Abstract
The anti-inflammatory roles of I7456 (1-isothiocyanato-7-(methylsulfonyl) heptane or 7-methylsulfonylheptyl isothiocyanate), a plant-derived and sulfur-containing isothiocyanate, were investigated. When macrophage cells (RAW 264.7) were challenged with lipopolysaccharide (LPS), nitric oxide (NO) increased. However, NO was remarkably reduced upon I7456 treatment. I7456 strongly reduced the expression of IL-6, IL-10, IL-1β, and iNOS. Interestingly, heme oxygenase-1 (HO-1) was strongly induced without LPS challenge. LPS-induced NFκ-B (nuclear factor kappa-light-chain-enhancer of activated B cells) translocation into the nucleus was inhibited by I7456 in a dose-dependent manner. I7456 markedly reduced the phosphorylation level of IκB, and NFκ-B remained inactivated. I7456 could play important roles in anti-inflammatory responses and have implications for anticancer treatments.
Introduction
Inflammatory responses as a defense mechanism against injuries and pathogens occur in macrophages, producing cytokines, chemokines, and other inflammatory factors. 1 The inflammatory response is beneficial, but an excessive response or a long period of exposure could lead to acute or chronic inflammatory diseases, potentially leading to cancer.2,3 Reducing excessive or uncontrolled inflammation is, thus, thought to be an effective strategy for curing diseases. Lipopolysaccharide (LPS), a component of the Gram-negative bacterial cell wall, triggers inflammation in macrophages via molecular pattern recognition, either pathogen-associated molecular patterns (PAMPs) or danger-associated molecular patterns (DAMPs).
Immune responses are intensified with nitric oxide (NO), cytokines, and prostaglandin E2 (PGE2), which is produced by cyclooxygenase-2 (COX-2). 4 In response to LPS or cytokines, such as IL-1β, IL-6, IL-10, and tumor necrosis factor-α (TNF-α), NO, a molecule that can diffuse through the vascular systems, is rapidly available via inducible NO synthase (iNOS).4,5 NO has many beneficial functions for hosts, but excess amounts result in inflammation. NFκ-B is an important transcription factor for pro-inflammatory mediators and iNOS in activated macrophages. It can be activated by various stimuli associated with stress and injury, including LPS. It can bind enhancer elements of the immunoglobulin κ light-chain gene and regulate many downstream genes involved in inflammation, such as iNOS, IL-1β, and TNF-α. 6 It remains inactive in the cytosol because of interactions with an inhibitor, IκB. Upon activation by extracellular signals, such as LPS or cytokines, IκB is phosphorylated by IκB kinase and it becomes vulnerable to ubiquitin-mediated proteasome degradation, which releases NFκ-B to enter the nucleus. In most known cancer types, NFκ-B is constitutive and is part of malignant cell proliferation. 7
Phytochemicals have been known to be effective in preventing oxidation, allergic reaction, inflammation, viral infection, and cancer.8,9 In particular, isothiocyanates are recognized as counteracting agents against neurodegenerative diseases. I7456 is one of the defense chemical compounds of plants against herbivores and pathogens and is found at high levels in the Brassicaceae family.5,10 It is widely known that it has antimicrobial and anticancer activities. Our study investigated the anti-inflammatory effects of I7456 on LPS-challenged macrophages and its possible implications for cancer therapy.
Materials and methods
Chemicals
I7456 was purchased from LKT Laboratories, United States. All other chemicals used in this study were of molecular biology grade.
Tetrazolium salt-based dehydrogenase assay (XTT assay)
Murine RAW 264.7 macrophages were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and 1% penicillin–streptomycin in a humidified 5% CO2 atmosphere at 37°C. A total of 5 × 104 cells were seeded in a 96-well microplate and incubated with different concentrations of I7456 in DMEM for 24 h. Cell viability was measured using an EZ-cytox cell viability assay kit according to the manufacturer’s instructions
Determination of NO production
The NO concentration in culture media was measured with Griess reagent. A total of 1 × 105 RAW264.7 cells in a 48-well plate were incubated with different concentrations of I7456 (1~5 μg/mL) for 1 h and stimulated with 1 μg/mL of LPS at 37°C for 24 h. The dimethyl sulphonte (DMSO) concentration in each well was 0.0123%. Supernatants (100 μL) were mixed with 100 μL of the Griess reagent and incubated at room temperature for 10 min. Absorbance of the mixture was determined at 540 nm using a microplate reader. All measurements were performed in triplicate.
