Abstract
Keywords
Introduction
Sepsis is a dysregulated host response syndrome triggered by infection that leads to progressive organ dysfunction.1,2 It is a life-threatening disease with a high fatality rate, and the 30-days mortality rate of patients with sepsis remains as high as 47% in intensive care units because of the lack of effective methods. 3 Organ failure is one of the most common complications of sepsis, and one of the first organs to fail is the lungs. 4 Moreover, about half of the patients with severe sepsis can develop acute lung injury (ALI) or its more severe form, known as acute respiratory distress syndrome (ARDS) in clinical cases.5,6 ALI is caused by indirect or direct lung injury with an increase in the permeability of the alveolar capillary membrane. 7 Epidemiological studies have revealed that the mortality rate of patients with ARDS remains as high as 40% in intensive care units in 50 countries.8–10 Although various animal experiments and clinical trials have been performed to improve the outcomes of sepsis-induced ALI, the morbidity and mortality rates remain high. 11 Thus, there is an urgent need to develop more effective therapeutic regimens to determine the mechanisms of sepsis-induced ALI.
MicroRNAs (miRNAs and miRs) are endogenous noncoding single-stranded RNAs with a length of 18–22 nucleotides that are transcribed in the nucleus and transported to the cytoplasm. 12 Commonly, miRNAs directly bind to the target genes by interacting with the 3′-UTR of target mRNAs to degrade them and suppress protein translation. Approximately 1000 miRNAs have been identified that regulate nearly 1/3 the coding genes in human cells and are involved in cell growth, proliferation, differentiation, apoptosis, metabolism and viral infection.13–16 Previous studies have revealed that multiple miRNAs act during inflammatory responses in ALI, such as miR-155, miR-146a, miR-34a, miR-454, and miR-21.17–19 MiR-21 inhibits the production of proinflammatory cytokines via the NF-κB signaling pathway.20–23 Moreover, miR‐21 increases cell viability and suppresses cellular apoptosis in non-small cell lung cancer by regulating the PI3K/Akt pathway. 24 Recent studies have confirmed that miR‐21‐5p overexpression can significantly decrease apoptosis in alveolar epithelial cell (AEC) II significantly via phosphatase and tensin homolog (PTEN).25,26 Futhermore, miR‐21‐5p decreased the severity of hyperemic ALI in rats. 27 Another study showed that miR-21 overexpression protects kidney cells from apoptosis induced by sepsis via the PTEN/PI3K/AKT signaling pathway. 28 However, it remains unclear whether miR-21 expression correlates with sepsis-induced ALI. Therefore, it is necessary to further investigate the connections and underlying mechanisms by which miR-21 regulates sepsis-induced ALI.
Materials and methods
Animals
All experimental procedures conformed to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (Publication No 85-23, revised 1996) and were approved by the Animal Ethics Committee of the Hebei General Hospital. Healthy Kunming mice (male; age: 7–8 weeks; weight: 25–30 g) were purchased from the Medical Laboratory Animal Centre of Hebei Medical University. Animals were supplied with food and water ad libitum under standardized laboratory conditions in a 12-h light-dark cycle room with a constant ambient temperature of 22±1°C and humidity of (50 ± 5)% for a week before the experiments were performed.
Sepsis acute lung injury model
The sepsis-induced ALI model was established by cecal ligation and puncture (CLP) as previously described. 29 Animals were anesthetized with isoflurane inhalation (induced at 3% and maintained at 0.5%) using a small animal ventilator. The abdomen was cut to 1.5 cm along a midline incision to expose the cecum. The cecum was ligated with a 5-0 sterile silk suture below the ileocecal valve and thoroughly punctured twice with an 18-gauge needle at the middle between the ligation and the tip of the cecum. A 1 cm drain was left across the puncture site. The cecum was placed back into the abdominal cavity. The abdominal wall incision was then sutured. The humane endpoints used to identify the animals that should be euthanized as a result of adverse effects from the experiments were a 20% loss of body weight or a body condition score <2/5 and persistent vomiting or diarrhea.
