Abstract
Molecular testing is fundamental to the modern precision oncology toolkit. However, set on a backdrop of inconsistent use of the term ‘value’ in healthcare literature, evidentiary limitations in what determines ‘high-value’ molecular testing have contributed to its variable adoption in the clinic. Key to an improved understanding of value in this setting is eliciting what outcomes (health and non-health) and costs of molecular testing are important to the relevant stakeholders and what contextual considerations influence this. This protocol describes the first stage of a multi-staged mixed methods study to elicit preferences and views on the value of molecular testing among the stakeholders relevant to an Aotearoa New Zealand societal perspective. Semi-structured interviews, focus groups, and questionnaires will elicit the elements that determine the value of molecular testing for patients and family | whānau (Māori and non-Māori), clinicians, testing providers, molecular medicine researchers, industry (pharmaceutical and diagnostic) members, and decision-makers. Interviews and focus groups will be analysed using thematic analysis and questionnaire data will be analysed using descriptive statistics. Identifying the elements of molecular testing in cancer that warrant consideration in value assessment discussion is a prerequisite to modernizing and improving health technology assessment and funding decision-making processes. Findings from this qualitative study will inform the second stage (presented in a subsequent paper) of this multi-staged mixed-methods study, which will use the identified elements in a discrete choice experiment to understand different stakeholders’ preferences and tradeoffs.
Keywords
Background
At the core of precision oncology, molecular testing supports diagnosing cancer, predicting response to genomically targeted drugs, and estimating prognosis. With a role in prevention and risk assessment, molecular testing can screen for inherited genes that increase an individual’s risk of developing cancer and inform the need for risk-modifying interventions. As such, molecular testing has expanding clinical indications across the cancer control continuum and patient and family | whānau care pathway.
However, the value of molecular testing in cancer is poorly understood (Marshall et al., 2020; Payne et al., 2018; Phillips et al., 2014). Set against a backdrop of ubiquitous and inconsistent use of the term ‘value’ in health literature more broadly the lack of a uniform approach to defining and measuring the ‘value-for-money’ (i.e., in the economic sense, the incremental costs relative to the incremental outcomes) of molecular testing in cancer across the technology life-cycle (i.e. from molecular testing research and development, regulatory approval, pricing and reimbursement, to optimal clinical implementation) (Regier et al., 2022) poses a challenge to decision-makers. What molecular test attributes (or features) are most important, such as benefits, risks, costs, or contextual considerations, is unclear. As is whether and how importance of these attributes varies for different stakeholders. Evidentiary and methodological limitations at the point of health resource allocation decisions contribute to unwarranted variation and inequitable access to molecular testing for patients. As more molecular tests come to market (The Business Research Company, 2022) and molecular testing increasingly feature in oncology clinical guidelines (Ettinger et al., 2022) the consequences of inadequate tools and insufficient data to measure value are expected to magnify.
Value in the clinical sense usually refers to the net benefits to a patient (e.g., health and personal benefits including improved quality or quantity of life) compared to the costs (including physical, financial and time spent toxicity) of a technology or intervention. The considered benefits intend to be those that matter most to patients. This value definition is analogous to clinical effectiveness, clinical value or clinical utility. ‘Value-based healthcare’ encompasses a range of factors (health improvement at the patient and population level, responsiveness of the health system to personal needs, financial protection, efficiency and equity) which define value as the contribution the healthcare system makes to societal wellbeing. With this perspective value should be passed on to patients and correspond to their interpretation of value. Value-for-money (or value for resource) in the economic sense is usually considered the incremental costs relative to the incremental benefits of a technology or intervention. The considered costs and benefits will depend on the perspective of the assessment. This value definition can be considered akin to cost-effectiveness where the marginal value of a technology or intervention reflects the willingness to pay for the improvement in benefits minus the opportunity cost of resources used to produce that improvement. From here in this paper the term value will be referring to value-for-money (or value for resource) with recognition that clinical effectiveness, incremental cost-effectiveness (with inclusion of non-health benefits) and contextual considerations (e.g., equity issues) all have potential to determine value and contribute to the goal of delivering value-based healthcare.
