Abstract
Lifestyle medicine is an emerging field that uses targeted behavioral interventions to prevent and manage chronic diseases. This review examines how its six pillars; nutrition, physical activity, stress management, avoidance of risky substances, restorative sleep, and social connection relate to dermatologic health. Through a comprehensive literature review, we identified evidence linking lifestyle factors to skin disease outcomes. Ultraviolet (UV) exposure, for example, accelerates photoaging and skin cancer risk, while photoprotection helps prevent this damage. Allergen and irritant avoidance are essential in conditions like atopic and contact dermatitis. Nutrition impacts inflammation, barrier function, and oxidative stress, influencing diseases such as psoriasis and acne. Physical activity improves skin appearance and reduces systemic inflammation. Sleep quality regulates immune function, with poor sleep linked to flares in eczema and urticaria. Risky behaviors like tobacco and alcohol use correlate with more severe disease and poorer outcomes. Social connection improves quality of life and treatment adherence, especially in visible or stigmatized conditions. Validated assessment tools, including questionnaires and biomarkers, can guide risk stratification and personalized care. When adapted to patient context, lifestyle interventions offer non-pharmacologic strategies that enhance dermatologic treatment. This review underscores the need to integrate lifestyle medicine into dermatology for a preventive, patient-centered approach.
Keywords
“The convergence of lifestyle medicine principles with dermatologic practice represents an evolution in how we conceptualize skin health and disease management.”
Introduction to Lifestyle Medicine
Lifestyle medicine is a medical specialty focused on using therapeutic lifestyle interventions to treat and reverse chronic conditions, as defined by the American College of Lifestyle Medicine founded in 2004. 1 Evidence-based strategies guide the prescription of lifestyle changes, shown to improve or reverse conditions like cardiovascular disease and metabolic syndrome.1,2 Integrating nutrition, physical medicine, rehabilitation, and psychology, lifestyle medicine promotes overall health and disease prevention.
These lifestyle modifications not only improve specific conditions but enhance general well-being. 3 For example, improving nutrition in a patient with hyperlipidemia can also improve sleep and energy levels. With chronic disease prevalence rising in the United States, growing research supports the relevance of lifestyle medicine in clinical care. 4 Chronic dermatologic diseases, such as psoriasis, eczema, and rosacea, are influenced by lifestyle factors. Thus, applying lifestyle medicine principles may aid in managing these conditions.
Lifestyle medicine promotes a whole-person, individualized approach grounded in six pillars: nutrition, physical activity, stress management, avoidance of risky substances, restorative sleep, and social connection. 1 The first pillar emphasizes that increasing intake of minimally processed foods helps prevent and reverse lifestyle-related chronic diseases. 5 The second pillar, physical activity, is crucial for preventing disease and improving health through 150-300 minutes of moderate or 75-150 minutes of vigorous aerobic activity weekly, plus strength training on at least two days. 5 Stress management is the third pillar, aimed at reducing chronic stress, which raises the risk for cardiovascular and mental health disorders.5–8 The fourth pillar focuses on avoiding risky substances like tobacco and alcohol, which are linked to chronic medical conditions. 9 The fifth pillar, restorative sleep, is vital for attention, hunger, and insulin resistance, all of which contribute to chronic disease. 5 The sixth pillar, social connection, is vital for emotional and physical health, as strong relationships improve cardiovascular markers like heart rate and blood pressure while social isolation increases morbidity and mortality.5,11 SMART goals (specific, measurable, achievable, realistic, and time-bound) are encouraged to help patients achieve their personal goals within each of these pillars.
The principles of lifestyle medicine are applicable across all specialties. This review focuses on the influence of lifestyle factors, especially the six pillars, on dermatologic diseases. Relevant factors include UV exposure, allergens, diet, exercise, sleep, substance use, stress, and social support (Figure 1). We aim to summarize evidence showing how these factors affect outcomes of several common skin diseases and are relevant in considering personalized approaches to the management of dermatologic disease overall. Modifiable lifestyle factors that influence dermatologic disease.
Methods
A comprehensive literature search was performed using PubMed and Google Scholar through June 1st, 2025. The following keywords and Medical Subject Headings (MeSH) terms were used in various combinations: “lifestyle medicine,” “nutrition,” “diet,” “exercise,” “stress,” “sleep,” “alcohol,” “tobacco,” “psoriasis,” “acne,” “rosacea,” “atopic dermatitis,” “hidradenitis suppurativa,” and “skin disease.”