Grouping and processing
A hundred and 40 mice were randomly divided into six groups: Sham group (n = 20), ALI group (n = 20), Pre-miR-21+ALI group (n = 20), Anti-miR-21+ALI group (n = 20), Pre-miR-21+ALI+PTEN inhibition group (Pre-miR-21+ALI+PI group, n = 20), Anti-miR-21+ALI+PTEN inhibition group (Anti-miR-21+ALI+PI group, n = 20). The Sham group was exposed to the cecum only, but was neither ligated nor punctured. The ALI group were induced by CLP. Pre-miR-21+ALI group and Anti-miR-21+ALI groups were treated with miR-21 precursor or miR-21 inhibitor in the caudal vein 4 days before CLP. Pre-miR-21+ALI+PI and Anti-miR-21+ALI+PI groups were treated with PTEN inhibition in the caudal vein 30 min before CLP. The remaining 20 mice were intravenously administered miR-21 precursor, miR-21 precursor control, miR-21 inhibition, and miR-21 inhibition control to examine the expression of miR-21.
RNA extraction and quantitative real-time polymerase chain reaction
Total RNA was isolated from the left upper lungs after CLP surgery at 6 h, 12 h, 24 h, and 48 h with Trizol (Invitrogen) according to the manufacturer’s instructions as well as from the sham group at the same time. The total RNA concentration and purity were detected at a wavelength of 260 nm using a NanoDrop 2000c spectrophotometer (Thermo Fisher). RNA was reverse transcribed into cDNA using poly A polymerase primers at 37°C for 60 min and 85°C for 5 min with a reverse transcription kit (GeneCopoeia). Quantitative real-time polymerase chain reaction (qRT-PCR) was performed according to the temperature profile (95°C for 10 min, followed by 40 cycles of 95°C for 10 s, 60°C for 27 s, and 72°C for 27 s) using a qRT-PCR kit and probes (GeneCopoeia). U6 expression was used as an internal normal reference. The primers used were provided by GeneCopoeia as follows: U6 forward, Cat# RmiRQP9003; miRNA 21 forward, Cat# RmiRQP1208; and reverse, Cat#Po1011 A. The Ct values of the target genes were normalized by subtracting of the U6 Ct values, thereby obtaining ΔCt values. The relative expression level was calculated using the following equation: relative gene expression = 2−(ΔCtsample-ΔCtcontrol). All qRT-PCR reactions were performed on an ABI7500 Real-Time PCR System (Applied Biosystems).
Transfection
Mice were transfected by injecting into the caudal vein with an adenovirus carrying the miR-21 precursor, miR-21 precursor control, miR-21 inhibitor, or miR-21 inhibitor control (0.2 mL, 2×108pfu, Invitrogen). Transfection efficiency was measured at 24 h after transfection. CLP surgery was conducted at 4 days after transfection. PTEN inhibitor (VO-OHpic, 10 μg/kg, Sigma) was injected into the caudal vein for 30 min.
Detection of inflammatory cytokines and oxidative stress indicators
After the mice were anesthetized with isoflurane, the right upper lungs were removed and homogenized on ice for 24 h in each group. Lung tissue supernatants were obtained by centrifugation at 10,000r/min for 15 min and stored at −80°C until analysis. The levels of lung tissue tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), reactive oxygen species (ROS), superoxide dismutase (SOD), malondialdehyde (MDA) and inducible nitric oxide synthase (iNOS) were detected using ELISA kits, according to the manufacturer’s protocols (Lianke, Hangzhou, China).
Evaluation of lung tissue cell apoptosis
After anesthesia with isoflurane, the right middle lungs were dissected to prepare a monoplast suspension at 24 h in each group. Annexin V (5 μL, B&D, USA) and PI (5 μL, B&D, USA) were added to the monoplast suspension and resuspended in 500 μL buffer. The mixtures were gently vortexed and incubated for 15 min at room temperature in the dark, and then analyzed by flow cytometry.
Assessment of oxygenation index and lung wet/dry weight ratio
Blood samples (about 2 mL) were collected from the carotid artery at 24 h after anesthesia with isoflurane for blood gas analysis, and the OI (oxygen partial pressure/fraction of inspired oxygen) was calculated. The right upper lungs were dissected, weighed, and dried at 70°C for 48 h. The lung wet/dry weight ratio was calculated as follows: lung water content = [(wet weight-dry weight)/wet weight] x100%.