RT-PCR
Total RNA was extracted using TRIzol reagent (Invitrogen, USA) following the manufacturer’s instructions, and measured using a Nanodrop reader. Total RNA (3 μg) was converted to first-strand complementary DNA (cDNA) by using MMLV reverse transcriptase (Invitrogen). cDNA was amplified with gene-specific primers using Go taq polymerase (Promega, USA). The primer sequences were as follows:
TNF-α F′-GGGACAGTGACCTGGACTGT, R′-GCAGAGGTTCAGTGATGTAG;
IL-6 F′-GAGACTTCCATCCAGTTGCC, R′-TACTCCAGAAGACCAGAGG;
IL-1β F′-AGCAGCACATCAACAAGAGC, R′-GGTGTGCCGTCTTTCATTAC;
Cox-2 F′-GCCTTCTCCAACCTCTCCTA, R′-ACCTCTCCACCAATGACCTG;
IL-10 F′-AGCCGGGAAGACAATAACTG, R′-TCTTCAGCTTCTCACCCAGG,
iNOS F′-GCTGTGCTCCATAGTTTCCA, R′-CTTGTCACCACCAGCAGTAG;
HO-1 F′-GACCAGAGC CCCTCACGG, R′-CCAGGCTCTGCTGCAGGA;
β-actin F′-ATCCTGCGTCTGGACCTGGCT, R′-CTGATCCACATCTGCTGGAAG.
β-Actin was used as an internal control.
Western blot analysis
RAW264.7 cells were plated at a density of 5 × 105 cells in six-well plates, pre-treated with various concentrations of I7456 (1~10 μg/mL) for 1 h, and stimulated with 1 μg/mL of LPS in DMEM at 37°C for 24 h. The cells were subsequently washed with phosphate-buffered saline (PBS), collected, suspended in lysis buffer (150 mM NaCl, 10 mM Tris (pH 7.5), 5 mM ethylenediaminetetraacetic acid (EDTA), 1% Triton X-100) containing protease inhibitors (1 μg/mL leupeptin and 100 μg/mL phenylmethane sulfonyl fluoride (PMSF)), and centrifuged at 12,000g for 20 min at 4°C to yield cell lysates. The protein level in each sample was measured using a bicinchoninic acid (BCA) protein assay kit (Thermo Scientific, Rockford, IL, USA). Equal amounts of protein obtained from the cells were subjected to electrophoresis on 10% sodium dodecyl sulfate (SDS)-polyacrylamide gels and were transferred onto nitrocellulose membrane. The membrane was blocked with 5% skimmed milk in Tris/HCl, pH 7.4, containing 159 mM NaCl and 1% Tween 20 (TBS-T) and incubated for 1 h at room temperature. The membrane was then incubated at room temperature with a 1:1000 dilution of polyclonal anti-iNOS, anti-COX-2, monoclonal NFκ-B, IκB-α, or β-actin antibody for 1 h. After washing three times with TBS-T, the membrane was incubated with a 1:2000 dilution of horseradish-conjugated anti-mouse and anti-rabbit IgG secondary antibodies for 1 h at room temperature. The membrane was again washed three times with TBS-T and developed with ECLTM detection reagents (Bio-Rad, USA). The intensity of each band was determined using the Multigauge V3.2 program (Fuji, Japan).
Confocal microscopy
RAW264.7 cells were grown on glass cover slips (18 mm in diameter; Marienfeld Laboratory Glassware, Germany). After 1 h of pretreatment with I7456 (10 μg/mL), they were incubated with LPS (1 μg/mL; Molecular Probes Inc., USA). Once fixed, they were stained with 4′-6-diamidino-2-phenylindole (DAPI; blue) and incubated with anti-NFκ-B and fluorescein isothiocyanate (FITC)-conjugated anti-rabbit IgG (green) antibodies. The slides were examined with an LSM710 confocal laser scanning microscope (Carl Zeiss, Germany) equipped with 200× objectives. Images were obtained using the ZEN2011 software (Carl Zeiss, Germany).
Statistical analysis
All data were presented with the means ± standard error of means (SEM) of at least three independent experiments. An analysis of variance (ANOVA) with Dunnett’s test was used for multiple comparisons.