We included peer-reviewed clinical trials, systematic reviews, meta-analyses, observational studies, and relevant expert opinions that explored the relationship between lifestyle interventions and dermatologic outcomes in adults and adolescents. Articles were included if they specifically addressed one or more lifestyle pillars and a skin condition of interest. Studies were selected based on their quality, relevance, and applicability to clinical practice. Key findings were extracted regarding the type of intervention, dermatologic outcome measured, and any reported statistical significance or clinical relevance. Preference was given to studies with clearly defined interventions and quantifiable skin outcomes.
This review is not systematic and does not aim to provide a meta-analysis of outcomes. Rather, it highlights significant trends and recurring evidence supporting the integration of lifestyle-based approaches in dermatologic care. Limitations include potential selection bias and the heterogeneity of study designs, populations, and outcome measures among included articles.
Results and Discussion
UV Exposure
Ultraviolet (UV) radiation contributes to both acute cutaneous effects, such as erythema and sunburn, and long-term consequences including photoaging, pigmentary disorders, and increased risk of skin cancer.12,13,14,15,16,17 UVB radiation primarily induces direct DNA damage through the formation of cyclobutene pyrimidine dimers and 6-4 photoproducts,15,18 while UVA penetrates deeper into the dermis and generates reactive oxygen species that damage cellular proteins, lipids, and DNA. 16 Together, these mechanisms accelerate skin aging and increase the risk of nonmelanoma skin cancers and melanoma.12,19 UV radiation also impairs skin barrier integrity, disrupts immunologic surveillance, and contributes to pigmentary changes.15,20
Given the wide variability in UV susceptibility and exposure, photoprotection strategies should be personalized based on both intrinsic factors (such as skin phototype, age, genetic predisposition, and other skin conditions) and extrinsic factors (such as occupational exposure, geographic location, and access to protective resources).14,17,20 Individuals with Fitzpatrick skin types IV-VI, while at lower risk for skin cancer, are more prone to pigmentary disorders such as melasma and post-inflammatory hyperpigmentation.14,21,22 For these individuals, tinted sunscreens with visible light protection (e.g., iron oxides) and non-nanosized inorganic filters offer both efficacy and cosmetic compatibility. 14 Similarly, children may benefit from oil-based inorganic sunscreens containing zinc oxide or titanium dioxide, which are less likely to cause irritation or systemic absorption. 14 Older adults may require heightened photoprotection due to cumulative photodamage and frequent use of photosensitizing drugs.14,23
People with occupations primarily located outdoors, including those in construction or landscaping, often experience high levels of occupational UV exposure and should receive individualized counseling that emphasizes consistent use of sunscreen, protective clothing, and scheduling outdoor tasks outside of peak UV hours.12,15,24,25,26 Geographic location also plays a role—UV intensity is greater at higher altitudes and closer to the equator, requiring more rigorous protection in these regions. 27 Patients with inflammatory or photosensitive skin conditions, or those undergoing phototherapy, may also require individualized adjustments to their photoprotection regimens.17,22 For instance, individuals with lupus or dermatomyositis may benefit from sunscreens that offer visible light and UVA protection to prevent flares, while those undergoing PUVA therapy require stricter sun avoidance outside treatment sessions compared to those receiving narrowband UVB.17,22
Emerging strategies, including sunscreens fortified with DNA repair enzymes like photolyase, have shown potential in improving photodamage markers among high-risk individuals with actinic keratoses.14,28,29 Beyond pharmacologic and topical interventions, behavioral modifications such as avoiding midday sun, seeking shade, and wearing protective clothing are fundamental. 14 Ultimately, precision photoprotection reinforces the broader goals of lifestyle-based dermatologic care, ensuring that interventions are adapted not only to an individual’s biology, but also to their daily environment, social determinants, and risk profile.
