Abstract
Ultrasonic vibrations were applied to the electrolyte in polarisation curve and chronoamperometry to observe the effect of anodic dissolution with the aim to apply knowledge to electrochemical machining. The application of the ultrasonic vibrations to the electrolyte allowed the dissolution potential to be lowered considerably and the machining rates were increased.
Introduction
Electrochemical machining (ECM) is a non-conventional machining technique capable of shaping hard-to-machine materials and complex shapes unattainable via conventional methods. 1 ECM utilises a process known as anodic dissolution in which the material which is to be machined is made the anode.2–5 A potential is applied between the workpiece (anode) and the tool (cathode), with the mirror image of the tool being replicated in the work piece.2–7 ECM has several advantages over conventional machining techniques; one of the major advantages being that the machining ability is not dependent on the mechanical properties of the material being machined, for example, the hardness of the material.2,6 This opened up the possibilities of using super alloys and other hard metals in a variety of industries, namely, the aerospace industry. 3
The development of ECM has allowed difficult-to-machine materials to be shaped using this technique, including titanium and its alloys.8,9 Titanium is a strong but light metal which has applications in both medical and aerospace industries, among others. 10 Titanium is difficult to machine electrochemically due to the formation of a corrosion-resistant oxide film, a property which makes it a very good choice for the above-mentioned industries, extending the life of the machined part. 11 Many researchers have developed methods to machine titanium with ECM; however, most report applied potentials over 20 V to machine titanium in acidic or saline solutions of sodium chloride or bromide which corresponds to machining in the transpassive region.2,8,9,12,13 Machining in the transpassive region uses more energy, first due to the higher potential required to break down the passive layer, and second a higher volume of material must be removed due to the formation of the passive layer. This makes machining less efficient than machining in the active dissolution potential region.
One of the disadvantages of ECM is that the machining rate decreases as the machining depth is increased due to the difficulties maintaining electrolyte flow in the inter-electrode gap (IEG). 12 Many researchers have investigated the effects of ultrasonic vibrations13–18 of the tool electrode or the electrolyte (only pulsed at 10 Hz) on mass transport in the ECM process and how it affected the machining results, with a higher proportion of research being conducted on the effects of applications of ultrasonic vibrations to the tool electrode.
Ultrasonic vibrations in ECM have been studied previously, with more focus on vibrations of the tool electrode. Vibrations of the tool electrode in the z-direction (parallel to the direction of tool movement) introduce pressure changes in the IEG. 17 As the tool is vibrated towards the workpiece surface, the pressure in the IEG is increased causing small bubbles in the electrolyte to collapse. As the electrode is vibrated away from the workpiece surface, the pressure drops and simultaneously the gaseous content in the IEG increases due to the formation of small bubbles. High concentrations of gas in the IEG can cause sparks to occur between the tool electrode and the workpiece. This is something that should be avoided as it can cause damage to both the tool and the workpiece. The magnitude of the pressure change is dependent on the ultrasonic vibration amplitude, as stated by Skoczypiec. 15 The changes in pressure draw fresh electrolyte into the IEG and force old electrolyte out. This increases the mass transport of the process, 15 increasing the overall machining rate and maintaining good mass transport as the machining depth is increased enabling the machining rate to be upheld.
Polarisation curves are used in preliminary research for ECM to determine whether a metal–electrolyte system is viable and at which potential machining should be carried out. 2
This article discusses the effects of ultrasonic vibrations of the electrolyte on the polarisation curves for titanium in sodium chloride solution.
Experimental method
Polarisation curves were conducted in a 1.0-M sodium chloride solution (NaCl) made with ACS Grade materials from Sigma–Aldrich and deionised water from an Elga Pure Lab Option Q water purifier (18.2 MΩ). The working electrode was a 1-mm-diameter titanium wire from Goodfellow with purity 99.6%. A three-electrode set-up was used with a Sigma–Aldrich silver/silver chloride double junction reference electrode and a homemade platinum flag counter electrode. An IviumStat potentiostat was used to control the potential and monitor the current response with a maximum voltage of 10.0 V being achievable.
The experiments were conducted at 60°C by half submerging the electrochemical cell in the heated water of a Skymen JP-031S heated ultrasonic bath. The ultrasonic bath was also used to apply the ultrasonic vibrations to the electrolyte with a frequency of 42 kHz and a power of 180 W.