Histological examination
The right lower lung was collected for histological analyses. Lung specimens were fixed in 4% paraformaldehyde, dehydrated in different concentrations of ethanol, and embeded in paraffin. Five-micron-thick sections were cut using a microtome and stained with hematoxylin and eosin for 3 min at room temperature. The tissue sections were observed under a light microscope (Olympus, Japan).
Experienced pathologists randomly selected 10 high-power visual fields under a light microscope to accsess the lung tissue injury. The scoring method was based on relevant literature standards. 30 The main scoring rules were as follows: under the microscope, five pathological injury indices were observed, including edema, hyperemia, alveolar hemorrhage or debris, neutrophil infiltration, and cell proliferation. Each index was divided into four grades: no injury, mild, moderate, and severe. The corresponding scores were 0, 1, 2, and 3, and the total score is 0–15. The higher the evaluation score, the more severe is the lung tissue injury.
Western blot analysis
Proteins isolated from the left lower lung of each group were analyzed by western blotting according to standard protocols. Equal amounts of protein were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis. Standard western blot analysis was conducted using the PTEN antibody (1:600 dilution, Epitomics), and GAPDH antibody (1:600 dilution, Epitomics) was used as a standard control.
Statistical analysis
The sample size calculation was based on the G* POWER3.1.9.7 software (bilateral α = 0.05, power of 80%), and the total sample size was calculated as N = 96. Considering a shedding rate of 10–20%, the total sample size was estimated to be 120 mice.
SPSS 19.0 software was used for the statistical analysis. All data are expressed as mean ± standard error of the mean. One-way analysis of variance was performed to compare differences between groups. Comparing the data of the two conditions was used by the post-hoc test. p < 0.05 was considered statistically significant.
Results
Cecal ligation and puncture-administration induces lung injury in mice
Compared with the sham group, CLP-treated mice had higher levels of inflammatory factors (Figure 1(a) and (b)), lung wet/dry weight ratio (Figure 1(c)), and low OI (Figure 1(d)). Additionally, the lung pathological damage were more severe in the ALI group (Figure 1(e)). Therefore, an ALI model was successfully established in this study. Inflammatory factors, lung wet/dry weight ratio, OI and lung pathological damage after CLP at every terminal. The levels of TNF-α (a) and IL-6 (b) were elevated in lung homogenates at 6 h, 12 h, 24 h, and 48 h post treatment by ELISA kits (*p < 0.05 vs. Shams). (c) The lung wet/dry weight ratio was increased at 12 h, 24 h, and 48 h after CLP (*p < 0.05 vs. Shams). (d) OI was reduced at 6 h, 12 h, 24 h, and 48 h after CLP (*p < 0.05 vs. Shams). (e) The lung pathological damage were more severe in the ALI group at 6 h, 12 h, 24 h, and 48 h.
MiR-21 is upregulated in cecal ligation and puncture-administered lungs
We examined the miR-21 expression in CLP-administered lungs at 6 h, 12 h, 24 h, and 48 h by qRT-PCR. MiR-21 expression was significantly higher in CLP mice than in sham mice (1.46-fold, p < 0.05 at 6 h; 1.92-fold, p < 0.05 at 12 h; 2.38-fold, p < 0.05 at 24 h; and 1.98-fold, p < 0.05 at 48 h) (Figure 2). MiR expression profile in mouse lungs after CLP surgery. MiR-21 expression was dramatically upregulated, which increased to 1.46-fold, 1.92-fold, 2.38-fold, and 1.98-fold in mouse lungs at 6 h, 12 h, 24 h, and 48 h post-CLP respectively (*p <0.05 vs. Shams).