Over recent decades, approaches that measure the net value of a technology relative to alternatives have become conventional. For example, cost-effectiveness analysis (CEA) focuses on incremental costs relative to incremental benefits. CEAs relate the resource use associated with a technology to the change in health/wellbeing achieved, expressed as an incremental cost-effectiveness ratio (ICER). The linked optimal decision rule is that the intervention with the lowest ICER is the best value (Palmer et al., 1999). The robustness of decision-making based on ICERs depends on whether full costs and benefits are accounted for. An adapted CEA, using the universal outcome metric, the Quality Adjusted Life Year (QALY), which measures a technology’s effect on both the quantitative and qualitative aspects of health (Pliskin et al., 1980; Zeckhauser & Shepard, 1976), is referred to as a cost-utility analysis (CUA). The QALY’s strength is enabling comparability across diverse interventions with different outcomes. However, with its focus on the health consequences of morbidity and mortality, the QALY cannot account for all potential outcomes and has clear limitations when applied to diagnostics. Unlike for cancer treatments where health effects are direct and more readily measured - and for which in Aotearoa New Zealand Pharmac (Home - Pharmac | New Zealand Government, n. d.) has a standardised approach to assessing value (Factors for Consideration - Pharmac | New Zealand Government, n. d.) - the health outcomes from cancer diagnostics, such as molecular testing, are largely indirect. The value is in the information derived from the diagnostic test rather than the test itself. These outcomes can be dynamic and not easily predicted (Marshall et al., 2020). Further, individuals beyond those tested (e.g. family | whānau) can be affected. From test information, multiple possible health and non-health outcomes can exist for multiple stakeholders. As such, approaches to capturing the broader elements of value have a natural appeal.
A recent shift has spotlighted these more novel, usually non-health, elements of value (Lakdawalla et al., 2018). Understanding the non-health elements of value (e.g., personal utility through reduction in uncertainty), or benefits beyond the individual tested (e.g., to whānau; akin to the value element family spillover (Garrison et al., 2018), or scientific spillover (Lakdawalla et al., 2018; Riley et al., 2016) is relevant to molecular testing in cancer to acknowledge the consequences outside of the obvious function of testing improving medical decision making and subsequent health outcomes. It is also recognised that conventional approaches have failed to incorporate contextual factors that determine value. Clinical factors (e.g., the rarity of the condition, line of treatment) can represent health need or influence the value of a subsequent intervention. Issues relating to social and health equity and the associated distributional benefit (and distributional costs) of health technologies are pertinent, especially in Aotearoa New Zealand, where there are stark and unjust existing inequities in health outcomes for indigenous Māori and Pacific peoples (e.g., Māori are 20% more likely to develop cancer than non-Māori and twice as likely to die from cancer (Te Aho o Te Kahu, 2020), and germane to molecular testing in cancer where access is notoriously variable.
Quantifying the costs of health technologies for value assessments is less debated than the quantification of outcomes. However, there is ambiguity, especially for costs beyond direct material costs of the testing technology (including costs of test processing, results interpretation, and downstream costs associated with the action informed by the test result information) and associated broader resource use e.g., salaries/wages of involved personnel. These costs may be perceived and experienced differently. The downstream costs of molecular testing can be numerous and within complex clinical pathways managed by siloed budgets. Consequently, cost data deficiency and evidentiary uncertainty are common challenges in the relatively new field of precision oncology (Pollard et al., 2022).
When a broader conceptualisation of value is emphasised, approaches that consider society’s views have obvious appeal, including efforts to incorporate the wide benefits of testing more holistically - at individual and population levels. For this, the views and preferences of all relevant stakeholders throughout the technology life-cycle are needed, including molecular medicine researchers, industry members (pharmaceutical and diagnostics), regulatory and health technology assessors, funding decision-makers, funders, the public, clinicians, and patients. Stakeholder views and preferences can complement existing value assessment decision criteria.
Published data on preferences of molecular testing in cancer are limited (Buchanan et al., 2016; Najafzadeh et al., 2013) and it is unknown to what extent views on the value of molecular testing in cancer differ between stakeholders. In international literature, indigenous views on in molecular testing in cancer are underrepresented.
In Aotearoa New Zealand a predominantly publicly funded healthcare system (New Zealand Health System | Ministry of Health NZ, n. d.), the healthcare system aims to maximise health outcomes and address inequitable health outcomes, particularly for indigenous Māori. (Pae Ora (Healthy Futures) Act 2022 No 30 (as at 27 July 2023), Public Act 3 Purpose of This Act – New Zealand Legislation, n. d.) As such, equity considerations must be front and foremost of all decisions in cancer control to honour Te Tiriti o Waitangi obligations, which have not been met historically (Ahuriri-Driscoll et al., 2022; O’sullivan et al., 2020). Previously, there has been no consistent approach to non-pharmaceutical health technology assessment. Diagnostic test funding decisions and payment for publicly funded tests have been made at an individual hospital level (former District Health Boards, DHBs), (Sampietro-Colom & Martin, 2017) usually initiated by separate laboratory-led business cases. Evidence to inform decision-making has typically focused on the intervention’s health benefits and direct costs, with emphasis on cost-minimisation (Sampietro-Colom & Martin, 2017). Recent national health reforms (New Zealand Health System | Ministry of Health NZ, n. d.) provide an opportunity for radical changes, including for New Zealand research to shed light on alternative approaches to more equitable health technology assessment and funding decision-making processes.