Allergens
Allergic and irritant exposures exemplify how environmental factors intersect with multiple lifestyle medicine pillars (nutrition, stress, sleep, risky substances, physical activity), influencing not only direct skin contact but also systemic immune reactivity. Understanding these interconnections is crucial for comprehensive dermatologic care, as allergic and irritant exposures are key contributors to a range of dermatologic conditions, particularly allergic contact dermatitis (ACD) and atopic dermatitis (AD). ACD, a type IV delayed hypersensitivity reaction, affects up to 20% of the population.30,31,32,33,34 Irritant contact dermatitis (ICD), which accounts for up to 80% of contact dermatitis cases, results from non-immunologic inflammation caused by repeated exposure to substances like solvents, surfactants, and alkalis. 32 Although distinct in pathophysiology, both ACD and ICD arise from a combination of environmental exposures and intrinsic susceptibility, particularly skin barrier integrity.31,33,34
Genetic disruptions in barrier function, most notably filaggrin (FLG) mutations, are strongly associated with AD and increased sensitization to allergens like nickel.31,35,36 Inflammation further comprises the skin barrier, allowing even weak allergens such as lanolin, parabens, tocopherol, acetate (vitamin E), and commensal antigens to provoke immune responses.33,37 Food proteins have also emerged as cutaneous sensitizers, especially in individuals with chronic hand dermatitis from wet work or in those with underlying AD.33,38
Occupational exposures vary widely across industries and are major contributors to allergen sensitization.39,40 High-risk professions include health care workers, hairdressers, agricultural laborers, construction workers, and those in manufacturing or food processing industry.39,40 In health care, common triggers include formaldehyde and rubber accelerators found in gloves, such as carba and thiuram mix.39,40 Hairdressers are frequently exposed to allergens like nickel in tools, p-phenylenediamine (PPD) in hair dyes, and ammonium persulfate in bleaches.39,40,41,42,43 Construction and manufacturing workers often encounter epoxy resins and chromates, while agricultural laborers may be sensitized to pesticides or plant proteins.39,44 In agriculture, dermatitis may result from both active ingredients and inactive components such as emulsifiers and surfactants, though standardized patch testing for these exposures remains lacking. 10 Jewelry-related allergens—including nickel, gold, and palladium—also remain common, particularly in individuals with piercings.40,45
Diagnosis and personalized management depend heavily on patch testing. Standardized panels like the Thin-layer Rapid-Use Epicutaneous Test are widely used, though custom trays are often required for occupational or uncommon allergens for individual patients. 32 Determining clinical relevance requires linking positive reactions to real-world exposures—a process guided by thorough occupational and personal histories, ingredient reviews, and product labeling. Resources like the Contact Allergen Management Program (CAMP) can generate personalized lists of safe products for each patient, improving adherence to allergen avoidance plans. 32
Lifestyle-based interventions remain foundational. Patients should minimize wet work, use gentle skincare products free of fragrances and preservatives, and apply emollients regularly to restore barrier function.32,34 In occupational settings, changing glove materials, substituting allergenic products, and adjusting workflows can prevent recurrences. 39 These strategies, when guided by patch testing and tailored to individual exposures, offer a precision-based approach to managing chronic dermatitis. Integrating allergen avoidance into dermatologic care reflects the principles of lifestyle medicine: empowering patients to improve skin health by identifying and mitigating modifiable environmental risks.
Diet
Diet affects overall skin health and a variety of dermatological conditions.46,47 Several nutrients and bioactive compounds are thought to contribute to skin health through diverse mechanisms, including antioxidant activity, barrier support, anti-inflammatory effects, and regulation of cellular repair and regeneration.48,49,50 Vitamin A and its derivatives, including carotenoids like β-carotene and lycopene, support immune function, reduce UV-induced damage, and improve conditions such as acne, atopic dermatitis, hidradenitis suppurativa (HS), and photoaging.51,52,53 Vitamin C enhances collagen and elastin synthesis, inhibits melanin production, and provides systemic antioxidant protection against UV radiation and inflammatory skin disorders.48,51,54 Vitamin D, synthesized in the skin, contributes to immune regulation, reduces inflammation in conditions like psoriasis and HS, and helps protect against UV-mediated cellular damage.53,54,55 Vitamin E acts as a lipid-soluble antioxidant that stabilizes cell membranes, supports collagen integrity, and may mitigate inflammation and photoaging.54,56 Essential fatty acids, particularly long-chain omega-3s such as EPA and DHA, play a key role in maintaining the skin barrier, modulating inflammation, and may reduce disease severity in atopic dermatitis, psoriasis, and acne.51,57,58 Polyphenols, including flavonoids like quercetin and kaempferol, protect against oxidative stress, preserve collagen and elastin, modulate inflammation, and may benefit skin structure and appearance when consumed in whole-food form.45,59,60 Zinc is concentrated in the epidermis and can support wound healing, immune defense, UV protection, and treatment of inflammatory dermatoses and HS.54,61–63 Copper facilitates collagen maturation, melanin synthesis, and angiogenesis, and copper peptides may enhance wound healing and skin regeneration.48,64 Selenium, through its role in antioxidant enzymes, can protect against UV-induced DNA damage and may reduce the risk of keratinocyte carcinomas while supporting anti-aging effects.50,51,53,65
Deficiencies in key micronutrients can result in characteristic cutaneous manifestations across multiple sites, including the scalp (alopecia: zinc, copper; hypopigmentation: selenium, copper; pili torti: copper), oral mucosa (gingival swelling or hemorrhage: vitamin C; angular cheilitis: zinc), nails (dystrophy: zinc; leukonychia or paronychia: selenium), and skin (seborrheic dermatitis: copper; follicular hyperkeratosis: vitamin A; perifollicular hemorrhage; hyperkeratotic papules: vitamin C; hidradenitis suppurativa: iron, zinc, vitamin A, vitamin D, vitamin B12).65,66,67 Dietary modifications may support management of inflammatory and autoimmune skin conditions such as psoriasis (address obesity, diabetes, dyslipidemia),47,68,69 atopic dermatitis (allergen avoidance),70,71 acne (low-glycemic, anti-inflammatory diet), and HS (low-glycemic, non-dairy, mediterranean diet).47,66,72 Moreover, evidence suggests that a diet rich in fruits and vegetables, along with the intake of vitamins E and A, selenium, polyphenols, curcumin, lycopene, and grape seed extract, may offer protective effects against nonmelanoma skin cancers.47,48,74
While several of these relationships are theorized or shown in preliminary data, there remains a lack of high-quality evidence to substantiate “gold standard” guidelines for a skin-healthy diet. One sample diet tailored to dermatology proposed target micronutrient levels that emphasize whole foods and limit ultra-processed items, mirroring the principles of the Mediterranean, DASH, and Healthy Eating Index diets. 48 Further research is needed to refine optimal maintenance intake levels and possible supplementation for specific dermatologic conditions.