Results
I7456 belongs to the isothiocyanates that have sulfur compounds that include –N = C = S as a functional group (Figure 1). These isothiocyanates are abundantly found in the Brassica family and act as defense molecules against herbivores and pathogens. 5 Many studies have demonstrated that they have anticancer, antioxidant, superoxide scavenger, and anti-inflammatory properties. We aimed to investigate anti-inflammatory effects of I7456 in this study.

Chemical structure of I7456.
I7456 toxicity was monitored to find the optimal concentrations for our studies. I7456 showed a better growth rate at 3 μM than did the control, and a relatively low toxicity was observed throughout the concentration range that we tested, up to 10 μM, where the control had a slightly higher survival rate (Figure 2). No side effects of I7456 were observed, so further experiments were pursued at this concentration range (1~10 μM).
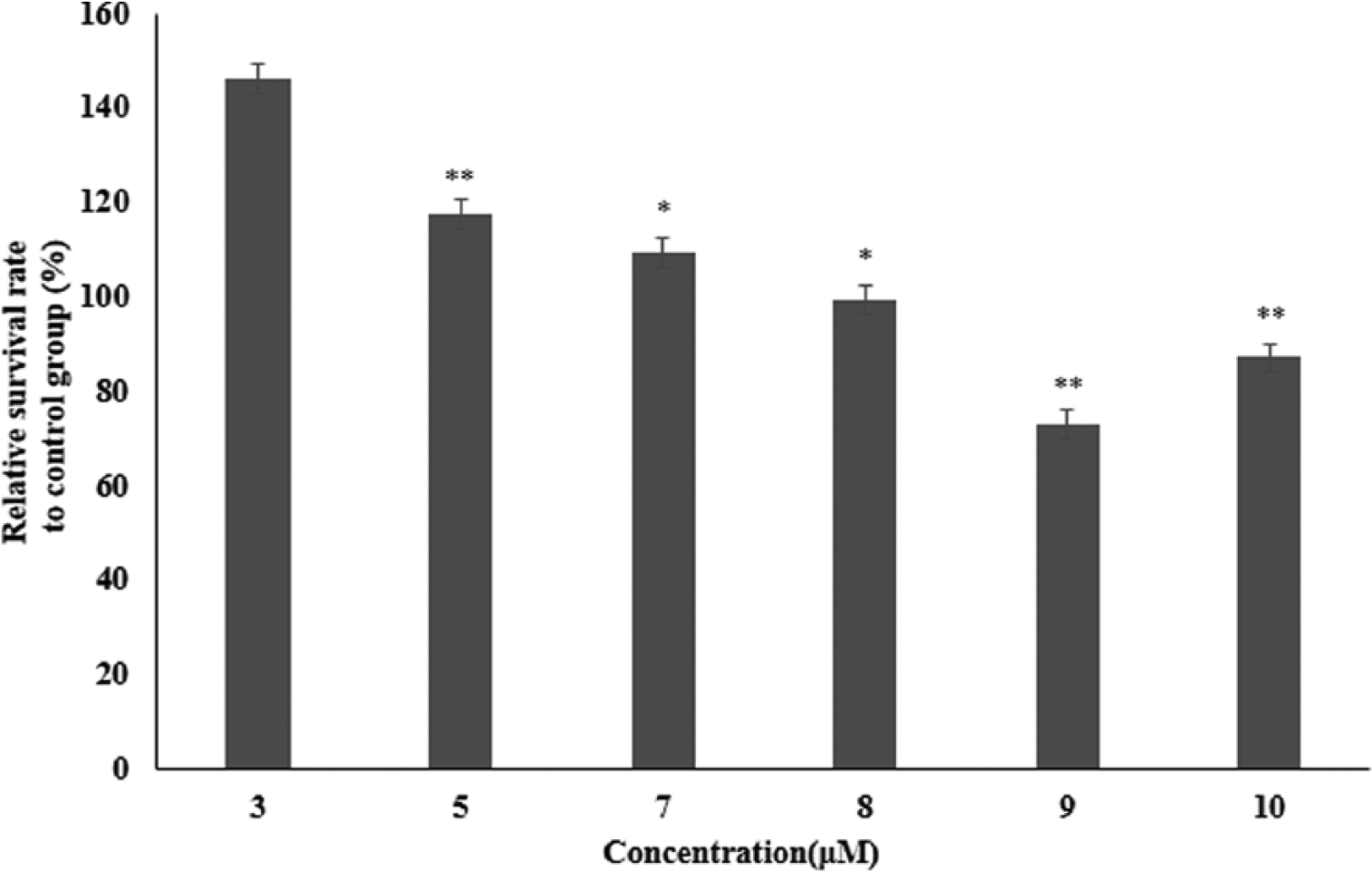
Survival rates of macrophages treated with I7456. Macrophage RAW264.7 cells were aliquoted into 96-well plates and cultured for 24 h. I7456 was applied for another 24 h. An XTT assay was used to measure cell toxicity and growth rates. Values are expressed as the mean ± SEM of determinations made in triplicate experiments. **
As inflammation is known to have a close connection with cancer, 5 we attempted to determine the anti-inflammatory effects of I7456. When macrophages were challenged with a well-known inflammatory factor, LPS, NO was increased (Figure 3). However, NO production was remarkably reduced with I7456 application, even at the lowest concentration (1 μM), and NO production continued to decrease to half at 2 μM I7456. The lowest NO was observed at 5 μM, where the NO level was almost the same as the control.