The focus of this work - to understand the value in molecular testing in cancer - is one example seeking to contribute to a more holistic, yet still methodologically achievable, approach to value assessment for molecular testing in cancer, with potential for broader application to other non-pharmaceutical health technologies.
This protocol describes the first stage of a multi-staged mixed methods study (Figure 1) to elicit preferences and views on the value of molecular testing among the stakeholders relevant to an Aotearoa New Zealand societal perspective. Here, the study aims to identify elements of molecular testing in cancer that warrant consideration in value assessment discussion. The second stage (presented in a subsequent paper) will use these elements in a preference elicitation exercise (yet to be designed) to understand different stakeholders’ preferences and tradeoffs. This Qualitative Research Protocol (Outlined in Red) Sits Within a Larger Multi-Staged Mixed-Methods Project Aiming to Understand the Value of Molecular Testing in Cancer
Objectives
The specific objectives from stage I are to: (1) Identify elements of molecular testing in cancer that warrant consideration in value assessment discussion. (2) To assign clinically and economically relevant levels to these value elements to inform the design of stage II’s discrete choice experiment. (3) to expand understanding of elements of molecular testing in cancer and the decision-making process used by current decision-makers.
Methods
This qualitative protocol informs research to be undertaken in Aotearoa New Zealand. It has been developed and described by following guidelines for using and reporting qualitative research in health economics and the steps recommended for attribute and level development in health care preference research (Bridges et al., 2011; Coast et al., 2011; Coast & De Allegri, 2018; Coast & Horrocks, 2007; Coast, 1999; Hensher et al., 2015; Hollin et al., 2020; Lacey & Luff).
Equity-Led, Anti-racist, Te Tiriti Guided, Ethical Study Design
Principles of equity, anti-racism, and Te Tiriti have guided the design of this study methodology. Previous healthcare funding decision-making has marginalised Māori views, breaching Māori tino rangitaranga (self-determination) entitlement to active, informed participation in systemic decision-making (O’sullivan et al., 2020). Further, philosophical underpinnings of health economic evaluation are traditionally Western-centric (Willing et al., 2019). This study’s use of select health economic methodologies must be acknowledged as a differential power relationship between knowledge systems and research disciplines. This study will take deliberate methods and processes to be Māori-centric and support a strong Māori voice. For example, whakapapa (genealogy) will be naturally built into engagement with Māori participants, and sampling will aim for as close to equal explanatory power (Röpü et al., 2002) as possible.
Selection of Data Collection Methods
A mix of qualitative data collection techniques have been selected to accommodate specific needs of stakeholder groups (e.g., conceptual complexity, potential topic sensitivity (Coast & Horrocks, 2007; Hollin et al., 2020), manage logistical/efficiency challenges (Coast et al., 2011) (e.g., geographically disperse individuals) and optimise quality and authenticity of responses (e.g., leveraging from group reflection, providing anonymity where required (Coast et al., 2011). The selected methods will serve the design needs of the planned preference elicitation exercise, which requires a deliberate and structured approach to develop attributes and attribute levels for improved reliability and content validity of the survey. (Coast & Horrocks, 2007; Vass et al., 2017)