Regulatory effect of miR-21 precursor or inhibitor in mouse lungs
To investigate the regulatory effect of miR-21 precursor or inhibitor on mouse lungs, we found that miR-21 precursor increased miR-21 expression by 1.88-fold and miR-21 inhibitor decreased by 67.5% in lung tissues compared to ALI mice after transfection with miR-21 precursor or inhibitor in CLP mice at 24 h. Moreover, there was no effect on miR-21 expression by transfecting with miR-21 precursor blank control or inhibitor blank control in the caudal vein. In addition, none of the transfections had an effect on healthy lung function in mice (Figure 3). MiR expression profile in mouse lungs after transfection. MiR-21 expression with injected miR-21 precursor increased by 1.88-fold and with miR-21 inhibitor decreased by 67.5% in mouse lungs compared with the ALI group at 24 h (*p < 0.05 vs. ALIs).
MiR-21 suppresses the inflammatory response in sepsis-induced ALI mice
First, we observed that the production of inflammatory cytokines, including TNF-α and IL-6, in the ALI group was significantly increased at 24 h post-CLP surgery compared that in the sham group. To confirm the underlying role of miR‐21 in the regulation of inflammation factors in ALI, we transfected with miR-21 mimic or miR-21 inhibitor before CLP surgery. TNF-α (Figure 4(a)) and IL-6 (Figure 4(b)) levels decreased in the Pre-miR-21+ALI group and increased in the Anti-miR-21+ALI group compared with those in the ALI group. Upregulation of miR-21 attenuates sepsis-induced inflammation. The levels of TNF-α (a) and IL-6 (b) were measured in lung homogenates at 24 h post treatment by ELISA kits (*p < 0.05 vs. Shams; #p < 0.05 vs. ALIs).
MiR-21 reduces oxidative stress response
The production of ROS (Figure 5(a)), SOD (Figure 5(b)), MDA (Figure 5(c)), and iNOS (Figure 5(d)) was notably increased at 24 h in the ALI group compared with the Sham group. Moreover, we found that the oxidative stress indicators were reduced in the Pre-miR-21+ALI group and were adversely raised in the Anti-miR-21+ALI group compared with those in the ALI group. Upregulation of miR-21 reduces oxidative stress response. The levels of ROS (a), SOD (b), MDA (c), and iNOS (d) were measured in lung homogenates at 24 h post treatment by ELISA kits (*p < 0.05 vs. Shams; #p < 0.05 vs. ALIs).
MiR-21 alleviates the lung injury
As shown in Figure 6(a) and (b), the OI decreased significantly, while the lung wet/dry weight ratio increased when mice were subjected to CLP surgery at 24 h. Administration of miR‐21 reversed the changes induced by CLP, as evidenced by the increased OI and decreased lung wet/dry weight ratio. On the contrary, we concluded that the OI decreased and the lung wet/dry weight ratio increased in the Anti-miR-21+ALI group compared with those in the ALI group. Upregulation of miR-21 attenuates sepsis-induced lung injury. (a) The OI was measured by arterial blood gas (*p < 0.05 vs. Shams; #p < 0.05 vs. ALIs). (b) The lung wet/dry weight ratio was obtained by weighing (*p < 0.05 vs. Shams; #p < 0.05 vs. ALIs). (c) Lung tissues obtained from the Sham, ALI, Pre-miR-21+ALI and Anti-miR+ALI mice with hematoxylin and eosin staining. Scale bar, 100 μm. (D) The quantified lung injury score in Sham, ALI, Pre-miR-21+ALI and Anti-miR+ALI group (*p < 0.05 vs. Shams; #p < 0.05 vs. ALIs).
The histological results of the lung tissue sections in the sham group at 24 h were revealed relatively normal. However, we observed that there is destruction in the alveolar wall, alveolar hyperemia, and interstitial inflammatory cells in mice subjected to CLP surgery at 24 h. We also found that lung injury was attenuated in the Pre-miR-21+ALI group but aggravated in the Anti-miR-21+ALI group (Figure 6(c)). The lung injury score increased at 24 h in the ALI group compared with that in the Sham group. The lung injury score was decreased in the Pre-miR-21+ALI group and was adversely increased in the Anti-miR-21+ALI group compared with that in the ALI group (Figure 6(d)).