Exercise
Exercise reduces the thinning of the stratum corneum and increases mitochondrial biogenesis, thus slowing skin cell aging.75,78 To elicit these effects, exercise should be aerobic and performed for at least four hours per week. Studies have also found exercise reduces the risk of psoriasis in women, potentially by modulating proinflammatory cytokines. 75 Exercise also affects dermatologic disease through enhanced wound healing. Increased healing rates were observed in people performing resistance exercises and physical activity for 90 minutes per week. 75 Regular exercise reduces free radical and oxidative stress damage in the skin, which may provide protection from skin neoplasms. 75 Additionally, aerobic and strength training exercise have long-term anti-inflammatory effects with regular practice, particularly in downregulation of TNF-alpha. 76 Psoriasis and dermatomyositis, two dermatologic diseases characterized by increased TNF-alpha, show marked improvement in patient function and reduced disease activity with exercise. 76 Overall, regular exercise of at least moderate intensity improves skin blood flow through vasodilation of cutaneous microvessels, enhances moisturization, strengthens skin structure, and improves skin appearance. 77
However, short-term, initial exercise produces a proinflammatory response that gradually subsides with consistent, regular training. 78 While regular exercise benefits dermatologic disease through the mechanisms described above, environmental factors can mitigate these beneficial effects. For instance, patients with eczema who meet their weekly exercise requirements in chlorinated swimming pools may experience worsening skin conditions due to the environmental exposure. 77 Additionally, patients who neglect post-exercise hygiene practices, including failing to wash their face and skin and allowing sweat, dirt, or makeup to remain on the skin, will experience skin irritation and develop dermatologic conditions such as acne vulgaris. 82 Therefore, patients should recognize that both irregular exercise patterns and environmental factors can promote skin inflammation. 77
Exercise intensity is measured using metabolic equivalents (METs), which quantify oxygen consumption relative to resting levels. 79 Regular beneficial exercise consists of more than four hours per week of high-intensity aerobic activity. Vigorous exercise corresponds to a MET score greater than 6, typically achieved through aerobic or calisthenic exercises. Longer workouts with higher MET scores correlate with greater reductions in inflammatory dermatologic disease risk, particularly psoriasis.75,80 Conversely, physical inactivity harms dermatologic outcomes. Atopic dermatitis, for example, has been specifically linked to lower average physical activity levels. 81 Thus, sedentary patients may be more susceptible to dermatologic diseases, as exercise serves as a protective factor.
Current physician consensus guidelines recommend at least 150 minutes weekly of moderate-intensity aerobic activity plus two additional days of muscle-strengthening exercises. In MET terms, this corresponds to scores ranging from 5-8+ for moderate-to-intense activity. 83 These activities include walking and running at a 10-minute mile pace. Physicians, including dermatologists, should discuss exercise as a lifestyle factor with patients given its significant impact on chronic disease management.