Sampling and Recruitment
Goal Participation, Mode of Recruitment, and Data Collection Method for Each Stakeholder Group
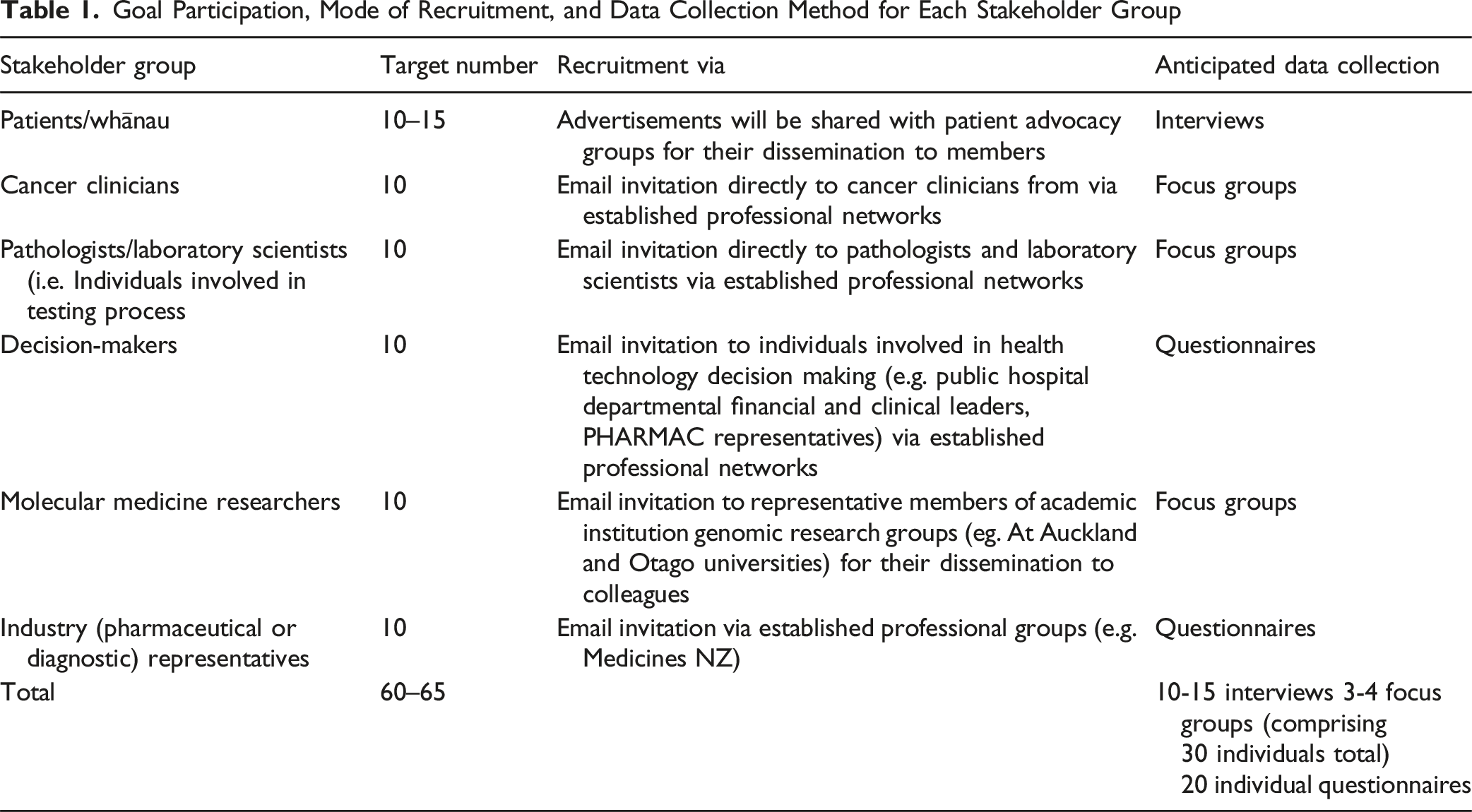
Interviews, focus groups, and questionnaires will stop when data saturation (Bazeley, 2013) is reached. Participants will be eligible if they are >18 years old, self-identify with one of the key stakeholder groups, and can provide informed consent to an interview, focus group, or questionnaire.
A study advertisement, with a link to participant information sheet and screening questionnaire, will be emailed to patient advocacy representative organisations to disseminate to their members. From the responses and registrations of interest received, interested participants purposefully selected for optimal spread and diversity by age, gender, ethnicity, and geographic location will be contacted. Clinicians, testing providers and molecular medicine researchers will be emailed an invitation to a focus group and asked to complete a screening questionnaire and register their interest in being contacted to schedule the focus group. For industry representatives and decision-makers, an invitation to complete the anonymised questionnaire will be emailed through established professional networks (Figure 2). Recruitment Pathways
Study Procedures
Conceptual value elements (and when available levels) will be identified from literature reviews (to be published separately) to provide the basis for the topic selection of the semi-structured interviews, focus groups, and questionnaires. One example of the conceptualisation of novel elements of value is the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) “value flower” (Lakdawalla et al., 2018) (Figure 3). ISPOR Special Task Force Value Flower
Interviews
Semi-structured interviews will be conducted in person, via telephone or a virtual meeting platform (audio or video depending on participant preference), lasting no longer than 60 min, and facilitated by a co-investigator trained in qualitative research using a topic guide informed by the literature review (see supplemental materials). Introductory material and key definitions will be provided at the beginning of the interview, and consent, including for audio-recording, will be obtained. Adjustments will be made as needed to promote cultural safety (offering in-person interviews or an interpreter) and the inclusion of disabled persons (e.g., the inclusion of breaks, simple language). First, using a ‘bottom-up’ approach to questioning, participants will be asked openly about what is most important, relevant or valued with molecular testing in cancer. Next, a ‘top-down’ approach will ask participants to reflect on elements of the value of molecular testing in cancer retrieved from the literature. Each interview participant will be gifted a grocery voucher in appreciation for their participation. Demographic information of participants will be collected. Interview audio-recordings will be transcribed verbatim before analysis. Participants will be offered an opportunity to check (and receive a copy of) their interview transcript.