MiR-21 restrains lung cell apoptosis
As shown in Figure 7(a) and (b), lung cell apoptosis was detected by flow cytometry in lung tissues at 24 h after CLP surgery. CLP treatment induced significant cell apoptosis, which was reversed by the administration of miR-21 precursor. However, pretreatment of mice with miR-21 inhibitor resulted in a firm increase in cell apoptosis, followed by CLP. Upregulation of miR-21 attenuates lung cell apoptosis. (a) The lung cell apoptosis in Sham, ALI, Pre-miR-21+ALI and Anti-miR+ALI group. (b) The profile of lung cell apoptosis was measured at 24 h post treatment by flow cytometry (*p < 0.05 vs. Shams; #p < 0.05 vs. ALIs).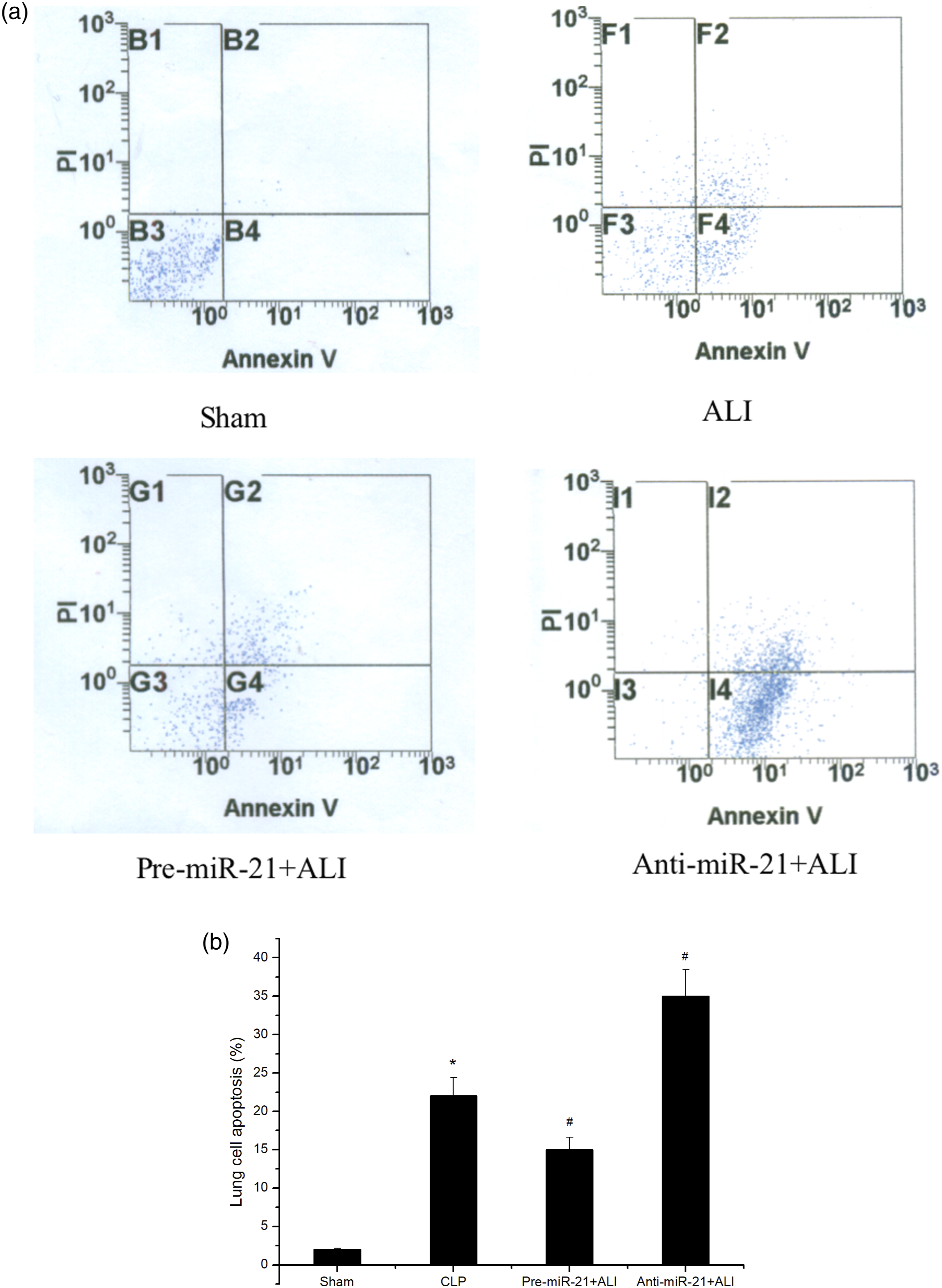
MiR-21 decreases PTEN protein expression in the lung
PTEN protein expression levels were determined using western blotting. We observed that PTEN protein expression was lower in the ALI group than in the Sham group. We also found that the PTEN protein expression levels were decreased in the Pre-miR-21+ALI group but increased in the Anti-miR-21+ALI group compared with the ALI group (Figure 8(a) and (b)). Upregulation of miR-21 decreases PTEN protein expression in the lung. (a) The protein level of PTEN was detected by western blot analysis 24 h post treatment. (b) Protein expression was quantified by densitometric analysis and normalized to GAPDH (*p < 0.05 vs. Shams; #p < 0.05 vs. ALIs).