Effects of I7456 on NO production of LPS-challenged RAW264.7. To investigate anti-inflammatory effects of I7456, NO production was measured. RAW 264.7 cells, treated with I7456, were incubated for 1 h and stimulated with LPS (1 μg/mL) for 24 h. The amount of NO released into the culture medium was measured by a Griess reagent assay. Values are expressed as the mean ± SEM of determinations made in triplicate experiments. *
The roles of inflammatory cytokines are known to be essential for inflammation-linked cancer development, so we investigated how the expression of inflammatory cytokines, such as IL-6, IL-10, IL-1β, COX-2, iNOS, and TNF-α, was affected by I7456. LPS treatments boosted the expression of inflammatory cytokines, but I7456 only had no effect without LPS challenge, which indicated that the triggering of inflammation by LPS was a prerequisite. I7456 strongly affected the expression of IL-6, IL-10, IL-1β, and iNOS in a dose-dependent manner, but COX-2 and TNF-α expressions were relatively less affected (Figure 4).

Expression of pro-inflammatory cytokines by I7456-treated RAW264.7 cells. Cells were pre-treated with various concentrations of I7456 (1~5 μg/mL) for 1 h before LPS (1 μg/mL) stimulation for 6 h. RNA was isolated using TRIzol (ThermoFisher, USA), and cDNA was synthesized. cDNA was amplified using primers specific for IL-6, IL-10, IL-1β, COX-2, iNOS, TNF-α, and β-actin. I7456 reduced LPS-induced mRNA accumulation of IL-6, IL-10, IL-1β, COX-2, iNOS, and TNF-α in a dose-dependent manner.
Since HO-1 has emerged as an important regulator of acute and chronic inflammation, its expression was examined with I7456 application (Figure 5). Interestingly, HO-1 was strongly induced without LPS challenge, which suggests that HO-1 could act as a signaling molecule for anti-inflammatory cytokines. Higher I7456 drove more HO-1 expression. Regardless of LPS challenge, HO-1 expression seemed to be directly influenced by I7456.

Expression of HO-1. The same procedure as in Figure 3 was used for RNA preparation. Macrophages were treated with I7456 for 1 h, and the LPS challenge was initiated. Semi-quantitative RT-PCR was performed to quantify HO-1 expression. The number of cycles and synthesized cDNA amount were optimized for quantification.
The expression level of inflammatory cytokines and HO-1 was quantified (Figure 6). HO-1 expression was slightly decreased after LPS challenge and remained at the same level up to 1 μM of I7456, but it increased significantly over 1 μM of I7456. Its highest expression was observed with I7456 treatment (5 μM). Sharp decreases due to I7456 treatment were found in IL-6 and IL-1β (11- and 20-fold, respectively) compared to LPS treatment, with modest decreases in IL-10, iNOS, COX-2, and TNF-α. Although I7456 alone had no effects on any expression changes of cytokines, it had an effective anti-inflammatory role upon LPS challenge. The protein level of iNOS, which is responsible for NO production, was significantly reduced by I7456, and a massive increase was observed only after LPS treatment (Figure 7). COX-2 showed a moderate reduction due to I7456 treatment.