Sleep
Immune function, which is tightly interlinked with inflammatory dermatologic diseases, is modulated by sleep and the circadian rhythm. Proinflammatory cytokines increase with sleep deprivation, serving as potential drivers of inflammatory skin disease. Natural killer cells are lost in sleep deprivation, making individuals prone to infections that can result in cutaneous manifestations. 84 During sleep, the skin is able to repair and regenerate, so accelerated skin aging and weak barrier functioning are associated with inadequate sleep. 85 Melatonin falls with increased inadequate sleep, which not only is essential for circadian rhythm but for antioxidant function within the skin barrier. 85 During sleep, there is a net transepidermal water loss which can lead to increased pruritus at night. 84 Overall, sleep and dermatologic disease have a complex relationship, intermediated by immunology and skin physiology. 85 Specifically, atopic dermatitis, psoriasis, chronic urticaria, skin infections, and pruritus are skin diseases associated with disorders that alter normal sleep, like obstructive sleep apnea, narcolepsy, and parasomnia. 84
Although sleep deprivation can amplify dermatologic diseases, dermatologic diseases can impede sleep. Nocturnal pruritus, a manifestation of skin infection (Sarcoptes scabiei, Staphylococcus aureus), atopic dermatitis, psoriasis, and urticaria, can result in sleep disturbances when at a high enough intensity for the individual. 84 Chronic pruritus significantly impairs sleep quality, productivity, and mental health, making it essential for dermatologists to assess these lifestyle impacts when treating affected patients. 84
Patients with poor sleep quality, as assessed through validated instruments like the Pittsburgh Sleep Quality Index, should receive comprehensive interventions including sleep hygiene counseling, lifestyle modifications, and when appropriate, pharmacological treatments such as melatonin or sedatives to reduce inflammation and prevent the onset or exacerbation of dermatologic conditions. The American College of Lifestyle Medicine recommends a full 7-8 hours of sleep for adults each night as the standard of self-care and for the prevention of chronic disease, although this measure increases in infants and childhood.5,10 For patients experiencing new-onset inadequate sleep due to the pruritus or other distressing symptoms of a dermatologic disease, comprehensive management may require a dual approach addressing both the underlying skin condition and the resulting sleep disturbance87,88. Dermatologic treatment may involve topical or systemic therapies (including hydrocortisone, triamcinolone, diphenhydramine) aimed at reducing inflammation, itch, and discomfort, while concurrent sleep-focused interventions, such as cognitive behavioral therapy for insomnia (CBT-I), sleep hygiene education, or short-term pharmacological aids, can help restore restful sleep and improve overall quality of life. This multidisciplinary approach is particularly important, as untreated sleep disruption can exacerbate dermatologic symptoms and contribute to a cycle of worsening disease severity and psychological distress. 85
Smoking
Tobacco use exerts widespread systemic effects through oxidative stress, immune dysregulation, and impaired angiogenesis, 5 all of which have direct implications for skin health and disease management. From a lifestyle medicine perspective, tobacco exposure should be considered a modifiable risk factor for numerous dermatologic conditions. Smoking through any delivery method—including cigarettes, vaping, or hookah—results in significant toxicant exposure that produces adverse cutaneous effects. 86 Among these methods, cigarettes pose the greatest risk due to their exceptionally high levels of harmful compounds, including particulate matter, reactive gases, heavy metals, and N-nitrosamines, which accelerate skin aging, promote chronic inflammation, and impair wound healing processes. 86
Clinically, smoking hastens skin aging through increased matrix metalloproteinase activity, depletion of antioxidants, and disruption of TGF-β signaling pathways.90,91,92 These changes result in collagen degradation, elastosis, and the characteristic “smoker’s face,” marked by periorbital wrinkling, atrophy, and yellowish discoloration. 92 Among current smokers in a study done in 1999, awareness of smoking’s link to premature facial aging was significantly lower than among former smokers, yet nearly 25% of smokers (especially younger ones) believed this information would influence quit decisions. 89 These findings suggest that incorporating premature aging messages into tobacco control campaigns could be an effective cessation strategy, particularly given smokers’ own recognition of its potential impact on quit attempts.89,92
Tobacco is also implicated in oncogenesis. Beyond its systemic carcinogenicity, it contributes to increased risk for squamous cell carcinoma and anogenital keratinocytic malignancies, particularly in HPV-positive individuals.86,94,95,96 Though associations with basal cell carcinoma and melanoma are inconsistent,86,97,98 the immunosuppressive effects of smoking are especially concerning in high-risk populations and patients should be counseled appropriately. Conversely, transdermal nicotine patches, while serving as valuable smoking cessation aids, may paradoxically undermine quit attempts by causing irritant or allergic contact dermatitis in some patients, potentially discouraging continued use of this therapeutic intervention. 94
