7 Relatively low lymphocyte count due to neutrophil predominance results in high NLR. Inflammation is related to the occurrence of and recovery from SSNHL. Patients with severe inflammatory status who suffer SSNHL, who may show high NLR, could show a poorer response to anti-inflammatory treatment with high dose steroids than patients with mild inflammatory status, who may show low NLR. Seo et al. commented that PLR could affect the occurrence of SSNHL, but it could not be associated with the prognosis of SSNHL. They explained that hearing loss at high frequencies due to vascular ischemia did not recover well, even in the recovered group, and patients with low NLR might have a loss of low tone which was easy to recover. 3 Therefore, based on their study, high tone loss may be reflected by PLR, although PLR may also be high in patients who recover well. Both inflammation and vascular ischemia are two major factors in the occurrence of and recovery from SSNHL. NLR and PLR should be further evaluated as biomarkers of the occurrence and treatment response in SSNHL.
There are major limitations of this study. First, because subjects with acute and chronic inflammatory conditions (which could influence the inflammatory biomarkers) were not included, the sample size was small. Second, power analysis for estimation of sample size was not performed. In addition, the study period was insufficient and shorter than one year, which could act as a confounding factor considering the possibility of seasonal variations in inflammatory factors. The subsequent inflammatory factors after initial oral or adjuvant intratympanic steroid treatment were not evaluated and compared with the initial values because this study focused on the initial inflammatory factors as prognostic factors. Therefore, generalizations from the results of this study will need to be undertaken with care.
Nonetheless, this study was prospective and evaluated multiple immunologic and hematologic inflammatory factors at the same time. Although the number of patients was small, NLR and neutrophil percentage were significantly higher and lymphocyte percentage was significantly lower in the poorer recovery group. The results of this study suggest that NLR is a reasonable and appropriate biomarker for the recovery of SSNHL. The results from this study could serve as preliminary characteristics for further study, and they suggest that study on the prognosis of SSNHL should be focused on inflammation using biomarkers such as NLR.
Conclusion
NLR may be a useful prognostic inflammatory biomarker of SSNHL. Further evaluation of the association of immunologic and hematologic inflammatory factors with the prognosis of SSNHL is necessary with respect to inflammation to reveal additional significant biomarkers because inflammation is related to SSNHL.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a Seoul Medical Center Research Institute Grant (2016-077).
Ethical approval
This study was approved by the Institutional Review Board of Seoul Medical Center (IRB 2016-077). Written informed consent was obtained from each participant.
Informed consent
Written informed consent was obtained from all subjects before the study.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