Quantification of the expression levels of HO-1 and inflammatory cytokines. The expression levels of HO-1 and inflammatory cytokines were estimated by quantifying band intensities. Total RNAs were prepared in the same way as in Figures 3 and 4. Values are expressed as the mean ± SEM of determinations made in triplicate experiments. *
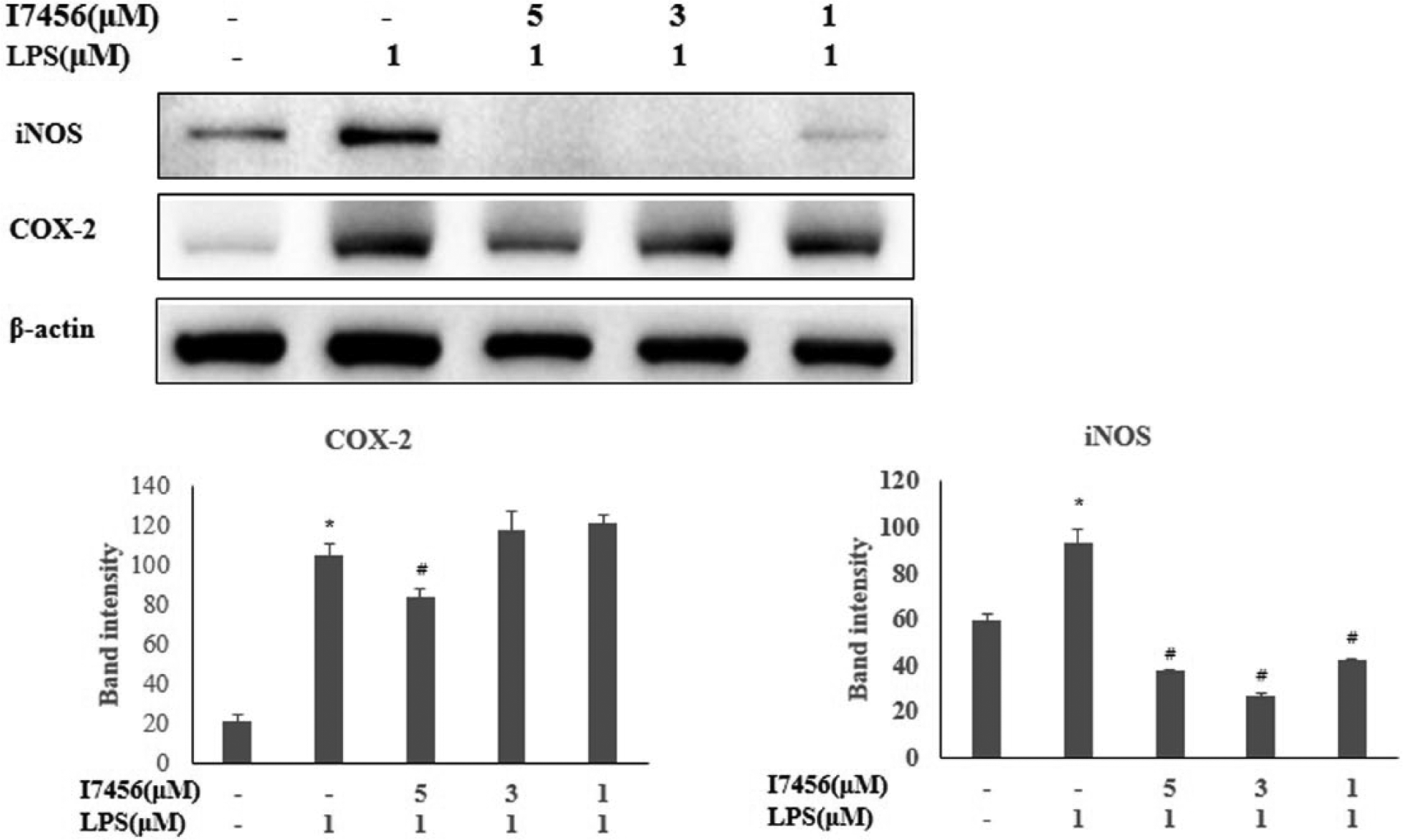
The protein level of iNOS and COX-2 after LPS challenge. Protein isolation and preparation for western blot and quantification are shown in detail in the Method section. Polyclonal antibodies to iNOS2 and COX2 (1:1000 dilution) were used, and for detection, horseradish-conjugated anti-mouse, and anti-rabbit IgG secondary antibodies (1:2000 dilution) were used for luminescence. Values are expressed as the mean ± SEM of determinations made in triplicate experiments. *
As NFκ-B translocation into the nucleus is considered critical for inflammation, the inhibitory effects of I7456 were investigated. LPS stimulation promoted massive translocation of NFκ-B, but increasing the concentration of I7456 reduced its translocation (Figure 8). To find target molecules involved in inflammation that could interact with I7456, the phosphorylation status of IκB-α was tested with phosphorylation-specific antibodies. I7456 markedly reduced the phosphorylation level of IκB-α to the same level as I0416, a known anti-inflammatory molecule 11 (Figure 9).