The potential was scanned from 0.5 to 10.0 V at a scan rate of 5 mV s−1. The current at the working electrode was recorded through the IviumStat using the software IviumSoft.
The titanium wire was pretreated by immersing it in concentrated nitric acid for 10 s and then rinsing with acetone to dry the surface. The mass of the titanium wire was recorded before and after the polarisation experiment to compare the change in mass during the polarisation curve. The titanium wire was inserted 1 cm into the electrolyte just 2 mm from the counter electrode to minimise the IR drop ([current-resistance] or voltage drop) due to electrolyte resistance. The distance between the reference electrode and the working electrode was maintained in both the experiments to ensure the potential applied to the working electrode is the same in each experiment.
In each case, the counter electrode and working electrode were connected to the potentiostat and the potential was applied before the electrodes were inserted into the electrolyte to prevent passivation of the titanium surface before a potential was applied due to contact with the electrolyte. The ultrasonic vibration was turned on before the electrodes were inserted into the solution also and maintained for the length of the experiment.
Chronoamperometry measurements were also conducted. A constant potential of 10.0 V was applied for 1 h in 1.0 M NaCl electrolyte heated to 60°C. Again, one experiment was conducted with ultrasonic vibrations and one without; the results were compared to one another. The titanium wire was treated the same as for the polarisation curves.
See Figure 1 for the experimental set-up for all experiments conducted with ultrasonic vibrations.

Experiment set-up with ultrasonic bath.
Results and discussion
In this research, ultrasonic vibrations were applied to the electrolyte during polarisation experiments of titanium in 1.0 M NaCl electrolyte and compared to the polarisation curves of titanium without the ultrasonic vibrations in the same electrolyte. Polarisation curves show the potential–current relationship. Polarisation curves are utilised by ECM users to determine ideal machining potentials for a given metal–electrolyte system.
Chronoamperometry experiments were also conducted which show the current–time relationship when a constant potential is applied. Two comparison experiments were conducted to observe how the ultrasonic vibrations affect the dissolution of titanium.
The experimental set-up limits the results somewhat as the IEG maintained during the experiments is larger than those maintained in ECM and micro-ECM. As such, only general conclusions can be made from the results but does provide important information on the electrochemical behaviour of titanium during anodic dissolution with insonation.
Polarisation curve results
Polarisation curves were conducted in 1.0 M NaCl electrolyte with and without the application of ultrasonic vibrations. Figure 2 shows the polarisation curves of both the experiments. The experiment conducted without the ultrasonic vibrations shows a slightly higher current in the potential range of 3–7 V. This has been attributed to the formation of an oxide layer which is unable to form when ultrasound is applied to the electrolyte. When the ultrasonic vibrations were applied, the concentrations of the active material were not able to reach values high enough to cause considerable oxidation, hence the lower observed current for the experiment conducted with ultrasonic vibrations. This is because the ultrasonic vibrations assist the transport of species in the electrolyte, both to and from the electrode surface. In this case, the oxidising species are carried away from the electrode surface with ultrasonic vibrations.
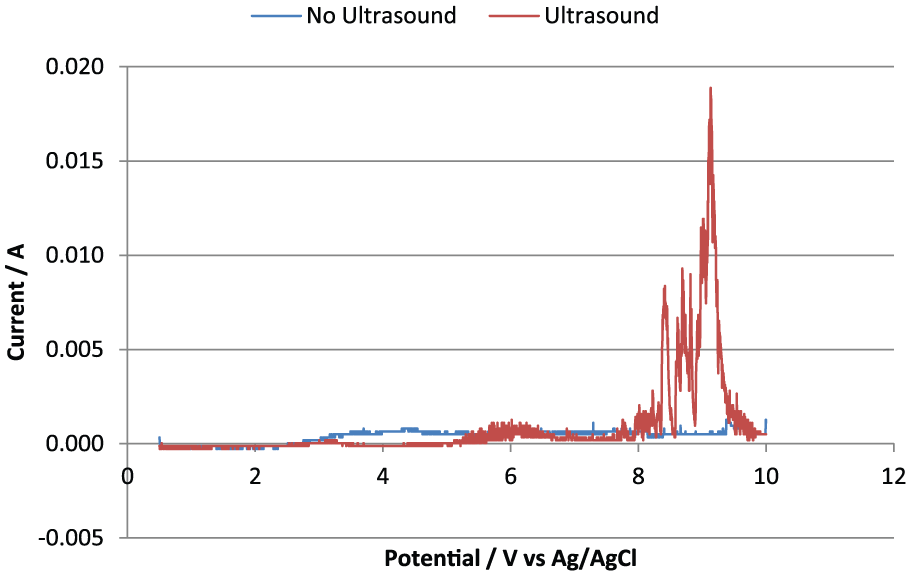
Polarisation curves of titanium in 1.0 M NaCl with (red) and without (blue) ultrasonic vibrations of the electrolyte at 60°C.