MiR-21 protects against sepsis induced acute lung injury by targeting PTEN
As shown in Figure 9(a)–(g), TNF-α, IL-6, oxidative stress indices, and lung tissue apoptosis were decreased in the Pre-miR-21+ALI+PI group compared with those in the Pre-miR-21+ALI group. However, the levels of inflammatory cytokines, oxidative stress indices and lung tissue apoptosis in the Anti-miR-21+ALI+PI group were not significantly different from those in the Pre-miR-21+ALI+PI group. The effect of miR-21 on anti-inflammatory, anti-oxidative stress and anti-apoptosis enhanced after PTEN inhibition. The effect of miR-21 on reducing the levels of TNF-α (a), IL-6 (b), ROS (c), SOD (d), MDA (e), iNOS (f), and lung tissue apoptosis (g) enhanced after PTEN inhibition (*p < 0.05 vs. Shams; #p < 0.05 vs. ALIs; &p < 0.05 vs. Pre-miR-21+ALIs).
Discussion
Sepsis is a systemic inflammatory response syndrome caused by infection, which leads to multiple organ failure and even death. ALI is a frequent complication of sepsis and is characterized by an uncontrolled inflammatory process, excessive oxidative stress, cell apoptosis, pulmonary edema, and lung tissue dysfunction.31,32 Recent studies have confirmed that decreasing excessive inflammation and oxidative stress, and inhibition of cell apoptosis may ameliorate the histopathologic damage and even alleviate ALI and ARDS in animal experiments. 33 Research has shown that miR-21 has a protective effect on type II alveolar epithelial cell apoptosis induced by H2O2 in vitro and ameliorates hyperoxic ALI in vivo. 34 In this study, we aimed to explore whether miR-21 expression correlates with sepsis-induced ALI.
In this study, we developed a sepsis model using classical CLP surgery. We observed that CLP mice had higher levels of inflammatory cytokines and presented pathological changes in the destructive alveolar wall, alveolar hyperemia, and infiltrated interstitial inflammatory cells. The sepsis-induced ALI model induced by CLP surgery was successful. We examined whether the miR-21 expression was upregulated in the lung tissue of CLP mice. We found that miR-21 expression reached the peak after CLP at 24 h. Therefore, we chose this time point to study each index. Moreover, transfection of miR-21 precursor or inhibition successfully induced miR-21 overexpression or downregulation.
First, miR-21 overexpression decreased the release of proinflammatory cytokines, which increased when miR-21 was downregulated. TNF-α and IL-6, which are critical inflammatory cytokines in sepsis, trigger uncontrolled inflammation by targeting the nuclear transcription factor (NF-κB). Experimental observations have also suggested that miR‐21 blocks NF‐κB activation and impedes the inflammatory response by inhibiting the proinflammatory mediator PDCD4. 35 Studies have shown that inhibition of these cytokines with neutralizing antibodies is beneficial for the treatment of sepsis. 36 Thus, the decrease in lung tissue TNF-α and IL-6 levels may be one of the protective mechanisms of sepsis-induced ALI.