Inhibitory effects of I7456 on LPS-induced activation of NFκ-B in RAW264.7 cells. Nuclear translocation of NFκ-B was assessed by confocal microscopy. RAW264.7 cells were pre-treated with I7456 (10 μg/mL) for 1 h and stimulated with LPS (1 μg/mL) for 30 min. Fixed cells were stained with 4′-6-diamidino-2-phenylindole (DAPI; blue) and anti-NFκ-B antibody and fluorescein isothiocyanate (FITC)-conjugated anti-rabbit IgG (green) antibody. Magnification 200×, (a) In untreated cells, NFκ-B is limited to the cytoplasm, (b) LPS-stimulated cells show NFκ-B (green) translocation into the nucleus, and (c) I7456 inhibited NFκ-B nuclear translocation. NC represents negative controls.

IκB-α phosphorylation was significantly reduced by I7456. Protein samples were prepared and blotted as in Figure 6. Specific antibodies raised against IκB-α were used to detect phosphorylated IκB-α. Actin protein was used as the control for protein loading. NC represents negative controls.
Discussion
Phytochemicals are used by plants to fight against herbivores and pathogens. They have been harnessed to boost the immune system. 12 Isothiocyanates are known to have antimicrobial and anticancer properties. Since prolonged inflammation can lead to cancer development, we focused on studying the anti-inflammatory effects of I7456 in LPS-challenged macrophage cells.
When macrophage cells were challenged with LPS, NO increased, but NO was remarkably reduced with I7456 application. I7456 decreased cytokines and iNOS remarkably, but it did not decrease COX-2 and TNF-α to the same extent as cytokines and iNOS. Our results were consistent with those of case studies on sulforaphane.13,14 They showed an inhibition of NO and prostaglandin E2 production and an inhibition of the expression of pro-inflammatory mediators, IL-1β, IL-6, and TNF-α.15,16 Therefore, it is suggested that sulforaphane and I7456 could have anti-inflammatory effects. 17
HO-1 expression seemed to be directly influenced by I7456 because its expression was observed only after I7456 treatment. From our studies, the anti-inflammatory properties of I7456 were more significant due to a strong induction of HO-1 without LPS challenge. Since the
A sharp decrease of inflammatory cytokines by I7456 was seen in IL-6 and IL-1β, and a modest decrease was seen in IL-10, iNOS, COX-2, and TNF-α.15,19,20 The protein level of iNOS, which is responsible for NO production, was significantly reduced by I7456. It is known that IκB-α is phosphorylated in inflammatory responses. When it is phosphorylated, its role of covering the nuclear localization sequence of NK-κB is disabled, and NK-κB can, thus, move freely into the nucleus to activate downstream genes. NFκ-B is an important transcription factor and can be activated by various stimuli associated with stress and injury. NFκ-B translocation was inhibited by I7456, and the IκB phosphorylation level was reduced by I7456, which suggests that the anti-inflammatory role of I7456 occurs upon suppressing the NFκ-B signaling pathway. We showed the reduced level of NFκ-B translocation and IκB phosphorylation but could not address the precise molecular actions of I7456. However, according to a recent report, 13 I7456 could interact with the cysteines of the NFκ-B kinase β subunit and drop the IκB phosphorylation level, thereby suppressing the NFκ-B signaling pathway.
We have been looking for effective anti-inflammatory agents. Traditionally, plant-derived compounds, which are commonly found in the Brassicaceae family, have antimicrobial, anti-inflammatory, and antitumor effects. 12 Analogs are also known to be very effective in antioxidation, anti-proliferation, and cancer amelioration. 8 I7456 reduced inflammatory cytokines and NO bursts markedly through suppressing the NFκ-B signaling pathway. 6
Footnotes
Authors’ contributions
HR, HJ and HN performed the experiments and statistical analysis. HR and HS drafted the article. All authors read and approved the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by a grant (HCRI 15012-1) from Chonnam National University Hwasun Hospital Institute for Biomedical Science.