As the potential is increased beyond 7 V, the current response for the experiment with no ultrasound applied remains virtually at 0, whereas the case with ultrasound applied to the electrolyte sees a dramatic increase in the observed current. This can be attributed to the start of dissolution of the titanium metal. The ultrasonic vibrations prevented the formation of the protective oxide layer at lower potentials leaving the surface active. The ultrasonic vibrations aid transport of dissolution products from the electrode and also bring fresh electrolyte to the electrode surface.
Chronoamperometry results
Chronoamperometry sees a constant potential applied for a set length of time. The chosen potential was 10 V, which was the highest available with the IviumStat potentiostat, with the potential being applied for 1 h (3600 s). The current was recorded for the length of the experiment. The chronoamperometry experiments are used to show how the application of ultrasound to the electrolyte facilitates the dissolution of titanium at a lower potential. The results of the chronoamperometry experiments are shown in Figure 3. The first 100 s of the experiment should be ignored as the current response is not a true representation of the conditions of the experiment; the titanium wire was not inserted into the electrolyte until the potential had been applied in order to prevent passivation of the titanium surface due to contact with the aqueous electrolyte. Higher currents are observed due to the movement of the electrode as it was inserted into the electrolyte and sealed. For the experiment which had no ultrasound applied, the current remains virtually at 0 for the length of the experiment. This is because a protective oxide film quickly forms on the surface preventing further reaction or dissolution.

Chronoamperometry of titanium in 1.0 M NaCl with (red) and without (blue) ultrasonic vibrations at 10.0 V versus Ag/AgCl at 60°C.
The surface finish of the titanium wire after the chronoamperometry experiment can be seen in Figure 4.

SEM image of titanium wire after chronoamperometry at 10.0 V at 60.0°C.
When the ultrasonic vibrations are applied to the electrolyte, the electrolyte conditions facilitate much higher mass transport rates both to and from the electrode surface. This renews electrolyte at the surface of the titanium, maintaining a high concentration of solvating ions, essential for dissolution. The electrolyte conditions also remove solvated ions from the diffusion layer at a high rate, meaning the concentration cannot build up to limiting saturation levels. Another effect that could be occurring is the destruction of the passive layer by cavitation. Ultrasonic vibrations cause nucleation and the subsequent collapse of gas bubbles within the electrolyte. The bubbles have a critical radius, dependent on the frequency of the sound wave. When the bubbles collapse, a large amount of energy is released as heat; if these bubbles collapse near the electrode surface, it can cause cavitation, increasing the machining rate further by mechanically removing the passive oxide layer.
The current range seen in the polarisation curves and chronoamperometry experiments here does not exceed 0.25 A. This current would not facilitate ECM but may be suitable for micro-ECM.
Conclusion
The application of ultrasonic vibrations to the electrolyte in ECM is very easy to achieve in comparison to vibrating the tool electrode.
The ultrasonic vibrations aided dissolution, as seen in the polarisation curves and chronoamperometry experiments. The machining potential was reduced from reported values of over 20 V to just 10.0 V, saving a considerable amount of energy. Lower voltages and temperatures will be investigated to reduce energy consumption further. The ultrasonic vibrations also increased the machining rates, reducing the processing time of each part and subsequently the processing costs are reduced.
It is still to be seen how insonation of the electrolyte will affect anodic dissolution of titanium under more realistic ECM conditions, but this study gives promising initial results.
Footnotes
Academic Editor: Hui Ding
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