In addition, upregulated miR-21 expression inhibited lung tissue cell apoptosis. Apoptosis of alveolar epithelial and vascular endothelial cells has been shown to induce increased vascular wall permeability and decreased alveolar diffusion. Studies have shown miR‐21 overexpression inhibited the apoptosis of nasopharyngeal carcinoma cells by targeting the 3′ untranslated region of PTEN mRNA. 37 Moreover, miR‐21‐5p has been confirmed to inhibit hypoxia/reoxygenation-induced lung microvascular endothelial cell apoptosis in vitro and decrease lung ischemia/reperfusion injury in vivo. 38 Additionally, a previous study indicated that the PTEN or the PI3K inhibitors efficiently attenuated the anti-apoptotic effect of miR‐21/PTEN/AKT signaling. 39 Interestingly, we observed that the expression of miR-21 inhibited lung cell apoptosis in mice during sepsis. However, lung tissue apoptosis in anti-miR-21-treated CLP mice was exacerbated in our study. Therefore, we inferred that miR‐21 ameliorated sepsis-induced ALI via PTEN/AKT-associated anti-apoptotic pathways.
Another protective mechanism of miR-21 during sepsis may be associated with the suppression of the oxidative stress response. Research has demonstrated that miR-214 exerts a protective effect against H2O2-induced cardiac myocyte injury. 40 Additionally, it has been confirmed that miR-21 is sensitive to H2O2 stimulation in cardiac myocytes and protect against H2O2-induced myocardial injury. 41 In our study, upregulation of miR-21 expression reduced the oxidative stress response in sepsis-induced ALI. Thus, we speculated that miR-21 alleviated mitochondrial dysfunction and energy deficits in lung tissue cells by reducing the production of oxidative stress and plays a protective role in sepsis induced ALI.
Finally, we confirmed that PTEN expression was decreased in CLP mice and was regulated by miR-21. Overexpression of miR-21 decreased PTEN expression, whereas miR-21 downexpression increased PTEN expression. These results suggested that PTEN is a potential target of miR-21. In cancer research, miR-21-5p has been shown to promote cell growth and mobility through PTEN/AKT. 42 Previous studies have demonstrated that miR-21-5p decreases ACE II apoptosis to protect lung tissue through the PTEN/AKT pathway. 27 To confirm the role of PTEN in the protective effects of miR-21, we used a PTEN inhibitor to reduce PTEN protein expression. We found that the effect of miR-21 on anti-inflammatory, antioxidative, and anti-apoptosis enhanced after PTEN inhibition, resulting in a cumulative effect. However, there was no significant difference in the effect of anti-inflammatory, anti-oxidative stress, and anti-apoptosis on the upregulation or downregulation of miR-21. Therefore, we speculated that miR-21 reduces lung injury in sepsis by inhibiting PTEN expression.
Conclusion
In conclusion, our study revealed that miR-21 alleviates lung injury by suppressing the inflammatory response, reducing the oxidative stress response, and restraining lung cell apoptosis in sepsis. These results may be related to the inhibition of miR-21 expression by PTEN.
Limitations
First, the samples were harvested 24 h after CLP during the acute phase of septic disease because the mortality increased with time and the peak time of miR-21 expression. This experimental design has limited relevance to the course of human sepsis. However, this is helpful in confirming that miR-21 is protective against sepsis-induced lung injury. In addition, the downstream pathway of PTEN has not been studied and will be investigated in subsequent studies.
Footnotes
Author’s note
The study was investigated that the role of microRNA-21 in inflammation, oxidative stress, and apoptosis in mice with acute lung injury, and the possible mechanism of microRNA-21 in acute lung injury.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Medical Science Research Key Project of Hebei Province (20180192).
Ethics approval
Ethical approval for this study was obtained from Animal Ethics Committee of the Hebei General Hospital (Approval Number 2022070).
Data availability
The data used to support the findings of the study are available from the corresponding author upon request.
Animal welfare
For all studies involving the use of animals, we have received permission from the Institutional Animal Care and Use Committee. All experimental procedures conformed to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (Publication No 85-23, revised 1996).