Focus Groups
Focus groups will be conducted in person or via a virtual meeting platform for approximately 60 min. Two co-investigators trained in qualitative research will facilitate using a discussion guide (see supplemental materials) to ensure procedural consistency across focus groups. Introductory material and key definitions will be provided at the beginning of the focus group, and participant consent will be obtained before the focus group discussion begins. Focus groups will include semi-structured discussion followed by a ranking exercise to identify value elements and (where possible) their levels relevant to molecular testing in the Aotearoa New Zealand setting for clinicians, pathologists/laboratory scientists, and molecular medicine researchers. A ′top-down’ approach will ask participants to reflect on elements of the value of molecular testing in cancer retrieved from the literature. Each focus group participant will be offered non-alcoholic refreshments and snacks in appreciation for their participation and gifted a grocery voucher in appreciation for their contribution. Demographic information of participants will be collected. One co-facilitator will take written notes during the focus group, supplemented by a group audio-recording transcribed verbatim before analysis.
Questionnaires
The questionnaire will be delivered electronically using Qualtrics (Qualtrics, Provo, UT) software. Questions will be based on the interview and focus group questions (including an open question) and contain a ranking exercise to ensure a minimum level of response (see supplemental materials for draft questionnaire). The questionnaire to funding decision-makers will also include questions about the current decision-making process and any criteria used in current decision-making. Consent will be implied by submission of the anonymised questionnaire. Each focus group participant will be offered an opportunity to enter a prize draw in appreciation for their participation.
Analysis Plan
Thematic analysis of the de-identified interview transcripts, focus group notes and transcripts and questionnaire responses for open-ended questions will be conducted using NVivo software (v 14.23.2, QSR International). Responses will be coded, then codes will be aggregated to construct themes. Careful attention will be made to any overlap in definitions/names of value elements. As a research group, coding and thematic analysis will be discussed to reach a consensus. The remainder of the questionnaire responses will be analysed with descriptive statistics. The outcomes of qualitative data analysis will be the determinants of value proposed to inform a societal perspective for decision-making. A specific output of this analysis is attributes and their levels for potential inclusion in a Discrete Choice Experiment (DCE) survey (Bahrampour et al., 2020). Qualitative data will also aid scenario design using whānau-friendly wording for the DCE survey.
Ethics Approval
Informed consent to be audio-recorded will be obtained from all interview and focus group participants. Consent will be implied by submission of the anonymised questionnaire. Data management and storage will be subject to the University of Auckland Research Data Management Policy (effective 1 July 2023). This requires research data management consistent with FAIR data (Wilkinson et al., 2016), Te Tiriti obligations, and the people and purpose-oriented CARE principles (Carroll et al., 2020). Ethical approval has been obtained from Auckland Health Research Ethics Committee (AH26739).
Dissemination
This study will be reported according to the Consolidated Criteria for Reporting Qualitative Research (COREQ) (Tong et al., 2007) and guided by the Consolidated Criteria for Strengthening Reporting of Health Research Involving Indigenous peoples (CONSIDER) (Huria et al., 2019).
Study results are planned for wide dissemination to relevant stakeholders via scheduled meetings and hui, webinars, conference presentations, and peer-reviewed journal manuscripts. Sharing and dissemination of results will strive to be mana-enhancing (strengths-based) supporting national cultural growth and development. For example, invitation for feedback and criticism will be welcomed during community results sharing.