In inflammatory skin diseases, tobacco use is a well-established risk factor. Smokers are significantly more likely to develop psoriasis and palmoplantar pustulosis, with a dose-dependent relationship.99,100 Hidradenitis suppurativa is strongly linked to smoking, and while cessation may not fully reverse disease risk, it can lead to symptomatic improvement.101,102 There is also evidence to suggest that smoking is associated with atopic dermatitis and allergic and irritant contact dermatitis.102,103 Autoimmune diseases like systemic and cutaneous lupus erythematosus are also exacerbated by smoking, which is associated with higher disease activity and reduced response to standard treatments such as hydroxychloroquine.104,105,106,107
While a few conditions, including pemphigus vulgaris, Behçet disease, and pyoderma gangrenosum, have shown paradoxical improvements with tobacco use,86,108,109 these findings are preliminary and should not alter cessation recommendations. In clinical practice, smoking cessation should be framed not only as a cardiovascular or oncologic intervention but also as a dermatologic one, as visible skin improvements and reduced disease burden can serve as powerful motivators. 89 While dedicated tobacco cessation counseling led by dermatologists may not be feasible given high patient volumes and time constraints, every physician has an ethical obligation to remind patients about smoking’s detrimental effects on health and encourage cessation. Even brief interventions in the dermatology setting can be impactful, particularly when patients can directly observe smoking-related skin damage and understand the potential for visible improvement upon quitting.86,89,92
Alcohol
Alcohol disrupts cardiovascular, immune, and hemostatic systems through both acute and chronic effects, contributing to cutaneous manifestations. 110 It alters vascular tone and peripheral circulation, impairs cellular immunity by reducing T cell and neutrophil function, and disrupts coagulation through defective platelet production and aggregation.111,112 These systemic changes help explain the diverse skin findings observed in individuals with alcohol use. Chronic use is associated with cutaneous signs such as jaundice, pruritus, pigmentary changes, urticaria, alterations in hair and nails, and oral mucosal changes. 113 Alcohol-related liver dysfunction may underlie many of these findings (particularly jaundice, pruritus, and spider angiomas) while vascular abnormalities such as telangiectasias, palmar erythema, caput medusae, and flushing may result from hepatic decompensation or direct vasodilatory effects of ethanol. 110 Additional vascular manifestations include plethoric facies, unilateral nevoid telangiectasia, and corkscrew scleral vessels. Nail abnormalities commonly observed include Terry’s nails, red lunulae, koilonychia, and clubbing, while hyperpigmentation can reflect systemic effects or nutritional deficiencies. 110
Oral manifestations of chronic alcohol use include black hairy tongue, glossitis, leukoplakia, and parotid swelling, often resulting from poor oral hygiene, nutritional deficiencies, or immune suppression.110,113 Other alcohol-associated conditions with skin involvement include Dupuytren’s contracture, Madelung’s disease, and coagulopathy. 106
Alcohol is a known exacerbating factor for chronic inflammatory skin diseases such as psoriasis, rosacea, and discoid eczema.110,113,114,115,116 Patients with chronic inflammatory conditions such as psoriasis or eczema are at increased risk for alcohol use disorder, with studies reporting prevalence as high as 30% based on CAGE (Cut down, Annoyed, Guilty, Eye-Opener), AUDIT-C (Alcohol Use Disorders Identification Test-Consumption), and MAST (Michigan Alcohol Screening Test) questionnaires.117,118 Alcohol use in these populations is not merely incidental but may reflect attempts to cope with psychological distress, and impaired quality of life. Evidence suggests alcohol may also contribute to decreased response to treatment, including biologics. 119 Given these findings, routine alcohol screening using tools such as AUDIT-C, CAGE, or the single alcohol screening question should be integrated into dermatologic care for patients with chronic inflammatory conditions or other dermatologic disease with high burden. Additionally, alcohol is an established risk factor for skin infections and certain malignancies, including basal cell carcinoma and melanoma, potentially due to immunosuppression and oxidative stress associated with chronic intake.120,122 Physicians should be aware of the systemic and cutaneous effects of alcohol use, incorporate appropriate screening and counseling into dermatologic care, and communicate findings of unhealthy alcohol consumption to the patient’s primary care provider to ensure coordinated, multidisciplinary management that addresses both dermatologic and systemic health concerns.
Stress and Trauma
Stress plays a significant role in skin health through dysregulation of the neuroendocrine-immune (NEI) axis. Activation of the hypothalamic–pituitary–adrenal (HPA) axis and sympathetic nervous system leads to elevated cortisol, catecholamines, and proinflammatory cytokines such as IL-1β, IL-6, and TNF-α.122,123 These mediators impair skin barrier function, delay wound healing, and disrupt keratinocyte differentiation, increasing susceptibility to inflammation and infection. 124 Acute stress can heighten itch via mast cell activation and neuropeptide release, while chronic stress contributes to sustained immune dysregulation and disease flares.