Rigor
The study’s design for scientific rigour reflects the contribution of co-investigators with complementary and multidisciplinary skills, including health economics, qualitative research, te Ao Māori and medical oncology expertise. The team are committed to making (and reporting) iterations to research design as the research progresses in response to analysis of early data and feedback from research participants. This may include iterations to sampling and data collection (Coast & De Allegri, 2017). Engaging in reflexivity practices, in particular by the lead interviewer and focus group co-facilitator who is an oncologist, aims to acknowledge and leverage from any influence of their subjectivity (Olmos-Vega et al., 2023). During qualitative analysis, there will be an iterative process for coding whereby frequent discussion with members of the research group will inform iterative development of the coding framework. Similarly, generation of themes will be based on discussion as a research group to strengthen reliability and validity of final themes. As mentioned in ‘Selection of data collection methods’, the deliberate and structured approach to attribute selection using qualitative research improves the reliability and content validity of the DCE. The publication of this protocol paper and the subsequent DCE protocol will demonstrate transparency of scientific method.
Discussion
This qualitative study intends to achieve depth of knowledge on what determines the value for money of molecular testing in cancer represented by the views and preferences of a range of relevant stakeholders. These data on determinants of the value of molecular testing in cancer may be helpful to inform the co-creation of improved value assessment that could be useful for decision-making (e.g., health care resource allocation).
Drawing on the views of those directly impacted by molecular testing in cancer funding decisions will complete the two-stage process for developing and refining DCE attributes and levels. DCE surveys are a well-established preference elicitation method. To date no DCE survey has attempted to measure the value of molecular testing in cancer. This qualitative research will directly inform the design of a DCE that aims to quantify preferences and tradeoffs of the elicited value elements and then examine heterogeneity between stakeholder groups.
Increasingly, DCEs have a role in capturing and measuring the value of attributes of health and healthcare (McIntosh, 2006; Ryan, 2004). The capability of a DCE survey to quantify and present the contribution of value of each attribute as a willingness to pay allows measurement of benefits beyond health gain (Ryan et al., 2008) and examine the influence of contextual factors on value (Moro et al., 2022). The design of a DCE survey requires first selecting appropriate attributes and assigning attribute levels (Lancsar & Louviere, 2008; Louviere et al., 2010). A challenge is balancing adequate representation of all attributes and a practical number of attributes-levels well aligned with random utility theory (Veldwijk et al., 2023). Recent attention has turned to the importance of rigor in implementing and reporting on the processes of developing attributes and attribute levels. A deliberate and structured approach to attribute selection using qualitative research – typically focus groups and interviews - improves the reliability and content validity of the DCE survey (Coast et al., 2011; Coast & Horrocks, 2007; Vass et al., 2017).
Methodological consensus and practical guidance on using qualitative research for the robust design of DCEs that aim to measure benefits beyond health gain and contextual considerations and present these as willingness to pay values are limited. This emphasises the importance of testing and reporting on qualitative methods used in this setting.
Limitations
The usefulness of the data is dependent on input that is reflective of stakeholders’ views relevant to molecular testing of cancer in Aotearoa New Zealand. We acknowledge that multiple competing realities and views may differ across time and context, and the analysis findings may be limited by the small sample size, and to the time and context of this study.
Conclusion
Our study will use interviews, focus groups, and questionnaires to elicit preferences and views on the value of molecular testing in cancer. These findings will inform the design of the planned DCE. We hope that this mixed-methods, multi-staged study will inform an improved approach to measuring the value of molecular testing in cancer from the societal perspective. We hope that publishing this protocol contributes to the growing body of literature on the use of qualitative research to inform quantitative preference studies in healthcare.
Supplemental Material
Supplemental Material - Understanding the Value of Molecular Testing in Cancer in Aotearoa New Zealand: A Qualitative Study Protocol to Elicit Stakeholder Preferences
Supplemental Material for Understanding the Value of Molecular Testing in Cancer in Aotearoa New Zealand: A Qualitative Study Protocol to Elicit Stakeholder Preferences by Alice Minhinnick, Michelle Wilson, Rob McNeill, George Laking, and Paula Lorgelly in International Journal of Qualitative Methods
Footnotes
Acknowledgements
The authors gratefully acknowledge the future participants of this study for the sharing their time, experience, and opinions for the purpose of this research.
Ethical Considerations
Approved by Auckland Health Research Ethics Committee (Ref AH26739).
Consent to Participate
Informed written consent to participate for interviews and focus groups.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is to be conducted during tenure of a Health Research Council of New Zealand Clinical Research Training Fellowship and so the research is supported by funding from the Health Research Council of New Zealand (Grant No. 23/175).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