Dermatoses like psoriasis, atopic dermatitis, and alopecia areata are particularly sensitive to stress. In psoriasis, increased expression of corticotropin-releasing hormone (CRH) and CRH receptor 1 in lesional skin suggests local HPA axis dysregulation may perpetuate inflammation. 125 Psychological stress may also promote disease chronicity through expansion of tissue-resident memory T cells. 127 Moreover, skin disease itself can cause psychosocial distress, creating a feedback loop that worsens both psychological and dermatologic outcomes. 126
Stress can be assessed using self-report tools such as the Perceived Stress Scale (PSS) and Dermatology Life Quality Index (DLQI), which measure emotional burden and quality of life related to dermatologic disease.128,129,130 Biomarkers like salivary cortisol and heart rate variability provide physiologic insights. 131 These measures also assist with risk stratification, helping identify patients more likely to experience flares or impaired treatment response under stress.122,123
Management strategies can be personalized based on stress sensitivity. Patients with high perceived stress may benefit from cognitive behavioral therapy, mindfulness-based stress reduction, or guided imagery. Early studies have demonstrated that mindfulness-based interventions may improve outcomes in psoriasis. One pilot study evaluated a structured eight-week mindfulness-based cognitive therapy program among individuals with psoriasis and found that participation led to improved illness perceptions and reduced distress, supporting the role of mindfulness in addressing the psychosocial burden of the disease. 132 Another randomized controlled trial tested an eight-week mindfulness-based stress reduction program delivered alongside standard phototherapy or photochemotherapy for moderate-to-severe psoriasis. Participants in the mindfulness-based stress reduction program showed significantly faster rates of skin clearing compared to those receiving phototherapy alone, suggesting that stress reduction can meaningfully augment treatment response through psychoneuroimmunologic mechanisms. 133
Such interventions are increasingly recognized as effective complements to medical therapy. Incorporating these approaches into dermatologic care may help disrupt the stress–skin disease cycle and enhance both cutaneous and mental health outcomes.134,135
Social Support
Social support significantly influences skin health and disease outcomes. High levels of support can buffer psychosocial stress, a known trigger for flares in chronic inflammatory conditions such as psoriasis and atopic dermatitis. 135 Patients with strong support networks often report better quality of life, fewer depressive symptoms, and greater acceptance of their disease. 136 Conversely, individuals with limited or poor-quality support tend to experience increased psychological burden. For example, vitiligo patients with low perceived support exhibit significantly higher levels of depression and distress, underscoring the psychosocial weight of visible skin conditions. 138 Social support likely functions as a protective factor within the brain–skin axis, mitigating inflammatory pathways through stress modulation.
Validated tools such as the Multidimensional Scale of Perceived Social Support (MSPSS) and the UCLA Loneliness Scale can help quantify patients’ social environments. 138 In dermatology, quality-of-life measures like the Dermatology Life Quality Index (DLQI) capture the emotional and social dimensions of disease burden, indirectly reflecting the impact of support systems. 137 Monitoring these indices in clinical settings can help identify individuals whose psychosocial well-being is declining. Support level also informs clinical risk stratification. Patients with low support show consistently worse quality of life and more severe depressive symptoms, particularly in psoriasis and vitiligo.136,138,139, 140 Crucially, inadequate support can undermine treatment adherence. Studies show that tangible forms of assistance, such as help with medication routines, are associated with higher adherence rates and better therapeutic outcomes. 136
Personalizing dermatologic care based on social support can improve both mental and physical health. For patients with minimal support, referrals to support groups, psychoeducational programs, or digital communities may foster connection and resilience. Reinforcing strong support networks in well-connected patients can likewise enhance adherence and engagement, supporting a more holistic, lifestyle-informed model of dermatologic care.
Conclusion
Summary of Impact(s) of Lifestyle Factors on Dermatologic Disease.
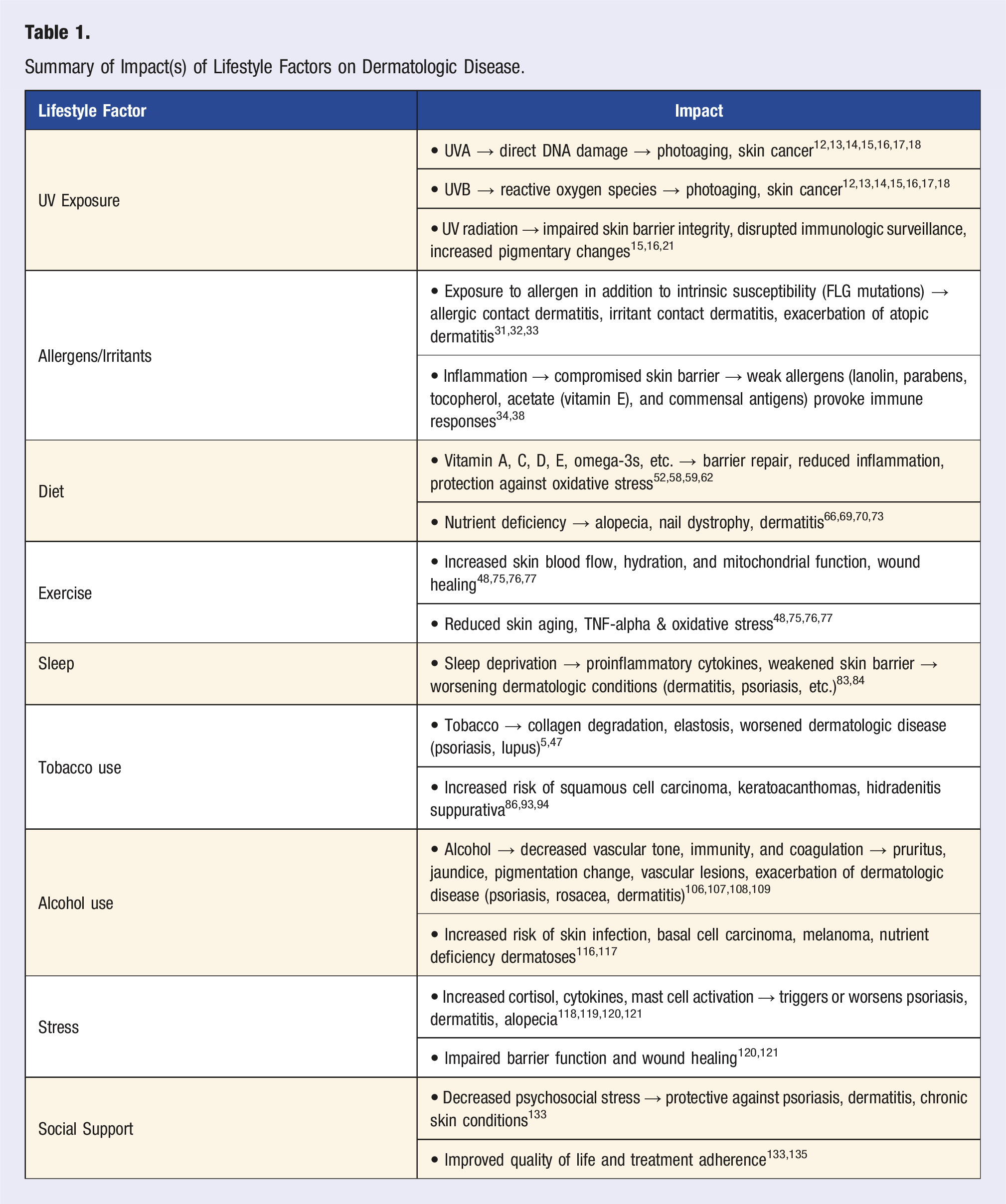
The integration of lifestyle medicine principles into dermatologic practice requires a shift from purely pharmacologic approaches toward personalized, multidisciplinary care models. Practical implementation begins with incorporating validated assessment tools such as the DLQI, PSQI, and stress scales to identify high-risk patients who may benefit from lifestyle interventions. For patients with chronic inflammatory conditions like psoriasis or atopic dermatitis, addressing modifiable lifestyle factors—through dietary modification, stress management techniques, exercise counseling, and substance cessation—can meaningfully improve treatment outcomes while simultaneously addressing common comorbidities.
The therapeutic multiplicity of lifestyle interventions offers particular advantages in dermatology, where conditions often have systemic implications. Dietary interventions targeting inflammatory pathways benefit both skin disease and cardiometabolic health. Exercise recommendations reduce inflammatory markers while supporting wound healing and photoprotection. Stress management techniques can break the cycle of psychological distress and disease flares that characterizes many chronic dermatoses.
Clinical implementation challenges include the need for interdisciplinary collaboration, extended consultation times, and patient adherence to long-term behavioral changes. However, the potential for sustained disease improvement, reduced medication dependence, and enhanced quality of life justifies investment in these approaches. Digital health platforms and community resources can extend therapeutic reach beyond traditional clinical encounters, supporting sustained behavioral change.
Future research priorities should focus on standardized lifestyle intervention protocols and long-term cohort studies incorporating real-world data to establish optimal dosing and timing of interventions. The development of precision medicine approaches that account for genetic susceptibility, environmental factors, and individual stress reactivity represents a particularly promising direction.
Dermatologists are uniquely positioned to champion lifestyle medicine integration given the visible nature of skin disease and its profound psychosocial impact. By addressing lifestyle factors alongside conventional treatments, dermatologic practice can evolve toward a more comprehensive, preventive model that treats the whole person rather than isolated cutaneous manifestations. This approach not only improves dermatologic outcomes but positions the specialty at the forefront of patient-centered, sustainable health care delivery.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article
Data Availability Statement
The information pulled that support the findings of this study are openly available in PubMed as listed in the reference section.
